Abstract
Abstract
Introduction
Intravenous fosphenytoin is widely used for acute exacerbation of trigeminal neuralgia, however, few studies have investigated this treatment. We aimed to examine the efficacy and side effects of initial intravenous fosphenytoin plus oral tapering of phenytoin for exacerbation of trigeminal neuralgia.
Methods
Consecutive patients with primary trigeminal neuralgia were included in this prospective observational 90-days follow-up study. Data were collected using standardized interviews before, at 24 hours, day 7, 30 and 90 post loading dose. The primary outcome was the proportion of responders defined as a 50% reduction in pain intensity 24 hours post loading dose.
Results
We included 15 patients. Nine patients (60%) were responders. Pain intensity 24 hours post loading dose was reduced by 5.00 points on the numerical rating scale (p < 0.001), and at day 7 by 5.5 points (p < 0.001). The most common side effects were hypotension and dizziness.
Conclusion
Intravenous fosphenytoin relieves trigeminal neuralgia pain in most patients and provides a window for titrating prophylactic trigeminal neuralgia medications or planning neurosurgery. The decision to administer intravenous fosphenytoin should be taken with support from trigeminal neuralgia experts and involves considerations of co-morbidities and other treatment options for acute exacerbation of trigeminal neuralgia.
Keywords
Introduction
Trigeminal neuralgia (TN) is highly debilitating due to its severe trigger-evoked pain and unpredictable nature (1,2). According to clinical experience, the natural history of TN is fluctuating in pain intensity and attack frequency. Patients can have periods of complete remission of pain as well as periods of excessive pain (3).
First-line prophylactic treatment is carbamazepine or oxcarbazepine (4). These treatments are often hampered by side effects and may not provide sufficient pain relief (3,4). In case of severe exacerbation of pain, patients with TN can become dehydrated and anorectic as drinking and eating will evoke pain (3). Approximately 30% of patients with TN followed in pain clinics are medically refractory and undergo neurosurgical treatments including microvascular decompression or ablative procedures such as balloon compression (5).
Phenytoin was the first drug used as treatment for TN and is currently a second-line prophylactic drug for TN (4). In contrast to other TN drugs, phenytoin can be administered intravenously as the prodrug fosphenytoin. Clinical experience has led to intravenous fosphenytoin becoming one of the drugs of choice for TN exacerbation, and current treatment guidelines recommend intravenous infusion of fosphenytoin or lidocaine in acute exacerbations of pain, despite the quality of evidence for use being very low (4).
To the best of our knowledge there are no prospective studies on acute treatment of patients with TN with intravenous fosphenytoin loading dose. We aimed to examine the efficacy and side effects of intravenous loading dose of fosphenytoin for the treatment of exacerbation of TN.
Methods
Study design and population
This prospective observational study was conducted at the Danish Headache Center, Rigshospitalet – Glostrup in Denmark in collaboration with the Department of Anaesthesiology, Rigshospitalet – Glostrup. We followed the usual treatment course of patients who were admitted with acute exacerbations of TN. The study did not involve any changes to routine clinical practice. The decision of administering fosphenytoin was made by the neurologist on call. All patients who were to be treated with intravenous fosphenytoin for facial pain were screened for inclusion. Intravenous fosphenytoin was considered in patients with severe exacerbation of TN pain, i.e. multiple and intense daily paroxysms severely debilitating the affected patient, and where other options for pain control had been tried out or considered. Such other options typically involved titration of current oral prophylactic treatment for TN according to international guidelines (4), add on of other oral prophylactic treatments for TN or accelerated referral to neurosurgery. Referral to neurosurgery was considered in patients willing to undergo neurosurgery and without co-morbidities that could pose a contraindication to neurosurgery. Due to a tight schedule at the neurosurgical department, it was usually not possible to plan the date for neurosurgery any earlier than 1–2 weeks after referral.
Inclusion criteria were a) diagnosis of either classic or idiopathic TN according to the diagnostic criteria of the 3rd edition of the International Classification of Headache (1), b) aged 18 years or older, c) MRI of the brain and brainstem and d) informed consent. Exclusion criteria were a) psychiatric or mental illness that interfered with the ability to give an informed consent, b) language barrier, and c) lack of compliance. Patients admitted for fosphenytoin loading dose came either directly from the neurological emergency room or from the neurological ward at Rigshospitalet – Glostrup, a large university hospital covering a population of approximately 1 million.
