Abstract
Background:
The migraine-specific monoclonal antibody Erenumab targeting the calcitonin gene related peptide receptor is an effective and well tolerated preventive treatment of episodic and chronic migraine. However, its price limits its use as a first line therapy against migraine. Therefore, identifying patients who will adequately respond to such treatment is paramount.
Methods:
In this retrospective, real-life cohort study, 172 adult patients with refractory episodic or chronic migraine treated with Erenumab were included. To identify the predictors of response to Erenumab, bivariate subgroup analysis of several potential factors was performed, and multivariate logistic regression modeling was done to obtain Odds Ratio (OR).
Results:
Of the 172 patients, 57.0% achieved a successful treatment response (reduction of monthly migraine days by ≥50%). Statistically significant predictors of a treatment response were the presence of chronic migraine, tension-type headache, and a positive response to triptan with an odd ratio of 0.473 (95% CI, 0.235–0.952), 0.485 (95% CI, 0.245–0.962) and 3.985 (95% CI, 1.811–8.770), respectively (P < 0.05).
Conclusions:
Successful Erenumab treatment response rate was 57.0% in this retrospective cohort. As chronic migraine and tension-type headache were negative predictors of Erenumab response while triptan response was a positive predictor, this data suggests the potential for Erenumab monotherapy without the need for traditional preventive treatment in refractory migraine sufferers improving side effect profile and treatment adherence for a cohort of patients difficult to treat.
Introduction
Several preventive treatments are available for patient suffering from episodic and chronic migraine. 1 –6 However, many patients do not improve with the traditional preventive therapies and see their quality of life deteriorate. The treatment with Onabotulinumtoxin A (BTX) injections and, more recently, Erenumab showed improvement in the management of patients with refractory chronic migraine. 7,8
Erenumab is a humanized monoclonal IgG2 antibody anti-calcitonin gene related peptide (CGRP) receptor approved for migraine prevention. Erenumab reduced the monthly migraine days (MMD) in patients with episodic and chronic migraine who failed at least one preventive treatment (including the ARISE, STRIVE and LIBERTY clinical trials). 8 –11 While there was a high success rate in achieving at least 50% reduction in MMD, some patients had over 75% reduction in MMD while others failed to respond to the treatment. 11 A post-hoc analysis of two pivotal studies 8,9 demonstrated that patients with episodic and chronic migraine treated with Erenumab demonstrated a sustained effect of reduced migraine attacks as early as 1 week after the first monthly injection. 12
While the efficacy and long-term tolerability of Erenumab is supported by the current literature, 8 –11 its price limits its use as a first line therapy against migraine. Therefore, identifying patients who will adequately respond to such treatment is a judicious health care spending. To our knowledge, there are no formal studies evaluating the predictors of response to Erenumab. To date, only one recent report showed a positive association between a triptan response and a successful Erenumab treatment. 13 Further, in patients with chronic migraine treated with BTX injections, cranial allodynia, unilateral migraine and short disease duration, absence of depression, and absence of medication overuse were associated with a positive treatment response, although these findings were not consistent across studies. 14 –18 Thus, this study aimed to identify predictors of treatment response to Erenumab in a cohort of patients with episodic and chronic refractory migraine.
Methods
Subjects
This retrospective cohort study included 172 Quebec City patients who attended the neurology clinic between December 2018 and January 2020 for either episodic or chronic migraine and were treated with Erenumab (70 mg or 140 mg monthly). Eligible patients were aged at least 18 years of age with episodic and/or chronic migraine for at least 1 year, who failed to respond to at least two of the following approved oral preventive treatments: propranolol, metroprolol, topiramate, valproic acid and/or amitriptyline and returned for a 3-month follow-up after the first monthly injection of Erenumab.
We chose a 3-month time point because it has been previously demonstrated in randomized controlled clinical trials that patients with either episodic and chronic migraine showed a successful treatment response at this end-point. 8,10,11,19 Patients receiving either a 70 or 140 mg dose of Erenumab and those with chronic migraine who previously (but not during Erenumab treatment) benefited from BTX were included. We included patients who had both doses of Erenumab as there was no significant difference in the response rate to Erenumab treatment with the 70 mg or the 140 mg dose in patients with episodic and chronic migraine. 8,9,19
Patients were asked to log prospectively on a personal calendar taking a migraine-specific abortive drug during an episode of aura and/or headache as well as their daily migraines with characteristics fulfilling the ICHD-3 criteria. The baseline MMD represent the MMD days in the month preceding the first dose of Erenumab.
