Abstract
Introduction
Incomplete evaluation of stroke patients may result in an unclear diagnosis. Our objective was to determine if older stroke patients more often undergo incomplete diagnostic evaluations versus younger patients in an international cohort.
Patients and methods
The Embolic Stroke of Undetermined Source Global Registry was a retrospective cohort of consecutive stroke patients evaluated at 19 stroke centers in 19 countries. Diagnostic evaluation was considered as complete if the patient had, at a minimum, brain computed tomography or magnetic resonance imaging with evidence of infarction, extracranial and intracranial vascular imaging, electrocardiography, ≥24 h of cardiac rhythm monitoring, and echocardiography. Patients were diagnosed with Embolic Stroke of Undetermined Source if brain imaging confirmed a nonlacunar infarction and no stroke etiology was determined after complete evaluation. Completeness of evaluation was compared between patients ≥75 versus <75 years old.
Results
The registry included 2132 patients with recent ischemic stroke during 2013–2014, of which 349 were diagnosed with Embolic Stroke of Undetermined Source. Embolic Stroke of Undetermined Source patients ≥75 years were less likely to undergo brain magnetic resonance imaging (74% versus 89%, p = 0.001), transesophageal echocardiography (22% versus 39%, p = 0.005), and combination transthoracic and transesophageal echocardiography (16% versus 32%, p = 0.005) compared with Embolic Stroke of Undetermined Source patients <75 years.
Discussion
Our study has identified an international age disparity in fundamental diagnostic testing for older patients with stroke of unknown etiology. Some testing biases were affected by geographic location (e.g., brain MRI was less frequently used in European ESUS patients), whereas other testing was implemented less frequently in the elderly regardless of location (e.g., transesophageal echocardiogram).
Conclusion
Older patients in this international cohort had less sophisticated diagnostic testing for stroke, despite advanced age being well established as an independent risk factor for recurrent stroke. This was a global problem and further investigations are warranted to explore the cause.
Introduction
Cryptogenic stroke is one of the major etiologic stroke subtypes, accounting for as many as one in three strokes. 1 It is a diagnosis of exclusion and requires thorough evaluation in order to exclude the more well-defined stroke subtypes. However, cryptogenic stroke also includes patients with incomplete diagnostic testing. Recently, embolic stroke of undetermined source (ESUS) was defined as an ischemic stroke, nonlacunar in radiographic appearance, without evidence of stenosis detected by carotid imaging or obvious cardioembolic source after electrocardiography (EKG), transthoracic echocardiography, and at least 24 h of cardiac rhythm monitoring have been completed. 2 This definition sets a minimum standard for diagnostic evaluation but leaves further testing to the discretion of the treating clinician and patient. Given how invasive, potentially risky, costly, and/or time consuming, several additionally available diagnostic tools (including transesophageal echocardiography, implantable cardiac event monitors, intracranial vascular imaging, blood tests, and others) may be, it stands to reason that some may choose against extensive evaluation.3–6 In contrast, since cryptogenic stroke has a substantial rate of recurrence 7 and a known etiology of stroke can be discovered in a considerable proportion of these patients,4,8,9 these tests may be valuable in the prevention of recurrent stroke. Elderly patients are at particularly high risk for recurrent stroke,10–12 which makes etiologic identification critical to their management. Several treatable risk factors, such as paroxysmal atrial fibrillation (PAF), are associated with older age and with more severe strokes.13,14 Consequently, patients and providers may believe that thorough diagnostic testing may not be worthwhile. However, the discovery of a treatable cause of stroke (such as PAF) and optimization of secondary prophylaxis (oral anticoagulation) could dramatically reduce the risk among these patients who would otherwise have been misclassified and treated as cryptogenic stroke. 15
In the present study, we aimed to identify if age was associated with incomplete evaluation for cryptogenic strokes using a multinational ESUS global stroke registry. 2 We hypothesized that elderly patients would be at greater risk for incompleteness in their stroke evaluation than younger patients, both in the overall stroke population and in the subset with ESUS.
Methods
Patient population
The ESUS Global Registry is a retrospective cohort of consecutive ischemic stroke patients evaluated at 19 stroke centers in 19 countries. 16 Briefly, a nine-page case report form was completed for each patient by a physician provided with specific instructions and definitions. Data were entered into a central database via online data entry or via an automated fax-based data entry system. Missing data were queried, and final data were exported to SAS software, version 9.2 (SAS Institute Inc., Cary, NC) for statistical analysis.