Fosphenytoin
Fosphenytoin is the prodrug of phenytoin, which acts by blocking voltage-gated sodium channels, (6) and thereby prolongs the refractory period of the neurons. Conversion from fosphenytoin to the active drug phenytoin has a half-life between 7–15 minutes. The maximal plasma concentration of phenytoin derived from fosphenytoin is reached approximately 30–60 minutes after intravenous loading dose (7). The half-life period for phenytoin in adults is 30–100 hours due to nonlinear pharmacokinetics (8).
Study procedures
Treatment and monitoring
Upon decision of administering intravenous fosphenytoin loading dose, treatment procedures were conducted according to local guidelines. The patient had ECG and laboratory work (liver and kidney function, electrolytes, and blood glucose) to ensure that potential contraindications were identified before infusion. Once a neurologist approved the ECG and laboratory work results, the patient was transferred to an intensive care unit for monitoring of blood pressure and heart rate due to the risk of cardiac arrythmia and hypotension during administration of the loading dose. Any event of hypotension was recorded. Hypotension was defined as: a) ≥20 mm Hg decrease in systolic blood pressure or a ≥10 mm Hg decrease in diastolic blood pressure (9), or b) a systolic blood pressure less than 90 mm Hg (10). Other side effects such as nausea, dizziness, nystagmus, loss of coordination and confusion were also recorded systematically.
Fosphenytoin was given as Pro-Epanutin (fosphenytoin sodium equivalents [PE]) relative to patient weight (20 mg PE per kilo bodyweight) diluted in isotonic saline 0.9% containing a total volume of 140 mL and given over a period of 14 minutes. During the infusion and 30–45 minutes following the infusion, the patient was continuously monitored with ECG, heart rate and oxygen saturation, and blood pressure every two minutes. If the patient was stable one-hour post loading, the patient was transferred to the neurological ward. The next day, peroral treatment of phenytoin was initiated with typically 5 mg phenytoin per kg body weight divided in two daily doses. Peroral phenytoin was slowly tapered on an individual basis and a plan for tapering was made on the day of discharge. The patients were discharged when stable on either new medication or increased doses of previous medication and when side effects were tolerable. The number of days of hospitalization were individualized.
Pain assessments
Just before administration of the loading dose the patient was asked about the average pain intensity for the last 24 hours and the last 7 days. The pain intensity was assessed using an 11-point numerical rating scale (NRS), where 0 is no pain, and 10 is the worst imaginable pain. The patient was also asked about the average daily frequency of paroxysms during the last 7 days and the last 24 hours according to predefined intervals: 0 paroxysms, 1 paroxysm, 2–5 paroxysms, 6–50 paroxysms or above 50 paroxysms per day.
To measure pain influence on activities of daily living (ADL), the patient was asked to fill out the two latter domains of the Brief Pain Inventory-Facial (BPI-Facial) covering pain interference with ADL and face-specific activities (11). The BPI-Facial uses an 11-point Likert scale, ranging from 0 to 10 for all questions. The more points, the more pain has interfered with a specific activity.
Side effects to usual TN medication were recorded before loading dose. All new side effects that occurred during and after loading dose were also recorded. During and 30 minutes after loading dose the lowest blood pressure was noted.
Follow-up
The patients were followed up 24 hours post-loading dose and at day 7, 30 and 90. At 24 hours post-loading dose a physician conducted a semi-structured-interview for information including; a) clinical characteristics and demographic data, b) history of previous and current medication and surgical treatment and their efficacy, and c) patient’s self-reported assumed cause for exacerbation of pain (e.g. physical or psychological stress, non-compliance etc.).
Before loading and at day 7, the BPI-Facial was also recorded. At day 7 the patient’s impression of change after fosphenytoin loading dose was recorded using the Patient Global Impression of Change (PGIC) scale, a validated tool used for other TN trials (12). A patient was classified as a being a PGIC responder if there was a PGIC response of “much improved” or “very much improved” at day 7. At day 7, 30 and 90 the following was recorded; a) pain intensity (NRS) and the average daily number of paroxysms, and b) current TN medication and side effects. At day 90 we further recorded whether the patient had been referred to or had undergone neurosurgery, or had received new medical treatments for TN.
