Abstract
Background:
Signs of distinct brain dysfunction in patients where migraine intersects with vertigo (i.e. vestibular migraine (VM)), remain elusive. As migraine and vertigo can both independently modulate attentional processes, here we seek the utility of the attentional network to functionally differentiate patients.
Methods:
We used the Attentional Network Task (ANT) to elucidate three separate functional networks: Alerting, orienting and resolving conflict. 120 participants had to attend to the direction of a target visual stimulus, while other parameters were simultaneously manipulated. Reaction times across the networks were assessed in, (i) 30 healthy controls, (ii) 30 VM patients, (iii) 30 patients with migraine without vertigo, and (iv) 30 patients with benign paroxysmal positional vertigo (BPPV) but no migraine.
Results:
Patients with VM (mean = 737.1 ms, SEM = 28), migraine (mean = 735.3 ms, SEM = 36.4), and BPPV (mean = 720.3 ms SEM = 24.3) all exhibited significantly delayed ANT reaction times compared to healthy controls (mean = 661.3 ms, SEM = 23.4). Specific attentional network deficits were observed for resolving conflict in VM, alerting in migraine and orienting in BPPV.
Conclusion:
VM patients displayed deficits in executive function characterized by an inability to focus attentional resources and suppress peripheral distractors, whereas migraineurs without vertigo exhibited changes in the alerting network that reflects hypervigilance.
Introduction
Vestibular migraine (VM) is a relatively new diagnostic entity that has been accepted by the International Classification of Headache Disorders (ICHD) as the unifying term for patients that typically complain of both episodic headache and vestibular symptoms. 1 VM is among the commonest cause of spontaneous (non-positional) episodic vertigo, affecting between 1% and 2.7% of the general population. 2,3 It accounts for 11% of all patient referrals to specialized dizzy clinics 4 and 13% of all referrals to tertiary headache clinics. 5
The clinical presentation of VM is diverse. Episodes of dizziness in VM usually last between 5 minutes and 72 hours, although shorter and longer episodes have been reported. 1 Symptoms include spontaneous and/or motion-triggered vertigo, spatial disorientation and unsteadiness which are typically accompanied by history of headache, photophobia, phonophobia and nausea. 1 In addition, a proportion of patients additionally report experiences of spatial misperceptions, impaired concentration, and foggy headiness, during the attacks. 1
Recent neuroimaging studies have revealed altered functional connectivity and reduction in grey matter volume in VM patients. 6 Furthermore, a range of perceptual (i.e. motion discrimination, 7 motion detection, 8,9 and spatial orientation) 10,11 and cognitive deficits have been reported interictally in VM patients, as well as in patients with episodic vertigo due to a peripheral aetiology. 12,13 Moreover, a recent meta-analysis has objectively confirmed that cognitive deficits can occur in migraine patients during the interictal period that span a range of domains including attention, spatial cognition, memory and executive function. 14 Taken together, the presence of such persisting dysfunction during the interictal period has been attributed to possibly reflect increased noise within the sensory circuits of patients with episodic migraine and/or vertigo. 7
Despite the aforementioned behavioural and neuroimaging findings, the underlying pathophysiology of VM remains largely unknown. This lack of knowledge is reflected by the fact that we currently do not have any robust behavioural markers that can objectively signify interictal brain dysfunction in VM patients. Establishing such a sign is imperative to improve diagnostic certainty as well as aid our understanding of the pathophysiology. Critically, the marker necessitates the ability to be able to dissociate VM patients from those patients with either, (a) migraine without vertigo, or (b) episodic vertigo that is attributable to inner ear pathology such as benign positional paroxysmal vertigo (BPPV).
