Abstract
Purpose:
To evaluate the anatomic changes in the cornea and anterior segment following intravitreal aflibercept loading dose for neovascular age-related macular degeneration.
Methods:
The study included 40 eyes of 40 patients with neovascular age-related macular degeneration. Each patient underwent a loading dose of one injection per month for three consecutive doses of aflibercept (0.05 ml/2 mg). Before and after the loading dose, a record was made for each patient of corneal topography, anterior segment, corneal densitometry, and lens densitometry parameters with the Pentacam HR and specular microscopy parameters with a non-contact specular microscope. The data before and after the aflibercept loading dose were compared.
Results:
Corneal densitometry parameters in the 0- to 2-mm and 2- to 6-mm concentric zones of the posterior layer were significantly higher after the loading dose compared with baseline (
Conclusion:
The aflibercept loading dose caused slightly increased densitometric values in some corneal regions while it did not affect the corneal topography, anterior segment, lens densitometry, and specular microscopy parameters.
Keywords
Introduction
Vascular endothelial growth factor (VEGF) plays a crucial role in the pathogenesis of many retinal diseases including neovascular age-related macular degeneration (nAMD) and anti-VEGF agents are used in its treatment. The discovery of the ocular effect of bevacizumab (Avastin; Genentech, South San Francisco, CA, USA), which is actually used in colon cancer, has made it very popular in the treatment of nAMD despite its off-label use in ophthalmology.1,2 Then, ranibizumab (Lucentis; Genentech, Inc., South San Francisco, CA), which has been approved by the Food and Drug administration (FDA), started to be used safely in ophthalmological treatments. 3 As a relatively new anti-VEGF agent, intravitreal aflibercept (Eylea; Regeneron, Tarrytown, NY, ABD and Bayer, Leverkusen, Germany) has emerged as an effective alternative for primary treatment of nAMD or as ‘salvage’ therapy for inadequate treatment response to other anti-VEGF agents.4,5 The differences between anti-VEGF agents originate from their efficacy mechanism. Bevacizumab binds to all subtypes of VEGF to prevent new vessel formation while ranibizumab blocks all biologically active isoforms of VEGF-A. Unlike other anti-VEGF agents, aflibercept is a soluble fusion protein that can also bind to placental-induced growth factor, which is a critical factor involved in pathological neoangiogenesis. 6
Endophthalmitis and other ocular complications together with systemic cardiovascular complications from the use of anti-VEGFs have been studied at various times. Recently, possible anterior segment complications have also attracted the attention of many researchers. Bevacizumab was shown to be present in the anterior chamber of rabbit eyes even 1 month after intravitreal injection. 7 VEGF and its receptors have been detected in the corneal endothelium. 8 Some studies have also demonstrated the passage of intravitreally injected bevacizumab into the aqueous humor.9,10 In a recent study by Arslan and colleagues 11 of the short-term effects of three anti-VEGFs (bevacizumab, ranibizumab, aflibercept) on the cornea, anterior chamber, and intraocular pressure (IOP), a significant reduction was detected in anterior chamber volume (ACV) and endothelial cell density (ECD) values after two intravitreal injections despite no significant difference in IOP and central corneal thickness (CCT) values. In a part of the RE-VIEW Study, the effects of repeated intravitreal aflibercept injection on the corneal endothelium were reported and no apparent corneal endothelial toxicity was noted on specular microscopy in patients treated for nAMD. 12
The aim of this study was to investigate the anatomic changes in the cornea and anterior segment with Scheimpflug tomography together with specular microscopy following aflibercept loading dose for nAMD.
Methods
This prospective study was conducted with 40 eyes of 40 patients with nAMD who were administered an aflibercept loading dose of three consecutive intravitreal aflibercept (0.05 ml/2 mg) injections. The study was conducted in accordance with the tenets of the Declaration of Helsinki at a tertiary referral hospital and was approved by Ankara Numune Training and Research Hospital Ethics Committee (E-18-1754). Informed consent was obtained from all patients included in the study.
