Abstract
Background:
Patients with cerebral or spinal cavernous malformations (CM) and a primary headache disorder are often limited in medication options due to concern for bleeding risk.
Methods:
From a prospective cohort of CM patients (2015–2020), demographics, mode of clinical presentation, and radiographic data were collected. Follow up of patients was performed with electronic medical record review, in person visits and/or written surveys. Select medication use was ascertained from the time of the CM diagnosis to a censor date of first prospective symptomatic hemorrhage, complete surgical excision of sporadic form CM, or death. The influence of non-aspirin NSAID (NA-NSAID), triptan, or OnabotulinumtoxinA on prospective hemorrhage risk was assessed.
Results:
As of August 20, 2020, 329 patients with spinal or cerebral CM (58% female; 20.1% familial; 42.2% initial presentation due to hemorrhage; 27.4% brainstem) were included. During a follow-up of 1799.9 patient years, 92 prospective hemorrhages occurred. Use of NA-NSAIDs, triptans, and OnabotulinumtoxinA after the diagnosis of CM was unassociated with an increased risk of prospective hemorrhage.
Conclusions:
Use of triptans and NA-NSAIDs, does not precipitate CM hemorrhage. Similarly, we did not find that OnabotulinumtoxinA precipitated CM hemorrhage in a limited number of patients at doses <200 units per session.
Background
Cavernous malformations (CM) are vascular malformations of the brain and spinal cord consisting of endothelial lined caverns devoid of smooth muscle. Patients with CM may develop secondary headaches due to hemorrhage of the CM, edema, or hydrocephalus. 1 However, it is also common that patients with CM have concomitant primary headache disorders as well. 2 Given the concern for bleeding and the vascular nature of the lesion, uncertainty about the use of non-aspirin non-steroidal anti-inflammatory drugs (NA-NSAIDs) and triptans have arisen leaving few abortive options for tension-type and migraine headaches. 3 In addition, a single case report raised concern that OnabotulinumtoxinA, used for chronic migraine purposes, led to bleeding from a CM in that single patient. 4
In this study, we aimed to determine the safety of NA-NSAID, triptan and OnabotulinumtoxinA medications in a large, prospective cohort of patients with cerebral and spinal CM. Use of antithrombotics including aspirin in patients with CM has been previously been reported 5 and not included here, but demonstrates a lower risk of hemorrhage in people on antithrombotic medications. Propranolol, which is commonly used for migraine prophylaxis, has been reported to possibly reduce risk of bleeding in CM. 6 –8 While not the focus of this study, data on propranolol and beta-blocker use was included.
Methods
With Institutional Review Board approval, we have maintained a prospective data base of patients with radiographic evidence of cerebral or spinal CM seen at our institution beginning in January of 2015. All adult patients with CM of the spinal cord or brain were screened. Those who consented to participation were included. Patients were excluded if they were under 18 at the time of their visit to our institution, pathology at surgery demonstrated an alternative etiology, or they had a Zabramski Type 4 lesion 9 without an associated DVA and no other characteristic CM lesions as Zabramski type 4 lesions can mimic other etiologies. 10
Initial clinical information
Medical records of each patient were reviewed. Patients also filled out an initial written survey. Data collected included: demographic information, familial versus sporadic form CM, co-morbid conditions, medication use leading up to the diagnosis, and mode of initial presentation. Patients were considered to have the familial form if they were genotyped, or they had multiple CM lesions without associated DVA. The mode of presentation was divided into those with symptomatic hemorrhage, focal neurologic deficit without hemorrhage, seizure without hemorrhage or incidental findings based on standards for reporting put forth by the Angioma Alliance. 11
Initial imaging
The initial MRI scan used to make the diagnosis of CM was reviewed by clinical neuroradiology and the lead author (KDF). The number of CMs was recorded based on hemosiderin sensitive sequences (gradient recall echo or susceptibility weighted imaging). The location of the symptomatic lesion was recorded. If no lesion was symptomatic, then the largest lesion size was recorded. Locations were divided into: supratentorial-cortical, supratentorial-subcortical, brainstem, cerebellum, spinal cord and other. Presence or absence of developmental venous anomaly (DVA) was recorded. In patients for whom the initial diagnostic MRI was not available, the location of the CM was recorded based on subsequent imaging available for review.
