Abstract
Introduction:
Post-traumatic headaches (PTH) are common following mild traumatic brain injury (mTBI). There is evidence of altered central pain processing in adult PTH; however, little is known about how children with PTH process pain. The anterior cingulate cortex (ACC) plays a critical role in descending central pain modulation. In this study, we explored whether the functional connectivity (FC) of the ACC is altered in children with PTH.
Methods:
In this case-control study, we investigated resting-state FC of 5 ACC seeds (caudal, dorsal, rostral, perigenual, and subgenual) in children with PTH (n = 73) and without PTH (n = 29) following mTBI, and healthy controls (n = 27). Post-concussion symptoms were assessed using the Post-Concussion Symptom Inventory and the Child Health Questionnaire. Resting-state functional Magnetic Resonance Imaging (fMRI) data were used to generate maps of ACC FC. Group-level comparisons were performed within a target mask comprised of pain-related regions using FSL Randomise.
Results:
We found decreased FC between the right perigenual ACC and the left cerebellum, and increased FC between the right subgenual ACC and the left dorsolateral prefrontal cortex in children with PTH compared to healthy controls. The ACC FC in children without PTH following mTBI did not differ from the group with PTH or healthy controls. FC between rostral and perigenual ACC seeds and the cerebellum was increased in children with PTH with pre-injury headaches compared to those with PTH without pre-injury headaches. There was a positive relationship between PTH severity and rostral ACC FC with the bilateral thalamus, right hippocampus and periaqueductal gray.
Conclusions:
Central pain processing is altered in children with PTH. Pre-existing headaches help to drive this process.
Trial registration:
The PlayGame Trial was registered in ClinicalTrials.gov database (ClinicalTrials.gov Identifier: NCT01874847).
Keywords
Introduction
Traumatic brain injury (TBI) occurs in about 1.7 million individuals in the United States annually, 700,000 of whom are children and adolescents (0–19 years old). 1 Although approximately 90% of childhood TBIs are mild (mTBI), 2 half of these children will continue to experience persistent post-concussion symptoms (PPCS) one month following injury. 3 –5 Post-traumatic headache (PTH) is one of the most common PPCS symptoms, and is still problematic in 3,43 –58 % of children one month after mTBI. 3,4
The anterior cingulate cortex (ACC) is a critical region in the central pain processing networks and is involved in many pain-related functions, i.e. the sensory, cognitive, and affective aspects of pain processing, and pain modulation. It modulates connectivity within higher cortical (e.g., the dorsolateral prefrontal cortex, DLPFC; and medial prefrontal cortex, mPFC) and subcortical regions (e.g., periaqueductal grey matter (PAG)). 6 –10 This connectivity may be disrupted following adult and juvenile mTBI i.e. altered ACC and default mode network (DMN) connectivity. 11 –15
There is a significant knowledge gap about pain processing in PTH, especially in children. 16,17 Recent research suggests that functional connectivity within central pain-related regions relate to PPCS and may be particularly important in PTH. 13,18 Caudal-dorsal ACC connectivity with the secondary somatosensory cortex and inferior parietal lobe is significantly increased in adults with chronic PTH, 13 and early FC changes within pain networks (e.g., decreased PAG-DMN FC two days post-injury) significantly predict the persistence of chronic PTH. 18
In childhood, brain injury occurs during a period of rapid neuroanatomical and physiological change (i.e., ongoing myelination, synaptic pruning and functional integration). 19,20 Not only do developing brains respond differently to injury compared to adult brains, 21 but childhood headaches may also respond differently to treatment. 22,23 Given the critical role of the ACC in central pain processing, we examined the FC of the ACC with other pain-related brain regions in children with PTH following mTBI, and compared them to those without PTH, and healthy controls. As pre-injury headaches are a risk factor for PTH, 24 we also investigated how ACC FC differed in children with and without a prior history of headache. We hypothesized that children with PTH would have lower ACC FC with the DLPFC and PAG, and higher ACC FC with the insula. We also hypothesized that there would be a significant effect of prior headache history on ACC FC.
Methods
Participants in this case-control study were recruited from the PlayGame Trial (NCT01874847); a clinical trial investigating the effect of melatonin treatment on PPCS. 25 The study was conducted between 2014 and 2018 according to the Good Clinical Practice guidelines. Ethical approval was obtained from the Calgary Human Research Ethics Board (13-0372) Written informed consent and assent were provided by parents/guardians and participants, respectively. Neuroimaging and assessments for this study occurred at enrolment and before commencing medication. At enrolment, a comprehensive clinical assessment was performed by a physician with expertise in mTBI and data were collected on demographic and injury characteristics, post-concussive complaints including PTH, current headache characteristics, previous headache disorder, past medical history, and family history. PTH severity was verbally reported between 0 (no pain) to 10 (worst pain ever).
Participants
Participants (aged 8–18 years) were eligible if they had a medically diagnosed mTBI accordant with the American Academy of Neurology criteria. 26 Neuroimaging was not used to define mTBI, however, no intracranial abnormality was identified in the 10% of mTBI participants who had acute neuroimaging. Exclusion criteria included a loss of consciousness of greater than 30 min at time of injury, a Glasgow Coma Scale (GCS) score less than 13, injury due to assault, a previous mTBI within the last three months, neuroactive medications, any significant medical disorder, or contraindication to MRI.
Criteria for PTH were a current complaint of headache (either new-onset since mTBI or increased headache) at clinical interview, and an increase in the headache score on the Post-Concussion Symptom Inventory (PCSI) compared to pre-injury score (evaluated retrospectively at enrolment). Participants were further categorized as having a pre-injury history of headache (+history) or not (-history) at clinical interview. Pre-injury headaches were classified using the International Classification of Headache Disorders, second Edition (ICHD-II), and/or previous headache diagnosis by a neurologist or headache specialist.