Consecutive stroke patients were identified from stroke logs at each hospital once the local institutional review board or ethical committee approved this investigation. All consecutive patients were included in the final cohort of 2132 patients. Patients older than 18 years of age were included if they experienced focal neurologic deficits lasting ≥24 h and had neuroimaging without evidence of primary intracranial hemorrhage.
Patients were classified as having experienced ESUS if the stroke was identified as nonlacunar on neuroimaging, and no stroke etiology was determined despite at least 24 h of in-hospital cardiac rhythm monitoring, transthoracic echocardiography, and intracranial and cervical arterial imaging. 2 A lacunar infarction was defined as a subcortical ischemic infarct ≤1.5 cm in its greatest dimension which occurred in the distribution of small, penetrating cerebral arteries. 2 If other testing was performed beyond the minimal requirements, patients would be classified as having ESUS only if no other specific stroke etiology was identified.
Completeness of evaluation was compared between patients ≥75 years old and those <75 years old. Patients were further divided into age groups according to decade at the time of diagnosis (e.g. <30, 30–39, etc.), and according to geographic location (Europe, North America, Latin America, East Asia, and Pacific), for similar comparisons. Analyses were performed in the overall registry excluding patients with a reported history of atrial fibrillation, or atrial fibrillation detected on EKG or in-hospital telemetry, and in the subpopulation of ESUS patients.
The study complied with local institutional research board regulations. Informed consent was waived by each center’s institutional review board or ethics committee as these data were anonymized and retrospectively abstracted.
Statistics
Categorical data were summarized as percentages for all patients without atrial fibrillation as well as for ESUS patients. Comparisons between the two primary age groups and the various demographic and clinical characteristics were assessed using chi-square tests or Fisher’s exact test as appropriate. All analyses were performed using SAS software, version 9.2 (SAS Institute Inc., Cary, NC). A two-sided p-value of < 0.05 was considered statistically significant. Comparisons across geographic regions were also made. We consider these analyses to be exploratory as they were performed to describe the data among groups, so adjustments for multiple comparisons were not made.
Results
All patients without atrial fibrillation
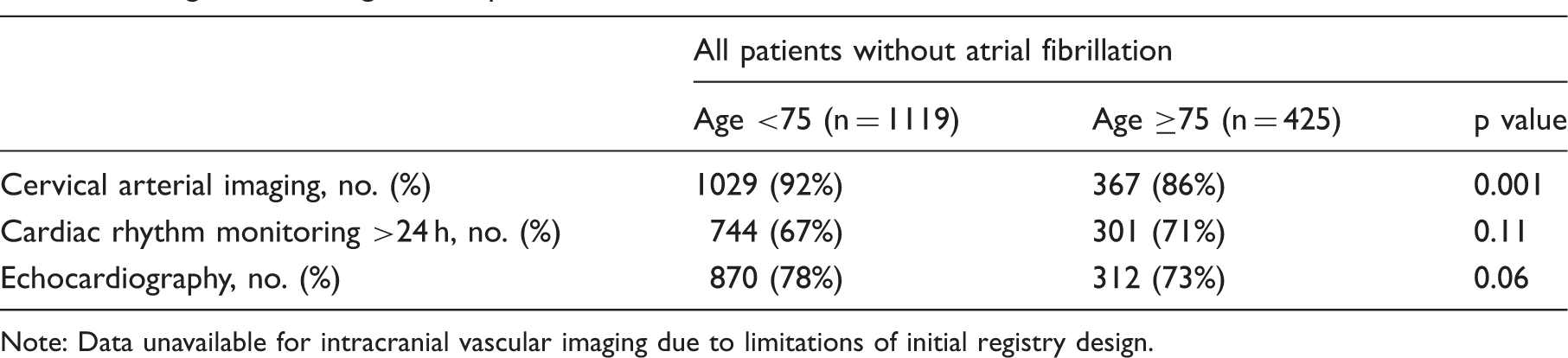
The ESUS global registry included 2132 consecutive patients with recent ischemic stroke during 2013–2014. After excluding 588 patients with either a history of atrial fibrillation, or atrial fibrillation on admission EKG or in-hospital telemetry, 1544 (72%) patients were analyzed according to age at time of presentation (Figure 1). Compared to patients younger than 75 years, a smaller proportion of patients ≥75 years underwent cervical arterial imaging (86% versus 92%, p = 0.001) and transthoracic echocardiography (73% versus 78%, p = 0.06), although the difference in echocardiography did not reach statistical significance (Table 1). Due to limitations of registry design, intracranial vascular imaging was only collected among patients with confirmed ESUS. Therefore, no comparisons were made among non-ESUS patients with regard to intracranial vascular imaging.
