Abstract
Background:
Burning mouth syndrome (BMS) is a chronic and debilitating oral pain of the normal oral mucosa. It mainly affects women in their fifth to seventh decade. Its aetiopathogenesis remains unclear and is probably of multifactorial origin, with increasing evidence that BMS may be a neuropathic disorder. BMS is classified as an idiopathic (nociplastic) orofacial pain with or without somatosensory changes by International Classification of Orofacial Pain (ICOP 2020). The diagnosis of BMS, having excluded ‘oral burning mouth symptoms’, has evolved from basic intraoral exclusion screening to extensive clinical and laboratory investigations, which include the screening of comorbidities and other chronic pains and somatosensory testing. There is no standardised treatment in managing BMS, but a proposed combination of supportive and pharmacological treatment has been recommended.
Aim:
To review the current concepts of BMS definitions, classifications, aetiopathogenesis, diagnosis techniques, and evidence-based treatments in managing BMS patients.
Conclusion:
As BMS is a diagnosis by exclusion, thus a stratified approach is required for assessment of patients presenting BMS. A BMS diagnosis protocol is desired using a standardised screening to distinguish BMS from patient’s presenting with ‘oral burning symptoms’, and evaluation of comorbid chronic pain disorders or other medical comorbidities, which will include haematological, fungal, salivary flow, and qualitative sensory testing. Axis II and other additional quantitative sensory testing may further elucidate the causes of this condition. For future BMS prediction and prevention, will be based upon research on the relationship between other chronic pain disorders and familial history, environmental and genetic information.
Introduction
Burning mouth syndrome (BMS) remains a challenging disorder for health care providers. It is a debilitating condition for the patients and is known for its persistent moderate to severe intensity of burning pain, with a pain score of 8/10. 1 BMS is probably of multifactorial origin with uncertain aetiopathogenesis. 73% of BMS patients experienced tingling, scalding, tender, or numbness of the oral mucosa. 2 Various terminologies have been used to describe the burning sensation in the mouth, especially on the tongue such as glossodynia, sore tongue, burning tongue, stomatopyrosis, glossopyrosis, and oral dysaesthesia. The most common site for BMS is the tongue (anterior two-third or tip), followed by the hard palate, gingivae, lower lips, and pharynx. BMS usually occurs bilaterally and symmetrically. 3 BMS may be associated with other comorbidities and is a diagnosis by exclusion with no objective clinical or laboratory findings. The complexity in diagnosing BMS increased the difficulties for both clinicians and patients to achieve a curative treatment for it. 1,4 –6 Advances in the research of BMS aetiopathogenesis have led to two clinical forms, which are ‘Primary BMS’ and ‘Secondary BMS’. Primary BMS (BMS) is described as essential idiopathic BMS for which no organic local or systemic causes can be identified. Secondary BMS (oral burning symptoms) is a result from local or systemic pathological conditions or factors such as nutritional deficiencies, dental-related trauma, hormonal (menopause), endocrine (diabetes mellitus) changes, medications related and allergy or hypersensitivity reactions. 2 There is debate about the appropriateness of BMS being called a syndrome as the condition does not fulfil syndromic criteria 7 and perhaps should be referred to as burning mouth disorder (primary and secondary).