A control group of healthy children of similar age and sex without a history of mTBI were recruited mainly from friends and siblings of participants, and underwent identical evaluations. We excluded children who reported having frequent headaches or headache disorders from the control group. We also recruited additional healthy controls from a separate study (International Collaboration on Neuroinflammation in Traumatic Brain Injury study (ICON-TBI, REB 16-2057)) with the same standardized clinical interview, neuropsychological testing (except the PCSI), and identical neuroimaging parameters.
Outcome assessments
Post-concussion symptoms
The PCSI was used to investigate post-concussion symptoms after mTBI. 27 –29 It consists of 26 symptoms in physical, cognitive, emotional and sleep domains and has moderate-to-high test-retest reliability and good convergent validity. 29,30 Using this, participants were asked to rate their headaches ‘over the last few days’. Pre-injury headaches were rated using the same questionnaire (baseline or pre-injury report). Headaches were rated between 0 (not a problem), to 6 (severe problem). The PCSI symptom total score ranges from 0 to 156.
Child Health Questionnaire (CHQ)
The Bodily Pain/Discomfort score from the Child Health Questionnaire (Child report, CHQ87). The CHQ is a validated measure of children’s general health, 31,32 and has been used to measure bodily pain in individuals with TBI and chronic pain conditions. 33 –38 The CHQ87 Bodily Pain/Discomfort score rates severity (“none” to “very severe”) and frequency (“none of the time” to “every/almost every day”) of bodily pain/discomfort in the past four weeks using a a 6-point scale. Scores are standardized and range from 0 (most severe) to 100 (none). Health-related Quality of Life (HRQOL) was reported based on other CHQ87 domains such as Global Health, Physical Functioning, Social Limitations due to emotional, and behavioral problems. 32,39,40
Neuroimaging data acquisition
Functional magnetic resonance imaging (fMRI) was conducted using a GE 3 Tesla 750w MRI with a 32-channel head coil located at the Alberta Children’s Hospital. To record blood oxygen level dependent (BOLD) responses during resting-state, children were asked to lie in the scanner with their eyes open and look at a fixation cross. Resting-state fMRI was recorded using a gradient echo, echo planar imaging sequence for 5 min and 10 s (TE = 30 ms, TR = 2000 ms, flip angle = 90, field of view (FOV) = 23 cm, matrix 64 × 64, 150 slices, slice thickness = 3.6, 150 volumes, voxel size = 3.6 × 3.6 × 3.6 mm). Structural T1-weighted anatomical images were used for registration (3D Ax FSPGR BRAVO, flip angle = 10, TI = 600, FOV = 24 cm, matrix 300 × 300, slice thickness = 0.8 mm, 230 slices, voxel size = 0.8 × 0.8 × 0.8 mm).
Statistical analyses
Demographic characteristics were compared between groups using SPSS 41 (IBM Corp. Released 2016. IBM SPSS Statistics for Macintosh, Version 24.0. Armonk, NY: IBM Corp). Normality was assessed using Shapiro-Wilk test. Group differences in age and motion outliers were analysed using the Kruskal-Wallis test (KW).
The effect of PTH across groups (controls, PTH or NoPTH) and pre-injury headache history on four HRQOL domains (CHQ 87) were analysed using linear regression controlling for age and sex. To account for multiple comparison among these four predictors, we used Bonferroni correction and corrected p-value was set as p < 0.0125.
Neuroimaging analyses
Preprocessing
The FMRIB Software Library (FSL) version 6.0.0 was used for fMRI pre-processing and analyses (www://fsl.fmrib.ox.ac.uk). 42 Pre-processing included skull removal for T1 images using the Brain Extraction Tool (BET), 43 motion correction using the six parameter affine transformation using MCFLIRT,44 slice-time correction, spatial smoothing (Gaussian kernel of 6 mm) at full-width half-max, linear registration of fMRI data to the structural images using six DOF and normalization of the data by linear registration to the MNI152 (2 mm) standard template using 12 DOF. 45 To mitigate the effect of head motion on the data, the FSL Motion Outliers was used (https://fsl.fmrib.ox.ac.uk/fsl/fslwiki/FSLMotionOutliers) to identify the volumes affected by significant head motion 46,47 with a frame-wise displacement (FD) threshold greater than 0.3 (mm). Scans with more than 25% outlier time-points were excluded (i.e., less than 113 out of 150 volumes remaining after excluding the volumes with motion). A nuisance model was generated using FEAT in FSL to account for the effects of motion and other nuisance factors including global signal (GS), white matter (WM) and cerebrospinal fluid (CSF) timeseries. Global signal regression (GSR) was used to mitigate the effects of non-neuronal noise factors such as respiration, cardiac impulses, motion, vasodilation, and scanner-related artifacts. 48 –54 As a complementary analysis, we further analysed the ACC FC maps without GSR (Supplementary Figures 2 to 4). 55 –57 WM and CSF masks were generated using the FSL FAST segmentation tool. 58 Masks were registered to the functional and standard space, thresholded at 0.4 for CSF and 0.66 for WM and binarized. Average time-series of WM, CSF, and GS were added as explanatory variables (EVs), alongside motion outliers (volumes), motion parameters and their derivatives as additional confound EVs. The FILM-prewhitening was activated in the analysis to account for the autocorrelation among the nuisance factors and to enhance the model’s efficacy. 59 The cleaned data was normalized via z-scoring and subsequently centered on 100, using the fslmaths command.
First-level analyses
Region-of-interest (ROI) analyses focused on the ACC. Using the coordinates reported by Margulies et al., Kelly et al. and others. 60 –63 we created five functionally differentiable seeds of the ACC: caudal, dorsal, rostral, perigenual, and subgenual. These ACC seeds have been found to have unique short-term and long-term connectivity with various pain-related regions. 60 –62,64 These seeds were generated for the right (R) hemisphere only as previous studies have shown that connectivity patterns of the ACC are similar for the right and left (L) hemispheres, 60 and pain processing is more lateralized toward the right hemisphere. 65 A sphere was created with a radius of 3.5 (mm) around the reported coordinates (see Figure 1). ACC seeds were then transformed into each participant’s native space and the mean time-series for each seed was extracted. For the first-level analysis, five separate models were generated within the FEAT model in FSL, by adding each seed time-series as an explanatory variable.

