Abstract
Objective:
To determine the rate of wearing-off effects of monthly and quarterly dosing of fremanezumab based on real-world data.
Methods:
We conducted an observational, single-center cohort study including 101 high-frequency episodic migraine or chronic migraine patients receiving fremanezumab over a 9-month observation period. Weekly migraine days (WMDs) were assessed by headache diaries. Regarding monthly dosing, comparisons of WMDs were performed between week (W) 1–2 and W3–4 and between W2 and W4 in months 3, 6 and 9. Regarding quarterly dosing, comparisons of WMDs were performed between W1–2 and W11–12 and between W2 and W12 in the first, second and third quarters. Wearing-off effects were defined as a >1 day increase in WMDs at any of the 3 time points.
Results:
There was no increase in mean WMDs at the end or the second half of the week in the monthly and quarterly dosing groups over 9 months. Wearing-off effects occurred in 6.7–11.7% of the 60 patients in the monthly dosing group and 9.8% of the 41 patients in the quarterly dosing group.
Conclusion:
Our findings of a low incidence of wearing-off effects support the sustainability of the effect of fremanezumab as headache prophylaxis.
Introduction
Fremanezumab, a humanized, IgG2 monoclonal antibody directed against calcitonin gene-related peptide (CGRP), has been approved for preventive treatment for migraine and is supplied with two dosage options: monthly (225 mg) and quarterly (675 mg). 1 Several randomized controlled trials 2 and real-world studies 3,4 of patients with episodic migraine (EM) and chronic migraine (CM) have shown the efficacy of fremanezumab monthly and quarterly for periods ranging from 3 to 12 months. In contrast, wearing-off effects, defined as the recurrence or worsening of clinical symptoms before the next dose and improvement after the next dose, can be observed in various drugs with different half-lives. Therefore, it is of interest to understand whether wearing-off effects with the recently available CGRP monoclonal antibodies (mAb) can be observed, and several studies have been conducted to analyze this topic. 5,6 Post hoc analysis of a 12-month randomized controlled trial showed no significant wearing-off effects in either the monthly or quarterly dosing for fremanezumab in patients with EM or CM. 5 However, data from real-world studies are still limited. The purpose of this study was to determine the rate of wearing-off effects with monthly or quarterly dosing of fremanezumab based on real-world data.
Methods
This was a retrospective, observational, single-center cohort study with a 9-month observation period. The patients in this study constituted a subgroup of the patients in a previous study 7 who received additional follow-up. This study was approved by the Institutional Review Board of Dokkyo Medical University Hospital. All participating patients were received information about this study and had the opportunity to opt out of participation in the study. Our institutional review board waived the written informed consent requirement for patients based on this retrospective, observational study setting.
Among the patients attending our outpatient headache clinic from October 2022 to January 2023, 113 patients with EM or CM aged 18 years and older received at least one dose of fremanezumab. Of the 113 patients, 101 high-frequency EM (HFEM) or CM patients with 3-month follow-up data available were finally included in the analysis. The study flowchart is shown in Figure 1. Migraine was diagnosed according to the International Classification of Headache Disorders, 3rd edition. CM was defined as headaches that occurred at least 15 days per month over a 3-month period during which migraine features were present for at least 8 days per month, while HFEM was defined as headaches that occurred 8–14 days per month.

Study flowchart. EM: episodic migraine; LFEM: low-frequency episodic migraine; HFEM: high-frequency episodic migraine; CM: chronic migraine.
The detailed study design is presented in Figure 2. Wearing off was evaluated for patients with HFEM and CM receiving fremanezumab either monthly or quarterly. Wearing-off was defined as an increase in weekly migraine days (WMDs) of >1 day at any time point after fremanezumab administration based on headache diaries. For the monthly dosing, WMDs were compared between weeks (W) 1–2 and W3–4 and between W2 and W4 in months 3, 6 and 9. For quarterly dosing, WMDs were compared between W1–2 and W11–12 and between W2 and W12 in the first, second and third quarters, in accordance with previous studies. 5,8 Baseline WMDs were determined using the average of the 4-week period prior to the initiation of fremanezumab treatment. The % decrease in WMDs at each time point from baseline was obtained by calculating the following formula. For example, the % decreases in WMDs at W1–2 = 100 − [(WMDs at W1–2/baseline WMDs) × 100].