Patients were excluded from the study based on the following criteria: a history of cataract surgery, known glaucoma, a history of previous intravitreal anti-VEGF injection and systemic anti-VEGF therapy, endothelial cell count <1000/mm2, a history of intraocular surgery, ocular trauma, inflammation (e.g. uveitis), corneal disease such as corneal scarring, Fuchs endothelial dystrophy, arcus senilis, pseudoexfoliation syndrome, refraction error above −2/+2 diopter, retinal diseases other than nAMD, and chronic topical medication usage. Any phakic patients with cortical, anterior, or posterior subcapsular, or pronounced nuclear cataracts were also excluded from the study because they would affect the reliability of lens density measurements and the evaluation of the fundus.
The neovascular type of AMD was confirmed with color fundus photography, optical coherence tomography, and fundus fluorescein angiography before beginning treatment. The intravitreal aflibercept injections were performed in clean room conditions. The eyelids and ocular surfaces of each patient were sterilized with 5% povidone-iodine, and a lid speculum was placed. Topical proparacaine hydrochloride ophthalmic solution (Alcaine®) 0.5% was instilled. A 30-gauge needle was filled with 0.05 ml of aflibercept, which was inserted up to the needle hub, and the injection was performed from the inferotemporal pars plana, 3.5 mm posterior to the limbus, as all the patients were phakic. A sterile cotton swab was applied as an anti-reflux system when the needle was withdrawn. Topical moxifloxacin 0.5% (Vigamox, Alcon) was administered four times a day for 1 week.
All patients underwent complete ophthalmologic examination including the best-corrected visual acuity (BCVA) with Snellen chart, slit-lamp biomicroscopy, IOP measurements using a pneumotonometer, and dilated fundoscopy. BCVA was recorded in Snellen decimal units. Optical coherence tomography measurements were performed at baseline and at every month throughout the follow-up period.
Scheimpflug measurements were performed with Pentacam HR (Pentacam HR, Oculus, Wetzlar, Germany) at baseline and 1 month after the aflibercept loading dose. The curtains of the room were closed to reduce light penetration and the room lights were also switched off for all examinations to be able to obtain a reflex-free image. All measurements were taken under standard dim-light conditions, between 8.00 and 12.00 a.m. and by the same experienced technician. Distorted images caused by high reflection that could not be evaluated were not included in the analysis. Each patient underwent two consecutive measurements. The first was without pupillary dilation and from these measurements, corneal densitometry and corneal topography values were recorded. The second was performed after full pupillary dilation with tropicamide eye drops to visualize the lens, and lens densitometry values were recorded from these measurements.
Corneal densitometry is an add-on to the standard software for the Pentacam HR and automatically locates the corneal apex and analyzes an area around it with a 12-mm diameter. This 12-mm corneal area is further divided into four concentric zones. The first zone consists of a circular area 2 mm in diameter at the center of the cornea. The second zone comprises a 2–6 mm annular area around it. The third zone consists of a 6–10 mm annular area, and the fourth zone is a 10–12 mm annular area. This analysis can be subdivided into anterior, central, and posterior layers based on corneal depth rather than on specific anatomic layers of the cornea; the anterior layer corresponds to the anterior 120 µm and the posterior layer to the most posterior 60 µm of the cornea. The central corneal layer has no fixed thickness, but is defined by subtraction of the anterior and posterior layers from the total thickness. Corneal densitometry values are expressed as the pixel luminance per unit volume in the Scheimpflug image, and these are given in gray scale units (GSU). The GSU is calibrated by proprietary software, which defines a minimum light scatter of 0 (maximum transparency) and a maximum light scatter of 100 (minimum transparency) according to backscattering light from the cornea. Backward light scatter intensities were evaluated in the 0–2, 2–6, 6–10, and 10–12 mm zones through the entire depth of the cornea.
Maximum keratometry (Kmax), corneal volume (CV), anterior chamber depth (ACD), ACV, anterior chamber angle (ACA), and pupil diameter (PD) values were recorded as corneal topographical and anterior segment parameters.