To identify objective behavioural markers of interictal brain dysfunction in VM patients, we assessed for behavioural changes associated within the attentional network, given that recent findings indicate that both migraine and vertigo can independently modulate distinct brain networks related to attentional processing. 15,16 Further supporting our view, are clinical observations that a common characteristic of patients who suffer from either migraine, vertigo or both is enhanced sensitivity to sensory stimuli. 17 –19 Such sensory hypersensitivity may potentially be mediated by changes in attentional processes that differentiate salient stimuli from other sensory distractors. 20
Theoretical frameworks and experimental data indicate that the attentional network is composed of three independent modules, which include alerting, orienting, and resolving conflict. Alerting assesses the ability to remain vigilant of upcoming sensory signals, whereas orienting involves spatially aligning attention with the salient source of a sensory signal. The resolving conflict or an executive aspect of the attentional network reflects the ability to readily focus on a task and suppress peripheral distractors. 21,22 All these processing aspects of attention can be examined via the validated Attentional Network Task (ANT). 23
Previous neuroimaging data has revealed that migraine patients without vestibular symptoms display changes in alertness associated with increased vigilance. 15 Here we hypothesized that such attentional dysfunction can be behaviourally quantified as changes in the altering network during performance of the ANT task. Contrastingly, as the vestibular system plays a pivotal role in spatial perception and patients with vestibular dysfunction often have spatial disorientation, 24,25 we hypothesized that patients with vertigo attributable to a peripheral vestibular aetiology (i.e. BPPV) may display a distortion associated with the orienting network during performance of the ANT task. In addition, we applied the same task in VM patients, who symptomatically experience both vestibular and migraine symptoms. We asked whether VM patients have a distinct locus of impairment in the attentional network, when compared to patients with either migraine or vestibular symptoms alone.
Materials and methods
Participants
Patients were recruited by Consultant Neurologists from neurology and neuro-otology clinics at Charing Cross Hospital and The National Hospital for Neurology and Neurosurgery. The study was approved by London Bridge NHS Ethics Research Committee (IRAS 72639). Written informed consent was obtained from each participant, and the study conformed with the World Medical Association Declaration of Helsinki.
Thirty VM patients were recruited (mean age 47.3 years; range 24–71; 19 F: mean days since the last episode: 22.7 days; range 5–240) who conformed to the diagnostic criteria of VM by International Committee for Classification of Vestibular Disorders (ICVD) of the Bárány Society and the 3rd edition of the International Classification of Headache Disorders (ICHD). 1 We also recruited a group of 30 migraine patients with aura but without vestibular symptoms (mean age 44.2; range 20–73; 19 F: mean days since the last episode: 34.5 days; range 7–331 days), who conformed to the ICHD-3 Beta clinical criteria for diagnosis of migraine with typical aura. 26,27 For peripheral vertigo, we recruited 30 patients who were diagnosed with posterior canal BPPV (mean age 55.8; range 32–71; 16 F: mean days since the last episode: 14.5 days; range 3–197 days), based on the ICVD clinical criteria. 28 Critically, none of the BPPV patients had any previous or current history of either migraine or VM. Finally, 30 participants (mean age 52.8; range 18–78; 19 F) were also recruited as a healthy control group. In both migraine and VM groups, the experiment was performed in the interictal period, and in BPPV patients it was performed one hour after the repositioning (treatment) manoeuvre. All participants were right-handed with normal vision and no history of ophthalmological or psychiatric disorders. There were (i) no other neurological disorder apart from migraine in the migraine and VM groups and (ii) no otological disorder apart from the active BPPV diagnosis in the peripheral vertigo group.
