Abstract
A transparent cornea is essential for the formation of a clear image on the retina. The human cornea is arranged into well-organized layers, and each layer plays a significant role in maintaining the transparency and viability of the tissue. The endothelium has both barrier and pump functions, which are important for the maintenance of corneal clarity. Many etiologies, including Fuchs’ endothelial corneal dystrophy, surgical trauma, and congenital hereditary endothelial dystrophy, lead to endothelial cell dysfunction. The main treatment for corneal decompensation is replacement of the abnormal corneal layers with normal donor tissue. Nowadays, the trend is to perform selective endothelial keratoplasty, including Descemet stripping automated endothelial keratoplasty and Descemet’s membrane endothelial keratoplasty, to manage corneal endothelial dysfunction. This selective approach has several advantages over penetrating keratoplasty, including rapid recovery of visual acuity, less likelihood of graft rejection, and better patient satisfaction. However, the global limitation in the supply of donor corneas is becoming an increasing challenge, necessitating alternatives to reduce this demand. Consequently,
Introduction
The human cornea is a transparent avascular tissue that transmits light to the retina. The cornea is arranged into well-organized layers, and each layer plays a significant role in maintaining the transparency and viability of the tissue. These layers include the epithelium, Bowman’s layer, the stroma, Descemet’s membrane, and the endothelium (Figure 1). Transparency of the corneal stroma is preserved by the critical spacing and crystalline organization of collagen fibers and a relative state of dehydration. 1 The corneal endothelium, located at the basement (Descemet’s) membrane, is the innermost corneal layer (Figure 1). 2 This layer has barrier and pump functions that are important for corneal clarity maintenance. 3 Unlike the epithelium, which has self-renewing capacity, the endothelium is not known to proliferate.4,5 Therefore, cell damage caused by different pathologies stimulates the remaining endothelial cells to enlarge and migrate to cover any defects, thereby maintaining corneal transparency.
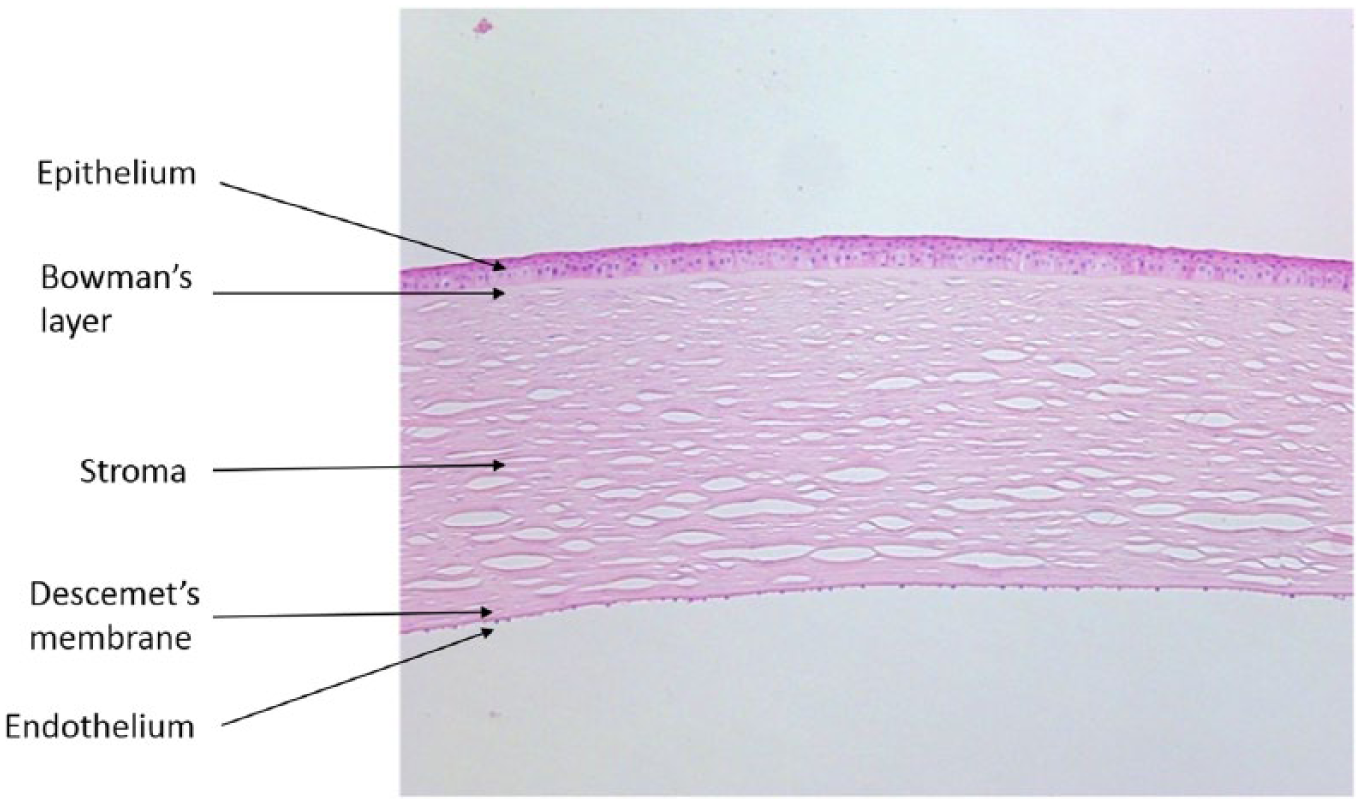
The normal cornea consists of five layers, including the epithelium, Bowman’s layer, stroma, Descemet’s membrane, and endothelium. The endothelial cells form a single hexagonal monolayer located in the posterior cornea (arrow; hematoxylin and eosin staining, 10×).