Patient flowchart. Diagnostic testing for “all patients without atrial fibrillation.” Note: Data unavailable for intracranial vascular imaging due to limitations of initial registry design.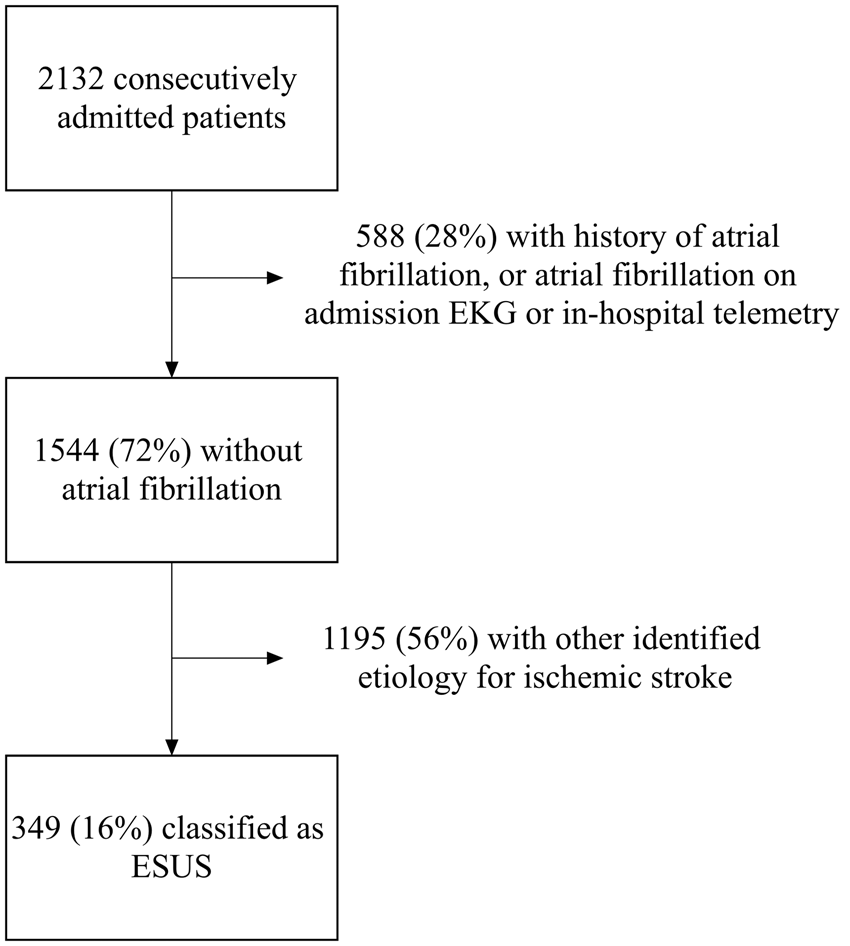
When evaluating diagnostic testing modalities among all patients without atrial fibrillation (n = 1544) according to age in decades, there were statistically significant differences in proportion of patients who underwent cervical arterial imaging (p = 0.03) and transthoracic echocardiography (p = 0.02), but not for cardiac rhythm monitoring (p = 0.24) (Figure 2). In these analyses, there was a downward trend in proportion of older patients after the age of 79 who received cervical arterial imaging and transthoracic echocardiography, whereas in-hospital cardiac rhythm monitoring was more routinely monitored regardless of age.