Outcomes
The primary outcome was the proportion of patients classified as responders defined as a reduction in pain intensity (NRS) of ≥50% at 24 hours post loading dose compared to the 24 hours leading up to admission. The secondary outcomes were; a) the proportion of patients with a reduction in pain intensity (NRS) ≥30% at 24 hours post loading dose, b) the proportion of patients with a PGIC response of “much improved” or “very much improved” at day 7, c) change in score on BPI-Facial comparing pre-loading rating and day 7, d) difference in 50% responder rate between patients with purely paroxysmal pain compared to patients with concomitant persistent pain, and e) prevalence and severity of side effects to intravenous fosphenytoin.
Side effects
Evaluation of side effects included pre-specified adverse events during administration of loading dose including dizziness, nausea, dysarthria, blurred vision, diplopia, rash, pruritus, loss of coordination and confusion. Additionally, we reported adverse events during the days of hospitalization.
Statistical methods
The number of included patients was determined by the inclusion period. Continuous and ranked data are summarized by descriptive statistics. Categorical variables are presented with frequency distributions (N, %), reported as numbers, means or medians, percentages and with 95% confidence intervals (CI). Nonparametric testing (Wilcoxon signed-rank test) was used to determine the significance of differences in patient-reported outcomes, e.g. NRS and BPI-Facial. Missing data were considered missing at random, based on the assumption that missingness was independent of the demography, treatment, and pain level. P-values are reported as two-tailed with a level of significance of 5%. To test for associations between being a 50% responder and the variable concomitant persistent pain (yes vs. no) we used 2 × 2 contingency Fisher’s exact test. Analyses were carried out using SAS 9.4 (SAS Institute Inc., Cary, NC, USA). We used STROBE reporting guidelines as the checklist for reporting of our findings (13).
Results
Patient population
