Abstract
Introduction
Acute trigeminal neuralgia exacerbation is a common reason for frequent emergency department visits, that often occurs while waiting for surgery, but evidence on effective drugs for acute trigeminal neuralgia is scant. Whether lidocaine aerosol could be a rescue option for the treatment of acute trigeminal neuralgia exacerbations is worth exploring. Positive predictors of the analgesic effects of lidocaine aerosol also warrant further investigation.
Methods
This is a retrospective study with a total of 152 patients. We analyzed the efficacy of lidocaine aerosol for the treatment of acute trigeminal neuralgia exacerbations. A positive response was considered a decrease in the VAS score of at least 50% at 30 min of treatment. Multivariable logistic analyses were performed to identify predictive factors for lidocaine aerosol response.
Results
In the group of 109 responders, the VAS score decreased from 8.3 ± 1.1 cm to 0.8 ± 1.0 cm at 15 min, and 1.7 ± 1.0 cm at 30 min. The effective rate at 15 min and 30 min were 77.6% and 70.4%, respectively. Multivariate logistic analyses showed the treatment may provide better clinical outcomes in V2 trigeminal neuralgia (OR 0.01, 95%Cl 0.001–0.15, p < 0.001), V3 trigeminal neuralgia (OR 0.02, 95%Cl 0.001–0.16, p = 0.001), and V2 + V3 trigeminal neuralgia (OR 0.01, 95%Cl 0.001–0.13, p < 0.001), patients who were taking carbamazepine or oxcarbazepine with a maximum dose (OR 6.15, 95%Cl 2.11–17.93, p = 0.001) were less likely to experience immediate pain relief.
Conclusion
Lidocaine aerosol sprayed on oral and/or nasal mucosa is beneficial for immediate pain relief in patients with acute trigeminal neuralgia exacerbations. It is expected to become a promising treatment option for patients with V2 and/or V3 trigeminal neuralgia.
Keywords
Introduction
Trigeminal neuralgia (TN) is characterized by recurrent severe shooting neuropathic pain in the territory of one or more divisions of the trigeminal nerve, which is triggered by innocuous stimuli such as touch, chewing, talking, and brushing teeth (1,2). In case of severe exacerbation of TN, patients can become dehydrated and anorexic as the intake of fluids and food can evoke severe pain (3). Recurrent TN exacerbations can impair daily activities and lead to depression, thus it is essential to control severe pain from the very first hour in patients with acute exacerbations of TN.
Acute TN exacerbation is a common reason for frequent visits to the emergency department, that often occur while waiting for surgery, but evidence on effective drugs in acute TN attacks is scant and no option is considered the gold standard up to now. Treatment options such as lidocaine (topical or systemic application), anticonvulsant (intravenous infusion), phenytoin or fosphenytoin (intravenous infusion), serotonin agonist (subcutaneous injection), sumatriptan (subcutaneous injection) and botulinum toxin (trigger point injection) are beneficial for acute exacerbations of TN (4–6), but evidence-based studies are scarce and future investigations should report on outcomes within 24h to improve knowledge of the acute TN exacerbation treatments.
