Abstract
Introduction
The management of acute exacerbations of trigeminal neuralgia remains a major clinical challenge due to the limited availability of clinical evidence. This retrospective case series aims to evaluate the effectiveness and safety of intravenous phenytoin in the treatment of these exacerbations.
Methods
We retrospectively analyzed the outcome of patients treated with intravenous phenytoin for acute exacerbations of trigeminal neuralgia at the University Hospital Zurich between January 2016 and December 2023. Statistical analyses were performed to assess the associations of patient and disease characteristics with phenytoin response.
Results
In our sample of 45 cases, treatment with phenytoin resulted in a significant or complete relief in 30 cases (66.7%), while 11 (24.4%) experienced slight relief and 4 (8.9%) none. Patient or disease characteristics were not associated with treatment response. Side effects were rare, with skin itching occurring in only one case.
Conclusion
This retrospective case series demonstrates, that phenytoin is a safe and effective treatment for acute exacerbations of trigeminal neuralgia. However, larger prospective studies are needed.
Introduction
Trigeminal neuralgia (TN) manifests as a recurring, paroxysmal and unilateral facial pain localized in the area of one or more branches of the trigeminal nerve. TN encompasses idiopathic, classical (caused by pathological neurovascular contact), and secondary forms. The secondary forms are associated with conditions such as multiple sclerosis, brainstem ischemia, or space-occupying lesions. 1
Triggered by innocuous stimuli such as chewing, drinking, talking, or brushing teeth, the pain significantly affects patients’ quality of life, often leading to serious complications such as dehydration and malnutrition. 2 Single attacks of TN are too brief for medical intervention, and the aim of acute treatment remains to reduce the remarkably high frequency and severity of attacks. 2 Although some medications have proven effective in long-term treatment, with sodium channel blockers typically recommended as the primary prophylactic therapy, the management of acute exacerbations of TN remains a clinical challenge due to limited clinical data and the severity of the clinical condition.2–4
Conventional analgesics such as paracetamol, non-steroidal anti-inflammatory drugs (NSAIDs) and opiates typically fail to provide adequate relief.2,3 Despite the limited available data, current guidelines - predominantly based on clinical experience and expert opinions - suggest intravenous phenytoin as a first-line treatment.2–5 Phenytoin exerts its therapeutic effects by inducing voltage- and frequency-dependent blockade of voltage-gated sodium channels (VGSCs), thereby reducing nerve hyperexcitability and alleviating pain. 6 The evidence supporting the use of this approach remains limited and it is derived from only one prospective observational series by Andersen et al. and several retrospective case series.7–9 Besides intravenous lidocaine and lacosamide, which may serve as treatment alternatives, other proposed treatments are less common and appear less effective.3,4,8
Given the limited availability of studies on the efficacy of phenytoin in this context and the need for more comprehensive data, we performed a retrospective analysis of patients with acute exacerbations of TN treated with intravenous phenytoin in our emergency department.
Methods
We conducted a retrospective study of patients coded as acute exacerbation of TN who were treated with intravenous phenytoin in the emergency department of the University Hospital Zurich between January 2016 and December 2023. Inclusion criteria were meeting the diagnostic criteria for TN according to section 13.1.1. of the International Classification of Headache Disorders, 3rd edition (ICHD-3), 1 treatment with intravenous phenytoin and availability of general consent for participation in a research project [Figure 1].