ACC seeds and target pain mask. Upper left panel: Five ACC seeds: (1) caudal ACC (cACC: pink), (2) dorsal ACC (dACC: red), (3) rostral ACC (rACC: light blue), (4) perigenual ACC (pACC: dark blue), (5) subgenual ACC (sACC: green). Lower left panel: target ROIs combined in the target pain mask. MNI-152 (2 mm) was used as a standard atlas; Right diagram shows summary of the analysis steps.
ACC seeds and target ROIs.
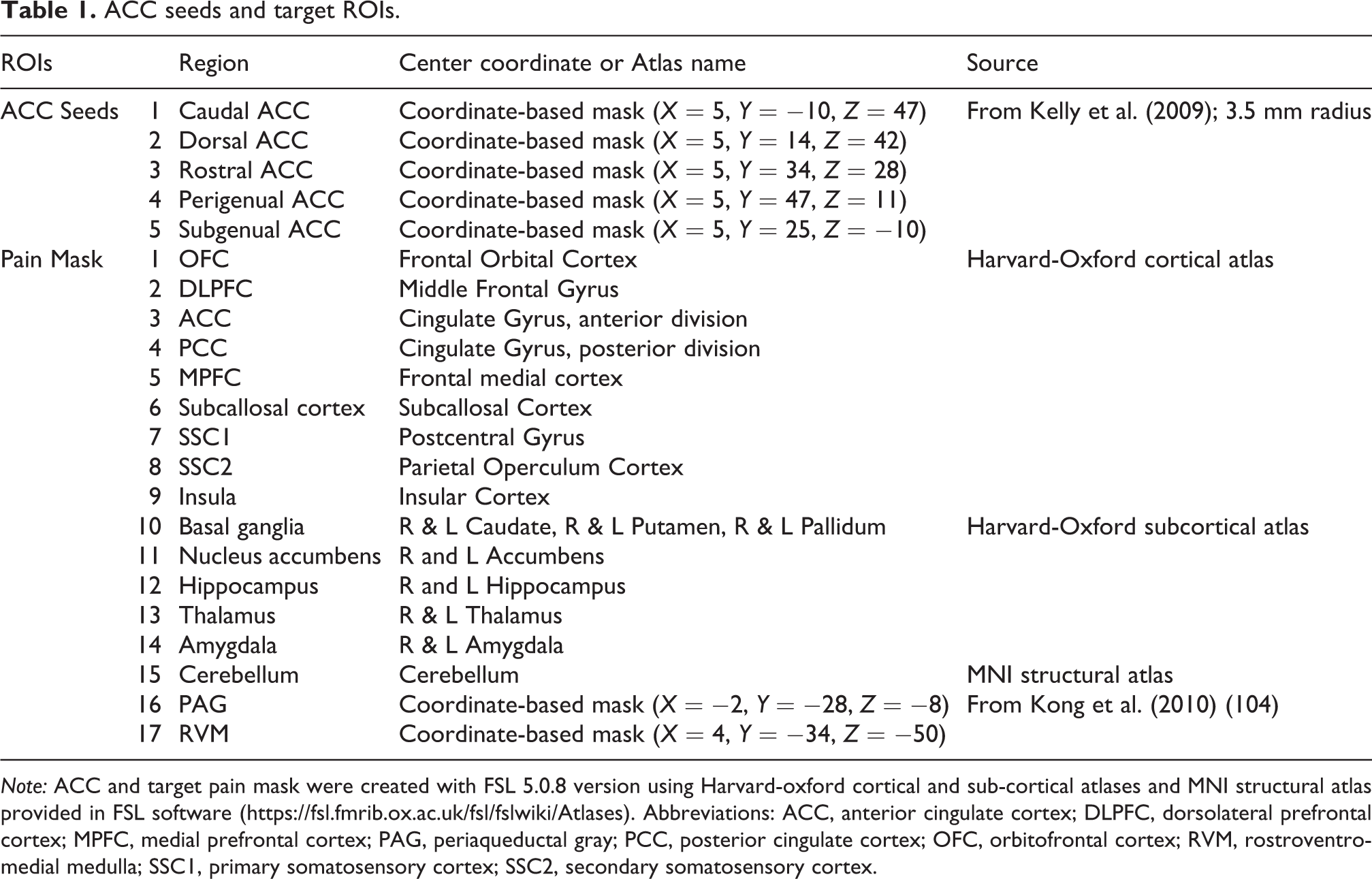
Note: ACC and target pain mask were created with FSL 5.0.8 version using Harvard-oxford cortical and sub-cortical atlases and MNI structural atlas provided in FSL software (https://fsl.fmrib.ox.ac.uk/fsl/fslwiki/Atlases). Abbreviations: ACC, anterior cingulate cortex; DLPFC, dorsolateral prefrontal cortex; MPFC, medial prefrontal cortex; PAG, periaqueductal gray; PCC, posterior cingulate cortex; OFC, orbitofrontal cortex; RVM, rostroventromedial medulla; SSC1, primary somatosensory cortex; SSC2, secondary somatosensory cortex.
Group-level analyses
The z-stat outputs from first-level analyses were transformed to standard space. Randomise was used to analyse group results using the threshold-free cluster enhancement (TFCE) statistic with 5000 random permutations (voxel-wise). 72 A nonparametric permutation test was used to reduce the level of false positives. 72 –74 The TFCE statistic provides a higher probability of finding true signal and better signal-to-noise ratio in the absence of a priori threshold for cluster identification. 75 Multiple comparison correction in Randomise is based on family-wise error rate (FWE) correction 76 and results of TFCE statistics in randomise are presented as ‘tstat’ with the p-values generated as ‘1-FWE corrected p.’ 72 We report on clusters of voxels with p-values less than p < 0.05 and their peak voxel values (Supplementary Table 1).