Lidocaine is a use-dependent sodium channel blocker. By blocking sodium channels, lidocaine stabilizes nerve membranes, delays nerve depolarization, and reduces ectopic discharges, which results in local analgesia (7,8). It has been reported that trigeminal nerve block (9), trigger point injection (10), and intravenous infusion with lidocaine (11) achieves immediate pain relief in acute TN patients. The aforementioned options require image guidance, skilled staff, operating theatre access, or all three, which limit their utility as a rescue option in TN exacerbation. Other lidocaine preparations have also been studied, including eye drops (12) and lidocaine meditated plaster (13,14). However, given the small number of cases investigated and the lack of randomized controlled trials (RCTs) with a larger sample size, it is therefore difficult to judge the validity of these options for TN exacerbations. An ideal treatment for TN exacerbations would be one that achieves complete and rapid analgesia, as much as possible, without any severe complications and could be easily administered. Lidocaine spray is an effective anesthetic of mucous membranes. The analgesic effect of this spray occurs within five minutes and lasts for 10–15 minutes (15). Studies have shown that 8% lidocaine spray applied on nasal or oral mucosa produces prompt analgesia without serious adverse reactions in patients with TN of second and third division (16,17). However, the number of cases were small and lidocaine spray was applied on patients’ oral mucosa with the fingers instead of spraying directly, which made it difficult to quantify the exact dose of lidocaine. So far, the effect of lidocaine sprayed directly on the oral and/or nasal mucosa for TN exacerbation has not been reported. In the present study, we retrospectively analyzed the efficacy of intraoral and/or intranasal 2.4% lidocaine aerosol to explore a safe, fast-acting, and convenient option for the relief of acute TN exacerbations.
Methods
Patients
This is a single-center, consecutive, retrospective, observational study. After approval from the Institutional Review Board of Beijing Tiantan Hospital, Capital Medical University, Beijing, China, the medical records of patients who received lidocaine aerosol sprayed on oral and/or nasal mucosa as treatment for TN exacerbations in the period between April 2020 and October 2022 were collected retrospectively from the hospital information systems (HIS).
The inclusion criteria for the study were as follows: 1) Patients who were over 18 years of age. 2) Patients referred to our clinic with the diagnosis of TN based on the International Classification of Headache Disorders 3rd Edition (ICHD-3) (18). 3) Patients who had ongoing severe TN pain with a pain intensity of more than 7 points on the visual analogue scale (VAS). 4) Patients who received treatment with lidocaine aerosol spray on oral and/or nasal mucosa for TN exacerbations. Patients with secondary TN or with missing data were excluded from the study.
Interventions
When receiving the aerosol, patients were asked to be in a sitting position with the neck extended and head slightly back to spray the aerosol on patients’ nasal mucosa, and mouths opened to spray the aerosol on patients’ oral mucosa. Physicians sprayed patients with two presses of 2.4% lidocaine aerosol (Likuaituo, Guangzhou Xiangxue Pharmaceutical Co., Ltd, Guangzhou, China), each spray lasted for one second (16 mg of one spray), on each specific area according to different affected division of the trigeminal nerve. The aerosol was sprayed in the affected nostril for pure V1 TN, V2 TN and V1 + V2 TN, on the painful mouth area and the perforation point of inferior alveolar nerve and lingual nerve block for V3 TN, and on both oral and nasal mucosa for V2 + V3 TN and V1 + V2 + V3 TN. We instructed patients who obtained effective pain relief to apply the prescribed lidocaine aerosol by themselves if recurrent TN exacerbations occurred, a maximum of up to 30 sprays per day was allowed (19).
Data recording and collection
Baseline variables collected for each patient were: age, gender, painful side, affected division of trigeminal nerve, location of trigger points, duration of TN, lidocaine aerosol treatment area, previous surgical treatment, ongoing medical treatment, the number of patients who were on carbamazepine or oxcarbazepine with a maximum daily dose of 1600 mg and 2400 mg respectively (20,21), VAS score at entry and clinical presentation (paroxysmal or continuous pain).
The clinical efficacy and adverse events on the day when patients received lidocaine aerosol recorded in the hospital information system (HIS) were collected. Patients were followed up after treatment due to the need for medical quality control. Routine follow-up at seven days after the treatment was done through telephone interviews or when patients revisited the clinic, in order to note whether the pain had returned, how long it had recurred after one application, and the efficacy of repeated applications within seven days. Pain intensity was assessed at pre-treatment, 15 min, and 30 min after the treatment on a VAS consisting of a nongraduated 10 cm horizontal line ranging from ‘0 = no pain at all’ to ‘10 = worst pain imaginable’. Response was defined as a reduction of at least 50% in the VAS score (22). Time required for a positive change were reported by patients as onset time. The effective rate at 15 min and 30 min after treatment were calculated as follows: the number of responders/total number of patients * 100%. The number of patients who had TN exacerbation relapse within seven days and the effectiveness of repeated applications were collected. Independent factors were screened from the factors that might have affected the clinical effects of TN exacerbation treated by lidocaine aerosol.