A successful treatment response required at least 50% reduction in MMD during the third month of the treatment. Patients with medication-overuse (fulfilling the ICHD-3 criteria and who used either NSAID, acetaminophen, triptan, or opioid alone or in combination) before the start of Erenumab were also included in the analysis.
Patients with cluster headache or other trigeminal autonomic cephalalgia, hemiplegic migraine or a new preventive treatment 2 months before Erenumab treatment were excluded from the analysis.
Objectives and endpoints
In this retrospective cohort study, we first ought to determine the proportion of patients who achieved over 50% reduction in monthly migraine days (MMD) during the third month of the treatment in all the 172 patients. This endpoint was considered as a successful response to Erenumab, in agreement with the primary 11 or secondary 8 –10 endpoints of the pivotal randomized controlled trials. A failure to treatment was considered if there was less than 50% reduction of MMD in the third month. MMD were assessed on daily self-reported migraine days during the follow-up evaluation at 3 months. A headache day was included where patients noted headaches with migraine characteristics fulfilling the ICHD-3 criteria or if the patient took a migraine-specific abortive drug during an episode of aura and/or headache.
Comorbidities were obtained from a retrospective chart review. To identify the predictors of response to Erenumab treatment, patients were stratified by: age, sex, years since diagnosis, type of migraine (episodic or chronic), characteristics of migraine (presence of aura, unilateral or pulsatile migraine), response to triptan (with at least three migraine attacks treated to reach a headache-free state within 2 h after treatment), prior BTX treatment response, concomitant preventive treatment (either propranolol, metroprolol, topiramate, valproic acid and/or amitriptyline), medication overuse (either NSAID, acetaminophen, triptan, or opioid alone or in combination), past traumatic brain injury, fibromyalgia, depression, obesity (body mass index or BMI ≥ 30 kg/m 2 ), occipital neuralgia (only if an occipital block previously alleviated their symptoms), cervicalgia, tension-type headache, and a family history of migraine.
Standard protocol approvals, registrations, and patients consents
The institutional ethics committee (Comité d’éthique de la recherche du CHU de Québec) reviewed and approved the study protocol. Specific written consent was not required for this retrospective study.
Statistical analysis
Descriptive statistics included the median, interquartile range, frequencies, and percentages. For continuous data comparisons authors employed the Wilcoxon-Mann-Whitney test while the Pearson chi-squared exact tests were used for categorical data. Univariate and multivariate (adjusted for age, sex, and dose of Erenumab treatment) logistic regression modeling was done to estimate the Odds Ratio (OR) along with the sociodemographic and clinical factors influence on the outcome: achieve at least 50% reduction in MMD. Statistical analyses were performed using SAS Statistical Software v.9.4 (SAS Institute, Cary, NC, USA), with a two-sided significance level set at p < 0.05. In the subgroup analysis of patients in the medication overuse, obesity, and occipital neuralgia groups, 1 patient information was missing. In patients in the unilateral migraine and pulsatile groups, 2 patients’ information was missing. They were not included in the analysis.
Results
Of 172 study patients, 89.5% were women. Table 1 shows the baseline characteristics of the cohort.
Baseline characteristics of the cohort.

The median age of the study patients was 42.5 years old and the median age since the first diagnostic of either episodic (n = 55) or chronic (n = 117) migraine was 22 years. Seventy-seven patients (44.8%) had at least one concomitant preventive treatment whilst being treated with Erenumab (59 patients had 1 (34%) while 18 had two preventives (10.5%)). In terms of migraine characteristics: 54 (31.4%) patients had migraine with aura, 116 (67.4%) patients had migraine with unilateral headache, 131 (76.2%) patients had migraine with a pulsatile headache and 146 (84.9%) study patients were treated with a triptan while 17 (10%) patients were previously treated and failed to respond to Onabotulinumtoxin A (BTX). Thirty-nine (22.7%) patients had a medication-overuse. In this cohort, 29 (16.9%) patients had a diagnosed depression, 9 (5.2%) with fibromyalgia, 47 (27.3%) with obesity, 14 (8.1%) with traumatic brain injury, 40 (23.3%) with great occipital neuralgia, 66 (38.4%) with cervicalgia, 48 (27.9%) with tension headache, and 121 (70.3%) with a family history of migraine. There was missing information from the chart of 1 patient in the medication overuse, obesity, and occipital neuralgia groups and 2 patients from the unilateral and pulsatile groups.
Of the 172 patients, 57.0% achieved a 50% reduction in MMD following treatment with Erenumab. Forty-three percent of patients (n = 74) achieved less than 50% reduction in MMD, which was considered a failure to treatment.
