Abstract
Objective:
The efficacy of galcanezumab was evaluated in patients with episodic cluster headache and history of preventive treatment failure.
Methods:
In the randomized, 8-week, double-blind study (CGAL), patients with episodic cluster headache received once-monthly subcutaneous injections of galcanezumab 300 mg or placebo. Patients who completed CGAL and enrolled in an open-label study were queried for preventive treatment history. In a subset of patients with a known history of failure of verapamil or any other prior preventive treatment, a post hoc analysis of least square mean change from baseline in weekly cluster headache attack frequency across Weeks 1 to 3 was assessed.
Results:
Fifteen patients provided data for known history of prior preventive treatment failure (6 placebo, 9 galcanezumab), of whom 11 failed verapamil. The mean reduction in the weekly frequency of cluster headache attacks was greater with galcanezumab compared to placebo among patients with prior preventive treatment failure (8.2 versus 2.4); mean difference 5.8 (95% confidence interval [CI] 2.0, 13.6) and among patients with verapamil failure (10.1 versus 1.6); mean difference 8.5 (95% CI 0.4, 16.7).
Conclusion:
In this exploratory analysis of patients with a known history of prior preventive treatment failures, treatment with galcanezumab resulted in greater mean reductions in weekly cluster headache attacks compared with placebo.
ClinicalTrials.gov:
NCT02397473 (I5Q-MC-CGAL)
NCT02797951 (I5Q-MC-CGAR)
Introduction
Cluster headache is a primary headache disorder characterized by attacks of intense unilateral headache and the frequent association of autonomic symptoms such as lacrimation, conjunctival injection, and nasal congestion. Episodic cluster headache (ECH) is a headache disorder in which cluster headache attacks occur in periods lasting from 7 days to 1 year, separated by pain-free periods lasting at least 3 months. 1 Patients with ECH can experience multiple excruciatingly painful unilateral headache attacks a day. 2 Consequently, this disease has a tremendous negative effect on overall functioning in occupational, educational, social, and daily activity domains of life. 3 Patients with ECH are under-prescribed preventive treatment and/or have low persistence and adherence to treatment regimens, in part due to efficacy and tolerability issues. 4 Most preventive medications described in the American Headache Society Guidelines have a Level C recommendation, including verapamil, which is the most commonly prescribed medication to date for cluster headache. 5,6
Preventive treatment failure is common in primary headache disorders. Patients with migraine disease who have a history of prior preventive treatment failure have described isolation, depression, difficulties in sleeping, and limitations on daily activities. 7 Though there is limited research specifically detailing challenges in this uniquely burdened population of patients with ECH who have experienced preventive treatment failure, clinical experience suggests many of the same challenges exist.
Galcanezumab is a humanized IgG4 monoclonal antibody that binds calcitonin gene-related peptide (CGRP) and prevents its biological activity without blocking the CGRP receptor and is indicated for the preventive treatment of migraine and treatment of ECH in the US. 8 In the Phase 3, double-blind, placebo-controlled study I5Q-MC-CGAL (hereafter, Study CGAL), after initial injection with galcanezumab, patients with ECH experienced significantly greater reduction in cluster headache attack frequency across Weeks 1 to 3 compared to placebo-treated patients. 9 However, it is unknown how patients with ECH having a history of prior preventive treatment failure will respond to galcanezumab.
The current study is a post hoc analysis of the efficacy of galcanezumab in a subset of Study CGAL patients with ECH and a known history of prior preventive treatment failure due to no or inadequate treatment response or intolerance to the medication. 9 In this exploratory study sample, the majority of patients with a known history of prior preventive treatment failure had failure of verapamil. As such, an additional stratification was performed to describe the efficacy of galcanezumab in the subgroup with a known history of prior verapamil failure to further inform future treatment strategies for a common clinical scenario observed in the management of ECH.