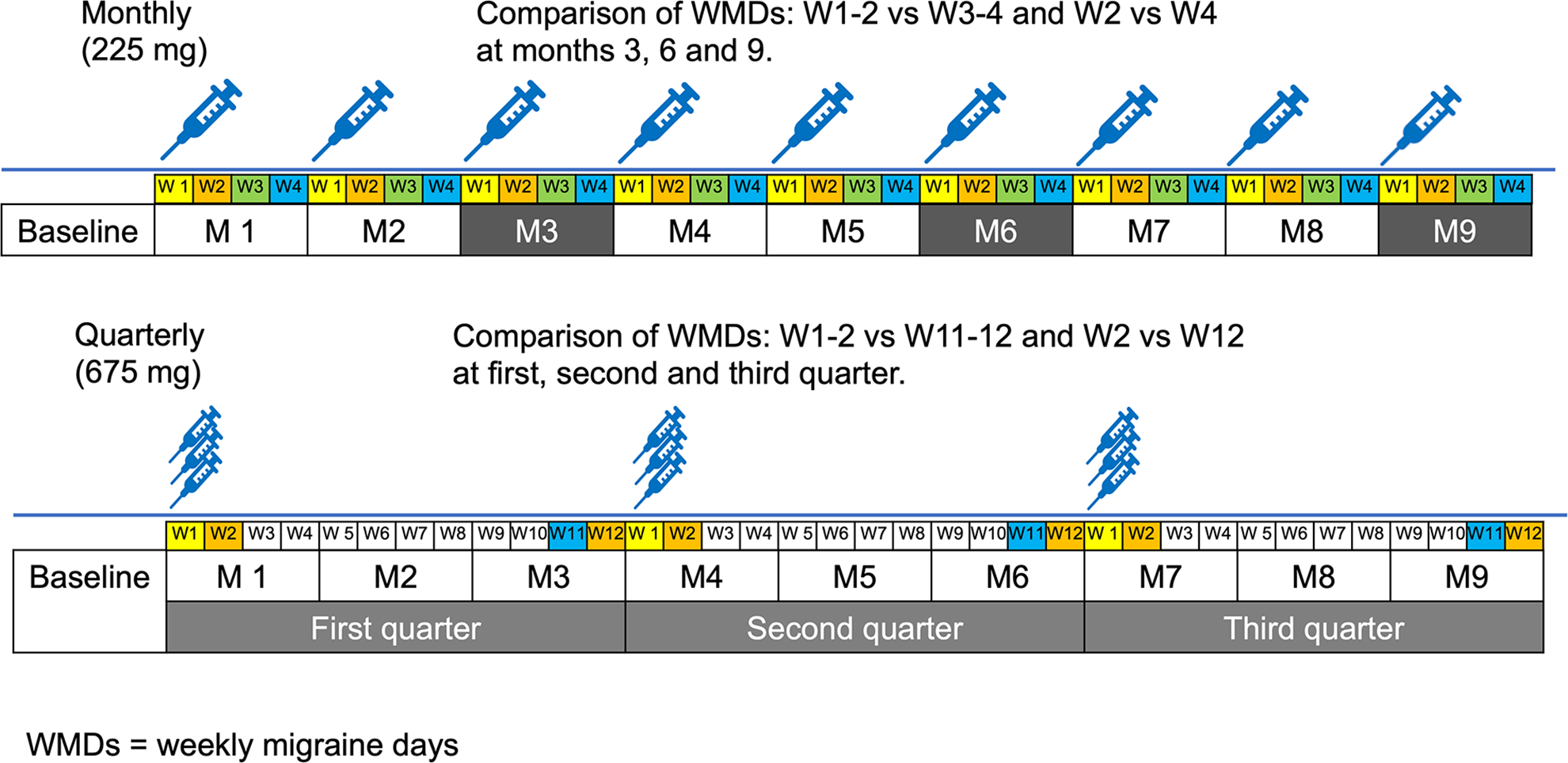
Time point of evaluation of wearing-off effects in the monthly and quarterly dosing groups.