Corneal endothelial decompensation leads to blurred vision and discomfort or even severe pain. Although medical therapy can be used to relieve symptoms, the only definitive treatment for corneal endothelial dysfunction is corneal transplantation, which can be performed in the form of full-thickness penetrating keratoplasty (PK) or selective keratoplasty. Selective endothelial keratoplasty has become popular in corneal endothelial dysfunction management owing to quicker visual rehabilitation and lower complication rate. Despite this, the global limitation in the supply of donor corneas is becoming a growing challenge, necessitating alternatives to reduce the demand. 6 One option involves culturing corneal endothelial cells in the laboratory and then transplanting these cells into patients. Herein, we describe the etiologies and clinical manifestations of human corneal endothelial cell dysfunction and summarize the options and recent developments in the management of corneal endothelial dysfunction.
Review criteria
A PubMed review was performed using the search terms ‘human cornea’, ‘endothelial cell’, ‘endothelium’, ‘dysfunction’, ‘corneal edema’, and ‘decompensated cornea’. All publications on ‘corneal endothelial dysfunction’ published from 1960 to 2017 were screened. This review includes human and animal studies that were published in full and in the English language.
Physiology and function of human corneal endothelial cells
Human corneal endothelial cells are located at the posterior cornea and form a single hexagonal monolayer, which is formed by the first wave of migration of neural crest cells that derive from the edge of the invaginating optic cup. 7 The two main roles of corneal endothelial cells are barrier function, which is mediated by proteins such as zonula occludens-1, and pump function, which is mediated by an active (Na+/K+-ATPase) pump. 8 The endothelial cell layer comprises an incomplete zonula occludens that allows molecules to enter the corneal stroma from the anterior chamber. The active Na+/K+-ATPase pump osmotically draws water and ions from the corneal stroma into the aqueous humor, which helps to maintain corneal thickness and transparency.9,10
Corneal endothelial cells get arrested in the G1 phase of the cell cycle and do not typically proliferate and regenerate in vivo.11,12 Therefore, loss of corneal endothelial cells results in compensatory enlargement and migration of the residual cells. It is possible that endothelial stem cells are located in the corneal periphery and retain a high regenerative capacity under laboratory conditions. 13
Clinical presentations
Corneal endothelial decompensation leads to ‘overhydration’ of the cornea, known as corneal edema, or in the advanced stage, bullous keratopathy. The patient may be asymptomatic in the early stage. As the corneal edema progresses, there may be glare or blurred vision caused by folds in Descemet’s membrane and increased stromal thickness. Eventually, a bulla forms, which leads to reduced visual acuity, and discomfort, and even severe pain. Long-standing corneal edema also predisposes to complications including corneal vascularization, infection, and scarring (Figure 2). 14

Long-standing corneal edema. Severe corneal opacity and scarring are evident and prevent the visualization of the details of the iris.
Etiologies
Etiologies that can cause corneal decompensation include Fuchs’ endothelial corneal dystrophy (FECD), posterior polymorphous corneal dystrophy (PPCD), aphakic or pseudophakic bullous keratopathy (ABK/PBK), endothelial dysfunction caused by penetrating or blunt trauma, congenital hereditary endothelial dystrophy (CHED), iridocorneal endothelial (ICE) syndrome, refractory glaucoma, previous failed corneal grafts, and herpes simplex virus endotheliitis. The most common primary etiology of corneal endothelial dysfunction is FECD.15–17 The most common secondary etiology of corneal edema is PBK, reflecting the popularity of cataract surgery and intraocular lens implantation in the past two to three decades. 18 Glaucoma and its treatment (medical and surgical) have deleterious effects on the corneal endothelium and can reduce the survival of corneal grafts.19,20
Fuchs’ endothelial corneal dystrophy
FECD is a dystrophy affecting the corneal endothelium. FECD has a regional prevalence that varies from 3.8% to 11% in individuals older than 40 years and is the primary indication for keratoplasty in the United States.15–17 This dystrophy is characterized by a progressive decrease in endothelial cell count, alterations in the shape and size of the residual cells, and formation of guttae (Figure 3). As the disease progresses, the endothelial cell count decreases until the residual cells are no longer capable of maintaining corneal deturgescence, resulting in corneal clouding and decreased vision. 21

Fuchs’ endothelial corneal dystrophy. (a) In the slit of light seen passing through the cornea from left (anterior surface) to right (posterior surface), the beaten-metal appearance of guttae is appreciated posteriorly in light reflected from Descemet’s membrane. (b) The anterior segment photograph of cornea with specular reflection illustrates the typical beaten-metal appearance. The dark spots in the photograph demonstrate the areas in which the endothelial cells have been lost.
FECD usually advances through four stages that span two to three decades.22,23 The patient is asymptomatic in stage 1 although slit-lamp biomicroscopy reveals nonconfluent guttae. In stage 2, the guttae coalesce, with an increase in polymegathism and pleomorphism along with loss of endothelial cells. In stage 3, the function of the endothelial pump is compromised and corneal edema is evident. In stage 4, long-standing edema results in corneal haziness and scarring that reduces visual acuity. Another grading scale devised by Krachmer and colleagues 24 scores disease severity based mainly on the number and distribution of guttae. A score of 1, which reflects asymptomatic disease, is defined as >12 central nonconfluent cornea guttae. A cluster (1–2 mm) of confluent central guttae is graded as 2. Grade 3 is defined as 2–5 mm of confluent central corneal guttae, and grade 4 is defined as >5 mm of confluent central guttae. Stromal or epithelial edema with >5 mm of confluent central guttae is graded as 5. 24
This dystrophy is caused by a complex combination of environmental and genetic factors. FECD can be categorized as early-onset or late-onset. Early-onset FECD, which is well defined both genetically and clinically, is a rare and almost always familial disease with autosomal-dominant inheritance.
