Abstract
Background
The hypothalamus has been posited to play a role in premonitory symptomatology in migraine. Many patients also report particular triggers for their migraine attacks. Identifying the relationship between these may provide insights into the potential role of hypothalamic dysfunction in migraine pathophysiology.
Methods
A single centre cross-sectional study was performed. Data was collected on the presence of premonitory symptoms and triggers from patients who attended a tertiary headache clinic from February 2024 to December 2024 and entered into a Headache Database. Statistical analysis with multivariate binomial logistic regression was performed to identify relationships between premonitory symptoms and triggers.
Results
Of the100 patients who were identified as fulfilling a diagnosis of migraine as per ICHD-3 criteria, 96% reported at least one premonitory symptom. Women reported a greater number of premonitory symptoms (6 [IQR 4–9] vs 4 [IQR2–7], P = 0.005) and triggers (4 [IQR 3–7] vs 3 [IQR 2–5], P = 0.046) compared to men. Linear regression showed that patients with more triggers were likely to report more premonitory symptoms (R2 = 0.466). Statistically significant correlations were found between certain triggers and premonitory symptoms such as poor sleep and photophobia (P = 0.001) and overcast weather and drowsiness (P = 0.003). Independent correlations were also found between certain premonitory symptoms, allowing for groupings of certain symptoms with each other.
Conclusions
The study demonstrated that patients who reported more triggers were likely to report more premonitory symptoms, and that women reported more premonitory symptoms compared to men. Certain triggers were also associated with the presence of certain premonitory symptoms, for example poor sleep and photophobia as well as overcast weather and drowsiness. The presence of independent associations between certain triggers and premonitory symptoms may suggest specific network dysfunctions in different networks that may account for interindividual variability in migraine triggers and symptomatology.
Introduction
Migraine is a neurological condition that has been described since ancient times and remains one of the most prevalent and disabling diseases globally, 1 however the pathophysiology of this disease remains incompletely understood.
Our knowledge regarding the biology of migraine has evolved over time, and considerable debate still remains regarding if migraine is a primarily peripherally-mediated process, centrally-mediated process or perhaps more likely, a combination of both. 2
The sentinel feature of migraine in medical discourse has been the throbbing or pulsating moderate to severe intensity headache, that is often lateralised and associated with a combination of either photophobia, phonophobia or nausea and vomiting. This is reflected in the International Classification of Headache Disorders, currently in its third version, that serves as the gold standard for the diagnostic criteria for headache disorders. 3 However, this does not entirely reflect the multi-phasic nature of the disease including premonitory and postdrome symptoms of migraine, which are increasingly understood to also be a part of the migraine attack rather than separate entities. 4
The array of different patient-reported triggers, premonitory and headache phase symptoms that patients with migraine can experience as well as the heterogeneity of combinations of these symptoms between migraine patients suggests a widespread network disorder involving multiple different parts of the brain.
Given the high prevalence of migraine, there have been several studies exploring the potential evolutionary reasons for its development. 5 It is possible that given the most common patient reported triggers for migraine are psychological stress and poor sleep and that the response of many migraine patients to aid in coping with symptoms during an attack is to lie down in a dark and quiet environment, that migraine reflects an aberrant homeostatic response.6,7 Much of our homeostatic response is mediated in the central nervous system by the hypothalamus, and thus this is potentially an important substrate in the pathophysiology of migraine.
The hypothalamus is a highly connected region of the brain with both afferents and efferents projecting to other areas that have been implicated in migraine pathophysiology, including the locus coeruleus, the thalamus, the trigeminovascular system, the sphenopalatine ganglion and the vagus nerve. It is also a sexually dimorphic area heavily influenced by and influential to hormonal function.8,9 Migraine affects females more than males and the disease can be influenced by hormonal changes such as in catamenial migraine, during pregnancy and as well as perimenopause. 10
Previous studies have primarily focused on the relationship between triggers and premonitory symptoms within the same sensory modality and which can often be conflated, for example food cravings as a premonitory symptom and certain foods as a trigger, as well as photophobia as a premonitory symptom and light as a trigger. 11 Cross-modal relationships between triggers and premonitory symptoms that are not conflated in this manner have not been explored as extensively. In the absence of definitive biomarkers for migraine, we look to explore if relationships between premonitory symptoms and patient reported triggers obtained through detailed history taking may provide further insights into the biology of migraine.