Classification
Over the past decade, the definition and classification of BMS have evolved in many ways that were dependant on its clinical features and response to treatments. Multivariate analysis has grouped ‘stomatodynia’ as a ‘persistent idiopathic orofacial pain’ based on their homogenous topography features. 8 The International Association for the Study of Pain (IASP) 9 has classified ‘glossodynia and sore mouth’ as chronic pain and defined it ‘as a burning pain in the tongue or other oral mucous membranes’. In 2016, IASP further described BMS as a ‘chronic intraoral burning sensation that has no identifiable cause either local or systemic condition or disease’. 10 The International Headache Society (IHS) in its classification of headache disorders (ICHD-3) has categorised BMS as ‘painful cranial neuropathies’ and defined BMS as ‘an intraoral burning or dysaesthesia sensation recurring daily for more than 2 hours per day over more than 3 months, without clinically evident causative lesions’. 11 The International Classification of Orofacial Pain (ICOP) is a collaborative effort between established orofacial and head pain groups that conformed to a universally accepted classification of orofacial pain disorders or diseases. 12 ICOP has classified BMS as an idiopathic orofacial pain and described it as ‘an intraoral burning or dysaesthesia sensation, recurring daily for more than 2 hours per day for more than 3 months, without evident causative lesions on clinical examination and investigation. ICOP has further suggested the used of somatosensory assessment to subgroup BMS into ‘with or without somatosensory changes’. 12 In this review, the author will define any intraoral burning sensation that does not fit into ICOP definition as ‘oral burning symptoms’
Epidemiology
Published prevalence data on BMS are still inadequate and are of poor quality in describing the impact of BMS in the population. The implementation of more precise BMS diagnosis criteria has narrowed the prevalence range between 0.1% to 3.7%. 13 –16 BMS predominantly affects middle-aged and older women, which are in the peri and post-menopausal stage 3,13,14 and the female to male ratio is 7:1. 1,2 The prevalence of BMS increased remarkably at the age after 60. 1,13 –15,17
Typically, BMS symptoms may persist for months or years without a period of remission. An earlier study has reported that 50% of BMS patients showed partial or complete remission with or without any treatment, and 20% achieved complete spontaneous remission within 6 to 7 years of onset. 18 Contrary to these findings, latter studies reported only 3% to 4% of patients have complete spontaneous remission within 5 to 6 years after the onset of BMS clinical manifestation, and less than 50% showed improvement of symptoms with treatments. 19,20 Reviews had reported a prevalence of 20% to 30% of patients with chronic pain to have suicidal intention in the past. 21,22 There were two case reports on BMS patients attempted suicide due to the unbearable mental stress from pain and feeling hopeless in their life. 23,24
Clinical symptoms
Along with the burning pain, BMS patients complain of accompanying symptoms such as xerostomia, alteration in taste, poor sleep quality, health issues, and psychological disorders. The onset of BMS can be spontaneous or triggered by a precipitating event such as dental procedures, medications, foods, and stressful life events. 25 –27 There have been reports that spicy or hot food and beverages, psychosocial stress and fatigue, and speech, increased pain intensity. 18,25 Patients have claimed that by chewing gums, sucking sweets or lozenges, drinking cold beverages, and relaxation and recreation activities were able to reduce the pain. 2,18,25 The pain is usually at its lowest in the morning upon awake, and once aggravated, it continuously reaching the maximum intensity by late evening. 25,28 However, the pain seldom interferes with sleep.
46% to 67% of BMS patients complain of subjective dryness of the mouth that affect their quality of life, with no signs of hyposalivation. 14,19,29,30 BMS patients exhibit a marked decrease in salivary flow rates 31 –33 and higher viscosity 31 of the unstimulated whole (parotid, submandibular and sublingual gland) saliva in relative to control. The unstimulated (resting) whole saliva (UWS) was accumulated by expectorating (spit or drool) the saliva into a wide test tube for an average of 5 minutes. Salivary flow rate per min was calculated from the total weight of collected saliva over time. UWS flow values were approximately same for various collection techniques such as drooling or draining, spitting and suction. 34,35 The submandibular gland is innervated by chorda tympani and continues to the submandibular ganglion by joining with the lingual nerve. It contributes 60 to 67% of UWS secretion, 36 with mucous as its primary secretion. The findings from the above studies and the relation of the submandibular gland and its nervous innervation, may suggest the possibility of dysfunction chorda tympani and/or lingual nerve contributes to the reduce submandibular gland secretion in BMS patients. There was no difference between BMS patients and control to stimulate whole saliva (SWS) flow rate. 32,37 These studies results showed that majority of the salivary glands continue to respond adequately upon stimulation. Parotid gland contributes 50% of the stimulated whole saliva. 36 Its secretion is purely serous, and its autonomic innervation is by the glossopharyngeal nerve. The contribution of sublingual glands to unstimulated and stimulated whole saliva is low. 36 A study performing scintigraphic imaging of BMS patients’ (with and without hyposalivation) parotid and submandibular gland reported no significant difference in the functions 32 of BMS patients.