A bivariate analysis (Pearson exact Chi-squared test and Wilcoxon-Mann-Whitney test) was performed to determine which migraine characteristics or comorbidities were associated with either a good response or Erenumab treatment failure. In Table 2, we compared the group of patients with a successful treatment response to Erenumab (at least a 50% reduction in MMD) to those who failed Erenumab treatment (less than 50% reduction in MMD).
Characteristics and proportion of patients with at least 50% reduction in MMD.
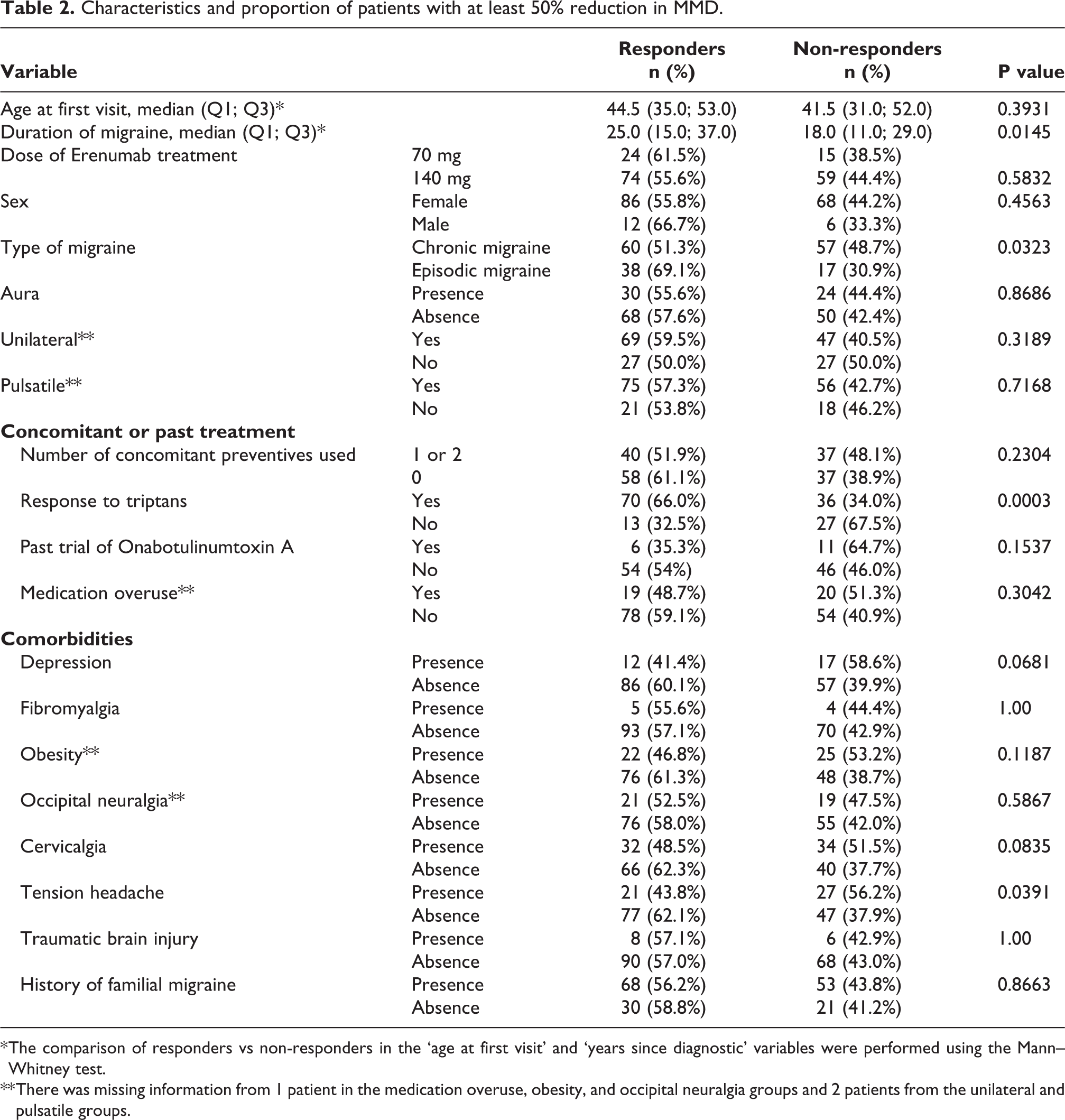
* The comparison of responders vs non-responders in the ‘age at first visit’ and ‘years since diagnostic’ variables were performed using the Mann–Whitney test.