Objectives
Primary: To characterise relationships between premonitory symptoms and patient reported triggers from patients in our tertiary headache clinic.
Secondary: To characterise relationships between different premonitory symptoms and thus describe certain groupings of premonitory symptoms that may suggest common biological pathways.
Methods
A cross-sectional single centre study was performed. Ethics approval was obtained by the St Vincent's Hospital Human Research Ethics Committee (2024/ETH0997). All participants were provided with written information about the study and provided informed consent.
Data was collected on consecutive patients seen in a tertiary headache clinic between February 2024 and November 2024. Patients fulfilling International Classification of Headache Disorders 3 (ICHD-3) criteria for migraine were included in the study. Patients with other concurrent headache disorders and headache phenotype that could not be distinguished from migraine were excluded from the study. Patients with less than 8 headache days a month were classified as having Low Frequency Episodic Migraine (LFEM), patients with 8–14 headache days a month as having High Frequency Episodic Migraine (HFEM) and patients with 15 or more headache days a month as having Chronic Migraine (CM).
Data collection and variables
Premonitory symptoms were defined as symptoms the patient identified prior to the headache phase of most of their migraine attacks. The timeframe of onset was between 48 h to immediately prior the headache phase of the attack. Patients were asked to identify premonitory symptoms they may have and subsequently prompted from a list of premonitory symptoms including food cravings, nausea, drowsiness, cognitive fogging, irritability, anxiety, depression, fatigue, photophobia, phonophobia, osmophobia, muscle soreness, neck soreness, yawning, lacrimation of the eyes and nasal congestion or rhinorrhoea.
Patient reported triggers were defined as potential external stimuli or circumstances patients encountered or experienced that would most of the time be followed by the onset of migraine symptoms within four hours, including premonitory symptoms. Patients were asked to identify these potential triggers and then subsequently prompted from a list of triggers including less sleep than usual, more sleep than usual, psychological stress, relaxation, concurrent illness, certain foods, overcast weather, heat, dehydration, change in season, missing meals, eating more than usual (labelled as excess eating), physical exertion and air travel. Data was then entered into a secure REDCAP database after clinic review.
Statistical analysis
The cohort of patients with HFEM was combined with the CM patients in our analysis. Linear regression was used to assess the relationship between number of triggers and number of premonitory symptoms. Multivariate binomial logistic regression was performed with each premonitory symptom as a dependent variable with all triggers as independent variables, to identify associations independent of the confounding influence of other triggers. Further multivariate binomial logistic regression was performed with each premonitory symptom as a dependent variable and all other premonitory symptoms as independent variables to assess if certain symptoms commonly occurred together in certain groups. Goodness of fit was assessed by Hosmer-Lemeshow P-value > 0.05 and model accuracy by correct classification rate. A likelihood ratio P-value <0.05 was considered statistically significant. Bonferroni correction was also applied, this was calculated manually by using a P-value threshold of 0.05 for statistical significance and then dividing this by the number of comparisons for each logistic regression analysis. Thus P < 0.0035 was the threshold for statistical significance after the correction for the logistic regression analysis of triggers and premonitory symptoms and P < 0.0031 was the threshold for statistical significance for the logistic regression analysis of premonitory symptoms alone.
As the data was non-parametrically distributed, statistical significance of numerical values between groups was calculated using Mann-Whitney two-tailed testing. Fisher's Exact Test was also performed to calculate statistical difference between demographic groups. All statistical analysis was performed using IBM SPSS Statistical Software (Version 29).