Methods
Study design
A subset of data of patients with ECH and a history of known prior preventive treatment failure from a Phase 3, double-blind, placebo-controlled Study CGAL were analyzed to characterize the efficacy of galcanezumab in this distinctive population (Figure 1). Patients who completed the double-blind and post-treatment follow-up phases of Study CGAL were eligible to enter the open-label safety study I5Q-MC-CGAR (hereafter Study CGAR) and receive galcanezumab 300 mg monthly. The subset of patients evaluated in the current post hoc analysis included those patients with a known history of failure of (1) verapamil and (2) any prior preventive treatment (including verapamil) that was due to no or inadequate response or intolerance to the medication. The protocol for Study CGAL did not include the collection of history of use or failure of any prior preventive treatments. Thus, during a point within Study CGAR, a query for the history of preventive treatment failures in patients with ECH was initiated. At that point in time, information regarding the history of preventive treatment failures was retroactively collected from patients currently participating in Study CGAR. In other words, not every patient who entered Study CGAR provided data for the history of preventive treatment failures as it was not initiated at the beginning of the open-label safety study.
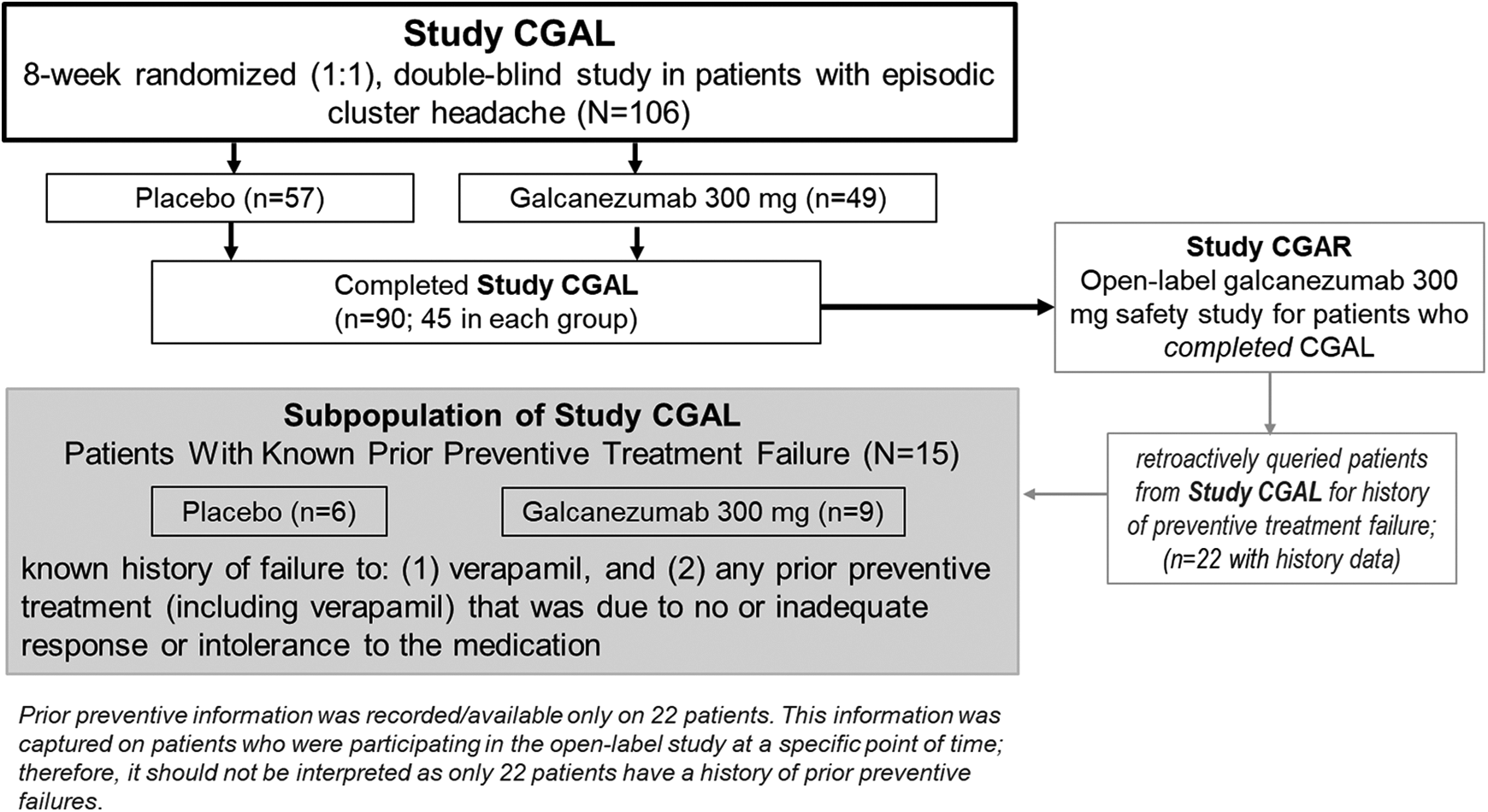
Study design.