Physical examination, including measurement of blood pressure and pulse rate were conducted pre-treatment and 30 mins after the treatment. All possible adverse events including local irritation (numbness and bitterness, burning, stinging), systematic complications (palpitation, heartburn, headache), anaphylactoid reactions and other discomforts (23,24), and duration to take effect were also collected.
Statistical analysis
This work represents a descriptive analysis of our sample population. According to patients’ response to lidocaine aerosol at 30 min after treatment, patients were further divided into two groups: effective group and ineffective group. Normally distributed variables were presented as mean ± standard deviation and compared by independent t-test. Non-normally distributed variables were reported as median and ranges and compared by Mann-Whitney U test. Categorical variables were presented using absolute frequencies and their respective proportions and compared by Pearson’ s χ2 or Fisher’ s exact tests (when the expected values were <5). All tests were 2-tailed and the baseline variables with P < 0.2 were included as potential predictors for initial efficacy of lidocaine aerosol in preliminary screening. Multivariate logistic regression analyses were then conducted to examine the predictors for immediate efficacy of lidocaine aerosol for patients with TN. We identified the final multivariate model for using a backwards stepwise approach with P < 0.05 of the likelihood ratio test for exclusion of excess factors. Odds ratios (ORs) and their 95% confidence intervals (CI) were calculated separately. Data were analyzed using the SPSS version 25.0 (SPSS Inc., Chicago, IL, USA).
Results
Patient characteristics
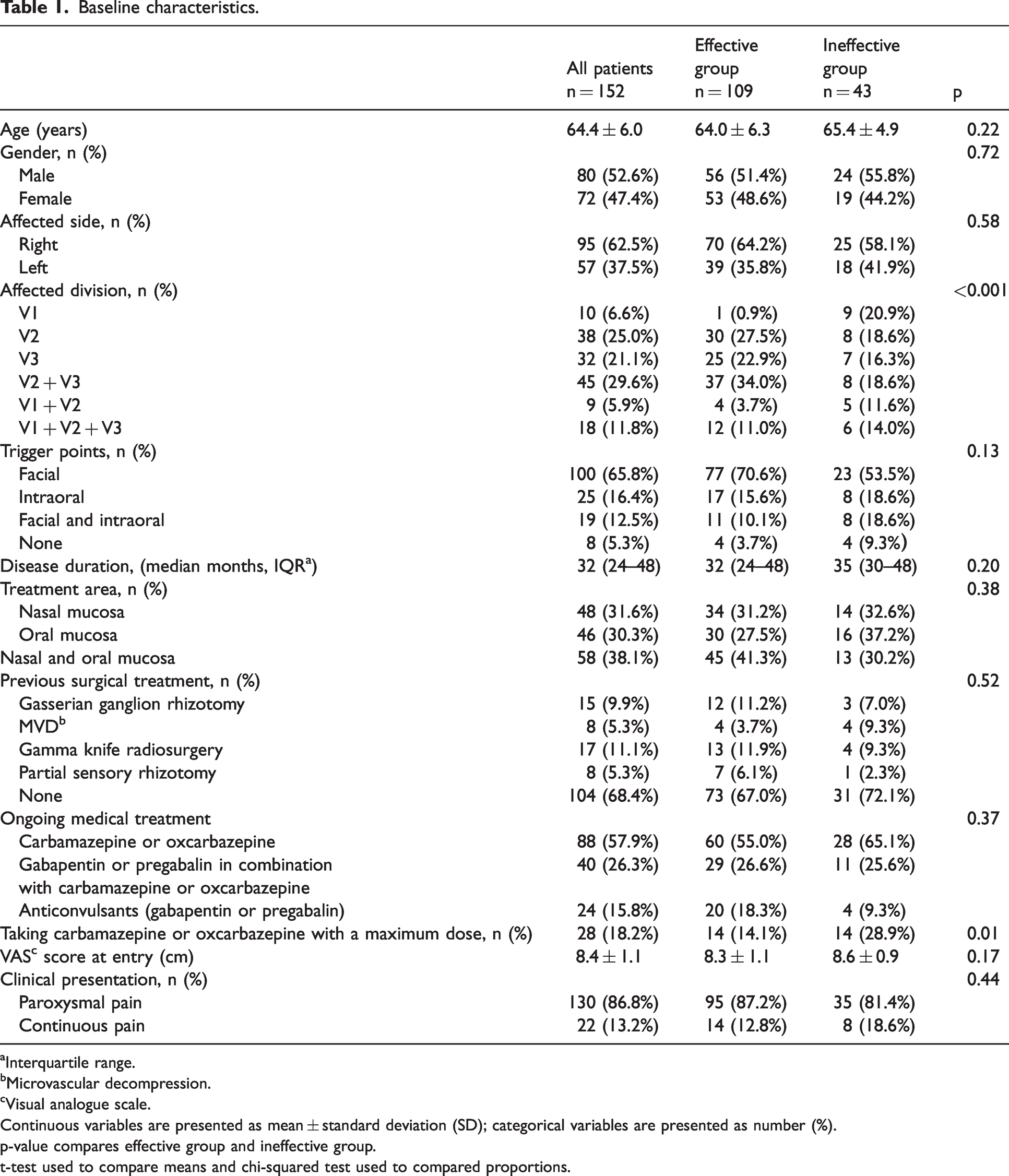