Xerostomia in BMS patients may be associated with the consequences of other systemic diseases, and the use of medications (Table 1). 37 –39 A meta-analysis of 47 studies reported that salivary flow rate was negatively affected with increased in age. UWS, and specific submandibular and sublingual saliva flow rate were significantly reduced in older adults, but parotid and minor salivary gland were not affected by age. 40 There was no influence of age and the use of drugs on salivary flow. 40 The ageing process may contribute and enhances the perceived oral dryness, as a majority of BMS patients are aged 60 and above. 40,41 Menopausal and post-menopausal females have reduced UWS flow rate too. 42
Possible causes of dry mouth symptom.

BMS patients may perceive the dry mouth effect due to the reduction of UWS that plays an essential role in lubricating and protecting the oral mucosa. 29,43 The high viscosity of the UWS may cause subjective frothy and sticky, uncomfortable sensation. 37 The lack of mucous concentration from parotid gland during the SWS may contribute to the subjective sensation of discomfort rough oral mucosa surface. 44
The salivary secretory IgA (SIgA) concentration of BMS patient showed no difference from the control group, but BMS patients have significant lower SIgA secretion rate in a minute. This may imply decrease immunity in BMS patients. 31 In responding to stress, the α-amylase activity was elevated in BMS patients. BMS patients have twice higher α-amylase activity than control, that explained by the high-stress occurrence in BMS patients. 31 Sodium (Na), total protein, albumin, IgA, IgG, IgM and lysozyme were significantly increased in BMS patient except for potassium (K). 33 Proinflammatory cytokines such as IL-2 and IL-6 were significantly increased in the whole saliva of BMS patients in comparison to healthy control and correlate with disease severity. These inflammatory cytokines can act as biological markers on the development and therapy response for BMS. 45
Saliva and taste enable each other in which both the transportation and dissolving of gustatory stimulants were dependent on salivary flow. 33 Approximately 70% of patients reported taste alteration such as dysgeusia and ‘phantom taste’ 3,30 and was thought to be related to the onset of a burning sensation. 46 BMS patients often complained of metallic and bitter taste in their mouth, and reduced taste sensitivity for salty and sweet. 30 Repeated topical application of bupivacaine reduced the pain, but it increased gustatory complaints in BMS patients. 47 The decreased in the gustatory sensitivities of the tongue in an electrogustometric test on the dorsal surface of the tongue has proposed degeneration of the chorda tympani nerve that leads to trigeminal neuropathy or glossopharyngeal nerve inhibition. 48 A chemogustometry whole mouth study reported a significant difference in taste disturbances for all four-basic taste between BMS patients and healthy controls. 33 However, Imura et al. reported no significant difference of sweet, salty, umami, and bitter taste between them, but the threshold for sour taste was significantly higher in BMS. 26,31 The taste threshold was independent of dry mouth, pain, or subjective taste alteration symptoms. 33 Disturbance in taste may give rise to a negative influence on oral health-related quality of life of BMS patients. 49
Comorbid medical conditions have been reported in patients with BMS (Table 2). Retrospective analysis of 102 BMS patients reported that 97% of patients have at least one comorbidity and life stressors (62.7%) being the most common. 59.8% of BMS patients have anxiety, 50% with depression, and 42.2% have low back pain. 1 However, many of these medical conditions may be symptomatic of the demographics of BMS patients. Gastrointestinal diseases are one of the most common associated risk factors of BMS. 15 The oral cavity is the second main reservoir of Helicobacter pylori (H. pylori) and commonly detected in dental plaque and saliva. 50 51.3% of BMS patients reported to have concurrent gastritis compared to 27.5% in the healthy control group, and 15 out of 19 of patients with burning mouth disorders has H. pylori infection. 51 A clear association of these diseases are essential to distinguish between a true BMS or ‘BMS like oral burning symptoms’.