Relationship between age and diagnostic testing (a) Cervical arterial imaging (p = 0.034). (b) In-hospital cardiac rhythm monitoring >24 h (p = 0.240). (c) Echocardiography (p = 0.024) (X-axis represents age in years at time of admission, Y-axis represents percentage of patients who completed the evaluation).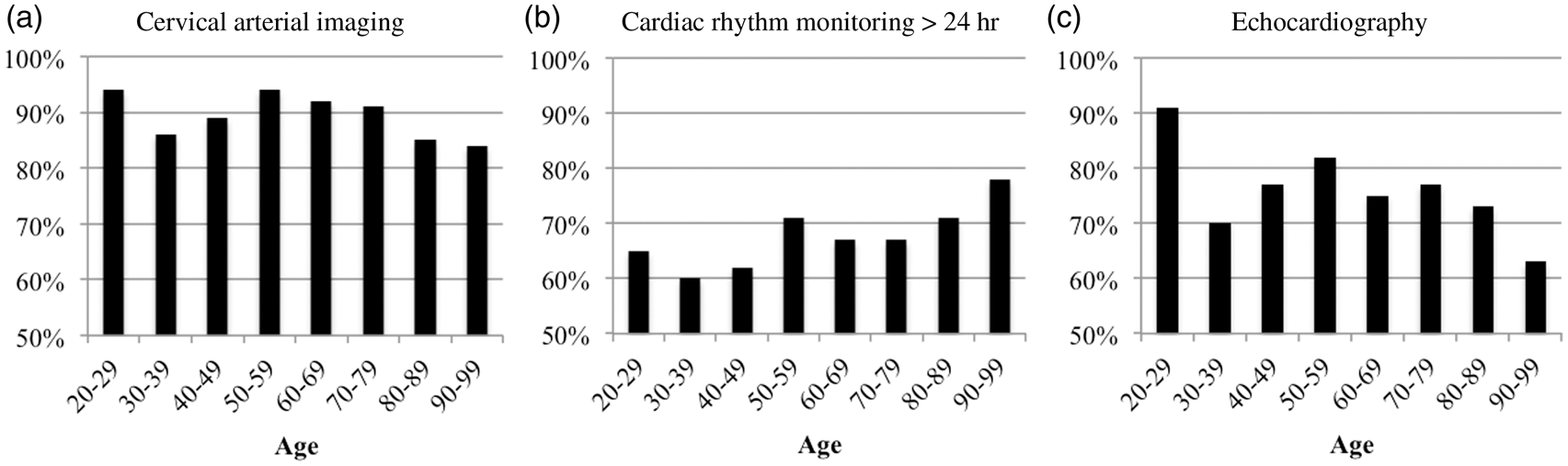
ESUS patients
Characteristics and diagnostic testing among patients with ESUS.
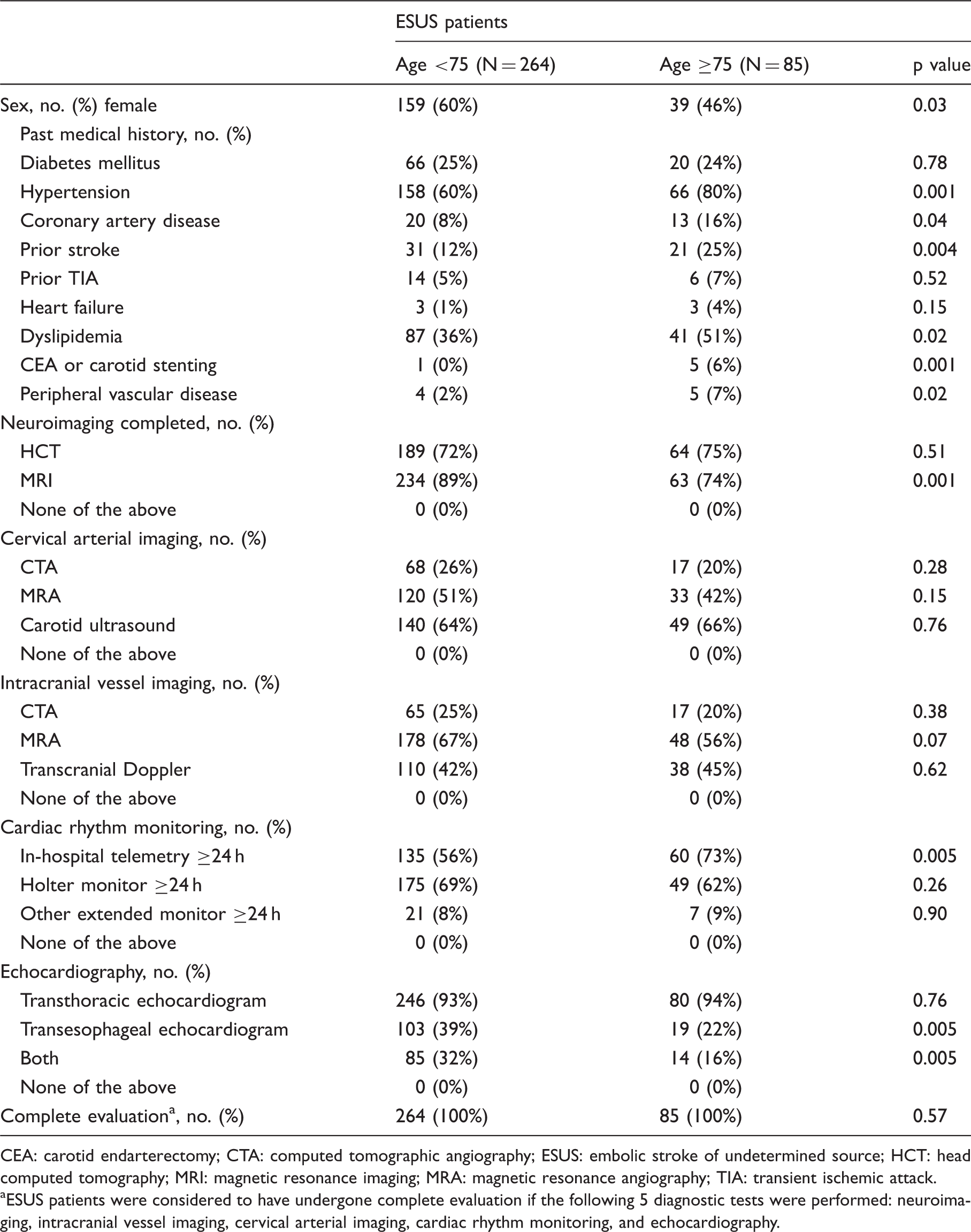
CEA: carotid endarterectomy; CTA: computed tomographic angiography; ESUS: embolic stroke of undetermined source; HCT: head computed tomography; MRI: magnetic resonance imaging; MRA: magnetic resonance angiography; TIA: transient ischemic attack.
ESUS patients were considered to have undergone complete evaluation if the following 5 diagnostic tests were performed: neuroimaging, intracranial vessel imaging, cervical arterial imaging, cardiac rhythm monitoring, and echocardiography.
Diagnostic testing among ESUS patients according to geographic location.
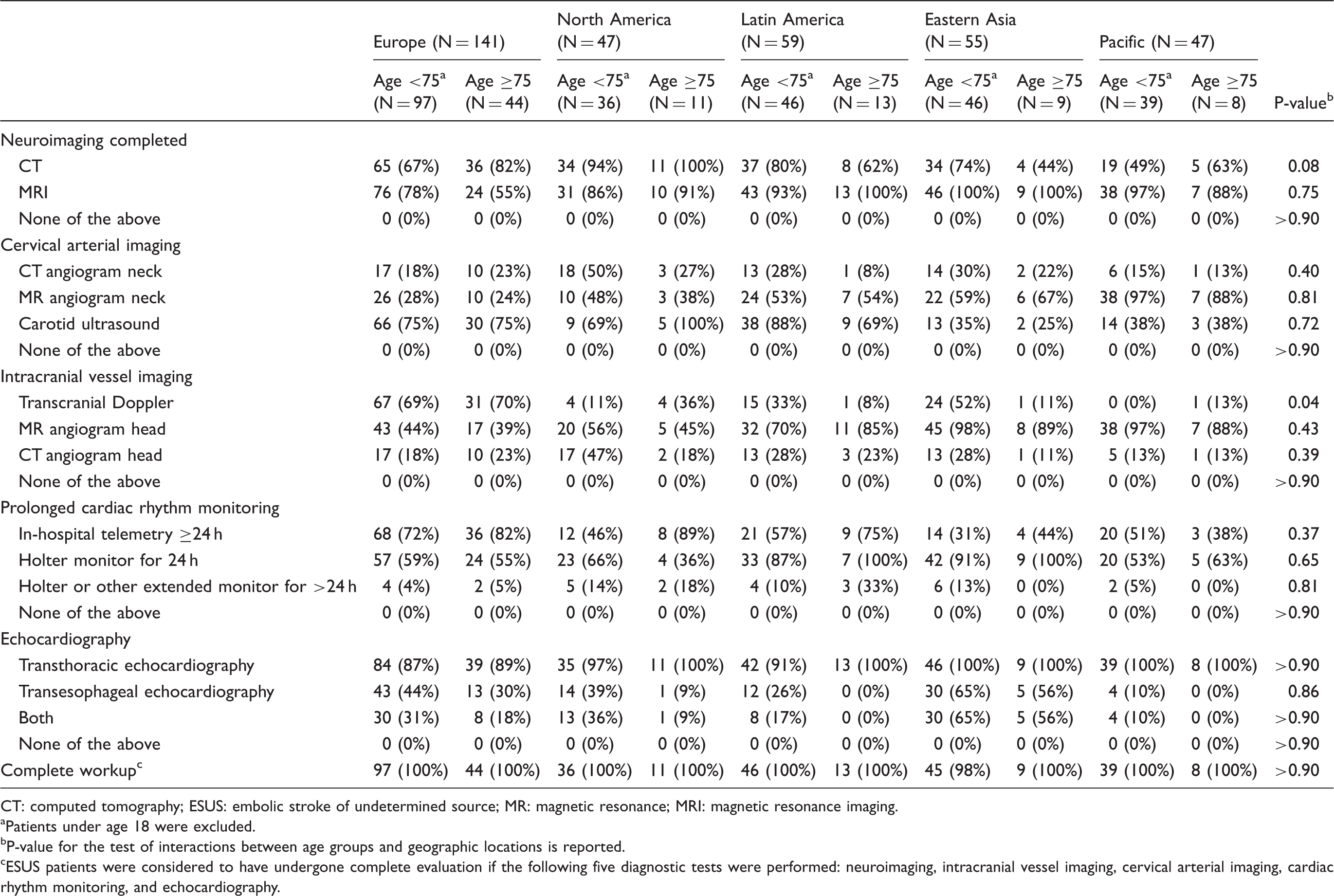
CT: computed tomography; ESUS: embolic stroke of undetermined source; MR: magnetic resonance; MRI: magnetic resonance imaging.
Patients under age 18 were excluded.
P-value for the test of interactions between age groups and geographic locations is reported.