Results
Demographics
One hundred patients seen between February 2024 and November 2024 were identified as fulfilling ICHD-3 criteria for migraine, 27% with LFEM (27/100) and 73% with HFEM/CM (73/100). In total 80% were women (80/100) and 20% were men (20/100). There was no statistically significant difference between the proportion of women with LFEM compared to men (26% [21/80] vs 30% [6/20], P = 0.781) or the proportion of women with HFEM/CM compared to men (74% [59/80] vs 70% [14/20], P = 0.781). The median age for the entire cohort was 45 years old [IQR 33–56], there was no statistically significant difference between the age of women in our study cohort compared to men (44 years old [IQR 32–56] vs 49 years old [IQR 37–63], P = 0.354). There was no statistically significant difference in age between patients with LFEM and patients with HFEM/CM (40 years old [IQR 31–50] vs 47 years old [35–60], P = 0.109).
The relationship between premonitory symptoms and triggers
The median number of premonitory symptoms reported amongst the study cohort was 6, with 96% (96/100) having at least one premonitory symptom. Women reported a greater number of premonitory symptoms compared to men (6 [IQR 4–9] vs 4 [IQR2–7], P = 0.005) and HFEM/CM patients reported a greater number of premonitory symptoms compared to LFEM patients (7 [IQR 4–9] vs 5 [IQR 2–6], P = 0.002). The most commonly reported triggers were less sleep (84%, [84/100]) and stress (83%, [83/100]), Table 1 shows the prevalence of all triggers in the study cohort. The most commonly reported premonitory symptoms were photophobia (78% [78/100]), cognitive fogging (64% [64/100]) and neck soreness (59% [59/100]), Table 2 shows the prevalence of all premonitory symptoms in the study cohort. The median number of triggers was 4 (IQR 3–6). Women reported more triggers than men (4 [IQR 3–7] vs 3 [IQR 2–5], P = 0.046). The median number of triggers reported by patients with HFEM/CM was higher than patients with LFEM (5 [IQR 3–7] vs 3 [IQR 2–4], P = 0.011). A linear regression model showed that a higher number of reported triggers was associated with a higher number of premonitory symptoms, as reflected in the scatter plot in Figure 1 (R2 = 0.466).

A linear regression model showed that a higher number of reported triggers were associated with a higher number of premonitory symptoms, as reflected in the scatter plot.
Prevalence of triggers.

Prevalence of premonitory symptoms.

The statistically significant trigger associations for each premonitory symptom after multivariate binomial logistic regression trigger as a predictor variable are shown in Table 3. Hosmer-Lemeshow P-value was in excess of 0.05 for each calculation indicating good fit models for all the logistic regression tests. After Bonferroni correction, statistically significant positive associations were found between less sleep as a trigger and photophobia as a premonitory symptom, excess sleep as a trigger and nausea as a premonitory symptom as well as overcast weather as a trigger and drowsiness as a premonitory symptom. A statistically significant negative association was found between excess sleep as a trigger and irritability as a premonitory symptom. No statistically significant relationship was found between lacrimation and other triggers.
Association between premonitory symptoms and triggers.

These results remained significant on Bonferroni correction. Missing Exp(B) values are because of quasi-separation of data as all the patients with the premonitory symptom had that trigger.
The relationship between different premonitory symptoms
The results of multivariate binomial logistic regression with each premonitory symptom as a dependent variable and the remaining symptoms as independent variables are shown in Table 4. Irritability did not demonstrate any associations with other premonitory symptoms. Hosmer-Lemeshow P-value was in excess of 0.05 for each calculation indicating good fit models for all the logistic regression tests.
Association between different premonitory symptoms.

*These results remained significant on Bonferroni correction.
Bidirectional positive associations were found between photophobia and phonophobia, osmophobia and phonophobia, osmophobia and fatigue, fatigue and drowsiness, drowsiness and muscle soreness, anxiety and depression as well as yawning and food cravings. Irritability did not have a statistically significant association with any other symptom. Figure 2 provides a diagrammatic representation of these relationships.