As summarized in Table 1, 28/30 of the migraine and 26/30 of the VM patients were taking analgesics (ibuprofen and paracetamol) as needed. 18/30 of the VM patients and 15/30 of the migraine patients respectively, were taking prophylaxis medications (VM: pizotifen n = 5, amitriptyline n = 8, propranolol n = 5); Migraine: (pizotifen n = 4, amitriptyline n = 11). 106/120 participants had normal hearing (threshold of less than 20 dBHL between 250 Hz and 8000 Hz), as tested with a pure tone audiogram. Fourteen participants had age-related hearing loss (three VM patients, three migraine without vertigo patients, five BPPV patients and three healthy controls). The average threshold of hearing in these participants was 45 dBHL (range of thresholds 25–70 dBHL: between 250 Hz and 8000 Hz. All patients had normal vestibular function assessed via caloric irrigation or when contraindicated due to a perforated ear drum (n = 1) via the video head impulse test (Table 1). Other exclusion criteria included an Abbreviated Mental Test Score (AMTS) of <8, Migraine 9.84 ± 0.11(SEM), VM 9.66 ± 0.10 (SEM), BPPV 9.91 ± 0.06 (SEM), healthy controls 9.80 ± 0.07 (SEM).
Subject groups characteristics and test results.

UW: unilateral weakness, SPV: slow phase velocity: vHIT: video head impulse testing, SCC: semi-circular canal: AMTS: Abbreviated Mental Test Score: DHI: dizziness handicap inventory.
Attentional network task
The attentional network consists of three independent modules, which include alerting, orienting, and resolving conflict. 23 The alerting module assesses the ability to remain vigilant of upcoming sensory signals, whereas the orienting module involves spatially aligning attention with the salient source of a sensory signal. The resolving conflict or an executive component reflects the ability to readily focus on a task and suppress peripheral distractors. These functional aspects can be examined via the validated Attentional Network Task (ANT). 23
This task requires subjects to visually fixate on a central dot and indicate the directionality (i.e. right/left) of a target arrow, presented either in the upper or lower visual hemifield on a screen placed 25 cm away (68° visual angle). Performance in the ANT task is assessed by reaction time (RT), and can be modulated by valid spatial (orienting) cues to the location of an upcoming stimulus which is a target arrow. Other modulatory features in the task include spatially non-informative (alerting) cues and directional cues (flankers) in either the same (congruent) or opposite (incongruent) direction of the target arrow (Figure 1). 23 The flankers allow assessment of the attentional ability to resolve conflict (i.e. executive function).

Attentional network task and overall performance (5). Subjects were instructed to indicate the direction (right/left) of a target arrow in one row of arrows that showed in the upper or lower visual hemifield. (a) Targets are preceded by one of four possible cue types: none, double, central, or spatial on each trial, (b) the central arrow is flanked by four a row arrows, pointing in the same (congruent), opposite (incongruent), or no (neutral) direction, as shown in the examples. Participants were required to respond to the direction that the central arrow is pointing in; (c) Timeline of experiment: (•) illustrates the fixation point which participants focus on throughout the experiment, one of the cue conditions presented is depicted which lasts for 200 ms, then the fixation circle is presented alone for 500 ms, after which one of the flanker target conditions appear either in the lower or upper hemifield. Participants have 2000 ms to respond. They are then given feedback after each response. (d) Measures for attentional network are derived as follows: (1) alerting network = RT (no cue) − RT (double cue); (2) orienting network = RT (central cue) − RT (spatial cue); (3) executive control = RT (incongruent) − RT (congruent).
Each experimental block consisted of 48 trials: 4 cue types (none, double, central, or spatial) × 2 target locations × 2 target directions × 3 flanker conditions (neutral, congruent, or non-congruent,) with two repetitions. The order of the presentation of trials was randomized. The visual and timing parameters of the stimulus employed in our protocol were identical to those previously described. 23 Participants pressed the right or left arrow keys always with their right hand.