Twenty patients with TN were treated with intravenous fosphenytoin in the inclusion period between November 2018–October 2021, and off those, 15 were included in the study (Figure 1). Seven patients (47%) were males (Table 1). The mean age of TN onset was 60 years (95% CI 52.2–67.6), and the mean age at time of loading dose was 68 years (95% CI 59.4–75.8). The mean disease duration was 7.8 years. Seven patients (47%) had classical TN and eight patients (53%) had idiopathic TN. In all but one case the patients were out-patients at the Danish Headache Center and therefore diagnosed by experts in TN before the onset of the acute exacerbations. The one patient was diagnosed by a TN expert at inclusion in the study. All patients had tried oral prophylactic medications to prevent TN attacks in years and months leading up to admission.
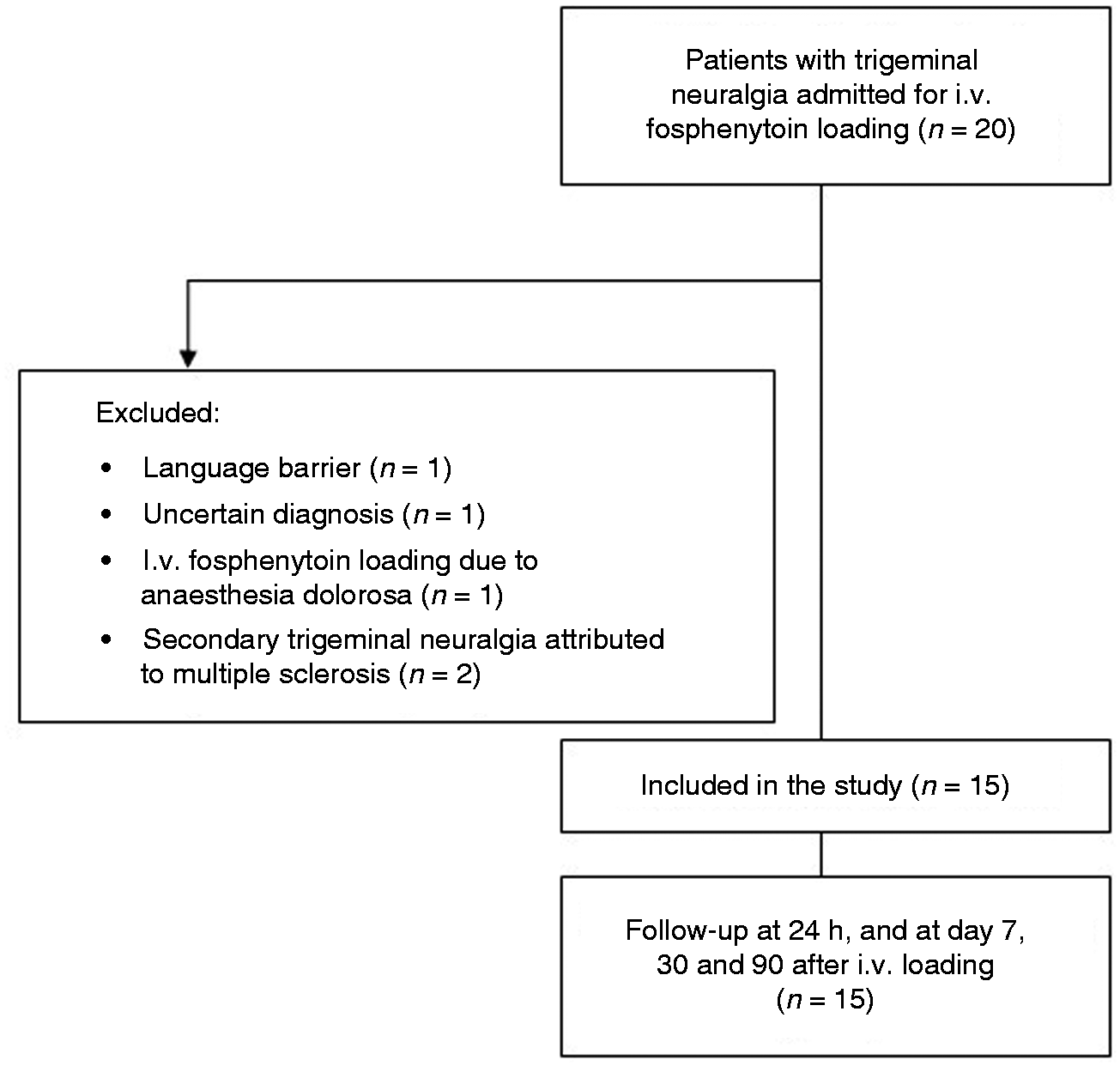
Flowchart of inclusion of patients with trigeminal neuralgia admitted for treatment with fosphenytoin loading dose.
Clinical characteristics, medication, and neurosurgical status 3 months post intravenous fosphenytoin loading dose for exacerbation of trigeminal neuralgia.
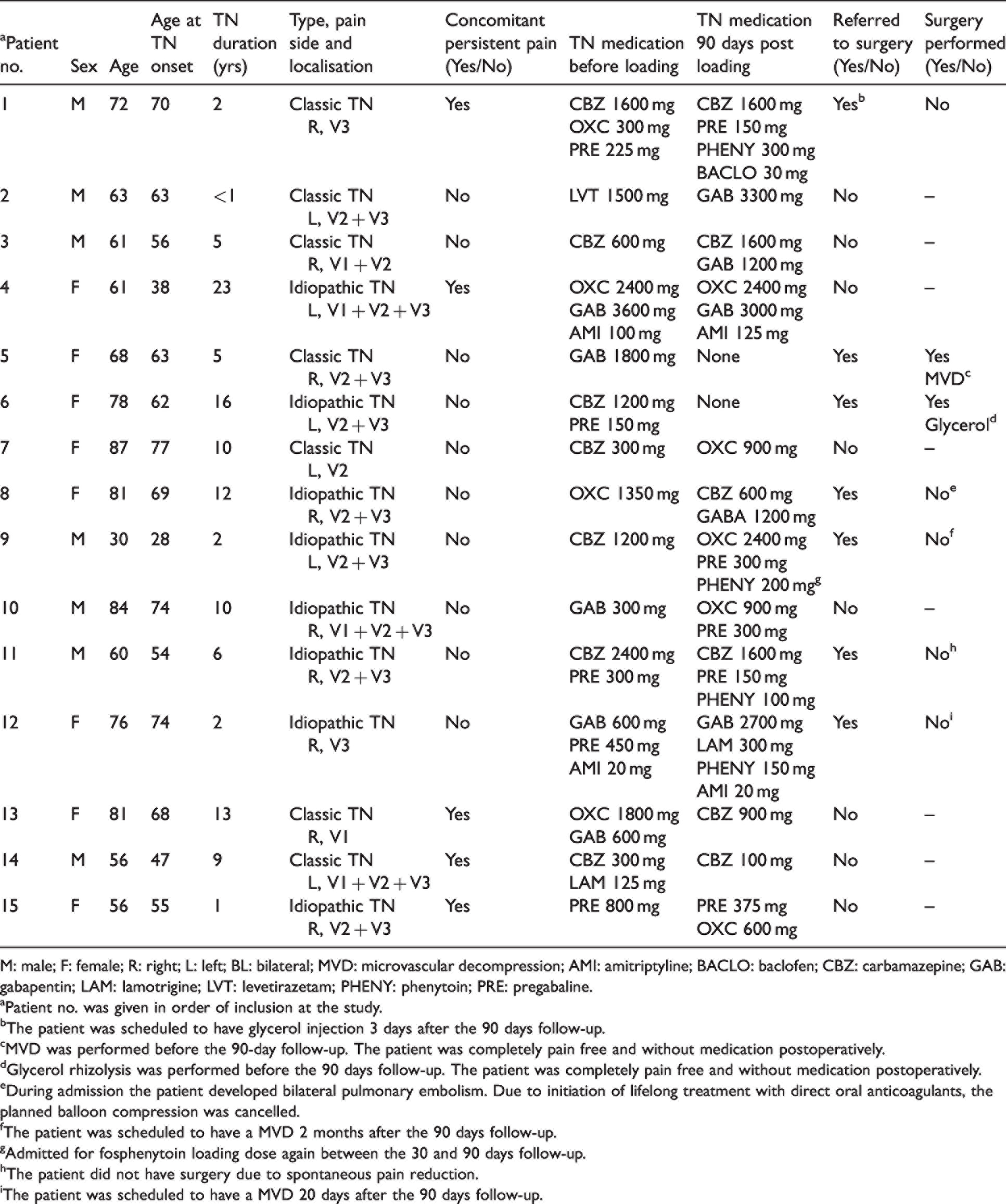
M: male; F: female; R: right; L: left; BL: bilateral; MVD: microvascular decompression; AMI: amitriptyline; BACLO: baclofen; CBZ: carbamazepine; GAB: gabapentin; LAM: lamotrigine; LVT: levetirazetam; PHENY: phenytoin; PRE: pregabaline.
aPatient no. was given in order of inclusion at the study.
bThe patient was scheduled to have glycerol injection 3 days after the 90 days follow-up.
cMVD was performed before the 90-day follow-up. The patient was completely pain free and without medication postoperatively.
dGlycerol rhizolysis was performed before the 90 days follow-up. The patient was completely pain free and without medication postoperatively.
eDuring admission the patient developed bilateral pulmonary embolism. Due to initiation of lifelong treatment with direct oral anticoagulants, the planned balloon compression was cancelled.
fThe patient was scheduled to have a MVD 2 months after the 90 days follow-up.
gAdmitted for fosphenytoin loading dose again between the 30 and 90 days follow-up.
hThe patient did not have surgery due to spontaneous pain reduction.