ESUS patients were considered to have undergone complete evaluation if the following five diagnostic tests were performed: neuroimaging, intracranial vessel imaging, cervical arterial imaging, cardiac rhythm monitoring, and echocardiography.
Discussion
Our results from the ESUS Global Registry have identified an international age disparity in stroke evaluation. Similar reports concerning the age disparity in stroke management have been published17–22; however, our results are unique in several ways. We found that older patients with stroke are less likely to receive a complete initial battery of diagnostic testing. This may represent an important contributing factor for undertreatment of the elderly. Furthermore, while other international investigations identified an age disparity in general stroke evaluation,
19
our results are novel in that we observed a persistent bias against etiologic evaluation among older patients with
Our results also expand on single country cohorts17,21,23 in that our global population represents many cultures, health behaviors, socioeconomic settings, and various levels of access to diagnostic testing and early treatment. When specifically analyzed by geographic location, transesophageal echocardiogram was ordered less frequently in patients ≥75 regardless of the patient’s location. This may be attributed to risk of sedation or intraoperative complications that increase with age and accumulated comorbidities. Other disparities in diagnostic testing for ESUS appear weighted heavily by geography. For example, MRI of the brain was ordered less frequently among European ESUS patients ≥75 compared to those <75, while noncontrast head CT remained the neuroimaging modality of choice in Europe. This effect appears to have driven the statistical significance of this finding within the global registry. Although MRI is more sensitive for identifying stroke than head CT, especially in hyperacute stroke, 24 all patients in this registry had confirmed infarction via neuroimaging regardless of the imaging modality. That being said, several studies have demonstrated utility of diffusion-weighted MRI in the etiologic diagnosis of stroke subtype.25–27 Therefore, brain MRI should be considered in stroke patients where the mechanism remains unclear, particularly if a cardioembolic source is suspected.26,27 Regarding intra- and extracranial vessel imaging, CT angiogram of the head and neck was ordered less frequently in North American patients ≥75 compared to those <75 (18% versus 47% and 27% versus 50%, respectively). Carotid ultrasound and transcranial Doppler remained the vascular imaging modalities of choice in this region despite the higher sensitivity for detecting cervical and intracranial atherosclerotic disease when using computed tomographic angiography or magnetic resonance imaging.28,29 This finding may reflect a North American aversion to risking contrast-associated injury or additional radiation exposure in the elderly.