Figure 1 shows how these responses were measured in this study by using either (a) one of four preceding cue targets: none, double, central, or spatial and (b) flanker arrows that were parallel to the target arrow. The flankers were absent in a neutral condition, pointing in the same direction as the target arrow in the congruent condition, and the opposite direction in the incongruent condition. 23 In this scheme, specific ANT measures could be derived for (1) the alerting module as the difference between the RT for double cue trials (alerting signal) and no cue trials, (2) the orienting module as the difference between the RT of spatial cues (orienting signal) and the trials with a central cue (non-spatially informative signal), and (3) the resolving conflict (executive function) as the difference in the RT between the incongruent and congruent flanker trials. 29
Clinical questionnaires
Participants completed the Dizziness Handicap Inventory, a 25-item questionnaire to assess: (i) physical (7 questions, 28 points), (ii) functional (9 questions, 36 points), and (iii) emotional (9 questions, 36 points) factors associated with dizziness. 30 Given the possibility that attentional function could be modulated by state and trait anxiety, we used The Spielberger Trait and State Anxiety Inventory in all participants to assess anxiety levels on a day-to-day basis (trait) and the short version in response to the experimental protocol (situational anxiety; state). 31 –33
Data and statistical analysis
Average RT for each stimulus and cue condition were calculated for each participant. To analyse the RT for the flanker conditions we implemented a two-way repeated measures ANOVA with factors including Flankers (three levels; neutral, congruent, and incongruent) and Group (four levels; healthy controls, VM, migraine without vertigo and BPPV). Additionally, to analyse the reaction time for the cue conditions we implemented a separate two-way repeated measures ANOVA with factors including Cue (four levels; none, central, double, spatial) and Group (four levels; healthy controls, VM, BPPV, migraine). For each of the derived network, a one-way ANOVA was performed to compare RT among the subject groups for each of the attentional networks (alerting, orienting and executive control). To assess for any differences in