Comorbidities related to BMS.

96.1% of BMS patients showed unexplained chronic extracephalic pain conditions like fibromyalgia and visceral pain. 52 33% of patients with fibromyalgia syndrome have reported having glossodynia with the average visual analogue score (VAS) of 5/10. 53 There have been case reports on vulvodynia coincided or preceded BMS, which both disorders commonly occur in menopausal and post-menopausal females. 54 Reports on the association of BMS with vulvodynia may be rare as it is not a common symptom that is questioned by the health practitioner and require further exploration. However, in contrast, a systematic review contradicted the claims that there was no evidence for the high co-occurring pain symptoms rate in BMS patients. 55
There is reported weak evidence between poor sleep quality and pain experienced in 80% of BMS patients with a mean global Pittsburg Sleep Quality Index (PSQI) score of 7.7 56 and Poor Sleep Quality (PSQI) Index of more than 5. 57
It was believed that psychological or psychopathological factors play a role in triggering or exacerbating the BMS symptoms. A meta-analysis reported BMS patients to have a high predisposition towards anxiety (odds ratio 2.64) and depression (odds ratio 3.18). 58 BMS patients may have other psychological profiles, such as social phobia, cancerphobia, hypochondria, and neuroticism. 58,59 The ongoing high-level oral burning pain will likely have a significant psychological burden on patients affected and the relationship being causative and or resultant of the condition requires clarification.
Aetiopathogenesis
The pathogenesis of BMS remains unclear, but advancement and development on neuoropathological and neurophysical studies have shifted the emphasis towards peripheral and central neuropathies rather than psychogenic factors. A better understanding of the pathophysiology of BMS could provide a basis for the development of more effective therapies.
Psychology factors
Prevalence of psychiatric disorders peculiarly anxiety and depression are high in BMS patients, but their role in the pathogenesis of BMS remains unclear. Anxiety and depression are commonly seen in patients affected by functional chronic pain disorders, such as irritable bowel syndrome, recurrent headache, fibromyalgia, chronic back pain, and chronic fatigue syndrome. 58 80% of BMS patients have depression, anxiety disorder, and other chronic pain conditions before the onset of BMS. 60
Anxiety and depression are thought to play a role in the pathogenesis of BMS as pain could be a somatic trait of BMS patients. 61 Psychological assessment using the General Health Questionnaire (GHQ) reported a significantly higher score, particularly in ‘somatic symptoms’ and ‘social dysfunction’ in the BMS group in comparison to control group. However, quantitative somatosensory testing revealed that there was no significant difference between painful and non-painful tongue site in the BMS group. 62 Besides, there were no differences in the tactile detection threshold (TDT) and filament-prick pain threshold (FPT) between BMS and healthy control. The GHQ scores did not correlate with the QST score on the painful or non-painful area of the BMS tongue, and the numerical rating scales (NRS) pain intensity. 62 A functional magnetic resonance imaging study has reported an increase in the functional activity of the neural circuits that regulate depression and anxiety symptoms and is correlated with the functional brain connectivity that regulates the burning pain. 63 Therefore, there may be feasibility link between psychological factors in BMS pathogenesis. Further studies are a desire to address the role of personality and cognitive factors in contribution to BMS.
Neuropathic disorders
Advancement in the neuroimaging studies of the brain has allowed a better understanding of the BMS pathophysiology. They have provided consistent evidence for BMS to be a neuropathic disorder of the peripheral and central nervous systems.