Statistical analysis
All continuous variables are presented as means ± standard deviations or medians with 25th–75th percentiles, and categorical variables are presented as frequencies (%). Since the WMDs at any time were nonnormally distributed based on the Shapiro–Wilk test (p < 0.05), Wilcoxon signed rank tests were used to compare WMDs at each time point. Time (36 weeks) and WMD changes were analyzed using a generalized linear mixed-effect model followed by restricted maximum likelihood, and a global test was analyzed by the type III analysis of variance table with Satterthwaite’s method. Two-tailed p < 0.05 was considered indicative of statistical significance. EZR (version 1.55) 9 and GraphPad Prism for Mac (version 8; GraphPad Software, San Diego, USA) were used for the statistical analyses.
Results
The baseline characteristics of the patients are shown in Table 1. The mean patient age was 47.3 ± 11.9 years, and the mean disease duration was 27.8 ± 11.0 years. Fifty-eight patients (57.4%) had CM, 60 patients (59.4%) received quarterly dosing, and 14 patients (13.9%) had comorbid MOH. The mean baseline for WMDs was 4.3 ± 1.6 days. There was a trend toward less aura, photophobia, and allodynia in the monthly dosing group compared with the quarterly dosing group, but there were no differences in other factors, including WMDs, between the two groups (Table 1). As shown in Figure 3, there were significant changes in the mean number of WMDs from baseline to 36 weeks (week’s fixed effect: intercept = −1.482 (SE = 0.102), β-estimate = −0.031 (SE = 0.002), and global test results, F(1, 3236.6) = 300.11, p < 0.001). The mean decreases in WMDs from baseline at 12, 24, and 36 weeks were −2.0 ± 1.4, −2.4 ± 1.4, and −2.4 ± 1.1 days, respectively.
Baseline characteristics of patients with migraine.a

HFEM: high-frequency episodic migraine; CM: chronic migraine; MOH: medication overuse headache; WMDs: weekly migraine days.
a Continuous variables are presented as means ± standard deviations or medians with 25th–75th percentiles, and categorical variables are presented as frequencies (%).

Change in the weekly migraine days in the monthly and quarterly dosing groups. Error bars represent standard errors of the mean.
In the monthly dosing group, wearing-off effects occurred in 4 (6.7%) and 7 (11.7%) out of 60 patients when WMDs in W1–2 were compared with those in W3–4 and when WMDs in W2 were compared with those in W4, respectively. At the third month, wearing-off effects occurred in 3 (5%) of 60 patients when WMDs in W1–2 were compared with those in W3–4 and in 5 (8.3%) of 60 patients when WMDs in W2 were compared with those in W4.
In the quarterly dosing group, a wearing-off effect occurred in 4 (9.8%) and 4 (9.8%) of 41 patients (W1–2 vs. W11–12 and W2 vs. W12, respectively). In the first quarter, wearing-off effects occurred in 2 (4.9%) of 41 patients when WMDs in W1–2 were compared to WMDs in W11–12 and in 2 (4.9%) of 41 patients when WMDs in W2 were compared those in W12.
In the monthly dosing group, mean changes in the number of WMDs did not differ between W1–2 and W3–4 or between W2 and W4 (Table 2(a) and Figure 4(a)). In the quarterly dosing group, there were significant decreases in WMDs in the first quarter between W1–2 and W11–12 and in the third quarter between W2 and W12 (Table 2(b) and Figure 4(b)). The number of WMDs in the second quarter did not significantly differ among any of the different conditions.