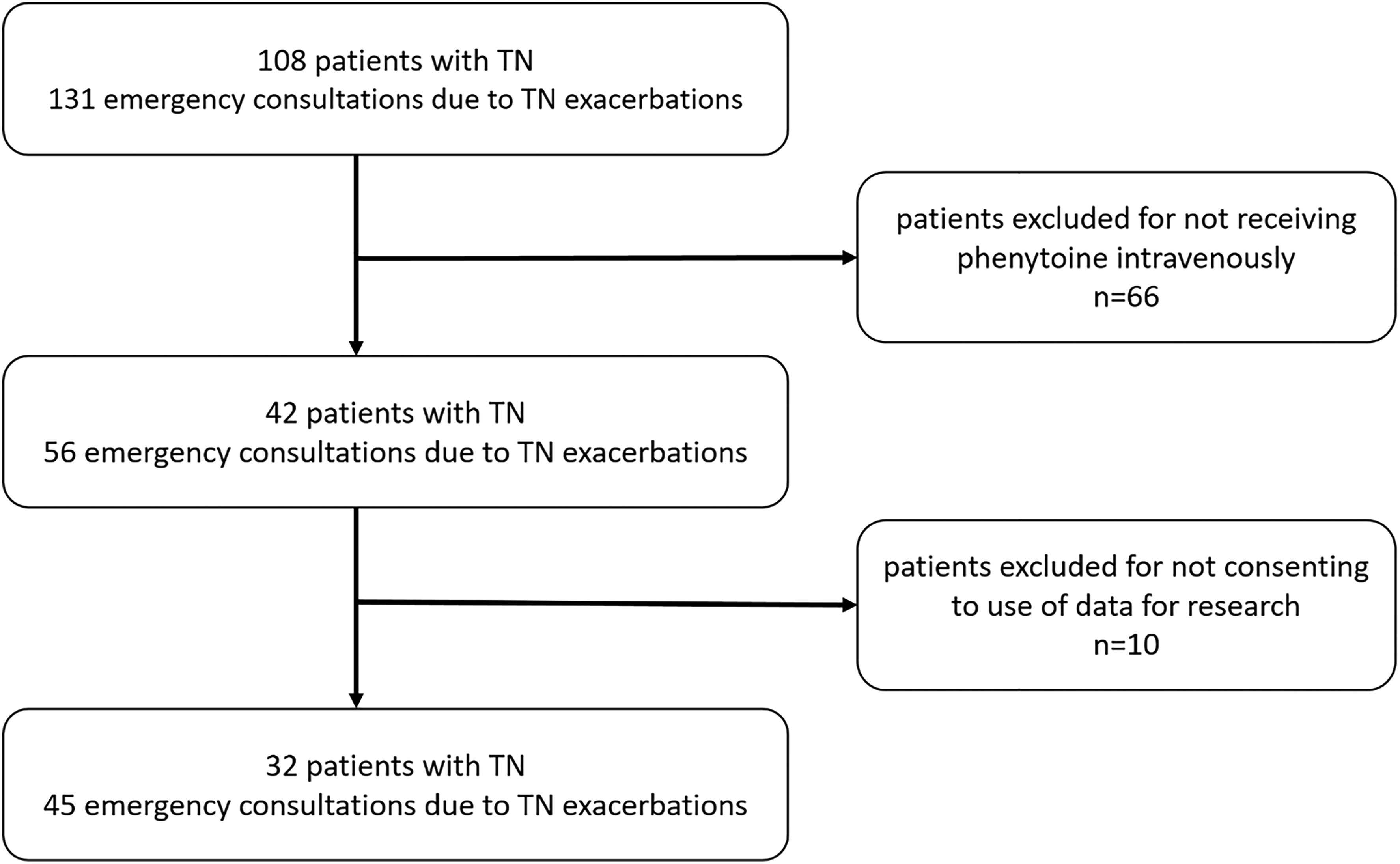
Flowchart outlining the inclusion and exclusion criteria for the sample.
The data were collected from electronic records stored in the University Hospital Zurich's medical record system. They included routine clinical data such as sex, age at TN diagnosis, age at emergency presentation, diagnosis, pain characteristics, drugs used in the treatment of TN with doses, and the patients’ outcome after treatment with phenytoin.