Peripheral neuropathy
Neuropathology
Multiple tongue mucosal biopsies studies have exhibited a significant decreased in the intraepithelial nerve fibres density in comparison to healthy control. 64 –66 Thus, this suggests of focal trigeminal small-fibres sensory neuropathy or atrophy of the oral mucosa. Immunohistochemical staining of the tongue biopsies revealed a significant increase in transient receptor potential vanilloid 1 (TRPV1), nerve growth factor (NGF) fibres, and purinergic receptor P2X3. 65,67 TRPV-1 fibres are derived from the trigeminal nerve and found in peripheral nociceptive terminals of Aδ and C fibres, and centrally in dorsal root and trigeminal ganglia. TRPV-1 is related to the conduction of heat or hot taste (capsaicin) and nociceptive signals. BMS patients clearly have shown a consistently decreased tolerance to noxious heat stimulation, such as capsaicin. 68 BMS patients’ tongue exhibited reduced in cannabinoid receptors CB1, while the expression of CB2 is increased. 69 CB2 receptors are involved in the antinociception role of the trigeminal system, and this suggests an accelerated role of TRPV1 receptors in BMS. The overexpression of TRPV1 receptors is associated with hypersensitivity, and neuropathic pain may be cause for BMS dysaesthesia symptoms. 68 P2X3 ion channel receptors are responsible for eliciting burning pain sensation when it is activated by adenosine triphosphate (ATP). 70
Non-neuronal cells such as astroglial, fibroblast, Schwann cells synthesised NGF and NGF is transported to the target tissues nerve fibres. 71 NGF plays an important role in peripheral pain modulation and is upregulated during inflammation. NGF sensitise the peripheral nociceptor and its responsible for TRPV1 and purigenic receptor of ATP (P2X3) regulation. 72 The increased in TRPV1 expression may be related to the increase of NGF uptake by target tissues nerve fibres. 65,72 Increased in the synthesis of NGF has shown to increase nociceptive heat response. 72 NGF exhibit significant neuroprotective activity in both peripheral and central nervous system neuron degeneration. 71 The decreased in intraepidermal innervation of tongue mucosa, 64 –66 along with the increased in TRPV1 expression and NGF positive nerve fibres 65 may strengthen BMS peripheral neuropathy mechanism hypothesis. Blocking of NGF actions in treating persistent pain has been demonstrated in animal study models. These studies used anti-NGF therapy in treating sciatic constriction pain model and reported a reduction of mechanical and thermal hyperalgesia. 73,74
It has been hypothesised that chorda tympani nerve hypofunction plays a role in the BMS pathophysiology. 46 Damage to the peripheral nervous system gustatory fibres affects the central inhibitory mechanism, leading to hypersensitivity trigeminal input and could cause the dysgeusia or ‘phantom taste’. 46,75 Grushka and Bartoshuk reported that the density of fungiform papillae was higher in BMS patients than in control and suggested that these high fungiform papillae density could be a risk factor for BMS. 75 The increased in taste buds’ density may explain for ‘Supertasters’ in BMS patients. However, the opposite has been reported in other studies with no significant difference in the fungiform papillae density between BMS and controls. 46,76 The absence of chorda tympani nerve innervation was associated with atrophy of fungiform papillae, but there was no substantial loss of taste or tactile in the tongue. 76
Neurophysiology
BMS patients often complained of taste loss, and this symptom was demonstrated in a study that compared BMS patients and healthy control gustatory sensitivity and capsaicin threshold test. A whole mouth chemogustatory test revealed a reduction in the gustatory sensitivity (sweet, sour, salty and bitter) and increased in lateral tongue capsaicin burning pain threshold in BMS patients. 77 This supports the hypothesis that peripheral taste nerve fibres damage may contribute to BMS pain following the loss of trigeminal nociceptive transmission. Therefore, taste dysfunction may be an indication of small afferent nerve fibres disorders. 30,75
Thermal quantitative sensory testing (QST) has described an increase in heat pain detection thresholds 64,78 and increased sensitivity to heat pain on the tongue mucosa of BMS patients. 79 Due to the heterogeneity of BMS patients, QST profiling of BMS abnormalities may be more for an individual patient level. 80 QST studies highlighted the dysfunction of intraepithelial small nerve fibres by expressing a more pronounce damage of Aδ fibres than C fibres. It is suggested that the degeneration of Aδ fibres led to the loss of noxious inhibition of C fibres that were responsible for the persistent burning pain sensation 80 similar to diabetic neuropathy. Another hypothesis is that damage of Aδ taste afferents fibres, which travelled along in the chorda tympani nerve, can cause the pain related to BMS. Hypofunction of Aδ fibres of the chorda tympani was revealed by an electrogustatometry study that described an elevation of taste detection thresholds of BMS patients’ tongue mucosa. 79,81 The reduction of taste fibres inhibitory control on the afferent stimuli may cause burning pain. The burning phantom pain may be caused by a reduction in the inhibitory control customarily exerted by taste fibres on somatic afferents, acting either peripherally or centrally.