** There was missing information from 1 patient in the medication overuse, obesity, and occipital neuralgia groups and 2 patients from the unilateral and pulsatile groups.
Patients with episodic migraine versus chronic migraine (69.1% vs 51.3%, respectively, P = 0.0323), without tension-type headache versus with (62.1% vs 43.8%, respectively, P = 0.0391) and those that responded to triptan medication versus non-responders (66.0% vs 32.5%, respectively, P = 0.0003) were significantly more likely to benefit from Erenumab treatment.
The odds ratio estimated by multivariate logistic regression adjusted for age, sex and dose of Erenumab treatment for a 50% decrease in MMD with the Erenumab treatment in patients with chronic migraine, tension-type headache and those who respond to triptan were 0.473 (95% CI, 0.235–0.952), 0.485 (95% CI, 0.245–0.962) and 3.985 (95% CI, 1.811–8.770, respectively (see Figure 1). An odd ratio below 1 is associated with a negative response to Erenumab.

Factors studied evaluating their association with the response to Erenumab treatment, their odds ratio and 95% confidence intervals. Multivariate logistic regression modeling was performed (adjusted for age, sex, and dose of Erenumab treatment). Univariate logistic regression modeling was done to estimate the odds ratio for the age, sex, and dose of Erenumab factors. Odds ratio superior to 1 are associated with a successful Erenumab response while values inferior to 1 are associated with a failed treatment response. P < 0.05.
All the other migraine characteristics, age, duration of migraine, comorbidities, or the concomitant use of either one or two preventive treatments did not show a significant association in the treatment response to Erenumab. However, there was a trend toward reduced Erenumab treatment success (slight reduction) in patients with depression or cervicalgia.
Discussion
In this real-life retrospective cohort study of 172 patients, Erenumab treatment decreased the MMD by at least 50% in 57.1% of patients. A recent real-life retrospective study of 43 patients showed a similar treatment success rate (54.8%) at the 6 months of follow-up. 20 Patients with episodic migraine showed a higher proportion of treatment success when compared to chronic migraine (69.1% vs 51.3%, respectively). Furthermore, patients suffering from chronic migraine or tension-type headache had reduced treatment success to Erenumab at 3 months whereas the opposite was true in patients who respond to triptan. Interestingly, patients with cervicalgia or depression seem to respond less frequently to Erenumab, although the results were not significant. The concomitant use of preventive treatments did not differ between the responders and non-responders. To our knowledge, this is the first study evaluating several predictors of response to Erenumab.
There are few reports evaluating the predictors of response to BTX in patients with chronic migraine and only one evaluating the association between triptan and Erenumab treatment response. In a previous retrospective observational study in patients with multiple complex pain problems and chronic migraine treated with BTX, depression and pain interference were associated with decreased treatment response. 21 A prospective study of 84 patients with refractory chronic migraine treated with BTX for 1 year also showed that depression and medication overuse were associated with a reduced response to treatment. 22 Our study demonstrated similar findings in patients with depression in term of treatment response to Erenumab, but we did not observe this association in patients with medication overuse.
A study of an adolescent cohort showed that the persistence of migraine was associated with obesity and adiposity whereas a weight loss intervention was associated with a better migraine outcome. 23,24 Moreover, higher BMI was shown to be associated with higher migraine frequency and severity. 25 This study showed that obesity did not alter the response to Erenumab in patients with at least 50% reduction in MMD. Therefore, Erenumab could be a valuable alternative in patients with obesity and refractory migraine, in addition to weight loss targeted to decrease future cardiovascular risk factors.
Our results showed an association between a positive triptan response and a successful Erenumab treatment which is reflected in a single study earlier this year. 13 The failure of triptans was also associated with a lower response to BTX 26 in a small cohort study of 44 patients with refractory chronic migraine. Thus, the response to triptan in patients with migraine seems to be an important factor in predicting the successful response to a preventive treatment. Since one of the actions of triptan is to inhibit the release of pro-inflammatory neuropeptides including CGRP, and the effect of Erenumab in antagonizing the binding of CGRP to its receptor is another way in reducing the action of this migraine-inducing neuropeptide, the mechanism supports this association. Furthermore, cranial allodynia was shown in an observational study of acute migraine sufferers to be associated with a failure to triptan treatment. 27 Although not studied in this cohort, cranial allodynia should be evaluated and considered in potential candidates for Erenumab therapy as they might not properly respond to Erenumab knowing the negative association between allodynia and the response to triptan.