The design and results of Study CGAL have been detailed previously. 9 In brief, patients enrolled in the study were 18 to 65 years of age and had a history of ECH with cluster headache attacks that were distinguishable from other headache disorders. 1,9 Further, patients were required to have ≥1 cluster headache attack every other day, ≥4 total cluster headache attacks, ≤8 attacks per day during 7 consecutive days of the prospective baseline period, and a history of cluster headache period that had lasted ≥6 weeks. Patients were allowed to use only the following medications to treat their cluster headache attacks: subcutaneous, intranasal, or oral triptans; high-flow oxygen; acetaminophen (paracetamol); and nonsteroidal anti-inflammatory drugs. No concomitant preventive medications for cluster headache were permitted. 9
Patients were randomized (1:1) to subcutaneous injections of galcanezumab 300 mg/month or placebo. During the prospective baseline and double-blind treatment periods, patients used electronic diaries daily to record the number of cluster headache attacks (regardless of attack duration), the average attack severity and duration over a 24-hour period, and the use of acute treatments for attacks. The attack severity was rated on a five-point scale (0 = no pain, 1 = mild pain, 2 = moderate pain, 3 = severe pain, 4 = very severe pain). 10 The primary endpoint for Study CGAL and the endpoint of this current post hoc analysis for the subset of patients with a known failure of a preventive medication was the overall mean change from baseline in the weekly frequency of cluster headache attacks across Week 1 through Week 3. 9
The study protocols were reviewed and approved by the appropriate institutional review board for each of the study sites. The studies were conducted according to Good Clinical Practice and the Declaration of Helsinki guidelines. Patients provided written informed consent before undergoing study procedures. The trials are registered with ClinicalTrials.gov (NCT02397473, Study CGAL and NCT02797951, Study CGAR).
Statistical methods
The objective of this post hoc analysis was to analyze the primary and key secondary outcomes among a subset of patients from Study CGAL with a known failure (either due to lack of efficacy or intolerance to medication) to at least one prior preventive treatment. Reasons that counted as lack of efficacy included inadequate response and no response. Further, these outcomes were also analyzed among patients with a known history of failure of verapamil.
The change from baseline in weekly cluster headache attacks during the first 3 weeks of the double-blind period was analyzed using a mixed model repeated measures (MMRM) analysis. The model included the fixed categorical effects of sex, treatment group, week, treatment group-by-week interaction, and the continuous baseline weekly cluster headache attacks as a covariate. An unstructured covariance matrix was imposed on the residuals to model the correlation among repeated measurements observed on the same patient. The number and percentage of patients with ≥50% reduction in weekly cluster headache attacks from baseline at Week 3 were also summarized.
Baseline characteristics were summarized using means and standard deviations (SD) or medians and quartiles for numeric or ordinal variables and frequency and percentages for categorical variables. Treatment effects from MMRM models are presented using differences in least squares (LS) means between treatment groups and associated 95% confidence intervals (CI). All analyses done were post hoc and thus should be considered exploratory in nature. No adjustments for multiple testing were conducted. All analyses were performed using SAS® Enterprise Guide® Version 7.1.