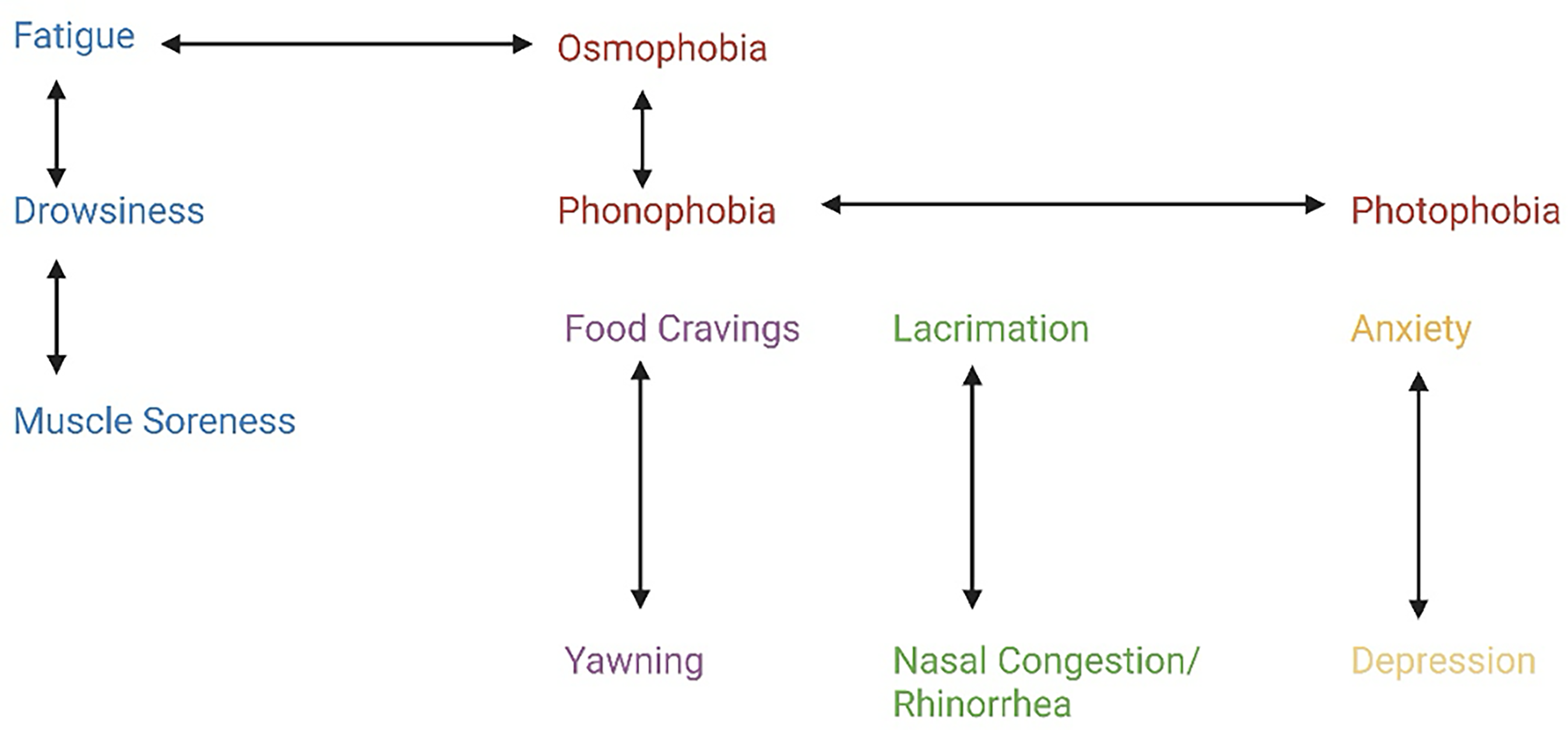
Provides a diagrammatic representation of these relationships. Solid lines indicate bidirectional positive correlations. Red = sensory hypersensitivity group, Blue = Generalised somatic symptoms, Purple = symptom group attributed to cortisol increase, Green = cranial autonomic symptom group, Yellow = mood disturbance group. The red group consists of symptoms that are considered cardinal for the diagnosis of migraine. The blue group consists of symptoms commonly seen in patients with chronic pain disorders and fibromyalgia, which are often comorbid with migraine.12,13 The purple group consists of symptoms that have been shown to arise from activation of the hypothalamic-pituitary-axis (HPA), mediated by the paraventricular nucleus.14,15 The green group consists of cranial autonomic symptomatology and the yellow group consists of mood disturbance symptoms. Created with Biorender.com
Discussion
Prevalence of triggers and premonitory symptoms in relation to headache frequency and sex
There was a high prevalence of premonitory symptoms and triggers in our study cohort, with most patients reporting at least one premonitory symptom and at least one trigger. Patients with HFEM/CM reported more triggers and premonitory symptoms compared to those with LFEM, suggesting a positive relationship between headache frequencies, premonitory symptoms and number of triggers. Furthermore, our study showed that patients with more triggers were likely to report more premonitory symptoms.