Changes in the WMDs and % WMD decrease in the fremanezumab monthly and quarterly dosing groups.

WMDs: weekly migraine days.
a p < 0.01, using the Wilcoxon signed rank test.
b p < 0.05, using the Wilcoxon signed rank test.

Mean changes in weekly migraine days in the fremanezumab monthly and dosing groups. *p < 0.05, **p < 0.01, Wilcoxon signed rank test. (a) Fremanezumab monthly dosing group. (b) Fremanezumab quarterly dosing group. Error bars represent standard errors of the mean.
In total, the rates of clinical responders, defined as experiencing a ≥50% reduction in monthly migraine days during months 1 and 2, were similar between the wearing-off positive group (38.5%, 5/13) and the wearing-off negative group (38.6%, 34/88, p = 0.990).
Discussion
We found no increase in the mean number of WMDs at the end or the second half of the week in the HFEM and CM patients receiving monthly or quarterly fremanezumab dosing over 9 months. The percent decreases in WMDs were also relatively stable during the observation period. For the monthly dosing group, WMDs were compared in two settings based on previous studies (W1–2 vs. W3–4 and W2 vs. W4). 5,6,8,10 Among all the settings, we did not observe a diminished effect of fremanezumab at 3, 6, or 9 months. In the quarterly dosing group, we performed comparisons of WMDs in W1–2 versus W11–12 and in W2 versus W12 in the first, second and third quarters to determine whether the efficacy of fremanezumab diminished before the next dosing. As with the monthly dosing, we did not observe a decreased efficacy of fremanezumab at the end of dosing. Interestingly, a significant decrease in WMDs was observed in the first quarter between W1–2 and W11–12 and in the third quarter between W2 and W12. Although the fremanezumab dosing interval was not strictly determined because this was a real-world study, it is possible that the quarterly dosing group more closely followed the scheduled dosing interval than did the monthly dosing group, which may have led to the decrease in WMDs in the first and third quarters. The effect of the mean concentration of fremanezumab on the reduction in migraine days has been reported to be similar to exposure-response models for both EM and CM patients receiving monthly doses of 225 mg and quarterly doses of 675 mg. 11 However, the pattern of increase in the fremanezumab blood concentration after fremanezumab administration differed between the quarterly dosing group (675 mg) and the monthly dosing group (225 mg). This difference may have contributed to the significant difference in WMD values in the first quarter between the start and end of dosing in the quarterly dosing groups in our study.
In our study, wearing-off effects occurred in 6.7–11.7% of the monthly dosing group and 9.8% in the quarterly dosing group. In a real-world retrospective study including patients with migraine treated with erenumab, one-third of patients with migraine experienced wearing-off effects; of these, 80% reported that these effects appeared 1 week before the next injection. 10 In a questionnaire-based survey including 145 patients with HFEM or CM receiving erenumab, 13.8% reported decreasing efficacy with every injection, and 37.8% experienced some wearing-off effects between injections. 12 These rates of wearing-off effects were higher than those seen in our study in the monthly and quarterly dosing groups. One of the reasons for this difference could be that the wearing-off effect was not well defined in the other studies. 10,12 Additionally, it should be noted that questionnaire-based surveys on wearing-off effects may suffer from a nocebo effect, which is described as a reported symptom or reaction not directly caused by drug action but related to patient expectations of side effects. 13 In contrast, our study assessed wearing off by a clear definition—an increase in WMDs of >1 day at any time point—which was assessed by the data recorded in headache diaries.