ACC FC differences between groups
To investigate differences in average FC between the ACC seeds and pain-related regions between participant groups (PTH: n = 65, NoPTH: n = 22, controls: n = 21), general linear model (GLM) design matrices were created for each seed and regressed against the z-stat FC maps. The GLM included three EVs for group intercepts together with age, sex, and number of volumes with motion outliers. Another EV was added to adjust for any variability related to differences in control groups (PlayGame Trial vs. ICON-TBI). Positive and negative group effects were also generated as within-group averages. A one-way ANOVA was used to compare the three groups, and t-contrasts compared pairs of groups. To examine ACC FC in participants with new-onset PTH (n = 35) compared to participants without PTH with no headache history (n = 16) and controls (n = 21), participants with a headache history were excluded (reference Figure 1 consort diagram). Differences in the ACC FC maps between groups (PTH, NoPTH, controls) were analysed using a GLM as above.
Effect of pre-injury headache on ACC FC
To investigate the effect of pre-injury headache history, we compared ACC FC between PTH participants with and without prior headaches (PTH + history: n = 30; PTH − history: n = 35). This was analysed using a GLM with two EVs (PTH − history and PTH + history) controlling for sex, age and motion outliers. T-tests were used for between-group contrasts. Exploratory analyses were performed to examine the specific effect of pre-injury headache history on ACC FC by comparing participants without PTH with a headache history (n = 6) to participants with PTH with a headache history. Results of these analyses are included in the Supplementary Figure 1.
Influence of PTH severity and bodily pain
FC ACC and pain-related regions was investigated in the PTH group using GLM with a covariate for PTH severity for each ACC seed. We entered one EV for the group intercept with PTH together with PTH severity as a covariate of interest, while controlling for sex, age and motion outliers. Positive and negative linear slopes of PTH severity were computed. Similarly, methods were used to investigate the effect of CHQ bodily pain score.
Results
One hundred and twenty-nine participants were eligible. Nineteen participants did not meet eligibility criteria (see Figure 2 for study details). Twenty-one scans were removed due to excessive motion. One hundred and eight scans were included in the final analyses: PTH (n = 65; mean age = 14.6 SD = 2.3 years; 40% male), No PTH (n = 22; mean age = 14.1 SD = 2.5 years; 60% male) and controls (n = 21; mean age = 14.5 SD = 2.8 years; 43% male). Groups did not differ significantly in age or sex distribution (see Table 2). Injury characteristics are presented in Table 3. Participants were between 22 and 54 days post-injury (mean = 37.4 days, SD = 5.9). The most common mechanism of injury was sport-related. Headache was the most common acute symptom.
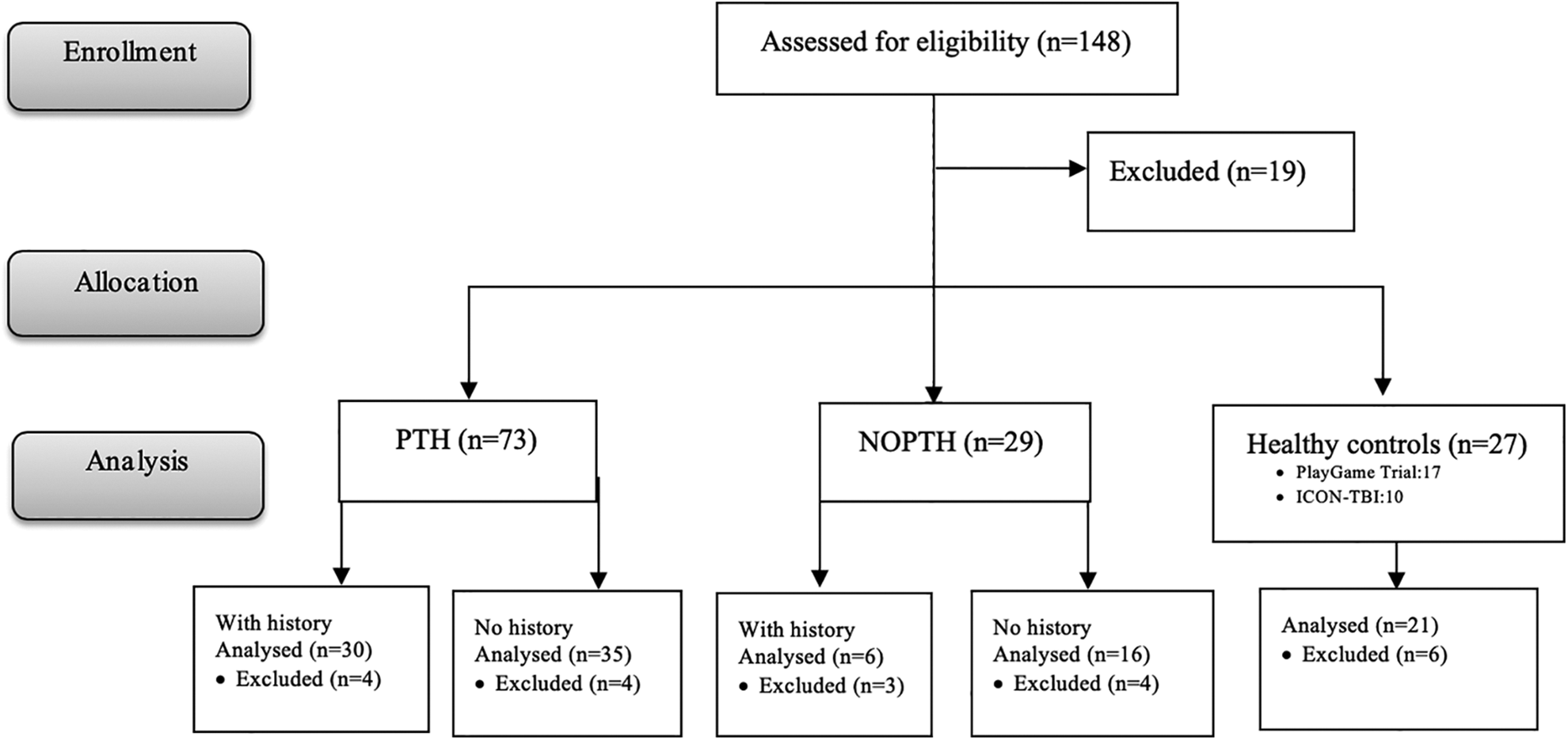
Details of the study participants.