Results
Patient population
In Study CGAL, 106 patients were randomized to galcanezumab 300 mg (n = 49) or placebo (n = 57). Patients were a mean age of 46.4 years, the majority were men (83%) and white (84.9%), and approximately two thirds of the patients were enrolled in Europe. In the prospective baseline phase, the mean number of weekly cluster headache attacks was 17.3 for placebo and 17.8 for galcanezumab, and the average severity of pain was moderate to severe. Demographic and clinical characteristics of the treatment groups in Study CGAL were well balanced between treatment groups.
Of the 106 patients from Study CGAL, 90 patients completed the study and 52 patients entered Study CGAR. At the time of the prior preventive medication query, 13 had already completed their final treatment visit in Study CGAR leaving 39 patients. Prior preventive treatment information was provided by 22 of the 39 patients. The subset of patients with known prior preventive treatment failure for which history was available included 15 patients: 9 in the galcanezumab group and 6 in the placebo group (Figure 1). As shown in Table 1, the baseline demographics and characteristics of patients with a known failure of prior preventive treatment were similar to the group without known failure of prior preventive treatments (n = 91). Additionally, the 7 of 22 patients who were retroactively queried, but did not have a prior preventive treatment failure, did not differ in demographics or characteristics from the subset of 15 patients included in this analysis. Of the seven patients, four had an adequate response to verapamil while three patients had taken treatments that were not classified as preventives. The subset of patients with a known history of failure of prior preventive treatment (n = 15) had an average of 1.9 treatments failures. This subset had a mean of 16.8 weekly cluster headache attacks and an average severity of pain that was moderate to severe and was similar to the overall Study CGAL group. Of the 15 patients with a known failure of prior preventive treatment, 11 failed verapamil (6 placebo, 5 galcanezumab) for reasons of lack of efficacy (n = 9) and intolerance to medication (n = 2).
Patient demographics and disease characteristics at baseline.

NSAID: nonsteroidal anti-inflammatory drugs; SD: standard deviation.
a Note that most patients were in the “unknown” category.
b0 = no pain, 1 = mild pain, 2 = moderate pain, 3 = severe pain, 4 = very severe pain.
Frequency of weekly cluster headache attacks
In the subset of patients with a known failure of prior preventive therapy, the mean reduction from baseline in the frequency of weekly cluster headache attacks over Week 1 to Week 3 was 8.2 in the galcanezumab group compared with 2.4 in the placebo group; the LS mean difference was 5.8 (95% CI −2.0, 13.6). The reductions observed in the subset of patients treated with galcanezumab were similar to that observed in galcanezumab-treated patients in Study CGAL (8.7). However, the reductions seen in the placebo group (5.2) were minimal compared with those seen in Study CGAL. Starting at Week 1, the LS mean reduction was numerically greater with galcanezumab compared with placebo and increased in magnitude at each subsequent week (Figure 2). Further, in the subset of patients with a known failure of verapamil, the mean reduction from baseline in the frequency of weekly cluster headache attacks over Week 1 to Week 3 was numerically greater in the galcanezumab group (10.1) compared with placebo (1.6), and the LS mean difference was 8.5 (95% CI 0.4, 16.7) (Figure 3).

Frequency of weekly cluster headache attacks among patients with a known failure of any prior preventive therapy. Mixed-model repeated measures: change from baseline in weekly attacks = baseline, sex, treatment, week, and treatment-by-week interaction. CI: confidence interval; LS: least squares.

Frequency of weekly cluster headache attacks among patients with a known failure of verapamil. Mixed-model repeated measures: change from baseline in weekly attacks = baseline, sex, treatment, week, and treatment-by-week interaction. CI: confidence interval; LS: least squares.