The emergency medical records lacked a standardized assessment of patient outcomes. The available descriptive treatment results were independently analyzed by two neurologists and categorized into three groups: “significant or complete relief”, “slight (insufficient) relief”, and “no relief”. “Significant or complete relief” indicated patients were either pain-free or had residual pain that did not disrupt them upon discharge from the emergency department post-infusion. “Slight (insufficient) relief” meant despite reduced attack intensity and frequency, pain still limited patients. “No relief” signified no perceived benefit from therapy.
Phenytoin treatment was considered for severe exacerbations that significantly impaired daily activities such as eating, drinking, and speaking. Dosages were individually determined by the treating neurologist after excluding contraindications based on electrocardiogram and blood test results.
The primary endpoints of the study were pain relief and the incidence of adverse effects during hospitalization. Secondary endpoints included the necessity for emergency readmission and the time to such readmission.
First, we describe the collected data using descriptive statistics. Continuous demographic and clinical variables are presented as means and standard deviation, depending on their distribution. Categorical variables are reported as frequencies and percentages [%].
To identify the association of patient or TN characteristics (etiology) or treatment (phenytoin dose) with the time to readmission after the last emergency treatment with phenytoin, we calculated the Kaplan-Meier estimator. Data were regarded as censored if patients did not return to the emergency department after 7 and 14 days after the treatment. We compared the curves using the log-rank test and set the significance level at 0.05.
Statistical analyses were performed using SPSS version 29.0.0.0 (IBM) and Microsoft Excel 2016. The coded data were collected and analysed in accordance with the approval granted by the Zurich Ethics Commission under BASEC number 2023-00755.
Results
Patient population
We identified 32 patients with 45 emergency consultations due to acute TN exacerbations between January 2016 and December 2023 that were treated with intravenous phenytoin. Six patients (18.8%) received the drug during more than one emergency consultation: 4 patients (12.5%) three times, 1 patient (3.1%) two times, and 1 patient (3.1%) five times. Each consultation entered the analysis separately.
Within our sample, 23 of 32 patients (71.9%) were female and the median age of the first manifestation of their TN was 56.1 years (SD = 15.1). On average, the emergency admission due to the first exacerbation occurred 4.1 years after the initial diagnosis. Ten out of 32 patients (31.3%) were diagnosed with classical TN, while 13 patients (40.6%) had a secondary TN, with the following specific etiologies: 10 (76.9%) were associated with multiple sclerosis, 2 (15.4%) with tumors, and 1 (7.7%) with arteriovenous malformation. In the remaining nine patients (28.1%), the etiology remained unknown (idiopathic). The majority (19/32, 59.4%) reported pain on the right side. The pain was most commonly localized in the V3 area (13/32, 40.6%), followed by an overlap of V2 and 3 (10/32, 31.3%), V2 (6/32, 18.8%), and an overlap of V1 and 2 in 3 cases (3/32, 9.4%). In addition to the paroxysmal attacks 6 patients (18.8%) suffered from concomitant continuous facial pain. The demographic and clinical characteristics of the patients are summarized in Table 1.
Demographic and clinical characteristics of patients in the analyzed groups.

SD = standard deviation; TN = trigeminal neuralgia, V1 = ophthalmic nerve, V2 = maxillary nerve, V3 = mandibular nerve.
The median age at presentation of the 45 emergency admissions was 60 years (SD = 15.8). At the time of infusion in 32 of 45 cases (71.1%), patients were receiving a long-term oral prophylaxis. Among these, 22 patients (48.8%) were on ongoing monotherapy, most frequently with sodium channel blockers (12/45, 26.7%), followed by lamotrigine (3/45, 6.7%), gabapentin (2/45, 4.4%), pregabalin (2/45, 4.4%), and phenytoin (2/45, 4.4%). Additionally, 7 patients (15.6%) were receiving polytherapy, which included sodium channel blockers (5/45, 11.1%), pregabalin (5/45, 11.1%), lamotrigine (3/45, 6.7%), phenytoin (3/45, 6.7%), and gabapentin (1/45, 2.2%). The median dose of intravenous phenytoin administered was 250 mg (range 100–500 mg), with the most common dose being 250 mg (37/45, 82%). This was followed by 200 mg (3/45, 7%), 100 mg (3/45, 7%), 300 mg (1/45, 2%), and 500 mg (1/45, 2%).