Demographics of participants in the control, PTH with a headache history, PTH with no headache history, without PTH with no headache history and without PTH with a headache history groups.

+ Pre-injury headache history.
– No pre-injury headache history.
NoPTH, No post-traumatic headache; PTH, post-traumatic headache; SD, Standard deviation; y, year.
Injury characteristics in the PTH and NoPTH groups.

+ Pre-injury headache history.
– No pre-injury headache history.
PTH, post-traumatic headache; GCS, Glasgow coma scale; MVA, motor vehicle accident; LOC, loss of consciousness; NoPTH, mTBI without PTH.
The clinical details and headache characteristics are reported in Tables 4 and 5. Migraine was the most common pre-injury headache disorder (90%). PTHs were often diffuse or occurred in multiple locations and were usually described as a pressure-like pain. Commonly associated symptoms were photophobia, phonophobia, nausea, and exacerbation with exercise.
Headache history and PTH characteristics in the control, PTH and NoPTH groups.
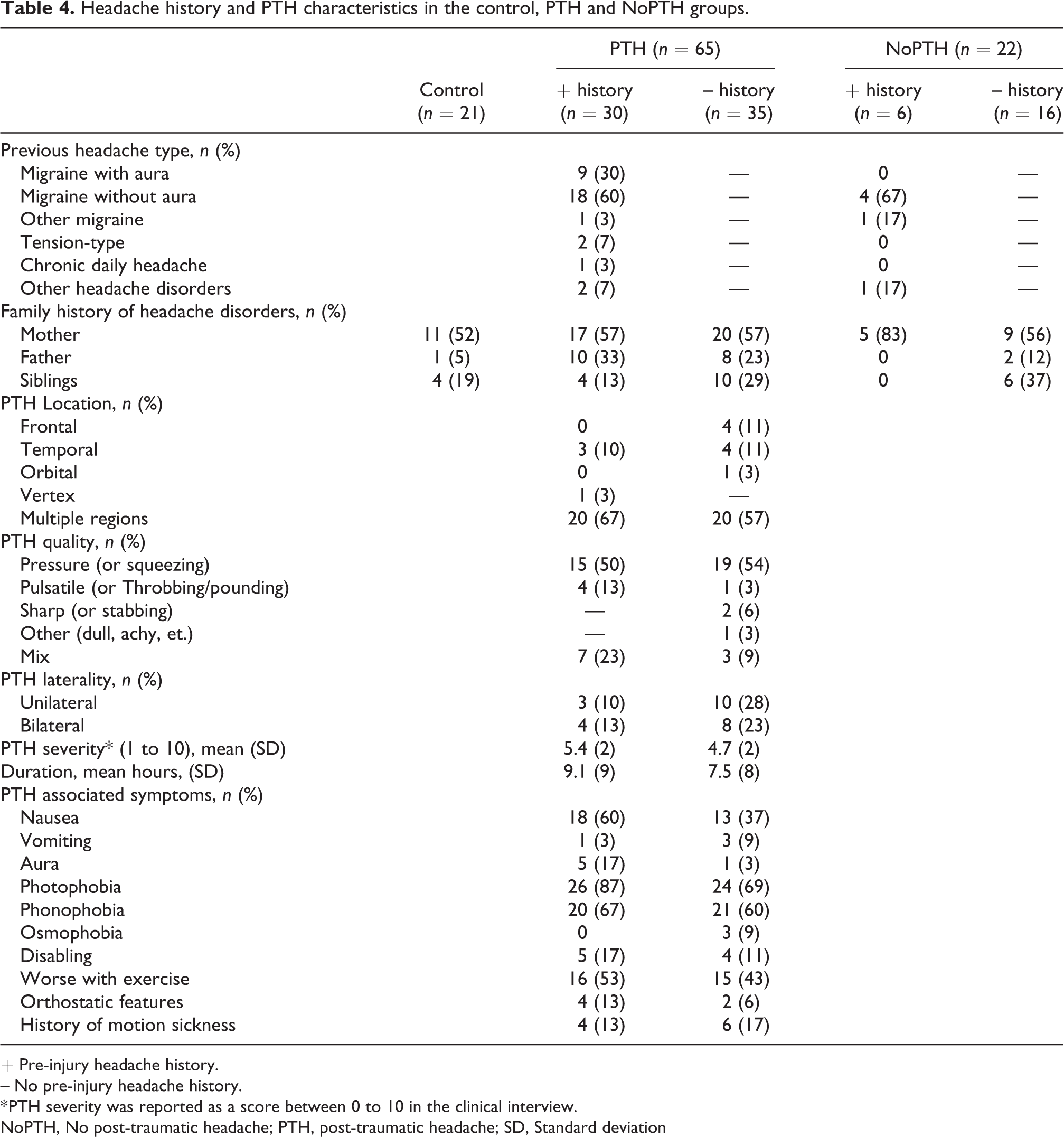
+ Pre-injury headache history.
– No pre-injury headache history.
* PTH severity was reported as a score between 0 to 10 in the clinical interview.
NoPTH, No post-traumatic headache; PTH, post-traumatic headache; SD, Standard deviation
PCSI scores and bodily pain in the control, PTH and NoPTH groups.

+ Pre-injury headache history.
– No pre-injury headache history.
CHQ, child health questionnaire-self report; NoPTH, No post-traumatic headache; PTH, post-traumatic headache.