At Week 3, in the subset of 15 patients with a known history of failure of prior preventive treatment, 1/6 (16.7%) for placebo and 7/9 (77.8%) for galcanezumab had a ≥50% reduction in weekly cluster headache attacks. Of the 11/15 patients with failure of verapamil, 1/6 (16.7%) for placebo and 4/5 (80.0%) for galcanezumab achieved a 50% response (Figure 4). This trend in achievement of 50% response was similar between treatment groups in the overall Study CGAL population.

Percentage of patients achieving ≥50% reduction from baseline in weekly cluster headache attack frequency at Week 3. Mixed-model repeated measures: change from baseline in weekly attacks = baseline, sex, treatment, week, and treatment-by-week interaction.
Discussion
In the select population of patients with known history of prior preventive treatment failure, galcanezumab treatment resulted in greater mean reductions in the frequency of weekly cluster headache attacks compared with placebo. With galcanezumab treatment, the magnitude of the change increased each week over the 3 weeks evaluated in this post hoc analysis. Likewise, a greater percentage of galcanezumab-treated patients achieved a ≥50% reduction in weekly cluster headache attacks at Week 3 compared to placebo. The reductions observed in the subset population were consistent with those observed in the overall study CGAL of 106 randomized patients.
Information available to guide clinicians in appropriate preventive treatment approaches for patients with ECH remains incomplete, particularly in light of the absence of published data regarding the characterization of patients with failure of prior therapy. Despite the limited understanding of the impact related to prior treatment failures in ECH, data emerging from other disabling primary headache disorders reinforce the importance of characterizing this unmet need. For example, in migraine, the reasons clinicians and patients switch or discontinue preventive treatments may include the lack of efficacy or intolerance to medication. 4 In a survey of patients with migraine for whom preventive treatments have failed, an overwhelming majority of patients reported negative aspects of living with migraine and feelings of helplessness, depression, and not feeling understood. 7 The results clearly conveyed a substantial burden of the illness. Furthering the burden, patients with migraine who have a history of switching or discontinuing preventive treatments have increased healthcare resource utilization. 11 –13 Clinical experience suggests that the population of patients with ECH experience similar frustration in the clinical journey to identify effective treatment for their disease. In this study population, patients had an average of nearly two prior preventive treatment failures, which likely mirrors the clinical scenario seen with migraine. Here, we have shown that regardless of whether a patient has failed prior preventive therapy or not, galcanezumab is effective in reducing weekly cluster headache attack frequency.
This analysis specifically examined patients with previous failure of verapamil. Treatment with verapamil involves consideration of dose titration, potential prescription of doses above the FDA-approved 480 mg/day to achieve efficacy, cardiovascular monitoring, and evaluation of significant drug interactions. 5,6,14 Despite a Level C recommendation from the American Headache Society, verapamil is the most commonly prescribed medication for ECH, owed in part to the general lack of available treatment options. 5,6,14,15 As reflected in the subset population of patients with previous failure of verapamil in our study, the most common reasons for treatment failure were lack of efficacy and intolerance to medication. In the subset population, the magnitude of the change in reduction of weekly cluster headache attack frequency in galcanezumab-treated patients increased each week over the 3 weeks and was similar to reductions seen in patients with known failure of any preventive therapy as well as the overall CGAL study population. 5,6
Although reductions in weekly cluster headache frequency for patients with a known failure of any preventive therapy and failure of verapamil were similar to the overall CGAL study population, the reductions in the placebo rates were lower. In patients who have failed prior preventive therapy, the reductions in cluster headache frequency tend to be lower in the placebo group and are potentially linked to the negative or reduced treatment expectation. Lower placebo response was also reported in the migraine trials of patients with migraine and multiple previous treatment failures (ie, CONQUER, 16 FOCUS, 17 and LIBERTY 18 ). This trend was also observed in this post hoc analysis, leading to speculation that a similar phenomenon could occur in cluster headache.