Medical records of 157 patients who met the inclusion criteria of the present study were reviewed. Five participants with secondary TN were excluded, 152 patients were included in this review (Figure 1). The overall condition of each patient was observed for at least 30 min and recorded after lidocaine aerosol was sprayed on their nasal and/or oral mucosa. The baseline characteristics of the participants are presented in Table 1.

Flowchart of inclusion of patients with TN admitted for treatment with lidocaine aerosol.
Baseline characteristics.
Interquartile range.
Microvascular decompression.
Visual analogue scale.
Continuous variables are presented as mean ± standard deviation (SD); categorical variables are presented as number (%).
p-value compares effective group and ineffective group.
t-test used to compare means and chi-squared test used to compared proportions.
The mean age at entry was 64.4 ± 6.0. Of the patients, 53.3% were male (n = 81), and 46.7% (n = 71) were female. The duration of trigeminal neuralgia ranged from six to 72 months with a median duration of 32 months. Among the 152 patients who were administered the lidocaine aerosol sprays, 53 patients sprayed on nasal cavity mucosa, and 46 patients sprayed on the oral mucosa, and 53 patients sprayed on both oral and nasal mucosa. All patients had previously received treatment with carbamazepine or oxcarbazepine. Ninety of these patients were on carbamazepine or oxcarbazepine only. Forty-one patients were being treated with gabapentin or pregabalin in combination with carbamazepine or oxcarbazepine. Twenty-one patients had to discontinue carbamazepine because of allergy or other severe side effects, and had been using gabapentin or pregabalin only. Among the 131 patients who were on carbamazepine or oxcarbazepine before the treatment of lidocaine aerosol, 28 were taking the maximum dose of the respective medicine (15 patients in the effective group and 13 in the ineffective group). The mean pain intensity at baseline was 8.4 ± 1.1 cm of VAS score when patients had ongoing TN exacerbations, despite previous medical or surgical treatments in all patients.