25
The late-onset form, which accounts for the majority of patients, seems to have an autosomal-dominant transmission pattern with incomplete penetrance. This form of the disease usually presents in the fifth decade of life and progresses over the subsequent two to three decades. Late-onset FECD is more genetically heterogeneous than the early-onset form, and only half of these patients show family clustering.26,27 The early-onset form of FECD has been linked to mutations in the
Pseudophakic bullous keratopathy
Although the introduction of new phacoemulsification techniques, optical viscoelastic materials, and intraocular lenses have decreased the risk of corneal edema following cataract surgery, PBK is still one of the most common causes of corneal edema. 18 Several mechanisms can cause damage to the endothelium during cataract surgery. Some patients have a known endothelial disease prior to undergoing cataract surgery, which increases the risk of developing persistent corneal edema immediately after surgery. 31 The type of surgery also influences postoperative corneal decompensation risk; this risk is lower for phacoemulsification than for other techniques used in cataract surgery, particularly extracapsular cataract extraction. 31 The incidence of PBK with the current technique used for cataract surgery and implantation of an intraocular lens in the posterior chamber ranges from 1% to 2%. 31 Certain intraocular lens designs, particularly angle-supported anterior chamber lenses, increase the risk of bullous keratopathy (Figure 4). The incidence of corneal decompensation caused by angle-supported anterior chamber lenses may be up to 10%. 32 Cell loss associated with this type of lens is probably caused by contact between the lens and the endothelial cells located at the corneal periphery as well as chronic inflammation.

Pseudophakic bullous keratopathy. Severe corneal edema in an eye implanted with an angle-supported anterior chamber intraocular lens.
Congenital hereditary endothelial dystrophy
CHED is a rare dystrophy of the corneal endothelial layer that causes corneal edema at an early age and consists of two types. 33 CHED1 is transmitted in an autosomal-dominant manner and starts within the first few years of life, presenting with progressive stromal opacity. CHED1 prevalence is <1/1,000,000. CHED2 is an autosomal-recessive disease and presents with stromal opacity at birth or shortly thereafter. Epidemiologic data regarding its incidence or prevalence are unavailable. It has been suggested that CHED1 is a type of PPCD with an early onset of corneal decompensation. 34 Gene analyses, including DNA extraction from peripheral blood samples and polymerase chain reaction for screening mutations, demonstrate that the majority of patients with CHED2 have mutations in a transmembrane protein in the family of bicarbonate transporters (SLC4A11).35,36
The hallmark of CHED2 is corneal opacification and edema that presents at birth or shortly thereafter (Figure 5). Varying degrees of amblyopia and nystagmus are usually present in patients with more severe forms of the disease. Inflammation, epiphora, and photophobia are not noticeable characteristics. In contrast, CHED1 presents with progressive stromal edema and opacification that starts in the first few years of life. Epiphora and photophobia are more common in CHED1. Both types of CHED include thickening of Descemet’s membrane. However, guttae are not evident. The normal morphology of the endothelial cells is changed or absent. When endothelium can be detected by confocal or specular microscopy, the endothelial cells are decreased in number and are fibrotic.

(a) Clinical photograph of a girl with congenital hereditary endothelial dystrophy type 2 demonstrating bluish-gray ground-glass appearance of the right cornea. The left eye that underwent PK demonstrates a failing graft. (b) The slit beam highlights the uniform thickening of the cornea in the right eye.
Posterior polymorphous corneal dystrophy
PPCD is a rare, bilateral, autosomal-dominant disease characterized by a number of corneal abnormalities, ranging from asymptomatic endothelial irregularities to significant corneal edema and glaucoma.37,38 The prevalence of this form of corneal dystrophy is unknown. However, it has been reported that at least 1 in 100,000 inhabitants of the Czech Republic are affected by this dystrophy. 39 Specular microscopy may show typical geographic-shaped, discrete, gray lesions as well as isolated grouped vesicles and broad bands with scalloped borders. Pupil abnormalities and alterations in the iris are also observed. The condition manifests variably, even in members of the same family. PPCD typically manifests within the first decade of life and is often asymptomatic. At any time later in life, depending on disease progresses, patients may develop varying degrees of photophobia, decreased vision, and sectorial corneal clouding, necessitating corneal transplantation in nearly 25% of cases.38,40,41 Although an abnormally thickened Descemet’s membrane and stromal edema may cause pseudoelevation of intraocular pressure, true glaucoma from angle closure may occur at any stage of life in 14% of affected patients.37,38
Endothelial cells from corneas with PPCD demonstrate epithelial-like features, including multicellular stratification and expression of epithelial cell markers.42,43 Descemet’s membrane shows variable abnormal thickening in this corneal dystrophy. Stromal and epithelial corneal edema may occur diffusely or sectorally because the dystrophic endothelial cells become inefficient at pumping fluid out of the corneal stroma.37,38
Several genetic mutations have been implicated in PPCD. PPCD1 is thought to result from a heterozygous mutation in the promoter of the
ICE syndrome
ICE syndrome is a rare corneal disease characterized by structural and proliferative abnormalities of endothelial cells, progressive iridocorneal adhesion, and iris anomalies, including atrophy and hole formation. 45 Common clinical findings are stromal edema, iris atrophy, secondary glaucoma, and pupillary anomalies that vary from distortion to polycoria (Figure 6). Corneal edema and secondary glaucoma are the most common causes of reduced vision in individuals with ICE syndrome. 46 The major subtypes of this disease include Chandler syndrome, Cogan-Reese syndrome, and progressive iris atrophy. 47 The syndrome that typically affects adults (more often women in the third to fifth decades of life) is sporadic in presentation and usually unilateral. Eventually, ICE syndrome severely compromises visual function if not correctly treated. 45 Even when patients with ICE syndrome are treated promptly, surgical interventions have variable success rates.