iThe patient was scheduled to have a MVD 20 days after the 90 days follow-up.
Primary and secondary outcomes
Nine patients (60% (95% CI 32.2–83.7%)) were responders as they had a 50% or greater reduction in pain intensity (NRS) 24 hours after loading dose (Table 2). Ten patients (67%) (95% CI 38.4–88.2) had a 30% or greater reduction in pain intensity 24 hours after loading dose. All 15 patients had pain reduction within the first hour after infusion. Twenty-four hours after loading dose the median reduction in pain intensity was 5.0 points (NRS) (from a median of 10 (range 6;10) to a median of 3 (range 0;10), p < 0.001). Seven days after loading dose the pain intensity was reduced by 5.5 points from a median of 10 to a median of 4.0, p < 0.001 (Figure 2). At day 30 and 90 the pain intensity was reduced to a median of 5.00 (p < 0.05) and 3.00 (p < 0.001), respectively, compared to before loading dose. Eleven patients (73%) were PGIC responders 7 days after loading dose.
Pain characteristics before and after intravenous fosphenytoin loading dose and treatment outcome.

aPatient no. was given in order of inclusion at the study. BPI-Facial: Brief Pain Inventory facial. PGIC score: Patient Global Impression of Change score.
bPatient no. 5 had microvascular decompression before the 90-day follow-up.
cPatient no. 6 had glycerol rhizolysis before 90-day follow-up.
dPatient no. 7 did not pick up the phone at follow-up 7 day.
ePatient no. 9 had not completed the questionnaire.
fPatient no. 10 was admitted due to infection not related to TN and was not able to answer the BPI-Facial.
gPatient no. 13 had not completed the questionnaire.

Pain intensity (NRS) before fosphenytoin loading dose, 24 hours, 7, 30, and 90 days after fosphenytoin loading dose. Time course of the numerical rating scale (NRS) scores during the 90 days following the intravenous fosphenytoin loading dose. The median NRS score before the fosphenytoin loading dose was 10. The median NRS scores (interquartile range) were 3.0, 4.0, 5.0, and 3.0 at 24 h, 7 days, 30 days, and 90 days. For all patients, the pain significantly decreased during the 24 hours following the fosphenytoin loading dose (p < 0.001) and during the following 7 days p < 0.001.