Visual analogue scale and effective rate
In the effective group, lidocaine sprayed on oral and/or nasal mucosa of patients significantly decreased the average VAS score from 8.3 ± 1.1 cm to 0.8 ± 1.0 cm at 15 min, and 1.7 ± 1.0 cm at 30 min after application, with the average onset time of 20.1 ± 5.7s. Based on patients’ feedback, the analgesic effect of one single treatment sustained for 43 (34,50) mins. The effective rate at 15 min and 30 min after treatment were 77.6% and 70.4%. In 102 (71.3%) out of 143 patients with trigger points before the treatment, the pain could not be triggered by touching or moving the face or the VAS score still decreased more than 50% compared to baseline. The change of VAS in all patients at 30 min is described in Table 2.
VAS change at 30 minutes.

Among 109 patients in the effective group, early pain relapse (within two hours) occurred in all patients, 105 (96.3%) of whom reported that the treatment efficacy of a repeated spray of lidocaine aerosol was as effective as the first application during the seven days after the first treatment. Four patients did not spray the lidocaine aerosol repetitively due to its bitter taste. Two patients obtained satisfactory pain relief only after multiple spray of lidocaine aerosol combined with oral medication. Fifty-five patients received percutaneous balloon compression or microvascular decompression (MVD) within seven days and the other 50 patients waited for further surgical interventions with lidocaine aerosol as a rescue option when recurrent TN exacerbations occurred.
Logistic regression analysis
Univariate statistical analyses showed that the patient’s age, gender, side of pain, disease duration, TN exacerbation duration, treatment area, pain presentation and history of previous neurosurgery were not significantly associated with the efficacy of mucosa lidocaine aerosol (P > 0.2, Table 1). Baseline variables shown in Table 1 with P < 0.2 were entered into multivariate logistic regression analyses, including location of trigger points (χ2 test, P = 0.09), affected division (χ2 test, P < 0.001), taking carbamazepine or oxcarbazepine with a maximum dose (χ2 test, P = 0.04) and baseline VAS score (χ2 test, P = 0.17). Multivariate logistic analyses showed that patients who were previously taking the maximum dose of carbamazepine or oxcarbazepine (OR 6.15, 95%Cl 2.11–17.93, p = 0.001) were less likely to experience immediate pain relief with lidocaine aerosol. Mucosa lidocaine aerosol may have better clinical outcomes in V2 TN (OR 0.01, 95%Cl 0.001–0.15, p < 0.001), V3 TN (OR 0.02, 95%Cl 0.001–0.16, p = 0.001), and V2 + V3 TN (OR 0.01, 95%Cl 0.001–0.13, p < 0.001) (Table 3).
Multivariate logistic analyses of lidocaine aerosol for patients with acute TN exacerbations.

p-value compares effective group and ineffective group.
Adverse effects
None of the patients experienced any serious side effects. No substantial changes in blood pressure or heart rate, no systematic adverse event or anaphylactoid reactions were reported in any patient. All side effects were limited to local irritation. In total, 100% patients reported numbness and bitterness in the area of application. Burning and stinging were the second most common adverse effect, reported in five (3.3%) patients. All the above adverse events were mild and disappeared without any medical intervention within a few hours.
Discussion
The findings of this study indicate that over 70% of patients treated with intraoral and/or intranasal lidocaine 2.4% spray reported a 50% or more reduction in pain intensity without any serious side effect for acute TN exacerbations. This rate is comparable with that reported for intravenous lacosamide and phenytoin (25), intravenous lidocaine (5,26) and lidocaine nerve block (9). To our knowledge, this is the first study that reports the clinical efficacy and safety of lidocaine sprayed directly to the oral and/or nasal mucosa for TN exacerbation, and the number of cases was the largest to this date. Besides, this is the first study that investigates the predictive factors of lidocaine aerosol sprayed on the oral and/or nasal mucosa in TN patients with all three divisions to date.