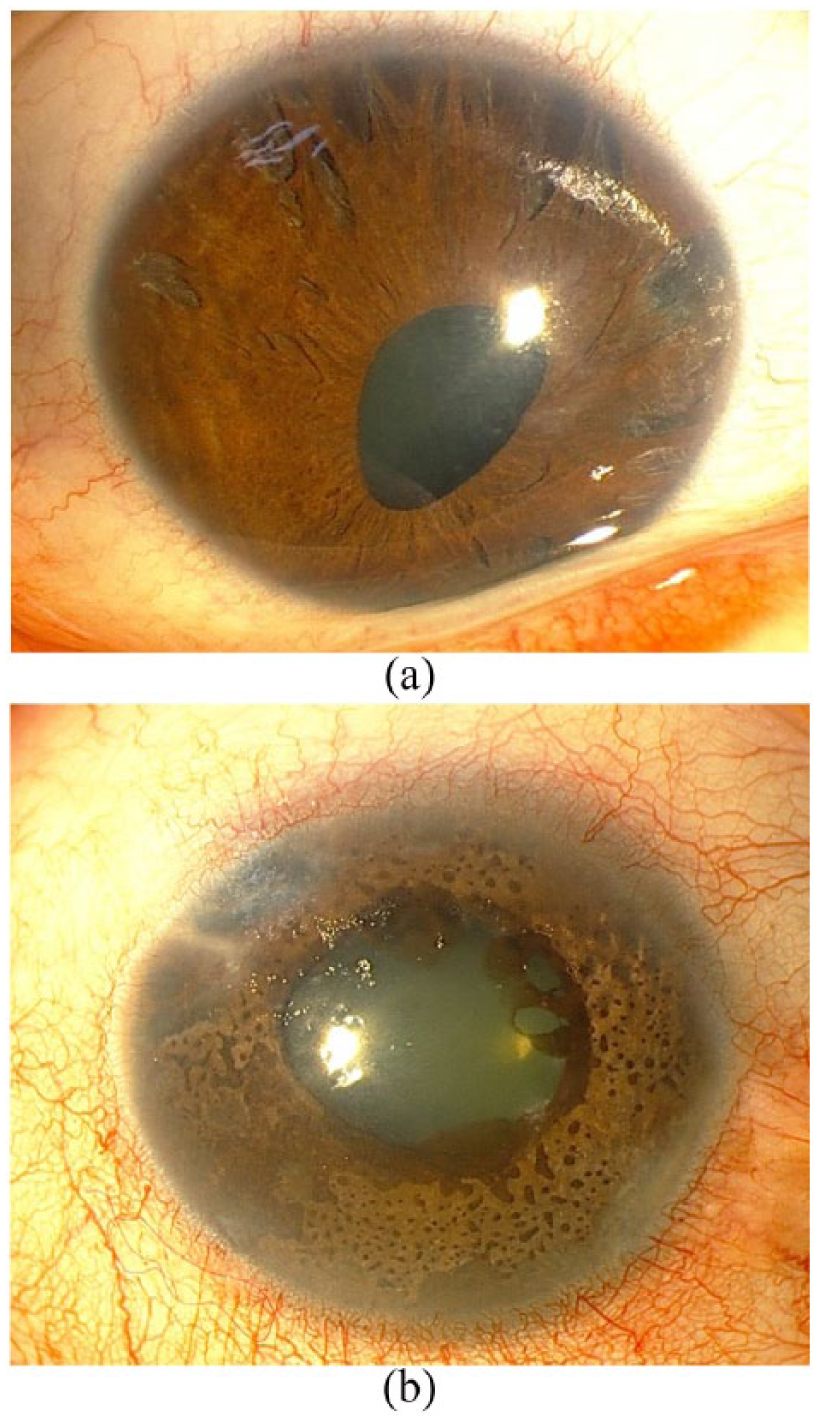
(a) Iridocorneal endothelial syndrome, characterized by atrophy of the iris, multiple atrophic holes, and corectopia. (b) Cogan-Reese syndrome, characterized by iridocorneal adhesion, diffuse nevi, and ectropion uveae.