Four types of CGRP mAbs are currently available for preventive treatment in migraine, among which erenumab, galcanezumab and fremanezumab are available in Japan. Differences in clearance and distribution volume may affect the pharmacokinetic parameters of anti-CGRP pathway mAbs. Fremanezumab has a half-life of 31 days and shows the lowest isoelectric point among CGRP mAbs, which may limit its distribution outside the blood compartment. 1 This characteristic may contribute to a longer half-life for fremanezumab than for other CGRP monoclonal antibodies, leading to a persistence of drug efficacy and minimum wearing-off effect. In a patient survey study including 557 patients with migraine, 69.2% of patients preferred quarterly over monthly fremanezumab dosing, suggesting that quarterly dosing options could provide benefits for patients who value their private lifestyles and seek the convenience of less frequent administration of prophylactic treatment. 14 Our study results suggest that fremanezumab may be a suitable CGRP monoclonal antibody for patient-centered therapy, as wearing-off effects occur with low frequency with either monthly or quarterly dosing approaches.
The main limitations of this real-world study were that we did not perform sample size calculations and there was inconsistency in the number of patients over 9 months; thus, the monthly and quarterly dosing group analysis respectively included 60 and 41 patients, 54 and 36 patients, and 42 and 33 patients at each of the 3 time points. According to the p value (0.05), power (0.8), and effect size (0.5) calculated from the Wilcoxon signed rank test, the needed sample size was at least 35 cases, at which the number of patients needed for comparative purposes was almost achieved. However, a larger-scale study will be conducted to validate these encouraging results. In our real-world study in Japan, migraine patients had a lower body mass index than in the previous study of fremanezumab in the U.S. 5,15 Higher body weight was associated with increased central clearance and volume of distribution of fremanezumab. 11 Therefore, the generalizability of the results of this study still needs to be verified. Further research focusing on evaluation methods regarding which weeks to compare and how many increased WMDs should be defined as wearing-off effects is needed. In addition, clinical factors that predict wearing-off erects should be identified in future studies.
In conclusion, in this real-world study, we found no significant increase in WMDs 1–2 weeks before the next dosing at any time point over 9 months. Our findings support sustainability in the effect of fremanezumab as headache prophylaxis.
Clinical implications
We determined the rate of wearing-off effects of monthly and quarterly dosing of fremanezumab, including patients with 101 HFEM and CM. In this real-world study, there was no significant increase in WMDs 1–2 weeks before the next dosing at any time point over 9 months. Our findings support sustainability in the effect of fremanezumab as headache prophylaxis in the real-world setting.
Footnotes
Abbreviations
Acknowledgments
The authors thank Ms Sanae Tani and Ms Kaori Soma, Department of Neurology, Dokkyo Medical University, for their help with this study.
Author contributions
All authors contributed to the interpretation of data for this study. SS and KS drafted the manuscript. SS, KS, TS, and KH contributed to the acquisition of data. KS and YH performed the statistical analysis. TS and KH performed a critical review for important intellectual content. All authors read and approved the final version of the manuscript.
Data availability statement
The datasets from this study are available from the corresponding author upon reasonable request.
Declaration of conflicting interests
The authors declare the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: S Suzuki received lecture fees from Eli Lilly Japan, Daiichi Sankyo, Amgen and Otsuka Pharmaceutical Co., Ltd, during the conduct of the study. K Suzuki received lecture fees from Eli Lilly Japan, Daiichi Sankyo and Otsuka Pharmaceutical Co., Ltd, during the conduct of the study. T Shiina received lecture fees from Eli Lilly Japan, Daiichi Sankyo, Amgen and Otsuka Pharmaceutical Co., Ltd, during the conduct of the study. Y Haruyama has nothing to disclose. K Hirata received lecture fees from Eli Lilly Japan, Daiichi Sankyo, Amgen and Otsuka Pharmaceutical Co., Ltd, during the conduct of the study.
Ethical approval
The study was conducted in compliance with the Declaration of Helsinki and was approved by the Institutional Review Board of the Dokkyo Medical University Hospital. The Institutional Review Board of the Dokkyo Medical University Hospital waived the need for patients to sign informed consent based on the retrospective, observational nature of the study.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