anxiety scores across the different participants groups we performed a 4 × 2 ANOVA with factors Group (four levels; healthy controls, VM, migraine without vertigo and BPPV) and Anxiety (two levels; state or trait). Post hoc comparisons were also performed throughout using Bonferroni corrections. To assess the relationship between the network measures and questionnaires, we performed Pearson’s correlations.
Results
Patients with VM, migraine without vestibular symptoms, and BPPV displayed longer RT compared to healthy controls (VM 737.1 [mean] ± 28 ms [SEM]; Migraine 735.3 ± 36.4 ms; BPPV 720.3 ± 24.3; healthy control 661.3 ± 23.4).
Repeated measures ANOVA revealed a significant difference among the flanker groups (df = 2; f = 171.271; p < 0.0001) (Figure 2(a)). There was a significant interaction among the subject groups (df = 3; f = 4.252, p = 0.008), and a significant interaction between the flanker groups and the subject groups (df = 6; f = 5.482; p < 0.0001) (Figure 2). Post hoc analysis revealed a significant difference for the neutral, congruent, and incongruent flankers between healthy controls and (i) vestibular migraine (p = 0.016), (ii) BPPV (p = 0.018) and (iii) migraine without vertigo (p = 0.032) patients. In other words, controls performed better than all of the three patient groups regardless of whether the flanker was congruent, incongruent or absent.
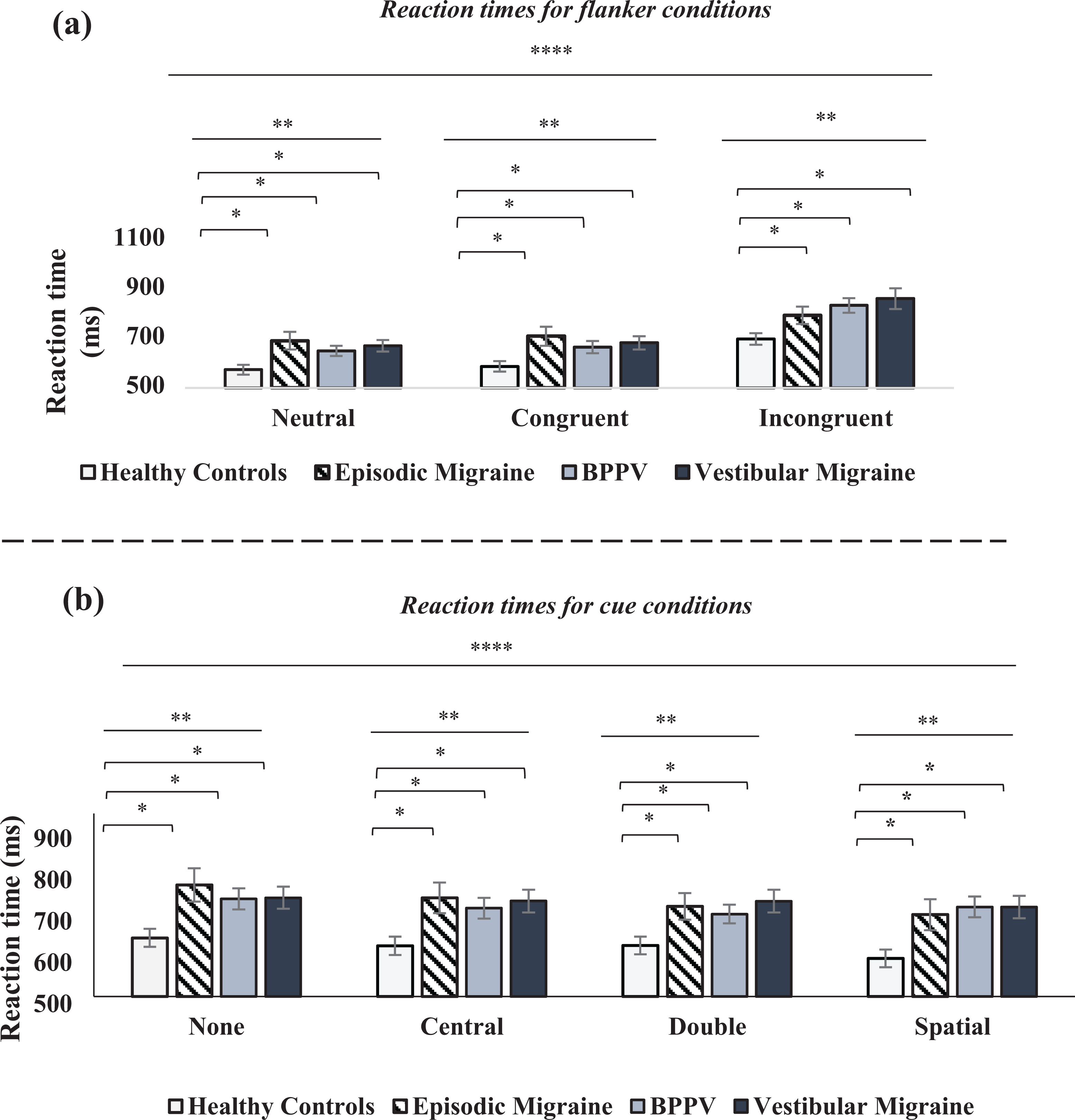
Results for the Flanker and Cue conditions. (a) Reaction times for flanker conditions; On the x-axis we represent the different flanker condition; neutral, congruent, and incongruent groups. Repeated measures ANOVA revealed a significant difference for the neutral, congruent, and incongruent flankers between healthy controls and (i) vestibular migraine (p = 0.016), (ii) BPPV (p = 0.018) and (iii) episodic migraine (p = 0.032) patients. (b) Reaction times for cue conditions; On the x-axis, we represent the different cue conditions, no cue, central, double and spatial cues. Repeated measures ANOVA revealed a significant difference between the healthy control versus; (i) BPPV was (p = 0.020) and (ii) VM was (p = 0.014), and (iii) migraine (p = 0.033). Significance levels represented are *= p < 0.05, **= p < 0.01, ***= p < 0.001, ****= p < 0.0001. Error bars represent standard error of the mean (SEM).