High-magnification slit-lamp biomicroscopy can show a fine, ‘hammered-silver’, or ‘beaten-bronze’ appearance of the endothelium. Changes in the endothelium in ICE syndrome may be visualized and further evaluated by specular microscopy and in vivo corneal confocal microscopy. 48
The etiology of ICE syndrome is still largely unknown. However, inflammation 49 and viral infections (e.g. Epstein-Barr virus and herpes simplex virus)50,51 have been suggested as the etiologies of the disease. The corneal endothelium is primarily affected in the ICE syndrome and shows proliferative and structural abnormalities and an ability to migrate into the surrounding tissues. Specular microscopy shows morphologic changes in the size and shape of endothelial cells, which resemble epithelial cells even at the earliest stages.52–54 Corneal edema is caused by altered endothelial cell function and abnormalities in the endothelial cell barrier. The abnormal endothelial cells in ICE syndrome migrate posteriorly beyond the Schwalbe line to obstruct the iridocorneal angle and into the anterior chamber to cover the iris, where they form an abnormal basement membrane that eventually contracts, triggering an abnormal pupil shape, atrophic damage to the iris, and formation of synechiae between adjacent structures (Figure 6). 55 The angle obstruction causes an increase in intraocular pressure and consequent development of glaucoma in 46–82% of patients with ICE syndrome. 46
Therapeutic approaches other than corneal transplantation
Keratoplasty is the standard treatment for patients with corneal decompensation because it provides visual recovery and symptomatic relief. However, in eyes with poor visual potential or when donor tissue is not accessible for keratoplasty, other approaches should be considered to reduce pain and discomfort. The options available in these cases are hypertonic saline eye drops, bandage contact lenses, phototherapeutic keratectomy (PTK), anterior stromal puncture, amniotic membrane transplantation (AMT), and conjunctival flaps. Depending on the severity of corneal edema, a combination of the above treatments can be used. New potential approaches consist of collagen cross-linking (CXL) and topical Rho-associated kinase inhibitors. These treatments aim to reduce the discomfort and pain caused by corneal edema and, if possible, improve vision. However, the efficacy and safety of these treatment options have not been evaluated in clinical trials.
Osmotic solutions
Topical 5% hypertonic sodium chloride (eye drops and ointment) is useful in the early stage of corneal decompensation to reduce corneal thickness and improve visual acuity but is not effective in the advanced stage of the disease. 56 Moreover, some patients cannot tolerate the eye drops. A combination of a bandage contact lens and frequent hypertonic saline eye drops may be a better choice for improving the patient’s symptoms as well as vision. 57 Other osmotic agents, such as colloidal dextran polysaccharide solution, are not effective in the management of corneal decompensation. 58
Bandage contact lenses
Bandage contact lenses have long been used in patients with bullous keratopathy.59,60 Contact lenses can be used in combination with hypertonic preservative-free saline to reduce patient discomfort and improve visual acuity more effectively. 57 Two types of silicone hydrogel bandage contact lenses have been compared with conventional (Sauflon 85%) lenses for their ability to alleviate pain and discomfort in patients with corneal decompensation. 61 After 1 month of fitting, the silicone hydrogel lenses were significantly better than the Sauflon 85% lenses with respect to patient comfort. 61 However, the three lenses were comparable in terms of pain relief. There was no significant difference in buildup of deposits, movement of the lens, or fit between the groups. 61
Anterior stromal puncture
Anterior stromal puncture is a rapid repeatable procedure that can significantly reduce ocular discomfort in patients with bullous keratopathy.62,63 However, it is effective in patients with localized stromal edema and can cause corneal vascularization and scarring. 63 Furthermore, the density and depth of all the punctures cannot be quantified. Although 25G and 26G needles have been used for anterior stromal puncture,62,63 some surgeons prefer to use a large-bore 20G needle for this purpose. 64 If corneal transplantation is to be performed in the future, the periphery of the cornea should be spared to prevent corneal vascularization. 64
This procedure leads to an increase in the expression of extracellular proteins, including fibronectin, laminin, and type IV collagen, in the corneal stroma. 65 These substances are essential for the adhesion of the epithelium to the underlying stroma. The efficacy and safety of this procedure have not been evaluated in patients with corneal decompensation. Moreover, the impact of this procedure on subsequent donor corneal graft has not been evaluated.
Phototherapeutic keratectomy
PTK is effective in reducing discomfort and pain in individuals with a decompensated cornea.66–68 The procedure can be repeated if indicated. It is assumed that PTK can remove the abnormal basement membrane, leading to better healing of the corneal epithelium. 69 Furthermore, corneal thinning after this procedure can decrease epithelial edema by reducing the osmotic load of the corneal stroma, thereby enhancing the dehydration efficacy of the residual endothelium. 70 Deep PTK (to a stromal thickness of 25%) was reported to be more effective than superficial PTK (8–25 µm) or intermediate PTK (50–100 µm) for pain reduction. 70 This effect is attributable to greater destruction of the corneal neural plexus or increased scar formation caused by a deeper ablation. 70 This procedure is an appropriate option for the management of patients with symptomatic corneal edema, especially as a temporary intervention for those awaiting keratoplasty. The main limitation of this treatment is its cost.
Gundersen conjunctival flap
A Gundersen conjunctival flap covers the entire cornea. In this procedure, the bulbar conjunctiva is dissected and mobilized from the underlying Tenon’s capsule (Figure 7). 71 This flap can significantly alleviate pain in patients with bullous keratopathy by covering the exposed corneal nerve endings with an intact surface. The procedure can be combined with AMT to alleviate pain in patients with symptomatic corneal edema. 72

A Gundersen conjunctival flap. The cornea is completely covered by an intact layer of bulbar conjunctiva.
Postoperative complications include a shortened fornix, retraction of the flap, and potential loss of healthy conjunctiva for subsequent trabeculectomy. The flap retraction rate varies from 10% to 15%. This complication results from excessive traction on the flap, buttonholes, and conjunctival melts because of epithelial ingrowth. 72 The conjunctival flap can be removed for a subsequent corneal transplant. However, the vascularized bed that remains after flap removal increases the risk of subsequent graft rejection. There is also a risk of limbal stem cell deficiency necessitating limbal autografts. 72 Therefore, a conjunctival flap is more appropriate for eyes with poor visual potential.