Four lens densitometry measurement options are available in Pentacam HR, which are peak (one point) scan, linear scan, area scan, and three-dimensional (3D) scan modes. In 3D scan mode, the mean lens density value is calculated in 3D zones located around the center of the pupil (zone 1: 2.0 mm, zone 2: 4.0 mm, and zone 3: 6 mm).
Corneal endothelial measurements were performed by the same experienced technician using a non-contact specular microscope (Tomey EM-4000, Tomey Co., Japan). Subjects were asked to look at the central fixation target, and the autoalignment function was used. At least 110 cells per measurement were included in each analysis automatically and the center method was used. Mean ECD, coefficient of variation (CoV) of cell size (standard deviation of the cell area/mean cell area), and percentage of hexagonal cells (HEX ratio) were recorded.
Statistical analysis
Statistical analysis was performed with SPSS (Statistical Package for Social Sciences) V22.0 for Windows software (SPSS Inc., Chicago, IL, USA). Conformity of the data to normal distribution was analyzed with the Kolmogorov–Smirnov test. Descriptive statistics were stated as mean ± standard deviation (SD) values. The paired-samples
Results
Evaluation was made of 20 males (50%) and 20 females (50%) with a mean age of 70.04 ± 6.4 years (range, 57–79 years). The mean BCVA values were 0.32 ± 0.20 (range, 0.01–0.7) and 0.42 ± 0.23 decimal (range, 0.01–0.80) before and after the aflibercept loading dose (