Furthermore, we observed a significant difference among the cue conditions (df = 3; f = 17.153; p < 0.0001) (Figure 2(b)). There was also a significant interaction among the subject groups (df = 3; f = 4.193; p = 0.009) (Figure 2), and a significant interaction among the cues and the subject groups (df = 9; f = 3.990; p < 0.0001). Post hoc analysis revealed a significant difference between healthy controls and (i) vestibular migraine (p = 0.014), (ii) BPPV (p = 0.020) and (iii) migraine without vertigo (p = 0.033) patients. That is, controls performed faster than all of the three patient groups regardless of whether the cue provided spatial information, alerting information or no information.
Attentional network
A one-way ANOVA was conducted to compare RT among the subject groups for each attentional function. A significant difference among the subject groups was observed for all three functions: (i) alerting (df = 3; f = 7.466; p < 0.0001) (Figure 3(a)), (ii) orienting (df = 3; f = 3.659; p = 0.015) (Figure 3(b)) and (iii) resolving conflict (df = 3; f = 3.970; p = 0.016) (Figure 3(c)). Post hoc analysis revealed a significant difference in the alerting function between healthy controls and migraine without vertigo (p = 0.038), and between migraine without vertigo and vestibular migraine (p = 0.012). For the orienting function, it revealed a significant difference between healthy controls and BPPV (p = 0.014) as well as BPPV and migraine without vertigo (p = 0.015). Finally, post hoc analysis also revealed a significant difference in resolving conflict between healthy controls and VM (p = 0.025) as well as VM and migraine without vertigo (p < 0.0001).

Results for the derived attentional network function. On the x-axis we represent the different participant groups and on the y-axis we represent the derived attentional networks for figures (a)–(c). For the altering network (a), there was a significant difference in the alerting network between healthy controls and migraine (p = 0.038), and migraine and vestibular migraine patients (p = 0.012). For the orienting network (b), there was a significant difference between healthy controls and BPPV patients (p = 0.014) as well as migraine and BPPV patients (p = 0.015). For the conflict resolution network (c), there was a significant difference between healthy controls and vestibular migraine patients (p = 0.025) as well as episodic migraine and vestibular migraine (p < 0.0001). Significance levels represented are *= p < 0.05, **= p < 0.01, ***= p < 0.001, ****= p < 0.0001. (d) Illustrates the significant positive correlation between the derived conflict resolution network (y-axis) and overall DHI score (x-axis) in VM patients (Pearson correlation r = 0.751; p = 0.0001).
Clinical significance
VM patients exhibited a positive correlation between attentional processing in resolving conflict and vestibular symptoms as measured by the overall DHI score (Pearson correlation r = 0.751; p = 0.0001) (Figure 3(d)). There was no relationship between attentional processing in the orienting network and vestibular symptoms as measured by the overall DHI in BPPV patients. No other relationship was observed in any of the other patient groups or networks, nor with respect to anxiety scores (range of r values: 0.007–0.11 (see Table 1 for clinical questionnaire scores). Regarding trait anxiety scores, the ANOVA revealed that the scores were significantly higher in the three patient cohorts compared to the control group (controls vs. VM (p < 0.000), controls vs. BPPV (p = 0.001) and control vs. migraine (p = 0.03)). There were no differences in trait anxiety scores between any of the three patient groups (p > 0.05). Regarding state anxiety scores, the ANOVA revealed that the (pre-experimental) scores were significantly higher in the control group versus VM (p = 0.002) and controls versus BPPV (p = 0.001) but not controls vs migraine (p = 0.888). Trait anxiety scores in the patient groups differed between migraine and both BPPV (p = 0.002) and VM (p = 0.005) patients, but there was no difference between VM and BPPV (p = 0.926).
Effect of medications
To rule out potential effects of medications on task performance during the ANT in VM patients, we performed an independent t-test on the average RT of patients on (n = 18) versus off (n = 12) prophylaxis medication. No significant difference was observed between the two subgroups in VM patients (p = 0.912). Similarly, in migraine patients there was also no difference between patients on (n = 15) versus off (n = 15) prophylaxis medication (p = 0.989).