Seven days after loading dose the total BPI-Facial score was reduced from a median of 110.5 (range 47;140) to a median of 33.0 (range 0;115), p = 0.005 (Table 2). At day 30, the total BPI-Facial was a median of 49 (range 0, 121) and at day 90 the median score was 10 (range 0;103).
There was no difference in the number of responders comparing patients with purely paroxysmal pain (6/10 (60%)) vs. patients with concomitant continuous pain ((3/5 (60%)), p = 1.0). There was no difference in the number of responders in patients with classical TN (3/7 (43%)) vs. patients with idiopathic TN ((6/8 (75%), p = 0.315)). In all but one case, where the patient stated that the exacerbation may have been due to stress, the patient-reported cause of exacerbation was unknown.
Medical and surgical status at 90 days follow-up
All patients were hospitalized in the neurology ward to secure that side effects had subsided and to obtain adequate pain control. The average duration of admission was 9 days (range 1; 28). The median time of discontinuing oral phenytoin was 23 days (range 9; 123). At the 90-day follow-up, four patients were still in the process of tapering oral phenytoin. Two (13%) patients had undergone neurosurgery (Table 1). Apart from these two patients who had no subsequent need for medication for TN, all other patients had had a change in the prophylactic medication for TN. There was a variety of individual changes of medical treatment characterized by addition or titration of oxcarbazepine, carbamazepine, gabapentin, pregabalin or lamotrigine or by a change from oxcarbazepine to carbamazepine or vice versa.
Side effects after fosphenytoin loading dose
All patients had side effects in relation to fosphenytoin loading dose. The three most frequent were hypotension (13 (87%)), dizziness (11 (73%)) and nausea (9 (60%)) (Figure 3). No patients had a systolic blood pressure under 90 mm Hg (Supplementary material A). There were no cases of cardiac arrythmia.
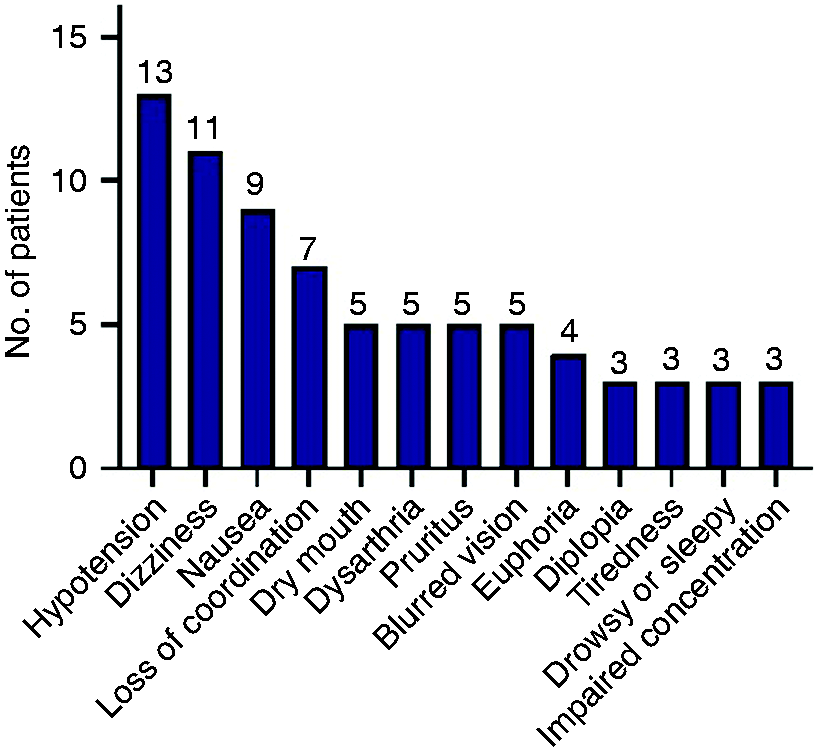
Most common side effects during the first 24 hours after fosphenytoin loading dose. In addition to the most common side effects, there were also cases of expressive aphasia (n = 2), confusion (n = 2), feeling drunk (n = 2), general weakness (n = 2), sensory disturbances (n = 2), skin rash (n = 2), tinnitus (n = 2), tremor (n = 2), auditory and visual hallucinations (n = 1), headache (n = 1), hearing loss (n = 1), memory problems (n = 1), left-sided neglect (n = 1) and nystagmus (n = 1).