Our study found that TN caused by the second and/or the third division of trigeminal nerve were independent predictors for analgesic efficacy of lidocaine aerosol. Previous reports of 8% lidocaine spray applied on either nasal or oral mucosa for V2 and/or V3 TN management had shown promising results (16,17). The findings of the present study with the treatment were in accordance with previous studies, but we used lidocaine aerosol with a lower concentration of 2.4%. Unlike other studies which used a single administration of lidocaine spray for a single episode of acute TN exacerbation, we prescribed lidocaine aerosol for responders to apply by themselves and found that repeated administrations showed the same effect as the first. In our study, the application of mucosa lidocaine aerosol was more likely to provide analgesia for V2 and/or V3 TN. Furthermore, different from previous studies which only examined the response to triggered episodes, we found that this treatment was also effective for continuous pain.
In the present study, 43 (28.3%) patients did not respond significantly to treatment at 30 mins. Multivariate logistic analyses showed that patients who were taking carbamazepine or oxcarbazepine with a maximum dose before (OR 6.15, 95%Cl 2.11–17.93, p = 0.001) tend to be refractory to our treatment. One possible reason is that lidocaine is a non-selective, voltage-gated sodium channel inhibitor, affecting both the generation and the conduction of nerve impulses; anticonvulsant drugs such as carbamazepine and oxcarbazepine also block sodium channels. If all the binding sites of Na+ channel are occupied by anticonvulsant, the highly active aromatic group of lidocaine cannot be suitably coordinated, and lidocaine can only bind to Na+ channels with tertiary amine chain of low affinity, thus leading to the ineffectiveness of the treatment (27).
Martini et al. (28), in an observational study, concluded that the use of lidocaine medicated plaster (LMP) may be useful for TN patients who were poorly responsive to pharmacotherapy. However, the plaster could be attached to the face only, and owing to the high muscle activity and movement, plaster attachment may be difficult. Besides, the plaster itself may also be a kind of stimuli which could trigger paroxysmal pain. A recent study reported the analgesic effect of 5% LMP for TN patients with the efficacy rate of 21% and patients with facial trigger points are more likely to benefit from LMP treatment (29). While in our study, lidocaine aerosol was sprayed directly to patients’ mucosa without the need to touch the face, and the penetration of drug through mucosa is better than transdermal route, thus contributing to a higher effective rate in patients with and without facial trigger points. Several studies supporting the efficacy of intravenous lidocaine (22,26) and nerve block with lidocaine (9) for the reduction of pain in TN exacerbations have been published. Different from these treatments, with mucosa lidocaine, the peak plasma levels are low and the delivery of the drug is painless and non-invasive, thus reducing possible side effects. Besides, intravenous infusion and nerve block should only be performed under specialist supervision because of the technical skill required and the need of hospital admission and cardiac monitoring, which limits their use as a rescue option in patients’ daily life.
The findings of our study indicates that lidocaine aerosol sprayed on oral and/or nasal mucosa can relive the pain of acute TN exacerbations in a very short period of time (20.1 ± 5.7s). We believe that the rapid analgesic effect of this treatment is due to the composition of the lidocaine aerosol solution. The lidocaine aerosol used in our study contains dimethyl ether as both solvent and propellant, which leads to greater cell membrane penetration of the active ingredients and thereby an increased bioavailability (30). A small number of alcohols accelerate the absorption of drugs into the mucosa, thus assisting better penetration of the therapeutic drug, lidocaine. Furthermore, we chose lidocaine in the alkaline solution; studies have shown that the rate of penetration of lidocaine would be faster at a higher pH level, as less lidocaine is in the ionized form (31), leading to increased effectiveness, prolonged action time, and a reduction in local irritation (32). Another advantage of lidocaine aerosol is convenience. It can be applied in patients visiting the emergency apartment, waiting for surgery, and other situations as a rescue technique for acute trigeminal neuralgia exacerbations.