The importance of the hypothalamus in premonitory symptomatology has been demonstrated via neuroimaging studies in humans. 16 Furthermore, hypothalamic nuclei play a vital role in the regulation of sleep, 17 fatigue, 18 cognition, 19 mood regulation, 20 nausea, 21 appetite, 22 sound sensitivity, 23 light sensitivity 24 and pain. 25 Similarly, it is the primary organ of homeostatic control, with evidence showing that it is influenced by environmental factors such as sleep, 17 stress, 26 changes in atmospheric pressure, 27 heat 28 and oral intake. 22
Our study also found women reported more premonitory symptoms and triggers compared to men. Multiple studies have demonstrated sex differences in hypothalamic structure and function, including differences in HPA response between males and females which suggest sex-related differences in paraventricular nucleus function specifically.8,9
Based off these previous findings and the positive relationship between number of premonitory symptoms and triggers as well as the positive relationship between headache frequency and migraine triggers found in our study, we propose a hypothesis to test in future studies: that a higher frequency of migraine attacks may reflect a greater degree of hypothalamic dysmodulation, resulting in increased sensitivity to more potential triggers and therefore increased activation of hypothalamic nuclei that project to central nervous system structures responsible for premonitory symptoms.
The relationship between triggers and premonitory symptoms
Our study identified several cross-modal associations between certain premonitory symptoms and triggers that are not easily conflated. The associations that withstood Bonferroni correction were between poor sleep as a trigger and photophobia as a premonitory symptom, excess sleep and premonitory symptoms of irritability and nausea and overcast weather as a trigger and drowsiness as a premonitory symptom. Our study also validates the relationship between food cravings and certain foods as a trigger that has previously been identified, although this relationship is difficult to analyse given possible conflation between the two concepts. 11
Photophobia and poor sleep
Poor sleep as a trigger was independently associated with photophobia as a premonitory symptom in the study cohort. This validates an association between photophobia and sleep quality found in a previous study, 29 although this did not specifically pertain to photophobia as a premonitory symptom. The mechanism for this link has not been definitively determined, however there are several hypotheses. Firstly, photophobia by definition entails the avoidance of light stimuli, and as such may be a homeostatic response to promote seeking out a darker environment for sleep. There is accumulating evidence of the chronobiological aspects of migraine and migraine patients commonly report improvement in their symptoms with sleep.30,31 The sleep cycle is modulated, in part, by the suprachiasmatic nucleus (SCN) and indirectly by the paraventricular nucleus (PVN). 32 The SCN receives input from but also feeds back to intrinsically photosensitive retinal ganglion cells (ipRGCs), which are considered to be the main cells responsible for photophobia via the melanopsin signalling system.33,34 This is further supported by the fact that ipRGCs function independently of rods and cones, with patients continuing to experience photophobia despite blindness from complete lack of rod and cone function.35,36
Whilst direct evidence of hypothalamic modulation of ipRGCs has been explored primarily in animal models, circadian fluctuations in ipRGC function independent of external light cues have been demonstrated in a study with humans, providing indirect evidence of possible ipRGC control from the SCN in human beings. 37
Nausea, irritability and excess sleep
Excess sleep as a trigger was independently associated with nausea as a premonitory symptom in the study cohort. Excess sleep has been identified as a trigger in previous studies, however the relationship with nausea has not been previously explored, although there are conceivable pathways through which they may be linked.