The corneal densitometry values of all the patients are shown in Table 1. Corneal densitometry parameters in the 0–2 mm and 2–6 mm concentric zones of the posterior layer were significantly higher after the aflibercept loading dose compared with baseline values (
Comparison of corneal densitometry parameters before and after aflibercept loading dose.
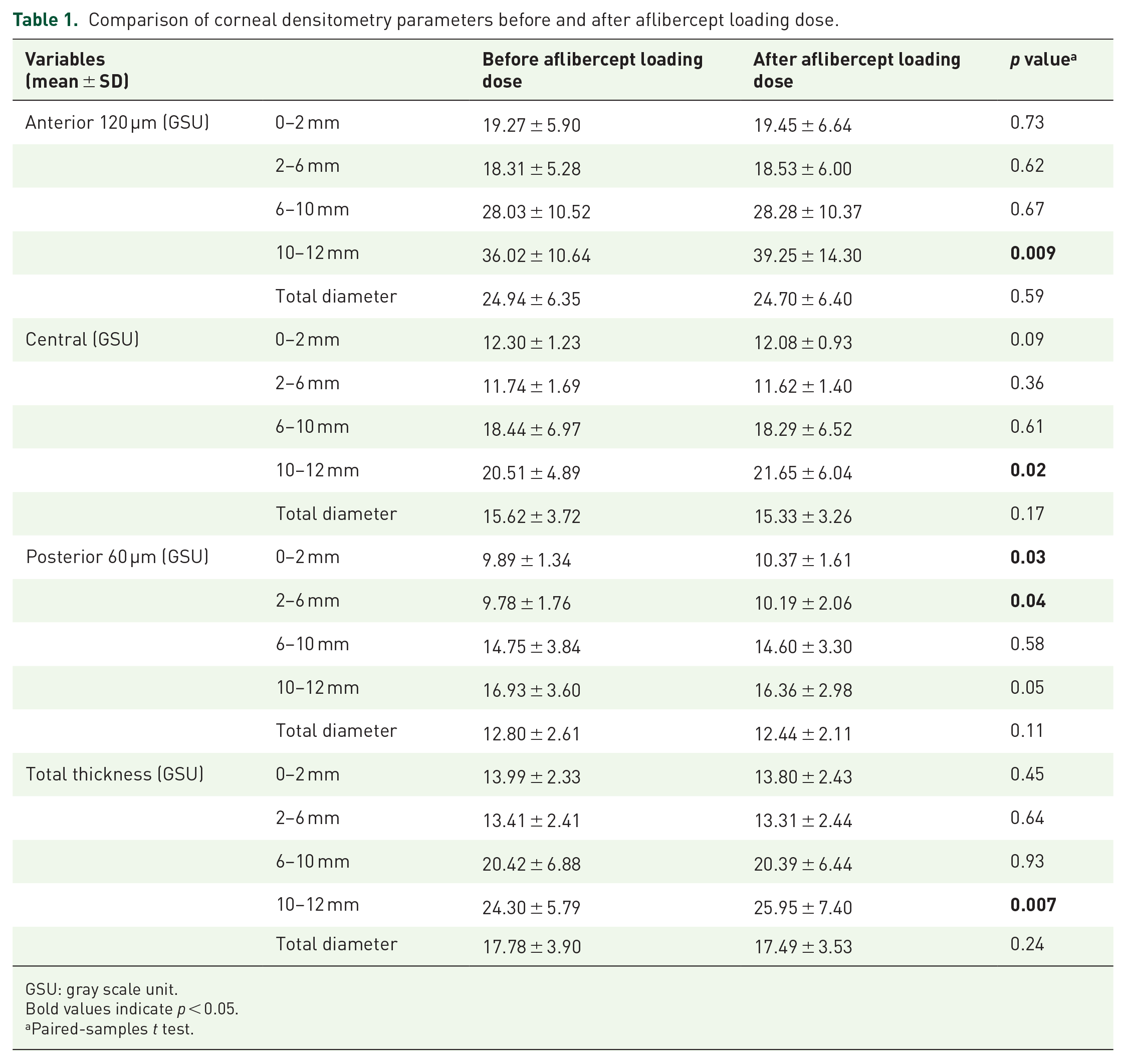
GSU: gray scale unit.
Bold values indicate
Paired-samples
The corneal topography parameters are shown in Table 2. There were no significant differences in Kmax, CV, ACD, ACV, ACA, and PD values after the aflibercept loading dose (
Comparison of corneal topographical and anterior segment parameters before and after aflibercept loading dose.

ACA: anterior chamber angle; ACD: anterior chamber depth; ACV: anterior chamber volume; CV: corneal volume; Kmax: maximum keratometry; PD: pupil diameter.
Paired-samples
The lens densitometry values of all the patients are shown in Table 3. There were no significant differences in the mean values of average and maximum lens densitometry measurements before and after the aflibercept loading dose (
Comparison of lens densitometry and specular microscopy parameters before and after aflibercept loading dose.
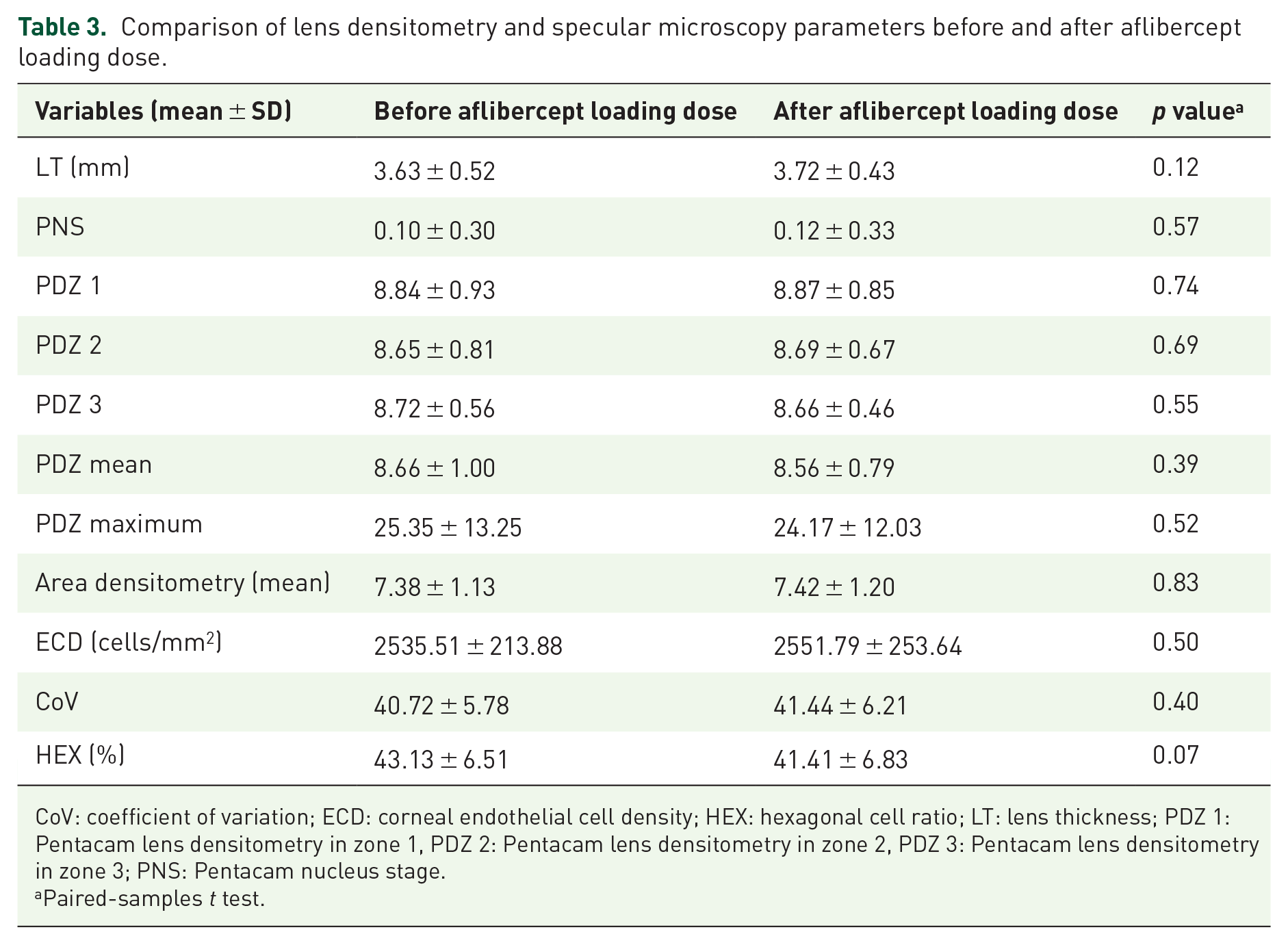