Discussion
Patients with VM, migraine without vestibular symptoms and BPPV exhibited impaired attentional processing as reflected by significantly delayed reaction times in the attentional network task when compared to healthy controls. Critically, discrete and specific attentional network impairments were observed for resolving conflict (i.e. executive functioning) in VM patients, alerting in migraine patients without vestibular symptoms, and orienting in BPPV patients. Notably, the observed deficit in VM patients was clinically relevant as it correlated with validated measures of symptom load.
Our results are in line with previous findings which have shown that both migraine and vestibular dysfunction can distinctly modulate separate components of the attentional network. That is, a decline in the orienting attentional network in patients with episodic vertigo due to BPPV tallies with our and other previous experimental data illustrating a clinically significant spatial disorientation in patients with peripheral vestibular disorders. 34,35 Furthermore, our observation of an altered alerting network in patients with migraine without vestibular symptom is in line with previous functional imaging data that indicates migraineurs display increased vigilance and inability to supress distractors. 15
In VM patients, where migraine and vertigo intersect, despite a significant handicap from both vestibular and migraine symptoms, the alerting and orienting functions were not altered. Rather, the locus of impairment was related to the ability to readily focus attentional resources on the task-relevant information (i.e. central arrow) and to inhibit the task-irrelevant peripheral stimuli (i.e. incongruent flankers).
Thus, VM patients are seemingly able to effectively prioritize and process sensory signals to stay vigilant and orientate to salient spatial information, but they show deficit in their attentional processing for resolving conflict. Such experimental observation tallies with the clinical reports of symptoms by VM patients, namely that they are triggered by irrelevant peripheral visual stimuli especially in busy sensory-rich environments. 36,37 This is further supported by the positive correlation we observed between deficits in the resolving conflict network and dizzy symptoms assessed by the validated DHI questionnaire.
It is also important to note some potential limitations of the present study. Migraine and vertigo are often associated with co-morbid psychiatric symptoms (i.e. anxiety), 38,39 which can modulate performance in the ANT task. 40 Moreover, recent research has shown that in VM patients, a DHI greater than 60 is unlikely to be caused by the neurological dysfunction alone but signifies an increased likelihood of co-existing functional or psychiatric disorders. 30,41 In our cohort, this accounted for 6 out of the 30 VM, which could potentially have impacted upon the attentional task performance. Reassuringly, the trait anxiety scores were comparable across all three patient groups in our study, and the state anxiety scores were comparable in the VM and BPPV patients. This taken together in the context of our findings, which highlights a distinct differential modulation of the attentional network in the three patient groups, argues against the notion that the findings we report are primarily driven by anxiety.
To conclude, our results show that patients with VM displayed an impaired ability to readily focus attentional resources and supress peripheral distractors. Contrastingly, in migraineurs without vertigo we observed changes that were associated with the alerting network, which is an observation that is consistent with previous findings of hypervigilance in migraine patients. Vertiginous (BPPV) patients exhibited features of disorientation as highlighted by a deficit in the orienting network. Indeed, spatial disorientation is a well-established phenomenon in patients with peripheral vestibular dysfunction. Taken together, our results illustrate that distinct deficits in the attentional network can differentiate different phenotypes of migraine with and without vertigo.
Footnotes
Acknowledgements
We would like to thank all the patients and control participants for their time and effort. We also would like to extend our thanks to Mr David Buckwell for technical assistance, and Professor John Golding for statistical advice.
Article highlights
*Migraine differentially modulates attentional mechanisms dependent upon the migraine phenotype.
*Migraine modulates the alerting component of the attentional network.
*Vestibular migraine modulates the executive component of the attentional network.
*Deficit in processing in vestibular migraine patients is clinically relevant as it correlates with symptom load.
Data availability statement
Data can be provided by emailing the corresponding author.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the National Institute for Health Research (NIHR) Imperial Biomedical Research Centre and the National Institute for Health (NIH). The work was also supported by the Grant R01DC018815 from the National Institute on Deafness and other Communication Disorders.