Prospective follow-up data collection
Follow-up medical data on patients were obtained by review of electronic medical record (EMR) in addition to mailed written surveys and, in some patients, in person follow-up (Figure 1). Patients received a written survey annually for the first 5 years after initial diagnosis or semi-annually if the diagnosis was made more than 5 years prior to the initial visit to our institution. We recorded any new symptomatic hemorrhages and verified any reported hemorrhage with ascertainment of medical records and/or imaging.

Methodology and cohort ascertainment (see text).
We assessed the use of select medications after the time of diagnosis to a censored date of first prospective, symptomatic hemorrhage, complete surgical removal of sporadic form CM, or death. Patients with familial form CM were not censored at surgical removal of a single lesion because they typically have multiple lesions and therefore the risk of further hemorrhage with ongoing select medication use continues. Headache treatment medications of interest included the use of NA-NSAIDs (either daily or intermittent use), triptans, and OnabotulinumtoxinA. Due to clinical data suggesting potential benefit in patients with CM, we also assessed concomitant use of daily aspirin, vitamin D supplementation, statin use, and beta-blocker use including propranolol. 12,13
Medications of interest were ascertained in four ways. First, we assessed the clinical note from the neurologist who saw the patient at our institution. Second, we searched individual neurologic notes from EMR system, EPIC (Epic.com). Care Everywhere in EPIC allowed verification of medications at other institutions in addition to our own. Third, we used the EPIC search term feature for individual medications of interest. This feature allows searching individual words from clinical documentation quickly and efficiently. Finally, due to known inaccuracies of medications in the EMR 14,15 and patients not always recording prn medications, we also included NA-NSAIDS, vitamin D supplementation, and statin on the initial and follow-up written survey filled out by the patient specifically for this study. Triptans, OnabotulinumtoxinA, and beta-blockers were only added to surveys in late 2020.
Statistical analysis
Descriptive statistics using means, standard deviations, and frequencies were used for patient clinical and radiologic characteristics. Univariate logistic regression models were used to assess risk factors for prospective hemorrhage and characteristics of NA-NSAID and triptan users. We report the odds ratios (OR), 95% confidence intervals (CIs) and likelihood ratio p values. JMP Pro Software (version 14.1.0; SAS Institute Inc., Cary, NC) was used for analysis.
Results
Between 2015 and October 1, 2020, we screened 535 patients, and 329 patients consented (Figure 2). Initial clinical and radiologic features are presented in Table 1. The cohort consisted of 191 women (58.0%) and 138 men (42.0%). Sixty-eight patients (20.1%) had presumptive familial CM (8 CCM1, 11 CCM2, 1 CCM3, the remainder not genotyped). The patients in this cohort initially sought medical attention for the following conditions: (1) clinically symptomatic hemorrhage, 139 (42.2%); focal neurologic deficit without hemorrhage, 22 (6.6%); seizure without hemorrhage, 36 (10.9%); and hydrocephalus, 1 patient (0.3%) The remaining patients (131; 40.0%) were felt to have an incidental CM. Table 2 displays the associated diagnosis of those in which the CM was felt to be incidental. The cohort included 90 (27.4%) patients with brainstem CM.