The most severe case of side effects involved a 60-year-old male (patient no. 11) who shortly after initiation of infusion developed a rash and pruritus. After five minutes the patient developed motor restlessness, dizziness, loss of coordination, diplopia and dysarthria and was severely nauseated. After a 20 minutes pause of the infusion, the infusion was resumed but ultimately had to be discontinued when nearly 90% of the loading dose had been administered. During the infusion the patient was given antiemetics. Six hours later the symptoms had gradually remitted. The patient continued with oral phenytoin and had not yet tapered fully off at the day 90 follow-up.
In two other cases (patient nos. 1 and 10) the nurse monitoring the patient decided to set up a sodium chloride 0.9% intravenous infusion.
It was the collective personal experience of the nurses monitoring the patients prior to, during and just after the administration of the loading dose, that it was helpful to prepare the patient for the expected side effects that would usually set in at around 10 minutes after onset of infusion, and then gradually wear off during the minutes and hours after the infusion. When the side effects would set in, the nurse would typically talk the patients through the symptoms and repeat that the side effects were typical and not dangerous which would calm the patients down. In some cases, the patients experienced the side effects as rather frightening.
Discussion
This is the first prospective and systematic study investigating the efficacy and side effects of intravenous fosphenytoin loading followed by oral phenytoin in consecutive patients with exacerbation of TN. The main finding was that 60% of TN patients responded to intravenous fosphenytoin loading with at least 50% reduction in pain intensity 24 hours after infusion. Pain was well-controlled for the following 90 days. The immediate pain relief from intravenous fosphenytoin and the following period of oral phenytoin, provides a window of opportunity for titration of other prophylactic medications for TN or for planning neurosurgery.
Until now the use of fosphenytoin and its efficacy for treatment of exacerbation of TN has been based on limited evidence from case reports and small retrospective case series. Previous case reports include a total of five patients and all report pain relief immediately post-infusion lasting for 1–3 days on average (14–16). A retrospective study of 39 patients with TN (both classic, idiopathic and secondary TN) treated with intravenous phenytoin infusion found that 89% of patients achieved pain relief of ≥50% immediately after infusion (17). Similar results were found in another retrospective observational study of 20 patients with classical TN (18). Common for all of the above-mentioned studies is that they are limited by a short follow-up period and great variability in the administration of fosphenytoin, both regarding loading dose and infusion time. Furthermore the case reports did not use any diagnostic criteria to secure correct inclusion of patients (14–16).
Pain relief after fosphenytoin loading dose
We show that the significant pain reduction following treatment with intravenous fosphenytoin and oral phenytoin is also reflected in improvements in the patient-related outcome measures. The long-term pain relief and improvement in patient-related outcome measures at the 30- and 90-day follow-up may be mediated by oral phenytoin, titration of other prophylactic medications, neurosurgical treatment, and regression to the mean. The study reflects that patients with TN that are severely affected need expert care for not only the acute treatment but also for installing a long-term plan for prophylactic medication and neurosurgical referral. We demonstrate that patients with TN when admitted due to severe pain and when treated with intravenous fosphenytoin together with close follow-up with optimization of medication have a favorable 90 days prognosis. With the caveat that this is a small numbers study, there was the same number of responders independent of etiology (classical vs. idiopathic TN) and pain phenotype (purely paroxysmal pain vs. concomitant continuous pain).
Pain relief after other rescue treatments
The evidence for other treatments of acute exacerbations of TN is weak (4,19). Several small randomized double-blind placebo-controlled cross-over trials have investigated whether lidocaine is effective. Lidocaine infusions (20), intranasal lidocaine 8% spray (21) and intraoral application of a metered dose-pump of 8% lidocaine (22) was reported to be superior to placebo. Similar effects have been described with the use of subcutaneous injection of sumatriptan in a randomized controlled cross-over double-blinded trial (23). The limitations of the rescue medications administered either intranasally, intraorally or subcutaneously are their short-term effect (2.8–7.9 hours), which require repetitive use to maintain pain relief (21–23). In the study of intravenous lidocaine, a single dose of 5 mg/kg was given and the treatment effect lasted for 24 hours (20). The advantage of fosphenytoin over the other drugs is the opportunity to taper off with oral phenytoin, providing the option of longer lasting effect.