Response to treatment
In 10 of 45 cases (22.2%), patients had been treated without success with other drugs (paracetamol, non-steroidal anti-inflammatory drugs (NSAIDs) and opiates) before receiving phenytoin. The outcomes of 45 emergency presentations were variable, but in 41 cases (41/45, 91.1%), patients experienced at least some relief after treatment with phenytoin; 66.7% of patients (30/45) reported significant or complete relief, while 24.4% (11/45) experienced only slight or inadequate relief. Only 8.9% (4/45) of patients reported no therapeutic benefit [Table 2].
Phenytoin treatment response rate.

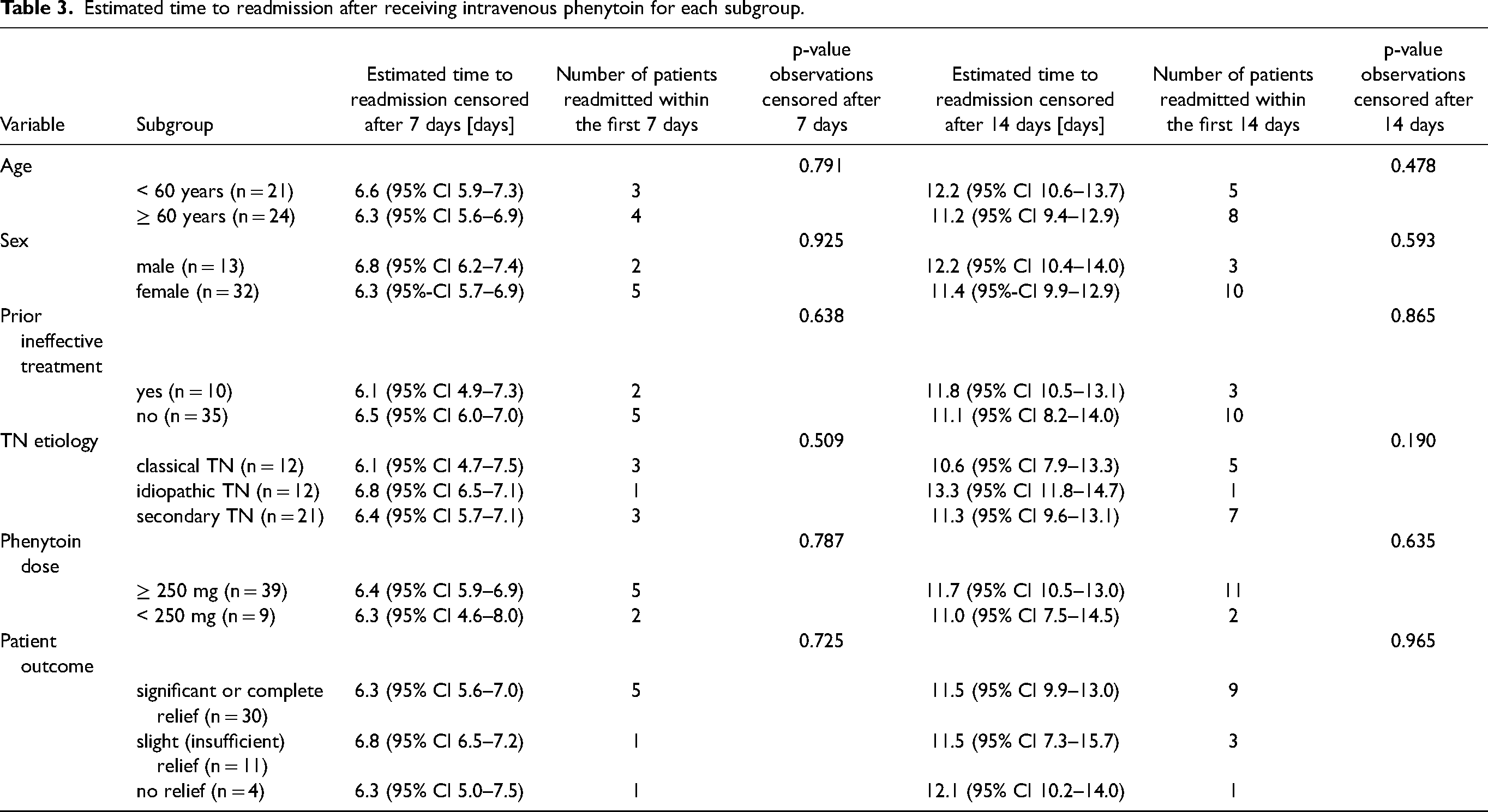
We further analyzed if time to readmission on days 7 or 14 after the initial presentation was different depending on certain patient or TN characteristics [Table 3]. However, we did not find a significant impact of patients age, sex, prior (ineffective) treatment, TN etiology, dose of phenytoin or patient outcomes on times to readmission [Figure 2A and 2B].

Kaplan-Meier estimator of the time to readmission within 14 days after receiving emergency treatment with phenytoin.
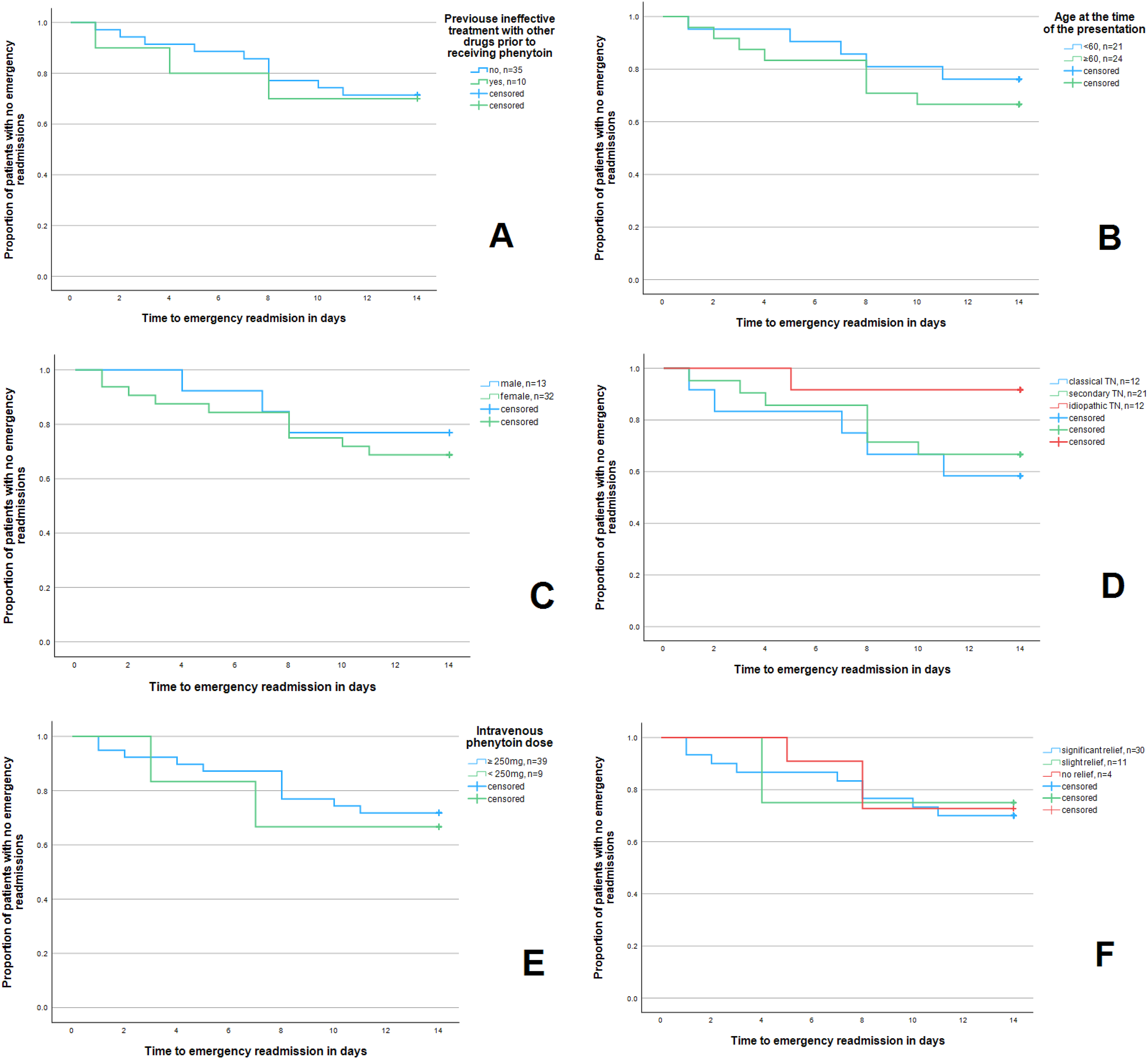
Kaplan-Meier estimator of differences in time to readmission within 14 days after emergency treatment with phenytoin in the group with and without previous ineffective treatment with other drugs before receiving phenytoin (A), in the group below and above middle age at presentation (B), according to patients’ sex (C), TN etiology (D), phenytoin dose (E) and patients’ outcome (F).