As this study was done retrospectively, it can-not evaluate beyond a reasonable doubt that the predictors of response to Erenumab are specific to this treatment. Methodologic limitations mean we can-not exclude that the response to triptan may have a confounding effect in the response to Erenumab treatment. To minimize these limitations, a double-blind randomized control trial is required.
Our cohort is composed of patients who suffered from a higher frequency and duration of migraine compared to the population studied in the clinical trials to date studying the effect Erenumab. 28 Despite this selection bias, the treatment success rate was similar to these randomized controlled trials supporting the idea that responders to Erenumab are also found in patients more severely affected by migraine.
Of note, patients with chronic migraine may suffer from tension headache semiology pain, therefore, to ensure the prevalence was similar among both migraine groups we reported patients with tension headache that was diagnosed independently from their migraine and compared the prevalence. The results demonstrated a similar prevalence (25% and 27%, respectively) of tension-type headache in patients suffering from episodic migraine compared to chronic migraine.
In this cohort, there is a high prevalence of occipital neuralgia. As the study is retrospective, it is possible that some patients may actually have suffered from an occipital headache versus true occipital neuralgia confounding the results. This being said, in this cohort, Erenumab seems to be equally effective in patients with or without occipital neuralgia and should not be delayed in patients with a concomitant migraine in addition to first line nerve block for the treatment of primary occipital neuralgia.
Seventy-seven patients maintained their preventive treatment during this study and this subgroup did not show an increase in the rate of response to Erenumab. This may suggest that Erenumab acts independently from the traditional preventive treatments used, whose mechanism of action in the prevention of migraines remains unclear. Although we did not show a synergistic effect in the prevention of migraine when combining Erenumab with another preventive agent, this possibility can-not be ruled out based on this study alone. Therefore, clinicians might consider gradually weaning other preventive treatments during Erenumab where possible on a case-by-case basis to decrease their side effects profile.
It is important to note the success of Erenumab treatment in patients with bilateral migraine and longer disease duration, presence of depression, and medication overuse in this study, as these characteristics are negatively associated with the response to other alternatives such as BTX treatment. 27 Our study suggest that Erenumab could be an effective alternative in patients with these characteristics. Unfortunately, patients with fibromyalgia (and chronic fatigue, data not shown) were not prevalent enough to evaluate appropriately.
Conclusions
The success rate of Erenumab treatment response in our retrospective cohort study of 172 severely affected refractory migraine patients in Quebec City was 57.1%, similar to previous randomized controlled trials. A positive triptan response was a positive predictor of response to Erenumab in this cohort while chronic migraine and tension-type headache (to a lesser extend patients with cervicalgia and depression) were negatively associated with a successful Erenumab treatment response. Further prospective research is needed to fully explain these findings. As the study results showed no synergistic effects with concomitant additional preventive migraine treatment, these agents could be gradually weaned during Erenumab treatment to reduced unwanted side effects. By providing clinicians with objectives factors needed to identify patients likely responsive to Erenumab therapy, this study will improve patient care in two ways: (1) It will allow early targeted therapy thus avoiding the negative side effects of traditional preventive therapies in patients likely to respond to Erenumab and (2) with effective targeted migraine treatment, potentially prevent patients from converting from an episodic to a chronic state of migraine.
Footnotes
Clinical implications
Patients with refractory episodic and chronic migraine should be treated with an anti-CGRP receptor antibody as soon as possible. A positive response to triptan was associated with a favorable response to Erenumab treatment. Negative predictors of response to Erenumab include chronic migraine and tension-type headache. A monotherapy with Erenumab should be favored to minimize the side effects of traditional preventive therapies.
Acknowledgments
We would like to acknowledge Michèle Tanguay, nurse, for her help collecting the data and Jennifer Charlesworth, MD, MSc, for comments on the manuscript.
Author contributions
H.S.-A.: drafting and revision of the manuscript for content, including medical writing for content; major role in the acquisition of data; study concept or design; analysis and interpretation of data. D.S.: analysis or interpretation of data; additional contributions: statistical analysis of the data and writing of the statistical section of the methods. J.P.: revision of the manuscript for content, including medical writing for content; major role in the acquisition of data; study concept or design; analysis and interpretation of data.
Availability of data and materials
Anonymized data can be made available to investigators upon request to the corresponding author.
Competing interests
J.P. reports personal fees from Novartis during and outside of the work submitted. H.S.-A. reports no disclosures. D.S. reports no disclosures.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics approval and consent to participate
The institutional ethics committee (Comité d’éthique de la recherche du CHU de Québec) reviewed and approved the study protocol. Specific written consent was not required for this retrospective study.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