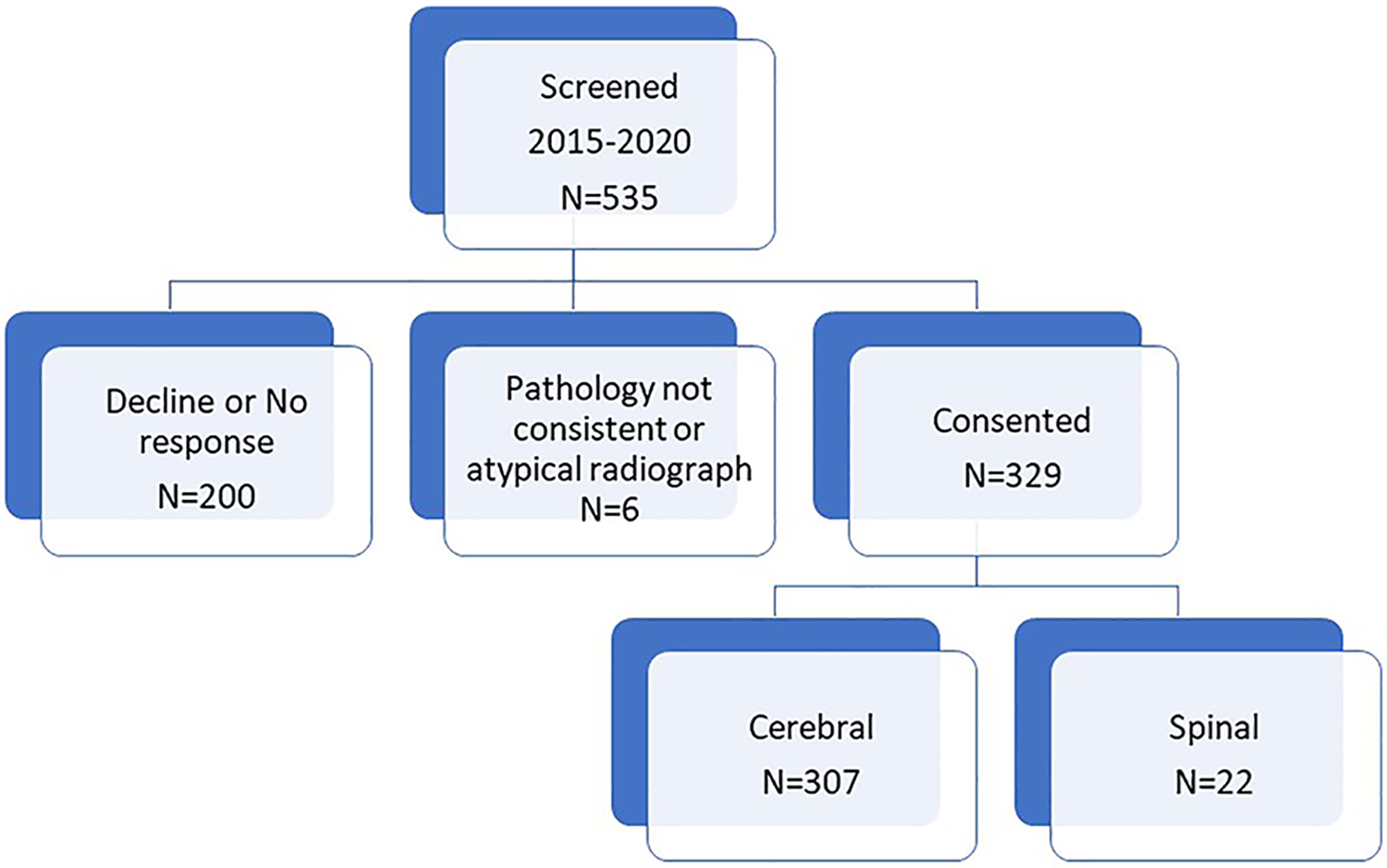
Cohort inclusion (see text).
Demographic, clinical and radiologic data of the cohort.

CM: cavernous malformation; DVA: developmental venous anomaly.
Patients designated as an incidental, non-symptomatic CM.

#These headaches were all deemed either primary headache disorders (most commonly migraine) without CM hemorrhage or an alternative cause other than the CM was found.
Medical data and/or written survey follow-up in the year 2020 was obtained in 261 patients (79.3%). 86% of patients returned at least one written survey after diagnosis and 56% returned multiple written surveys. 75.4% of patients were also seen in person at least one or multiple times by two of the authors (KDF; GL).