No patient in our study reported of experiencing systemic side effects related to lidocaine aerosol treatment. Systematic effects of intravenous lidocaine include central nervous system (CNS) and cardiovascular symptoms (33), but they are very uncommon for lidocaine aerosol because of minimal systemic absorption, which falls far below the threshold required for cardiac or CNS complications (34). The maximum safe dosage of lidocaine sprayed on airway mucosa has generally been considered to be 9 mg/kg (35), in our study, the dose of lidocaine aerosol used for TN was no more than four sprays (consisting of 16 mg lidocaine per spray) for one single attack of TN exacerbation, which was well below the toxic dosage of lidocaine. The primary safety concern associated with the use of topical lidocaine is the risk of systemic toxicity, particularly in the cardiovascular system. It is important to note that topical lidocaine should be used with caution in patients receiving class I antiarrhythmic agents because the effects may be additive and may result in toxicity (36). In-vitro studies have shown that both cytochrome P450 3A4 and 1A2 isoenzymes (CYP3A4 and CYP1A2) are important in the metabolism of lidocaine (37). Clinicians should be aware of the potential increase in toxicity of lidocaine when used with the combination of drugs which inhibit CYP1A2 or CYP3A4 enzymes such as statins (CYP3A4 inhibitor), fluvoxamine (CYP1A2 inhibitor) and erythromycin (CYP3A4 inhibitor) (38,39). Despite the ease of use, rapid-onset pain relief and good tolerability, numbness and bitter taste at the application sites were unavoidable, localized irritation was noted in a small number of patients, though reversible. Therefore, future efforts should focus on reducing local irritation and improving the taste of lidocaine aerosol to achieve high comfort and satisfaction among patients, thus increasing security and compliance for long-term and repeated applications.
Criticisms of this study include the limitations of a retrospective study without a control group and the short follow-up duration. Moreover, the only subjects included in our study were patients with serious TN conditions waiting for interventional or surgical treatments. In the future, we intend to perform a prospective observational study including all consecutive patients under the treatment of lidocaine aerosol spray with a clear description on how they were diagnosed and with a follow-up period of seven days or longer, for a more clinically relevant result. Besides, prospective randomized controlled studies with a long-term period of follow-up may clarify the clinical efficacy of lidocaine aerosol in acute TN exacerbations and help identify factors for clinical success. Additionally, considering that lidocaine aerosol contains other agents, we cannot exclude the analgesic effects of other components of the liquid spray (40). Another drawback is the bitter taste of the lidocaine aerosol, which leads to poor user experience, thus hindering the drug from becoming a long-term management tool for TN. Future efforts should focus on exploring the clinical efficacy and feasibility of lidocaine aerosol combined with carbamazepine or oxcarbazepine, or other medical treatment options to minimize adverse drug reactions and improve patient compliance for rescue of acute TN exacerbations in the long term.
Conclusion
Lidocaine aerosol sprayed on oral and/or nasal mucosa is likely to be beneficial for immediate pain relief in patients with acute TN exacerbations, which has an easy application technique and rarely has any complications. It is expected to become a promising treatment option for patients with V2 and/or V3 TN. Based on the results of this study, further randomized prospective comparative trials with an appropriate sample size are required to assess the analgesic effect of lidocaine aerosol, to determine the optimal dose and the most suitable site for application to achieve complete analgesia and provide more beneficial, long term treatment options for the rescue of acute TN exacerbations.
Public health relevance
Effect of mucosa lidocaiine for the rescue of acute trigeminal neuralgia exacerbations. Positive predictors of the analgesic effects of lidocaine aerosol.
Footnotes
Contribution statement
FL conceived the idea for this study and secured funding as well as doing the literature search plan and is responsible for the study design. Study conduction and data collection was led by YS and CMZ. Study analysis was done by XJZ, supervised by FL and YS. All authors were involved in data interpretation. Writing of the paper was led by FL and XJZ with assistance from CMZ.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by The Capital’s Funds for Health Improvement and Research (2020-2-2046). The sponsor had no role in the trial design, trial conduct, data handling, data analysis or writing and publication of the manuscript. All authors read and approved the final manuscript.