CoV: coefficient of variation; ECD: corneal endothelial cell density; HEX: hexagonal cell ratio; LT: lens thickness; PDZ 1: Pentacam lens densitometry in zone 1, PDZ 2: Pentacam lens densitometry in zone 2, PDZ 3: Pentacam lens densitometry in zone 3; PNS: Pentacam nucleus stage.
Paired-samples
The corneal endothelial parameters, ECD, CoV, and HEX ratio, did not change after the aflibercept loading dose (
Discussion
Age-related macular degeneration is the third leading cause of severe vision loss worldwide. 13 Neovascular AMD is the late phase of the disease in which the choroidal neovascular membrane (CNVM) is observed. 14 Bevacizumab, ranibizumab, and aflibercept, as a newer intravitreal anti-VEGF agent, are often used in the treatment of nAMD. Systemic and ocular safety is as important as the effectiveness of anti-VEGF agents, which are frequently used in the treatment of nAMD.
In both VIEW 1 and VIEW 2 studies, the most common ocular adverse events that were experienced with aflibercept and ranibizumab were conjunctival hemorrhages, eye pain, and vitreous floaters.4,15– 17 Fine and colleagues 18 also reported sterile inflammation after intravitreal aflibercept injection. In those studies, anterior segment effects were not defined in detail. In this study, the changes in corneal densitometry, corneal topography, lens densitometry, and specular microscopy parameters were investigated in detail after aflibercept loading dose.