The regression models across groups (PTH, NoPTH, and controls), pre-injury headache history, age and sex significantly predicted domains of the HRQOL including Physical functioning (F (5, 95) = 6.066, p < 0.0005, R 2 = 0.242), Role social limitations/emotional domain (F (5, 95) = 6.294, p < 0.0005, R 2 = 0.249), Role social limitations/behavioral domain (F (5, 95) = 2.584, p = 0.031, R 2 = 0.120), and Bodily pain and discomfort (F (5, 95) = 13.521, p < 0.0005, R 2 = 0.416). Overall, children with PTH reported significantly worse HRQOL compared to those without PTH following mTBI on all four HRQOL domains. Also, compared to healthy controls, the PTH group reported worse HRQOL in the Physical functioning, Role social limitations/emotional and Bodily pain. There was no significant effect of headache history on HRQOL domains, see Table 6.
Linear regression analyses exploring the effect of PTH and pre-injury headache history on HRQOL domains controlling for age and sex.

vs., versus. Beta (standardized coefficients) are reported here. *Significance level was set as p < 0.0125 Bonferroni corrected.
ACC FC differences in PTH
As demonstrated in Figure 3, right perigenual ACC FC with the left cerebellum was increased in controls compared to the PTH group. It is important to note that based on within-group averages, FC between right pACC and left cerebellum was negative; therefore, there was more negative FC between pACC and cerebellum in the PTH group compared to the healthy controls. Moreover, FC between right subgenual ACC FC and the left DLPFC was increased in the PTH group when compared to controls. Within-group averages showed that there was a significant negative FC between the subgenual ACC and DLPFC in controls, which was absent in the PTH group. There were no statistically significant differences between the NoPTH group and the PTH or control groups.
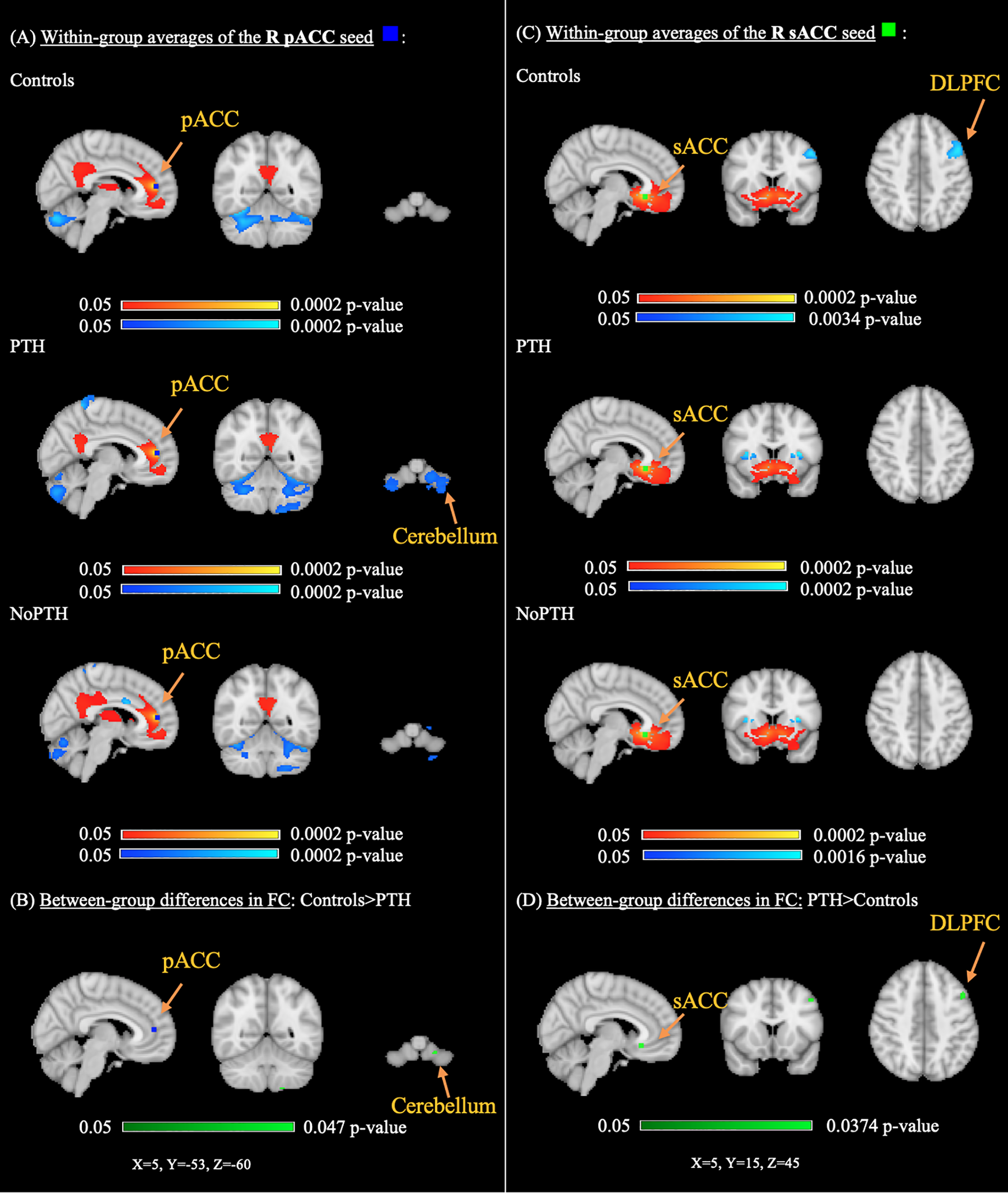
Significant differences in perigenual and subgenual ACC seeds among three groups: controls, PTH, NoPTH (Cluster threshold < 0.05). (A) The left column shows significant positive (yellow-red) and negative (light blue-blue) within-group averages for the R pACC FC with pain-related regions in controls, NoPTH and PTH groups. (B) The bottom left panel shows higher R pACC FC with the Left cerebellum in controls compared to PTH (voxels in cluster: 5, Max: L lobule VIIIb, tstat = 4.35, x = −18, y = −54, z = −60, p = 0.047). (C) The right column shows significant positive (yellow-red) and negative (light blue-blue) within-group averages for the R sACC with pain-related regions in controls, NoPTH and PTH. (D) The bottom right figure shows relatively higher R sACC FC with the left DLPFC in PTH compared to controls (voxels in cluster: 21, Max: L MFG, tstat = 4.83, x = −46, y = 12, z = 46, p = 0.037). MNI-152 standard atlas is used for presentation. ACC, anterior cingulate cortex; FC, functional connectivity; NoPTH, without post-traumatic headache; R, right; pACC, perigenual ACC; PTH, post-traumatic headache; sACC, subgenual ACC.