The total follow-up time from the time of diagnosis to time of first censor (first prospective hemorrhage, complete excision of a sporadic form CM, or death), was 1799.9 patient years. In that time, there were 92 prospective symptomatic hemorrhages. From the time of diagnosis to time of censor, 154 (46.8%), 29 (8.8%), and 18 (5.4%) of patients reported using NA-NSAIDS, triptans, or OnabotulinumtoxinA respectively. None of these medications were associated with an increased risk of prospective hemorrhage (Table 3). Age at presentation, initial presentation with hemorrhage and brainstem location were associated with increased risk of prospective hemorrhage as is consistent with prior literature. 16 –18
Prospective hemorrhage risk.

NA-NSAID: non-aspirin non-steroidal anti-inflammatory drugs.
*indicates significant P<0.05
Patients who reported taking NA-NSAIDS were more commonly women, had sporadic form CM, and a higher likelihood of having an incidental CM (Table 4). Patients who initially presented with hemorrhage took NA-NSAIDS at an unknown time interval after hemorrhage (n = 55). Patients taking NA-NSAIDS were more likely to have a chronic inflammatory disease 19 compared to those not taking NA-NSAIDs (48; 32.0% versus 37; 2.9%; p = 0.041) and were more likely to be taking concomitant vitamin D supplementation compared to those who reported not taking NA-NSAIDs (85; 55.2% versus 72; 41.1%; p = 0.011).
Characteristics of patients who used NA-NSAIDS after the CM diagnosis but before censor date.

*indicates significant P<0.05
NA-NSAID: non-aspirin non-steroidal anti-inflammatory drugs.
Patients who reported use of triptan medications (n = 29) after diagnosis of a CM were more commonly women and less likely to have had a prior CM hemorrhage (Table 5). Patients taking triptans more commonly used concurrent beta-blockers compared to those not taking triptan (9, 31.0% beta-blocker use in triptan users versus 29, 9.7% beta-blocker use in non-triptan users; p = 0.0006). There were seven patients who used triptans after initial CM presentation with hemorrhage, three of whom had a prospective hemorrhage. None of these three patients had CM hemorrhage temporarily related to a dose of triptan. Of the 22 patients not presenting with hemorrhage who used triptans, 2 had a prospective hemorrhage, none temporarily related to a dose of triptan.
Characteristics of patients who used triptans after the diagnosis of CM but before the censor date.
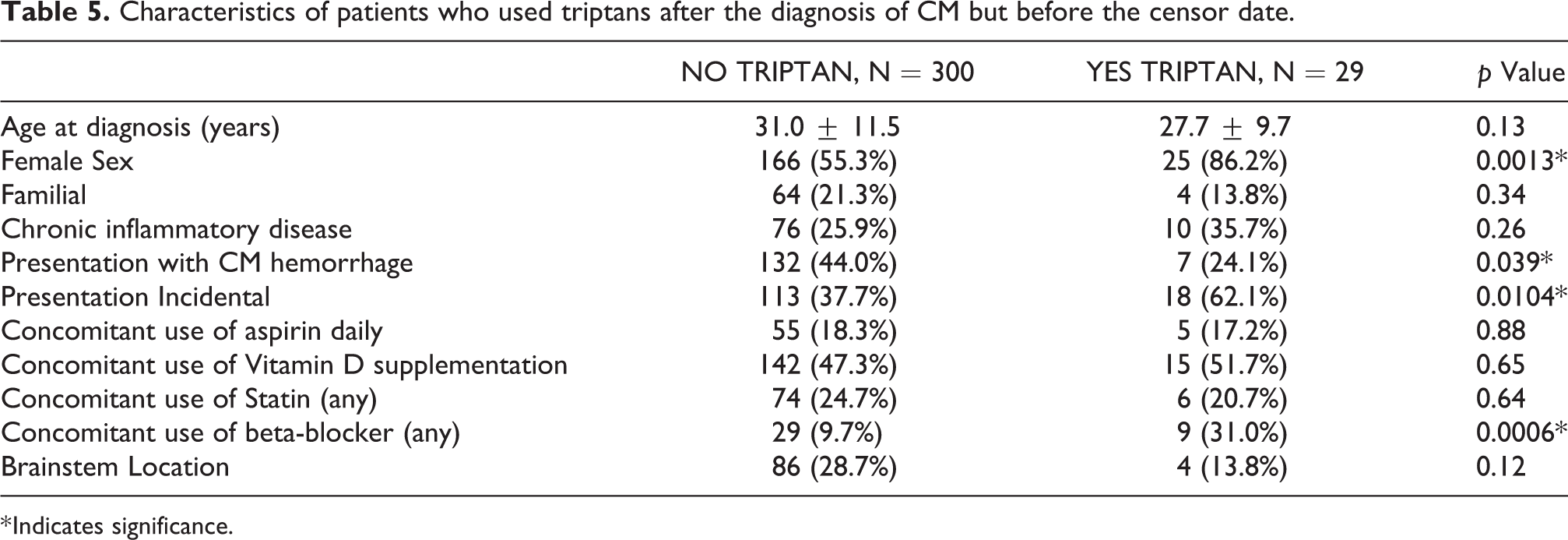
* Indicates significance.