Limitations
The most apparent limitations to this study were the small sample size and post hoc nature of the analyses, and as such, results should be considered exploratory. The sample size was limited because the collection of prior preventive failure history did not occur in the main study and was retroactively collected in the open-label safety study. Not every patient in Study CGAL entered Study CGAR. The point at which the collection of prior preventive information began was midway through Study CGAR. As such, there was no prior preventive information on every patient who entered Study CGAR. Therefore, the sample of patients who had prior preventive data may not be a random sample of patients seen in Study CGAL, thus leading to a potential selection bias. Further, as information was collected retroactively in Study CGAR rather than initially in Study CGAL, this could have led to a potential recall bias. To address some of these issues, baseline demographics of patients for whom preventive data were available were compared with those of the patients who did not have data. Additionally, comparisons were made for baseline demographics of the patients with prior preventive data available and with known prior preventive failures versus those without prior preventive failures confirmed during the retroactive data collection. Both of these comparisons revealed no significant differences and likely limit the impact of the potential biases.
Information collected on history of prior preventive failure in Study CGAR was based on patient report. Degree of reduction in cluster headache attack frequency or duration of response with prior preventive treatments were not collected. In addition, verapamil dose, duration of use, and frequency of use during cluster headache episodes were not specifically queried. As such, there was no specific criteria on the dose of verapamil that constituted failure due to lack of efficacy. Patients discontinued the use of verapamil under real-world conditions and could discontinue the medication after a short period of use in case of tolerability concerns.
Despite these limitations, the evaluation of those patients for whom prior preventive therapies have not been effective or tolerable is of great clinical relevance. To our knowledge, this is the first research that provides some evidence toward the general characterization of patients with ECH and prior preventive treatment failures. Specifically, this demonstrates that prior preventive treatment failure does not impact therapeutic benefit with galcanezumab in patients with ECH.
Conclusions
In this exploratory analysis of patients with episodic cluster headaches and a known history of prior preventive treatment failures (including verapamil, that was due to lack of efficacy or intolerance to medication), treatment with galcanezumab resulted in greater mean reduction in weekly cluster headache attacks compared with placebo.
Clinical implications
Novel characterization of patients with ECH who have a history of prior preventive treatment failures, including verapamil.
Galcanezumab treatment resulted in greater mean reduction in weekly cluster headache attacks compared with placebo in patients with ECH who have a history of prior preventive treatment failures, including verapamil.
With galcanezumab treatment, the magnitude of change compared to placebo increased each week over the 3 weeks.
Prior preventive treatment failure does not impact therapeutic benefit with galcanezumab.
Footnotes
Acknowledgements
Medical writing support was provided by Millie Hollandbeck (Synchrogenix), and Rohit Bhandari (Eli Lilly and Company) and was funded by Eli Lilly and Company, Indianapolis, IN, USA.
Author contributions
BP, JSA, MR, ATH, JB, RW, DK, and AA played a major role in the acquisition, analysis, or interpretation of the data. BP, JSA, MR, ATH, JB, RW, DK, and AA played a major role in the drafting, critical revision, and intellectual content of the manuscript.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: BP has received research support from Teva Pharmaceuticals, Electrocore (paid to employer), speaking or serving on an advisory board of Amgen, Allergan, Amgen/Novartis, Eli Lilly and Company and Biohaven. JSA is an ex-employee and minor shareholder of Eli Lilly and Company. MR, AT-H, JB, RW and DK are employees and minor shareholders of Eli Lilly and Company. AA has received research support from Novartis, Eli Lilly and Company and Teva, speaker honorarium from Novartis, Teva and Eli Lilly and Company, served on advisory board of Eli Lilly and Company, and received honorarium from Teva.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by Eli Lilly and Company, Indianapolis, IN, USA.
Institutional review board approval
The study protocols were reviewed and approved by the appropriate institutional review board for each of the study sites. The studies were conducted according to Good Clinical Practice and the Declaration of Helsinki guidelines. Patients provided written informed consent before undergoing study procedures.