Amniotic membrane transplantation
AMT is an option for individuals awaiting keratoplasty and can reduce pain in patients with bullous keratopathy.73–77 AMT is helpful in patients with persistent epithelial defects associated with long-standing corneal edema (Figure 8). The amniotic membrane allows the epithelial cells to grow and serves as a bandage for an abnormal cornea. 78 In this situation, the amniotic membrane melts within weeks. The limitation of AMT is that the amniotic membrane may remain in situ and reduce visualization of the anterior segment and retina because of its opacity. Studies have found that AMT is comparable with anterior stromal puncture 79 and PTK 80 in terms of reducing patient discomfort. AMT can be combined with other options, including PTK, to accelerate corneal epithelial healing, 81 the Gundersen conjunctival flap, 72 and anterior stromal puncture.82,83 It is not known whether AMT exacerbates corneal vascularization or whether it reduces the longevity of a future corneal transplant.

Amniotic membrane transplantation for management of pain and discomfort in a patient with bullous keratopathy.
Collagen cross-linking
CXL can improve visual acuity, minimize ocular discomfort, and delay the need for corneal transplantation in patients with corneal edema. 84 CXL makes the collagen fibers in the anterior corneal stroma more compact and organized, but this effect is diminished in severe corneal edema. 85 After CXL in a decompensated cornea, the transendothelial inflow and stromal imbibition pressure decrease, leading to a decrease in corneal edema. 86 Previous studies have demonstrated a significant increase in visual acuity and symptomatic improvement in individuals with PBK immediately following CXL.87–90 However, this effect was diminished by 3–6 months postoperatively.87–90 These results indicate that the effect of this treatment decreases with time and is dependent on the severity of edema. 88 Other studies have not achieved such good results and found CXL to be ineffective in the management of eyes with corneal edema.91,92
Rho-associated kinase inhibition
The Rho/Rho-kinase (ROCK) pathway regulates cell migration and proliferation as well as apoptosis.93–96 Y-27632, a selective ROCK inhibitor, can promote adhesion and proliferation of corneal endothelial cells by diminishing dissociation-induced apoptosis.97–99 This agent can be used in vivo as eye drops or ex vivo to expand human corneal endothelial cells in culture medium. In a clinical study, transcorneal freezing was performed using a stainless steel rod with a diameter of 2 mm in eight eyes in eight patients with corneal decompensation, caused by late-onset FECD, argon laser iridotomy-induced bullous keratopathy, or keratopathy in pseudoexfoliation syndrome. 100 Y-27632 eye drops were then applied six times daily for 1 week. Three out of four eyes with central corneal edema caused by FECD demonstrated a significant decrease in central pachymetry, which was maintained over time. 100 The remaining four eyes with diffuse corneal edema had no improvement in visual acuity or corneal pachymetry. 100 Human corneal endothelial cells did not demonstrate any cell alterations or toxicity after treatment with a ROCK inhibitor. 100 Therefore, topical ROCK inhibition can be used as an alternative to corneal transplantation in patients with early corneal decompensation.
Recently, cultured human corneal endothelial cells supplemented with a ROCK inhibitor were injected into the anterior chamber in 11 eyes with PBK. After 24 weeks of injection, all corneas were clear and nine eyes achieved an improvement in best-corrected visual acuity of more than two lines. 101 Use of Y-27632 may only be appropriate for cultures of human corneal endothelial cells harvested from younger donors, given that addition of this agent was not effective in cultures established using older donors.102,103 The combination of hyaluronic acid and Y-27632 can improve the efficiency of cell adhesion as a result of force attachment, enabling culture of endothelial cells from older donor corneas. 104
Corneal transplantation
PK has been the standard keratoplasty technique used to replace poor endothelium since corneal grafting became a routine operation in the 1950s. However, the undesirable complications of full-thickness keratoplasty are now well recognized and include prolonged visual rehabilitation, high postoperative astigmatism, and vulnerability to trauma. 105 Novel posterior lamellar keratoplasty techniques have recently been developed. These techniques include Descemet stripping automated endothelial keratoplasty (DSAEK) and Descemet’s membrane endothelial keratoplasty (DMEK). These methods of endothelial keratoplasty share the advantage of the lack of a large full-thickness wound created during PK, which results in a stable and less vulnerable eye with no corneal sutures, less induction of astigmatism, and more rapid visual rehabilitation. Currently, endothelial keratoplasty can be used to treat any cause of corneal endothelial dysfunction, including FECD, CHED, PPCD, ICE syndrome, ABK/PBK, and failed PK. PK may still be indicated in patients with severe end-stage corneal edema and deep stromal scarring.
Descemet stripping automated endothelial keratoplasty
DSAEK is currently the most commonly used endothelial keratoplasty technique (Figure 9). 106 DSAEK provides rapid and predictable visual rehabilitation, with better uncorrected and corrected distance visual acuity (CDVA) than PK. A mean CDVA of ⩾20/40 is usually obtained within 3–6 months of DSAEK. 107 The mean CDVA has been reported to vary between 20/33 and 20/66 in different reports, with postoperative examination between 3 and 30 months after DSAEK.108–117 In comparison, the proportions of individuals obtaining a CDVA ⩾20/40 have varied from 47% to 65% for FECD and from 20% to 40% for ABK/PBK in several large PK series that had a follow-up duration between 2 and 8 years.105,118,119 PK can lead to more eyes with a visual acuity correctable to 20/20. However, this level of CDVA is usually achieved with the use of a rigid gas-permeable contact lens.105,118,119

Descemet stripping automated endothelial keratoplasty was performed in an eye with pseudophakic bullous keratopathy. The graft and overlying recipient cornea are crystal clear.