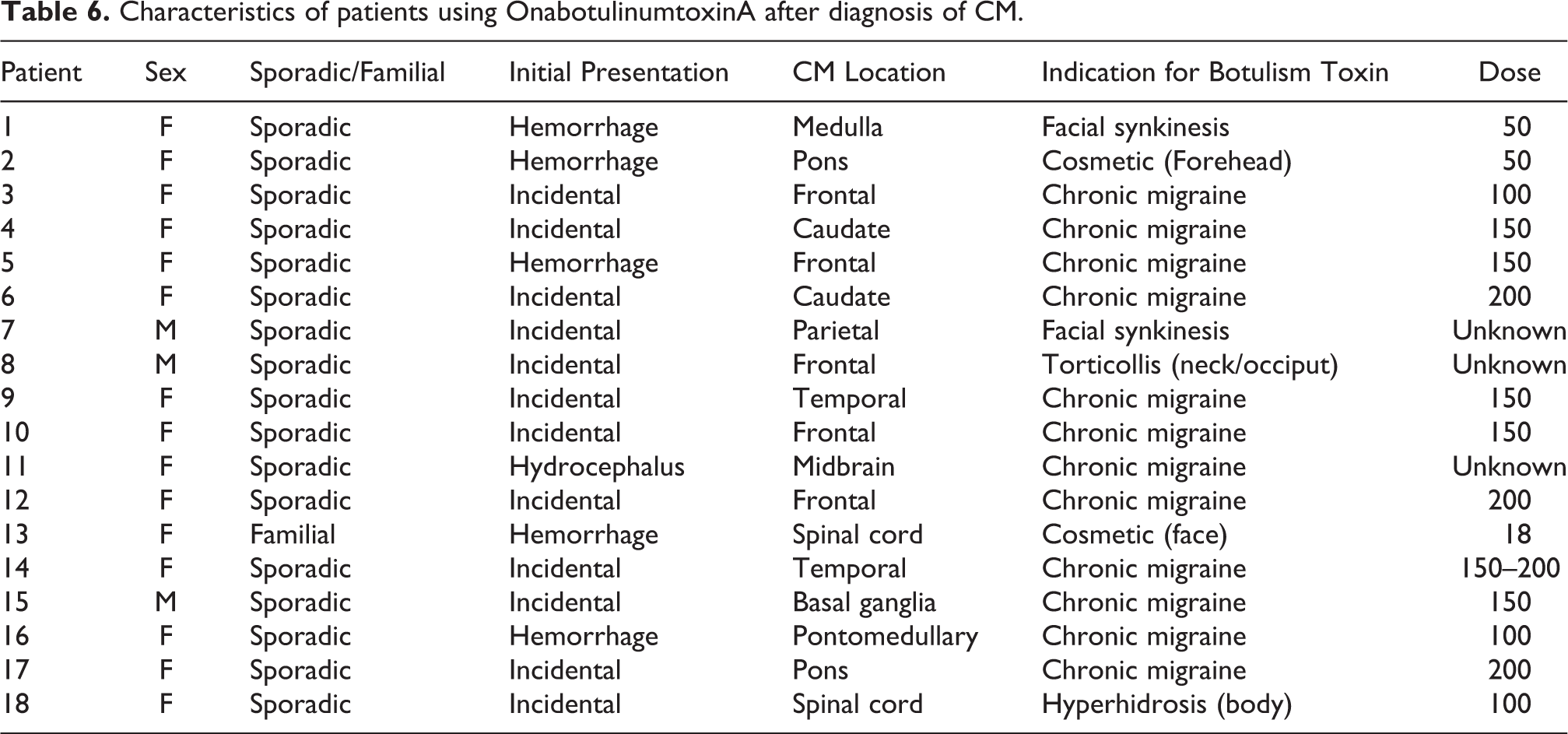
Eighteen patients used OnabotulinumtoxinA after the diagnosis of CM. Of these patients, the majority (n = 15) were women and only one patient had familial CM (Table 6). Five of these patients initially presented with CM hemorrhage, in 12 patients the CM was an incidental finding and 1 patient presented to medical attention with hydrocephalus. Indications for use of botulism toxin included: chronic migraine (12), cosmetic (2), facial synkinesis (2), torticollis (1) and hyperhidrosis (1). Total doses ranged from 18 units to 200 units with the most common dose for migraine patients being 150 units. None of these patients had a prospective hemorrhage after diagnosis.
Characteristics of patients using OnabotulinumtoxinA after diagnosis of CM.
We saw a trend toward patients taking beta-blockers having a lower risk of prospective hemorrhage (6; 6.5% versus 32; 13.5% p = 0.08). Only 11 of 38 patients taking beta-blockers were propranolol and there was no significant difference in prospective hemorrhage risk for those taking propranolol.
Discussion
Patients with cerebral or spinal CM present a unique population of patients in whom primary headache management may be difficult due to concerns over the safety of select medications. Our data suggest that NA-NSAIDS and triptans, taken intermittently, did not precipitate CM hemorrhage in this cohort. While the numbers of patients receiving OnabotulinumtoxinA were limited (n = 18), this medication did not precipitate CM hemorrhage at doses most commonly under 200 units per session.
NA-NSAIDs inhibit platelet thromboxane thereby reducing thrombotic activity and inhibit production of prostacyclin in endothelial cells thus increasing thrombotic activity. This has led to concerns of NA-NSAIDs increasing the risk of both ischemic stroke and intracerebral hemorrhage. 