Corneal densitometry parameters in the 0–2 mm and 2–6 mm concentric zones of the posterior layer were statistically higher after the loading dose compared with baseline. There are
The injection of the volume of 0.1 ml into the vitreous cavity, which is an enclosed area with little room for expansion, may cause a rise in IOP immediately after the procedure through the biomechanical properties of the sclera and the cornea, and expansion in the vitreous cavity may lead to changes in axial length, ACD, and ACV. 23 Unfortunately, there are few data about this issue in the literature. Alkin and colleagues 24 found that the changes in anterior chamber parameters returned to baseline values after the first few minutes of bevacizumab (0.05 ml) or bevacizumab-triamcinolone acetate combination (0.1 ml) injection. IOP may rise above 40 mmHg immediately after anti-VEGF and may fall in the first 15 min post-injection. 25 In the study by Arslan and colleagues, 11 the short-term anterior segment effects were investigated of three different types of anti-VEGF in the four different retinal diseases of diabetic retinopathy, nAMD, central retinal vein occlusion, and central serous chorioretinopathy. This methodology prevented clear information about which agent and which diagnosis had caused which finding. Without making a comparison, that study concluded that anti-VEGF agents decreased ACD after the first and second injections compared with pre-injection while there was no change in IOP and CCT values. In this study, there were no significant changes after the first and second aflibercept injections in anterior segment parameters including ACD, ACV, ACA, Kmax, CV, and PD values. Aflibercept, like other anti-VEGF agents, might cause anterior segment changes in the very early phase due to vitreous expansion but after a loading period, no permanent change is expected in anterior segment parameters.
Cataract development in patients receiving intravitreal corticosteroids has been investigated in many studies. Anayol and colleagues 26 reported that the mean lens densitometry value in zone 1 increased significantly 3 months after intravitreal dexamethasone implant injection. Similarly, cataract is also an ocular complication of intravitreal anti-VEGF injection. 15 In a study which reported 12-month outcomes with as-needed aflibercept, the most common ocular adverse events were progressive diabetic retinopathy (12%) and worsening cataract (10%). 27 To the best of our knowledge, there is no study in the literature about the lens densitometry change after aflibercept injection. The results of this study showed that there were no significant differences in any of the three lens densitometry zones after the aflibercept loading dose compared with baseline. Further prospective studies with longer follow-up period are needed to correlate the densitometric changes of lens with injection number and duration.
Corneal endothelium expresses VEGF receptors 8 and anti-VEGFs injected intravitreally have been detected in the aqueous humor.28,29 Hence, intravitreal anti-VEGFs may affect the functions of VEGF in the corneal endothelium. Intravitreal bevacizumab injection has been reported to have no harmful effects on the corneal endothelium. 30 Pérez-Rico and colleagues 31 found no significant differences in central CD, CV, and the percentage of HEX at 7 days and 6 months after the first intravitreal ranibizumab injection. The RE-VIEW study investigated the corneal endothelial safety of repeated injections of aflibercept in patients with unilateral nAMD and reported that ECD, CoV, and HEX ratio values remained unchanged at 52 weeks. 12 In a study by Muto and Machida, 32 the effect of aflibercept injection on the corneal endothelium was investigated at 1, 3, and 6 months after a single dose of aflibercept injection, and no significant difference was found in ECD, CoV, and HEX ratio values. Similar to the aforementioned studies, in this study, there were no significant changes in ECD, CoV, and HEX ratio values after the aflibercept loading dose compared with baseline. When the findings are analyzed together, the corneal densitometry alteration could not be explained by corneal endothelial dysfunction. Therefore, this may also support the hypothesis that altered corneal densitometry after an aflibercept loading dose might be due to altered corneal tight junctions.
This study has some limitations, primarily the relatively short follow-up period and small sample size. Nevertheless, the aim of the study was to show the effect of aflibercept loading dose on cornea and anterior segment parameters.
In conclusion, aflibercept loading dose can affect corneal densitometry values leading to slightly decreased corneal transparency. Aflibercept loading dose may be safe for the anterior chamber, lens, and corneal endothelium as it was not seen to have any effect on anterior chamber, lens densitometry, and corneal endothelium parameters. Long-term follow-up studies with larger sample sizes may provide additional contributions to the literature.
Footnotes
Acknowledgements
The paper was reviewed by a native English speaker, Dr Caroline J Walker, PhD.
Conflict of interest statement
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