A chief advantage of DSAEK is that it causes minimal changes in spherical equivalent and astigmatism. 120 DSAEK usually induces hyperopia between 0.7 and 1.5 D, with a median of 1.2 D.106,115,116 This hyperopic shift is likely caused by the variation in thickness of the lenticule, which results in a reduction in total corneal power.121,122 The mean surgically induced astigmatism after DSAEK is minimal at a median of +0.1 D.106,113,115,116,121–123
Endothelial cell loss varies from 13% to 54% in the 6 months after DSAEK109,116,124–128 and from 15.6% to 61% in the first year after the procedure.108,124,129–131 Endothelial cell loss can be as high as 89% at 5 years. 132 In comparison, the mean reduction in endothelial cell density following PK varies from 11% to 29% in the first 6 months,133–135 from 16% to 45% at 1 year,133–136 from 29% to 54% at 2 years,133,136,137 and is 70% at 5 years. 138 The majority of the endothelial cell loss in DSAEK takes place during the first 6 months as a result of surgical trauma. 132 Other factors can also influence postoperative endothelial cell density after DSAEK. The Cornea Preservation Time Study Group investigated the effect of duration of donor preservation in cold storage medium on endothelial cell loss 3 years after successful DSAEK and found that cell loss 3 years after DSAEK was greater with longer preservation time. 139 However, cell loss was not affected by preservation for up to 13 days. 139
Early DSAEK studies indicate that the graft survival rate is comparable with that of PK and may be even higher. 140 Graft survival rates between 55% and 100% one year after DSAEK have been reported.106,108,110,113,123,130,131,141,142 This wide range of clear grafts reflects the results reported by surgeons who were in their learning curve. Excluding those reports, the range of graft survival at 1 year is 94–100%. 132 This range of rates is comparable with that reported for PK at 1 year (89–95%).105,143–146 The graft survival rate after DSAEK is lower in eyes with ABK/PBK than in those with FECD and in eyes in which operative complications such as inverted graft occurred. 147 Furthermore, the risk of graft failure may be higher in recipient eyes that receive corneas from donors with diabetes. 147 Diabetes has adverse biochemical, morphologic, and functional effects on the corneal endothelium, resulting in a decrease in the graft survival rate after DSAEK. 147 Therefore, the increasing frequency of diabetes in the aging population may affect the donor pool. 148 It has also been noted that diabetes may make preparation of the tissue for DMEK more difficult. 149
The most common reason for regrafting after DSAEK is unsatisfactory vision (2.7%). 150 In contrast, the most common reasons for PK graft failure are ocular surface disease, glaucoma, and graft rejection. 145 The reasons for low CDVA after DSAEK include donor folds with visual axis involvement, nonuniform donor graft thickness, and subepithelial, stromal, or interface opacities.151,152
Donor dislocation, that is, lack of attachment between the recipient stroma and donor lenticule, is the most frequently reported complication after DSAEK and occurs at a rate that ranges from as low as 0% to as high as 82%.108–112,121,141 A repeat air injection is generally required to manage this complication. Endothelial cell loss may be greater in dislocated grafts than in grafts that remain attached.130,131 Rebubbling is usually performed for complete detachments because partial detachment of the donor lenticule may resolve spontaneously. 153
Graft rejection rates after DSAEK vary from 0% to 45.5% during follow-up of 3–24 months.108–112,115,116,154 The estimated risk of a first rejection episode after DSAEK is 7.6% by 1 year and 12% by 2 years. 155 The graft rejection rate in endothelial keratoplasty is significantly lower than that in PK, which is attributable to the prolonged use of corticosteroid eye drops following endothelial keratoplasty. 154 Corticosteroids are frequently tapered off within the months following PK to allow wound healing before suture removal, whereas this is not an issue with DSAEK.