The downstream effects of these biases in stroke evaluation are clear. First and foremost, incomplete etiologic evaluation for stroke has been shown to impact the proportion of elderly patients who receive guideline-based therapies,19–22,30 and may put these patients at greater risk of recurrent stroke, other major vascular events, and mortality.10–12 Second, there are a significant number of elderly patients with treatable causes of stroke who would benefit from such targeted intervention.20,31 Third, life expectancy is increasing, which means a growing number of elderly patients who may suffer from stroke as well as other comorbid diseases. 32 Although patients with ESUS have no specific treatable etiology of stroke, their risk of future ischemic stroke remains high in cryptogenic patients—especially the risk of recurrent cryptogenic stroke. 7 According to recent results from the Oxford Vascular study, the risk of recurrent infarct in cryptogenic stroke patients is just as great as strokes due to cardioembolic and large vessel disease. 33 Therefore, the etiologic identification and appropriate management of these patients may reduce long-term health costs associated with the morbidity of recurrent or progressive cerebro- and cardiovascular disease. 34
The explanation for the age disparity in stroke evaluations remains unclear and is a major limitation of the present study. Prior investigators have argued that physician uncertainty of the risks and benefits from standard treatment exists because randomized trials of stroke and other major vascular events underrepresent the elderly.19,20,35,36 Older patients who suffer stroke may also have either known comorbidities that contraindicate certain standard-of-care measures (e.g. progressive liver disease and statin use, or frequent falls and anticoagulant use) that reassure providers against pursuing additional testing which may be deemed irrelevant or inappropriate for the patient. Furthermore, older patients with higher disability and mortality rates may undergo limited evaluations. Older stroke patients and/or family may decide against additional testing/treatment in order to improve quality of remaining life.37–40 While many such reasons are justifiable, they may not fully explain the prominent age effect observed in this study. Elderly patients with good functional status, life expectancy, and adherence to treatment would likely benefit from thorough evaluation and treatment.
Despite the global scale of this investigation, this study has limitations. Notably, the definition of ESUS utilized in this stroke registry is relatively novel, but is more concrete than some of the earlier classifications for cryptogenic stroke.41–43 At the time the ESUS methodology was originally described, it was undecided whether the presence of intracranial arterial stenosis would be considered as an exclusion criterion, and therefore criteria for the ESUS Global Registry Project only later integrated intracranial imaging results within the definition of ESUS. Further, we are limited in that no data were collected regarding assessment of code status or goals of patient care. The data collected in our global registry did not include socioeconomic variables or data pertaining to level of education, which might have impacted the extent of diagnostic testing and level of care. Additionally, for patients without an ESUS diagnosis demographic variables were incompletely reported from affiliated sites, thereby prohibiting comparisons of baseline differences between “all patients without atrial fibrillation.” Finally, it is unclear whether certain sites may be more significant contributors to the age disparity in diagnostic evaluation (e.g. those with comparatively fewer resources or personnel) when compared to other sites.
Of note, a significantly smaller proportion of patients aged 30–39 received complete evaluation (and evaluation according to specific diagnostic test category) compared to other patients. Because of the small number of patients in the youngest age groups, we speculate that the incomplete evaluation of these patients stems from two sources: First, sampling error given the small number of subjects. Second, the likely diagnosis of stroke etiology may have been made at admission (e.g. cervical artery dissection) and thereby preempted further evaluation.
Conclusions
This is the first study to demonstrate an international bias toward less complete diagnostic evaluation of patients with embolic stroke of unclear etiology according to patient age. Underdiagnosis persists and likely contributes to undertreatment and excessive recurrent stroke in this population. Further investigations are warranted to explore and resolve the potential bias in age-associated stroke evaluation completeness.
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: SK receives consulting fees from Bayer, Boehringer-Ingelheim, Bristol Meyers Squibb, Medtronic and Daiichi Sankyo. RGH receives research support and serves on advisory committees for Bayer Healthcare and Bristol Myers-Squibb. JB receives research support from Bayer Healthcare.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by grants from Bayer Healthcare, Canadian Stroke Prevention Intervention Network, and the Hamilton Health Sciences Research Strategic Initiatives.
Ethical approval
The study was compliant with local institutional research board regulations of each participating center.
Informed consent
Informed consent was not required at any site for this anonymized data collection based on medical record review.
Guarantor
SEK.
Contributorship
JES and SEK conceived the idea of the project and performed the literature review. MG, JB, RGH, and SEK were responsible for data management and insured ethical approval was obtained by participating centers. BS performed all data analysis. JES wrote the first draft of the manuscript. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
Acknowledgements
None.