Orexin-A, a neuropeptide in hypothalamic circuits, reduces in sleep, and thus levels may be particularly low in cases of oversleeping. 38 Exogenous orexin-A has anti-emetic effects in animal models of nausea, and orexin-A has been shown to modulate appetite and feeding behaviour. 39
There is reduced firing of dopaminergic neurons in the lateral hypothalamus during prolonged NREM sleep in animal models as well as reduced dopamine receptor sensitivity.40,41 PET studies in humans have shown that excitation of dopamine receptors in the context of reduced dopamine receptor sensitivity can be followed by a compensatory dopamine surge. 42 As such, it is possible that oversleeping can be followed by a compensatory dopamine surge, given the prolonged reduction in dopamine receptor sensitivity in this context. Dopamine agonism with apomorphine has been shown to induce nausea in migraineurs. 43
This hypothesis regarding the compensatory dopamine surge after excess sleep may also account for the inverse relationship between excess sleep and irritability as a trigger that was found in our study, as elevated dopamine levels are associated with improved mood and may conceivably result in lower irritability. 44
Drowsiness and overcast weather
Overcast weather as a trigger was independently associated with drowsiness as a premonitory symptom in the study cohort, this relationship has not been reported previously.
Atmospheric pressure changes have been found to cause noradrenergic activation of the sympathetic nervous system, possibly through baroreceptor activation. 45 These sympathetic pathways are largely mediated by the nucleus tractus solitarius, which can modulate cortical arousal as demonstrated by EEG through three possible pathways; the posterior hypothalamus, the ventrolateral medulla and locus ceruleus or through thalamo-brainstem pathways. 46 However, it remains unclear which of these pathways is most implicated in this particular relationship.
The relationship between different premonitory symptoms
Our results highlighted associations between different premonitory symptoms, suggesting that certain premonitory symptoms are independently linked. These findings are illustrated in Figure 2 and align with these symptoms occurring in groups that could share common biological mechanisms.
This generates the hypothesis that interindividual variability in migraine symptoms may suggest differences in central nervous system networks that are affected in that particular patient's migraine biology. In combination with the hypothesis generated earlier in the paper, this may specifically reflect variability in central nervous system, including hypothalamic, brainstem and thalamic pathways, that are dysmodulated in patients with migraine, suggesting there exist distinct patterns of dysmodulation between patients, accounting for differences in premonitory symptoms and triggers.
Limitations
As the data is based on patient history, it is subject to recall bias, although given the cohort included mostly chronic migraine patients, it is likely that their last attack was quite recent to the clinic appointment. However, this also means recall bias may be more pronounced in patients in the LFEM group, as their attacks may be more remote to the clinical review when data was collected. There was no inter-observer variability as a single clinician collected the history for all the study participants. The study cohort size was quite small as well, reflecting the clinic being newly established and the paucity of public hospital Headache Clinics in Australia. There may also be a referral bias for more complex cases given the hospital is a metropolitan tertiary referral centre. Some values had a very high Exp(B) and very large confidence intervals, suggesting small subgroup size and there was some quasi-separation of data that should be interpreted with caution. Although many associations were found, a significant proportion of results did not continue to have statistical significance after Bonferroni correction, which may reflect the smaller sample size as well as the exploratory nature of the study.
Some migraine triggers and premonitory symptoms may be difficult to distinguish, and the history may not accurately reflect this. Previous studies have shown that many patients who report certain foods as a trigger are in fact experiencing food cravings rather than having attacks directly attributable to consuming certain foods. 11 This association was validated in our data. Similarly, physical exertion may lead to muscle soreness independent of migraine biology. However, the statistically significant correlations found in the study are between triggers and premonitory symptoms that are unlikely to be conflated in this manner, including poor sleep and photophobia, excess sleep and nausea and overcast weather and drowsiness. As such, we do not think this affects our findings significantly. Data regarding premonitory symptoms including increased urinary frequency, gastrointestinal disturbance and other cranial autonomic symptoms as well as some triggers such as certain smells were not collected in our study and future studies may be more comprehensive in this regard.