Influence of pre-injury headache and PTH severity
After excluding participants with pre-injury headaches, there were no significant group (PTH-history; NoPTH-history; controls) differences in ACC FC, suggesting a significant influence of pre-injury headache. Participants with PTH with a prior headache history had significantly higher ACC FC (rACC and pACC) and the cerebellum compared to the PTH without prior headache group, see Figure 4. Within-group ACC maps demonstrated that there was a less pronounced negative FC between rostral and perigenual ACC seeds and the cerebellum in participants with PTH+headache history.

Significant differences in the perigenual and rostral ACC seeds FC patterns between PTH without headache history and PTH with a headache history groups are demonstrated (Cluster threshold < 0.05). (A) The left column shows significant positive (yellow-red) and negative (light blue-blue) within-group averages for the R pACC with pain-related regions in PTH-history and PTH+history. (B) The bottom left figure shows higher between-group differences R pACC FC with bilateral cerebellum in PTH+history compared to PTH-history (voxels in cluster: 722, Max: L deep cerebellum nuclei (dentate nucleus), tstat = 3.8, x = −22, y = −52, z = −34, p = 0.016). (C) The right column shows significant positive (yellow-red) and negative (light blue-blue) within-group averages for the R rACC with pain-related regions in PTH-history and PTH+history. (D) The bottom right figure shows higher R rACC FC with right cerebellum in PTH+history compared to PTH-history (voxels in cluster: 101, Max: R cerebellum VI b, tstat = 5.12, x = 40, y = −54, z = −24, p = 0.018). MNI-152 standard atlas is used for presentation. ACC, anterior cingulate cortex; FC, functional connectivity; pACC, perigenual ACC; PTH, post-traumatic headache; PTH + history, post-traumatic headache with a headache history; PTH-history, post-traumatic headache with no headache history; R, right; rACC, rostral ACC.
PTH severity positively correlated with higher rACC FC with central pain regions (i.e., bilateral thalamus, right hippocampus and PAG) as demonstrated in Figure 5. No significant relationship was found between bodily pain and any ACC FC seeds to pain-related regions in participants with PTH.

The significant relationship between PTH severity and R rACC FC in the PTH group (Cluster threshold < 0.05). This figure shows positive relationship between PTH severity and rACC FC with the bilateral thalamus, R hippocampus and PAG in participants with PTH (voxels in cluster: 371, Max: right thalamus, tstat = 6.13, x = 18, y = −26, z = −2, p = 0.005). MNI-152 standard atlas is used for presentation. ACC, anterior cingulate cortex; FC, functional connectivity; PTH, post-traumatic headache; R, right; rACC, rostral ACC.
Discussion
We investigated differences in ACC FC with other pain-related brain regions in children and adolescents with PTH following mTBI. Our findings supported our hypothesis that there would be significant differences in ACC FC in children with PTH following mTBI when compared to healthy controls. We also found that PTH was significantly associated with HRQOL. Although a history of pre-injury headache had no effect on HRQOL, it was significantly associated with ACC FC.
Altered ACC FC and central pain processing
The ventral portions of the ACC (rostral, perigenual and subgenual) play an important role in modulating pain while performing cognitive demanding tasks. This is mainly through the attentional modulation of pain, i.e. pain distraction. 77 –79 We found evidence of increased negative perigenual ACC FC with the cerebellum in children with PTH (compared to controls). As increased ventral ACC FC to the cerebellum has previously been associated with attention problems, 80 the increased pACC-cerebellum FC seen in our study could suggest dysregulated attentional modulation of pain.
Higher cortical areas, such as DLPFC, also modulate descending pain inhibitory mechanisms via other cortical and subcortical pain-related regions (e.g. ACC, insula and PAG). 81 –83 We found increased ACC FC between subgenual ACC and contralateral DLPFC in the PTH group (absence of negative FC) when compared to controls. This suggests a dysregulation in top-down inhibitory pain modulation i.e. attenuation of inhibitory DLPFC inputs to the sACC in children with PTH. Consistent with this, a study examining adults with migraine, reported increased FC between left ACC and right DLPFC, supporting alterations in pain modulation processes. 84
Influence of pre-injury headache and PTH severity
A pre-injury history of frequent headaches is present in 18–47% individuals with PTH. 23,24 We found weaker negative FC between ACC seeds (rostral and perigenual) and the cerebellum in children with PTH who had a prior headache history. Given that the rostral and perigenual ACC play important roles in descending pain inhibition and the modulation of the dorsal horn neurons, 71,85,86 our finding of lower negative ACC-cerebellum FC suggests a downregulation of descending pain inhibitory mechanisms.
Although there is a lack of consensus on the biological meaning/relevance of negative resting-state FC patterns in the literature, 57,87 –92 animal and human data support its validity; the anti-correlated hemodynamic pattern originating from inhibitory synchronous neural activity. 93 –97 This is relevant to our data in that the cerebellum has major inhibitory functions via inhibitory GABAergic Purkinje cellsy 98,99 The posterior cerebellum is functionally connected (albeit indirectly) to frontal cortex, ACC and DMN regions and fMRI studies have demonstrated its role in pain inhibition and analgesia. 100 –103 The different rACC/pACC-cerebellar FC in PTH without a headache history found in our study further supports the role of the cerebellum as an important inhibitor of the rACC/pACC and central pain networks.