20 Studies assessing the risk of ischemic stroke or intracerebral hemorrhage in patients taking NA-NSAIDS for headache have yielded mixed results and are confounded by variable methodology, type of population studied, and types of NA-NSAIDs used. It is unknown if NA-NSAIDs influence CM bleeding, however patients are often cautioned about their use. 21 Contrary to this concern, limited CM animal data suggests potential benefit from one NSAID, sulindac. In that study, sulindac reduced the likelihood of CM formation in CCM3 knock-out mice. 22 In the present cohort, we similarly did not find that NA-NSAIDS precipitate CM hemorrhage in patients with known CM, and in fact, there was suggestion of benefit. That is, most patients taking NA-NSAIDs (80.5%) did not have a prospective hemorrhage after diagnosis. We cautiously interpret the potential benefit of NA-NSAIDS in reducing hemorrhage risk because the duration, dose, and frequency of NA-NSAID use were not specifically assessed. In addition, the risk of prospective hemorrhage in patients with incidental CM is significantly lower than those presenting with hemorrhage, which may account for some but not all benefit. Patients taking NA-NSAIDS were also more commonly taking vitamin D supplementation which has been suggested in preclinical and clinical data to play a potential protective role. 23 –25 Because many of the patients reporting use of NA-NSAIDs (48.7%) had presented with an incidental CM, our data may be best extrapolated to a similar demographic of patients; however, we do note that even among patients who used NA-NSAIDS after a prior CM hemorrhage, the risk of prospective CM hemorrhage remained low. We did not assess the timeframe at which a patient started taking NA-NSAIDS after a hemorrhage; however, it is our practice to avoid NA-NSAIDS in the immediate 3 to 6 months post-hemorrhage before considering intermittent, infrequent use (single standard doses less than four times monthly). We do not advocate daily or prolonged courses of NSAIDs because of remaining uncertainties in addition to other systemic side effects of such medications.