Furthermore, the majority of patients in our cohort had chronic migraine and at times may find it difficult to distinguish between different migraine attack phases i.e. report premonitory symptoms that may be occurring in the postdrome or during headache phases such as cognitive fogging. This may explain why the prevalence of certain premonitory symptoms was much higher in our cohort compared to other studies, although another factor is significant heterogeneity between studies in how premonitory symptom data is collected.47,48 This may result in some premonitory symptoms not in fact being purely premonitory, however the distinct phases do not necessitate distinct biological processes between premonitory symptoms occurring before the headache phase and premonitory symptoms that persist into headache phase and postdrome phase. Our definition of premonitory symptoms for this study, was different from the operational definition used in another recent study, where the latest cut-off was defined as two hours before headache phase. 49 This is based on our clinical experience where patients report symptoms before headache that may occur less than 2 h before the headache phase but clearly before the onset of pain. Furthermore, by defining premonitory symptomatology as happening between 2 and 24 h before headache phase, this leaves a gap of 2 h before headache where any symptoms experienced during this period of time remain unclassified. We note that whilst ICHD-3 mentions premonitory symptomatology, a strict time definition is not actually specified. The afore-mentioned study on the relationship between premonitory symptoms and triggers provided supporting data that premonitory symptoms do not exist solely in a discrete phase. This study also corroborated previously known findings about the relationship between same-modal premonitory symptoms and triggers, but did not explore cross-modal associations, which are the focus of our study. Indeed, neuroimaging data shows that hypothalamic and brainstem activation seen in the premonitory phase can persist into the headache phase. 34 There is also a paucity of evidence suggesting the same symptom in different phases of a migraine attack is due to distinct biological processes. Therefore, we do not feel this invalidates the findings of the study or the hypotheses generated.
Conclusion
This exploratory study found that patients with higher frequency of headache were likely to report a greater number of migraine triggers and premonitory symptoms and that patients with more migraine triggers were likely to report a greater number of premonitory symptoms. The study also found women were more likely to report a greater number of migraine triggers and premonitory symptoms compared to men. Furthermore, independent associations were found between poor sleep and photophobia, excess sleep and nausea and irritability as well as overcast weather with drowsiness. Certain premonitory symptoms were more likely to occur together, independent of other premonitory symptoms a patient may report. These findings suggest distinct patterns of triggers and premonitory symptomatology between patients. The authors hypothesise this may be due to migraine being a central nervous system network disorder, with dysmodulation of different hypothalamic, brainstem and thalamic pathways possibly explaining heterogeneity in migraine characteristics between patients. Further studies with larger cohorts would help validate the findings of this study and may also uncover other associations.
Article highlights
There was a positive association between the number of patient reported triggers and the number of premonitory symptoms patients experienced.
Certain premonitory symptoms (such as photophobia and drowsiness) were found to be independently correlated with certain triggers (poor sleep and overcast weather).
Particular premonitory symptoms were more likely to occur in particular groups with other premonitory symptoms, for example food cravings and yawning and sensory hypersensitivities with nausea.
This study generates the hypothesis that the interindividual variability in triggers and premonitory symptoms between migraine sufferers may reflect differential dysmodulation in the network of central nervous system structures relevant in migraine pathophysiology.
Footnotes
Acknowledgments
Not applicable.
Ethical considerations
Ethics approval was obtained by the St Vincent's Hospital Human Research Ethics Committee (2024/ETH0997).
Consent to participate
All participants were provided with written information about the study and provided informed consent.
Consent for publishing
Yes
Author contributions
Both authors equally contributed.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The information is provided upon reasonable request to the corresponding author.
Open practices
Not applicable.