Estimated time to readmission after receiving intravenous phenytoin for each subgroup.
Side effects
In one case (1/45, 2.2%), therapy had to be discontinued despite mild relief due to an allergic reaction (itchy skin).
Discussion
The management of acute exacerbations of TN remains a major clinical challenge due to the limited availability of clinical trials and data. Current guidelines are largely based on clinical experience and expert opinion. In an attempt to expand the evidence, we identified 45 emergency presentations and followed the outcomes after treatment with intravenous phenytoin for acute exacerbations of TN in our emergency department. This effort resulted in one of the largest study samples on this subject published to date.
Nearly all patients experienced some relief after treatment with phenytoin infusion and two thirds experienced substantial or complete relief of symptoms. We found no statistically significant differences in the efficacy of this therapy according to patient or disease characteristics, suggesting that the approach is potentially effective in all patients and TN types. Side effects were very rare, with skin itching occurring in only one case.
We observed that in the first 14 days, patients with idiopathic TN were less likely to present with a new exacerbation (p = 0.084) compared to those with other etiologies. Additionally, the prevalence of secondary TN in our sample (40.8%) is notably higher compared to rates reported in the literature. 2 These trends have not been discussed in previous publications on this subject and may be attributed to the small sample size. However, the slightly elevated proportion of 17.9% secondary TN in the case series on the same topic by Schnell et al. may indicate that these patients are more likely to present to the emergency department due to exacerbations of TN. 7
The presented results on phenytoin effectiveness align with previous publications, particularly with the only prospective study by Andersen et al. where 60% of the 15 patients with classic and idiopathic TN experienced at least a 50% reduction in pain intensity within 24 h post-infusion. 9 In the two largest available retrospective case series, which included patients with classic, idiopathic, and secondary TN, pain relief was documented in 72.8% and 89.2% from 81 and 65 receiving phenytoin infusions, respectively.7,8
Despite its efficacy, the use of phenytoin has several important limitations. The crucial aspect is the need for qualified medical professionals to supervise its use. It is advisable to administer the drug in a high dependency unit with continuous cardiac monitoring due to potential serious cardiac side effects. Phenytoin is contraindicated in individuals with a recent history of myocardial infarction, reduced left ventricular function, or second- and third-degree atrioventricular block.2,4 It is important to recognize that single intravenous phenytoin doses do not affect long-term management and outcome. However, effectively managing the acute exacerbation provides a chance to initiate or adjust appropriate long-term medical prophylaxis and greatly improves the patient's quality of life.