Triptans are 5-HT1B/1D-receptor agonists, and it is believed that triptans alleviate migraine by several mechanisms. Because of the potential for precipitating vasospasm, there has been concern regarding triptan use in patients with stroke or vascular disease. Cerebral hemorrhages and subarachnoid hemorrhages are listed in “warnings and precautions” in the package insert of most triptans. However, the relationship between triptans and intracranial hemorrhage remains debatable, as in most reported cases, the hemorrhagic events were misdiagnosed with migraine and treated with triptans rather than triptans being the direct cause of the cerebral hemorrhages. 26 Since CM are composed of endothelial lined cavernous that are devoid of smooth muscle, CM endothelial cells should not be prone to vasospasm. In addition, there are no reports of triptans precipitating CM hemorrhage. Our data support the safety of patients with CM using triptans after the CM diagnosis. The timeframe after hemorrhage in which patients used triptans was not assessed in our study.
OnabotulinumtoxinA reduces the frequency and severity of migraine through multiple potential mechanisms. 27 The PREEMPT protocol for chronic migraine recommends 155 to 195 Units of OnabotulinumtoxinA to be injected into 31 to 39 sites in seven muscles of the head and neck every 12 weeks. 28 It is thought to have minimal systemic side effects and drug-drug interaction. A recent case report has raised concern over the safety of OnabotulinumtoxinA in a patient with CM. 4 In that report, a 32-year-old patient with familial CM received increasing doses of OnabotulinumtoxinA for chronic migraine headache and had a first hemorrhage 8 days after a dose of 215 units. The patient resumed injections about 3 months after the initial CM hemorrhage. Patient had a second CM hemorrhage approximately 15 days after receiving 235 units of OnabotulinumtoxinA. Authors suggest the mechanism by which botulism toxin increases the risk of hemorrhage relates to the TLR2 inflammatory cascade that converges to a common pathway with the TLR4 path. The TLR4 inflammatory cascade has been recently implicated in the pathogenesis of CM. 29 In our cohort, we did not observe CM hemorrhage after OnabotulinumtoxinA injection in our 18 patients. This includes six patients who had initially presented with hemorrhage. The doses in our patients were below those administered in the case report.
Our study has several limitations. First, to detect a 5 to 10% increase in hemorrhage risk at 80% power, approximately 150 to 300 patients would be needed in each group, those taking versus not taking a medication. Therefore, our study may be underpowered for small changes in hemorrhage risk. Given the rarity of the disease, our cohort is the largest reporting on such data, and we are reassured that the select medications do not seem to immediately precipitate hemorrhage. The type, dose, duration of use and frequency of use for the medications was not assessed and would be difficult to collect based on recall bias. The EMR has limitations in gathering accurate medication lists. We thus obtained medication information from four different sources: a written survey, the neurologic evaluation at our institution, clinical notes, and searching EPIC Care Everywhere for specific terms. All but three patients received their OnabotulinumtoxinA doses at our institution, so procedures and dosing were captured. In those patients who initially presented with hemorrhage, we did not assess the time from hemorrhage to the timing of first taking the select medications due to recall bias. We thus urge caution in measuring the duration of the acute hemorrhagic period as to the applicability of this data. Notwithstanding these limitations, our study is the first to address the safety of common headache medications in a large, prospective cohort of patients with CM.
In conclusion, we found that NA-NSAIDS, triptans, and OnabotulinumtoxinA therapy (doses 200 units or less per session) did not precipitate hemorrhage in patients with CM in this cohort and can be considered in select patients with CM for management of primary headache disorders. Further data from ongoing registries may help assess ongoing safety of these and newer migraine medications in patients with CM.
Clinical implications
Primary headache disorders are common in patients with cavernous malformation (CM).
NA-NSAIDs and triptans do not increase CM hemorrhage in the patients studied.
Limited data exists on the use of OnabotulinumtoxinA in patients with CM
Our data do not show an increase in CM hemorrhage at doses <200 units.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