Donor lenticules for DSAEK can be prepared by surgeons intraoperatively or predissected by eye bank operators. Donor grafts prepared by surgeons can sometimes result in failure because of perforation or irregular cuts. Precut tissues prepared by an eye bank have the advantages of higher operating room efficiency and less wastage of tissue. 156 In addition, tissue for endothelial keratoplasty can be preloaded by an eye bank. Ruzza and colleagues 156 described a method of preserving and delivering posterior lenticules for DSAEK in which the donor tissue was precut, punched, loaded into a three-dimensional printed smart storage glide, and then preserved in transport medium. After 7 days of preservation, they demonstrated an average endothelial cell loss of 2.3% and an increase in lenticule thickness by 30%, with no apoptosis of endothelial cells. 156
Descemet’s membrane endothelial keratoplasty
DMEK is a further refinement of posterior lamellar keratoplasty in which only donor Descemet’s membrane and endothelial cells are transplanted. This procedure essentially substitutes for the same tissue that is removed from the recipient’s cornea and thus exactly replicates the corneal anatomy. 157 Donor Descemet’s membrane can be stripped manually from the stroma.157–159 However, the Descemet’s membrane along with endothelium can be pneumatically dissected from the posterior surface of the donor cornea. 160 Other methods of DMEK tissue preparation are slight modifications of the conventional stripping and bubble techniques. For example, liquid instead of air can be used to separate the Descemet’s membrane–endothelium complex from the stroma. 149 Studeny and colleagues 161 introduced a hybrid variation of DMEK that retains an outer rim of donor stroma with a centrally bared Descemet’s membrane. Parekh and colleagues 162 compared three stripping techniques and two bubble methods in terms of cost, preparation time, endothelial cell density, and endothelial cell death and morphology. They found a significantly higher cell death rate with pneumatic dissection and submerged hydroseparation than with stripping, which was attributed to the mechanical stress induced by pressure during bubble formation. 162 However, the preparation time and associated costs were the drawbacks of the stripping methods. 162
DMEK provides the most rapid visual rehabilitation of all the endothelial keratoplasty techniques. Compared with DSAEK, more patients obtain a CDVA ⩾20/30 after DMEK because of elimination of stroma-related optical issues. The early studies on DMEK reported that CDVA was 20/20 in 26% of patients, ⩾20/25 in 63%, and 20/40 or better in 94% at 3 months. The rates of CDVA ⩾20/25 by 3 months after DMEK surpassed the rates reported with DSAEK at 6 months and beyond.106,116,152 At 1 year, 39% of the eyes could be corrected to a CDVA of 20/20 or better, 79% recovered to 20/25 or better, and 97% had 20/30 or better. 163 Despite the refractive cylinder remaining unchanged, a small but statistically significant amount of hyperopic shift (⩽0.50 D) was noted following single DMEK procedures.159,163,164
Graft detachment necessitating reinjection of air may be encountered in 20% of cases after DMEK. 165 However, with experience, the rate of this complication tends to decrease. 166 A recent study demonstrated that cataract removal at the time of DMEK and air fill to ⩽75% of the anterior chamber height at 2–3 h postoperatively were independently associated with an increased risk of postoperative graft detachment. 167 Indications for intracameral air or gas injection include partial (>33% of the graft surface), central, scrolled, or complete DMEK–lenticule detachment. 166 It is believed that early rebubbling of the graft is necessary to obtain an early recovery rate and a visual acuity in the order of 20/20 and to prevent shrinkage and fibrosis of the donor Descemet’s membrane. 166
Endothelial cell loss, caused by intraoperative trauma to the donor tissue, is one of the most common complications reported after DMEK. The average endothelial cell loss is 32%158,159 at 6 months, 36% at 1 year,163,168 and 42% at 3 years 169 following DMEK. Excessive intraoperative manipulations caused primary graft failure in 9% of operated eyes.159,165 It has been reported that the risk of immunologic rejection may be lower after DMEK than after DSAEK or PK by 15-fold and 20-fold, respectively. 170
As surgeons transition to DMEK, eye banks have risen to the challenge of preparing tissue. Donor preparation by an eye bank can be a valuable option for surgeons because eye bank operators have experience preparing a significant volume of corneas on a daily basis, which reduces the graft preparation time, tissue wastage, and overall costs.171,172 Prestripped DMEK grafts can also be preloaded in an eye bank without additional endothelial cell loss, making the procedure more efficient.173,174 To prevent endothelial cell loss because of the graft scraping against the injector, DMEK tissue is manually tri-folded with the endothelial side inward before being inserted into the cartridge. 173 Prestripped and preloaded DMEK grafts can also be prestained with 0.06% Trypan blue with acceptable cell loss. 174
Cell-based approach for management of corneal endothelial dysfunction
At present, corneal transplantation is the only method that can cure corneal endothelial dysfunction. However, as a result of the worldwide global shortage of donor tissue, many affected individuals have no access to this treatment. Therefore, it is necessary to engineer corneal tissue that can be transplanted clinically. There are two problems associated with the development of such tissue engineering therapy, including
Culture medium
Different studies have used different types of human corneal endothelial cell culture medium, including human amniotic fluid, human bone marrow–derived mesenchymal stem cells, and conditioned medium from mouse embryonic stem cells.178,179 All types of conditioned medium have an animal origin or an animal-derived component, mainly serum. To avoid potential contamination by infectious agents such as viruses and bacteria, a xeno-free medium would be more suitable. However, the high cost of serum-free culture medium containing growth factors such as basic fibroblast growth factor limits their use for scalable expansion of human corneal endothelial cell cultures with clinical application.
Transplantation of cultured cells
The methods that can be used to transplant cultured corneal endothelial cells include transplantation of a cultured corneal endothelial sheet and injection of cultured corneal endothelial cells as a cell suspension. Several investigators have successfully transplanted a cultured corneal endothelial sheet in an animal model.180–183 However, transplantation of a flexible monolayer cell sheet to the anterior chamber of the eye is technically difficult. Furthermore, development of an artificial scaffold is a current problem for cell sheet transplantation. An alternative method is to regenerate corneal endothelium by cell injection to overcome the obstacles associated with cultured corneal endothelial sheet transplantation. The main drawback of this method is that the injected cells can be removed by the flow of aqueous humor, leading to poor adhesion of the cultured cells onto the posterior corneal surface. 184
Conclusion
Corneal endothelial dysfunction is one of the most common causes of corneal blindness. Although alternative approaches can be used to alleviate pain and discomfort, the most effective treatment is the replacement of the abnormal cornea with healthy donor tissue. With recent developments in lamellar keratoplasty techniques, endothelial keratoplasty, including DSAEK and DMEK, has become a popular corneal transplantation method in eyes with bullous keratopathy. However, the globally limited supply of human donor corneas is becoming an increasing challenge and necessitating a search for alternatives. Recent research has focused on addressing the challenges of culturing human corneal endothelial cells to allow transplantation of cultured cells to many recipients. However, standard culture methods and techniques of transplantation of cultured cells have not yet been well established for clinical purposes.
Footnotes
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of interest statement
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