Future studies on treatment of acute exacerbations in patients with TN should explore whether oral loading with phenytoin could have a similar effect as seen with an intravenous loading dose and with fewer side effects. Another perspective could be to consider an accelerated titration of standard TN medication in admitted TN patients.
Side effects after intravenous fosphenytoin loading dose
A retrospective cohort study with 39 TN patients who received a total of 65 phenytoin infusions reported side effects in 10 infusions (15.4%) out of which the most frequent were nystagmus, dysarthria, ataxia, hypotension (not specifically defined) and drowsiness. In 51 of the infusions the dosage was 10 mg/kg body weight (17), which is half the dose we used. A retrospective study of 164 patients with status epilepticus receiving intravenous fosphenytoin loading dose in the emergency department reported an incidence of hypotension of 5%, when defined as systolic blood pressure less than 90 mm Hg. There were no cases of cardiac arrythmia (10). Phenytoin doses were, however, not comparable to our study as the average loading dose was 775 mg (range 50–1000 mg) (10), as opposed to this study where the average loading dose was 1400 mg (range 800–2000 mg).
Strengths and limitations
The study is a single-centre experience and multicentre studies are warranted. Placebo-controlled verification of effects of fosphenytoin to treat TN exacerbations would be optimal, but we considered a placebo-controlled study unethical because of the severe pain associated with an exacerbation of TN.
Conclusions
Intravenous loading with fosphenytoin is efficacious in more than half of the patients with acute exacerbation of TN offering a window of opportunity for titration of other prophylactic medication for TN and referral to neurosurgery. Side effects to intravenous fosphenytoin are common but transient and include hypotension and dizziness. Ideally, the final decision to offer administration of intravenous loading dose with fosphenytoin should be taken with help of TN experts, and should involve considerations of co-morbidities and other viable treatment options for acute exacerbation of TN. Before discharge, a plan for tapering of oral phenytoin and titration of other prophylactic TN medication or referral to neurosurgery should be in place and follow-up should be offered.
Clinical implications
Intravenous fosphenytoin loading reduces pain intensity by at least 50% in 60% of the patients when measured 24 hours post infusion. Side effects are common and include hypotension and dizziness. Intravenous fosphenytoin loading provides a window for titration of other prophylactic medication for TN or planning neurosurgery. Follow-up by TN experts after discharge is mandatory.
Supplemental Material
sj-pdf-1-cep-10.1177_03331024221094536 - Supplemental material for Intravenous fosphenytoin as treatment for acute exacerbation of trigeminal neuralgia: A prospective systematic study of 15 patients
Supplemental material, sj-pdf-1-cep-10.1177_03331024221094536 for Intravenous fosphenytoin as treatment for acute exacerbation of trigeminal neuralgia: A prospective systematic study of 15 patients by Anne Sofie Schott Andersen, Tone Bruvik Heinskou, Mohammad Sohail Asghar, Birgitte Rossen, Navid Noory, Emil Andonov Smilkov, Lars Bendtsen and Stine Maarbjerg in Cephalalgia
Footnotes
Abbreviations
ADL: activities of daily living; BPI-Facial: Brief Pain Inventory-Facial; CI: confidence interval; DHC: Danish Headache Center; NRS: Numeric Rating Scale; PGIC scale: Patient Global Impression of Change scale; TN: trigeminal neuralgia
Acknowledgements
The authors thank the nursing staff at the department of anaesthesiology for their valuable help with this project.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval and patient consent
The study was registered at clinical.trials.gov, where only the primary outcome was preregistered (ClinicalTrials.gov Identifier: NCT03712254). The study was conducted in accordance with the guidelines of the declaration of Helsinki (![]() ). The Danish National Committee on Health Research Ethics confirmed that this project, ID nr. H-18023737, did not need ethical approval, as it was observational and based on routine clinical care and laboratory workup and therefore not defined as a scientific research project. All patients gave their written informed consent to participate in the study, and collection, archiving, and transmission of personal data was conducted according to the regulations of the Danish Data Protection Agency.
). The Danish National Committee on Health Research Ethics confirmed that this project, ID nr. H-18023737, did not need ethical approval, as it was observational and based on routine clinical care and laboratory workup and therefore not defined as a scientific research project. All patients gave their written informed consent to participate in the study, and collection, archiving, and transmission of personal data was conducted according to the regulations of the Danish Data Protection Agency.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the patient society Trigeminus Foreningen. The funding sources had no role in the study.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