In addition to altered descending pain inhibitory mechanisms, activation of pain facilitatory mechanisms in the brainstem may lead to increased sensitivity of the central pain networks. Regions such as the ACC, hippocampus, locus coeruleus, thalamus, PAG and RVM are critical elements of this mechanism and alterations of FC involving these regions may contribute to pain persistence. 67,104 –107 We observed higher rACC FC with the thalamus, hippocampus and PAG regions in PTH participants with higher pain burdens. As the interactions between inhibitory and facilitatory pain mechanisms are important in the development of chronic pain, 105,106,108 these findings could suggest increased activation of descending pain facilitatory mechanism in pediatric PTH. This finding may also be related to pain rumination tendencies, where inhibition of unwanted thoughts is impaired due alterations in cortico-thalamo-hippocampal circuits. 109 –112
Our contention of impaired descending pain modulation systems in pediatric PTH is partially supported by previous studies involving adults with PTH. 13,18 Leung et al. found increased ACC FC with the secondary somatosensory regions in adults with chronic PTH compared to healthy controls, 13 and Niu et al. found that diminished PAG-DMN FC predicted persistent PTH. 18
It is somewhat debatable whether our findings of altered ACC FC are associated with mTBI per se or the persistent PTH symptom itself. Although we found significant differences in ACC FC in PTH, which was driven (at least partially) by the presence of pre-injury headaches, we failed to find definitive evidence that mTBI played a significant role i.e. no differences in ACC FC were found between PTH groups when participants with pre-injury headaches were excluded. This also could have been due to a loss in power.
In our analyses, we used global signal as a nuissance regressor. Similar analyses without GSR are presented in the Supplementary materials for comparison. The usefulness of GSR in resting-state fMRI analysis is highly debated. 48 –54,56,113 Similar to others, 80 we found similar general group difference maps of ACC FC with and without GSR; however, the differences without GSR analysis, did not pass the threshold of p < 0.05. Other studies have also reported that GSR increases the likelihood of identifying significant group differences, especially where regions are negatively associated. 114,115 Our findings further support the use of GSR in rs fMRI to remove the effect of the nuisance factors.
The large sample size is a strength of our study and allowed some subgroup analyses. Other strengths include well-characterized clinical phenotypes, and GSR methodology. This study also has several limitations. As this was a pediatric population, we used a relatively short resting-state scan, which could have decreased the signal to noise ratio, 116 although others have demonstrated reliable FC with short 5-min scans. 117 Quantification of headache burden could have been improved by using a headache diary and specific headache questionnaire. It should be noted that we can only hypothesize about underlying inhibitory/facilitatory process as in FC studies it is not possible to identify the cause of the observed relationships. Finally, although we excluded healthy controls with headaches/headache disorders, about 75% of healthy controls had a family history of headache disorders.
This study has several important clinical implications. Our findings are in keeping with the common clinical scenario in which a head injury seems to be a trigger/stimulus that changes the presentation of pre-existing (and often infrequent) mild headaches to severe, chronic and migraine-like. 23 Simple analgesics seem to offer little or no relief to these patients. 3,118 Further, our findings of altered ACC connectivity in PTH are similar to those seen in animal and human studies of chronic headache disorders. 119 –122 A mTBI, therefore, seems to incite a shift from acute to chronic pain processing and supports the use of treatment and management strategies targeted toward chronic pain mechanisms. 23 Future research identifying the underlying mechanisms for this rapid shift from acute to chronic pain processing is likely to provide some valuable insights into potential therapeutic options for this clinically challenging problem. 123 Moreover, our findings offer potential targets for non-invasive brain stimulation to either inhibit facilitatory pain networks (e.g., cerebellum) or increase the activity of central pain attenuating regions (e.g., DLPFC) that could be used as a treatment for the challenging problem of post-traumatic headache. 124 –127
In summary, we found significant alterations in the functional connectivity of the perigenual and subgenual ACC seeds suggesting that PTH are associated with dysregulation in descending central pain modulatory systems. Pre-injury headaches play a significant role in the central modulation of pain processing. Further studies exploring differences in brain connectivity, especially in pain processing regions, during acute pain tasks could help in understanding the neurobiology of PTH.
Clinical implications
Altered functional connectivity of the perigenual and subgenual areas of the ACC in children with post-traumatic headache suggests dysregulation in descending pain modulation.
PTH severity is correlated with greater functional connectivity between the rostral ACC with thalamus, hippocampus and periaqueductal gray.
Children with pre-injury headaches may be prone to developing PTH due to downregulation of descending pain modulation (i.e., reduced negative functional connectivity between the rostral/perigenual ACC seeds and the cerebellum).
Supplemental material
Supplemental Material, sj-pdf-1-rep-10.1177_25158163211009477 - Functional connectivity of the anterior cingulate cortex with pain-related regions in children with post-traumatic headache
Supplemental Material, sj-pdf-1-rep-10.1177_25158163211009477 for Functional connectivity of the anterior cingulate cortex with pain-related regions in children with post-traumatic headache by Zahra Ofoghi, Christiane S Rohr, Deborah Dewey, Signe Bray, Keith Owen Yeates, Melanie Noel and Karen M Barlow in Cephalalgia Reports
Footnotes
Acknowledgements
We would like to acknowledge Brenda Turley, Tina Samuel, Aneesh Khetani and Regan King for their contributions. We thank all of the children and their families who participated in this study.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics or institutional review board approval
The PlayGame Trial (REB 13-0372) and ICON study (REB 16-2057) received local ethical approval.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Researchers acknowledge the support of the Canadian Institutes of Health Research funding (CIHR) (grant number: TBI-293375), the CIHR-Era-Net Neuron (JTC 2016, project number: 10016191).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