Considering the limitations and potential drawbacks of phenytoin, there is an urgent need for an effective and safe treatment that patients can also administer themselves. Recent studies suggest alternative intravenous drugs such as lacosamide and lidocaine as viable options. However, evidence regarding their efficacy remains weak, and administration of these drugs should also be restricted to qualified health professionals in a high dependency unit due to potential cardiac side effects.4,9 Notably, a previously cited retrospective analysis by Muñoz-Vendrell et al. found lacosamide superior to phenytoin regarding its effectiveness and side effects. 8 Additionally, some single studies indicate the usefulness of acute treatment of TN exacerbations with onabotulinum toxin A or subcutaneous sumatriptan, with minimal adverse effects observed at the doses used.4,10,11
For improved pain assessment in patients with TN, the use of standardized pain measurement scales such as the Penn Facial Pain Scale-Revised (Penn-FPS-R) and the Brief Pain Inventory-Facial (BPI-Facial) should be considered over traditional tools such as the Numerical Rating Scale (NRS) and the Visual Analog Scale (VAS).11,12 These specialized scales provide superior assessment by specifically addressing the unique characteristics of facial pain and its impact on daily functioning.
One limitation of our study is the small sample size and non-standardized reporting of pain at the emergency department records, underscoring the need for more comprehensive and prospective investigation. Larger comparative analyses and, particularly, randomized controlled trials are imperative to validate the observed results and to draw more definitive conclusions regarding the efficacy and safety of phenytoin and other drugs in the treatment of acute TN exacerbations. Additionally, it is imperative for the future to develop novel drugs that are well-tolerated, easy to administer, safe, and effective.
Conclusion
Our analysis shows that phenytoin is an effective and safe treatment option for acute TN exacerbations regardless of patient characteristics and disease etiology. Given the small sample size and retrospective nature of our study, further consideration in larger, prospective studies is required.
Highlights
Phenytoin is an effective and safe intravenous treatment option for acute exacerbations of trigeminal neuralgia.
The treatment can be used effectively regardless of trigeminal neuralgia characteristics and etiology.
Footnotes
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: SW has received research grants from Schweizerischer Nationalfond, UZH KFSP Stroke, Baugarten Stiftung, Hartmann Müller foundation, Betty and David Koetser foundation and Schweizerische Herzstiftung, as well as consulting fees (via institution for research) from Novartis, honoraria as a speaker (via institution for research) from TEVA, FOMF, ADVISIS, Springer Medizin Verlag GmbH; membership, leadership or fiduciary role in Zürcher Zentrum für Neurowissenschaften, Zürcher Neurologen Gesellschaft, Schweizerische Kopfwehgesellschaft, Schweizerische Hirnschlaggesellschaft, European Stroke Organization, Women in Neurology, Clinical Translational Neurosciences, JCBFM / ISCBFM, Schweizerischer Nationalfond, CAS Sex and Gender in Medicine, Brain Disease Foundation, Swiss Society of Neurosciences. There are no conflicts of interest in regard to this manuscript. HP has received research grants from Werner Dessauer Stiftung, support for travel from TEVA, as well as honoraria as a speaker from Lundbeck, Abbvie, Eli Lilly, TEVA and Novartis and for advisory boards from Novartis and Eli Lilly. There are no conflicts of interest in regard to this manuscript. LK und MTK report no conflict of interest.
Ethics approval and consent to participate
The coded data were collected and analysed in accordance with the approval granted by the Zurich Ethics Commission under BASEC number 2023-00755. Each included patient provided general consent to participate in a research project.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors received no financial support for publication of this article.
Universität Zürich, (grant number discount on the Open Access publication fees).