The trigeminal system blink reflex had decreased inhibition in 25% to 36% of BMS patients. 82 The increase in the electrical thresholds to elicit blink reflex in BMS patients indicate the hypofunction of the trigeminal tactile Aβ fibres. 83 There was a close relationship between the abnormalities of the blink reflex with the duration of disease.
A study using lingual nerve block with lidocaine on 20 BMS patients has 50% of their patients with decreased visual analogue scale (VAS) scores and seven patients reported with no changes or increased in their pain score. 79 The reduction in pain scores demonstrated the presence of peripheral neuropathy mechanism in BMS patients and the contrary for central neuropathy.
The perceived involvement of both peripheral and central nervous system in the pathogenesis of BMS is demonstrated with the used of topical anaesthetic. Application of 20% benzocaine in the burning area demonstrated significant relief of ‘oral burning symptoms’ pain, but BMS patients have the opposite outcome. 84 The topical application of local anaesthesia aggravated the pain in BMS patients. Topical application of dyclonine HCL anaesthetic has reported a mixed result with 21% patients have decreased pain score, and 26 patients experienced no changes or increase in the burning pain. 85
Central neuropathology
Nigrostriatal dopaminergic pathway plays an important role in central pain modulation, especially in the basal ganglion and sensory cortex. 86 Neurotransmitter positron emission tomography (PET) study has demonstrated the hypofunction of the nigrostriatal dopaminergic system in BMS patients that prompts the hypothesis of reduced efficacy of endogenous pain inhibitory control in the brain dopamine-opioid system. 87 –89 The increased availability in dopamine D2 receptors reflects the depletion of dopamine levels that may induce chronic neuropathic pain within the trigeminal distribution. 87,90 Dopamine D2 receptors (DRD2) gene 957C>T Single Nucleotide polymorphism has been associated with the increased availability of DRD2 in human striatum 91 and the presence of 957T allele leads to decrease in synaptic dopamine concentration and thus reducing dopamine receptors binding. 91 A study on the influence of DRD2 957TT genotype in orofacial neuropathic pain which consists of 5 BMS patients, 4 atypical facial pain and post-traumatic trigeminal nerve injury, have low painful and non-painful thermal detection threshold. 92 The prevalence of DRD2 957 TT genotype was significantly approximately twice higher in patients with orofacial neuropathic pain (50%) than the general Finnish population (27%). 92 Patients with 9577TT genotype encounter a twice higher mean numerical rating pain scores than patient with genotype 957CT and 957CC. It is a hypothesis that individuals with DRD2 9577TT genotype will have low striatal dopamine tone and are more affected with thermal stimuli.
Patients with Parkinson disease have a similar reduction of dopamine synthesis in their putamen. 93 PET scan imaging has demonstrated changes in the Parkinson disease patient cortical pain process sites such as the insula, anterior cingulate cortex and prefrontal cortex. 93 This similarity was seen in BMS patients in functional brain imaging. Hence, Parkinson’s disease patients may be susceptible to burning mouth syndrome, but the link between BMS and Parkinson disease varies from poor to moderate. The reported prevalence of BMS seen in Parkinson disease was between 4% to 24%. 94 –96 Kim et al. reported no correlation between patients with BMS and the risk of developing Parkinson disease. 97 Besides, the evidence on the association of Parkinson disease and BMS were exhibited in deficiency of habitual blink reflex in both groups. 82,83,98 The level of dopamine increased during the night sleep of Parkinson’s disease patient which improved their dopaminergic function during the sleep. 99 This may explain the increasing BMS pain in the day. 80 Four patients diagnosed with restless legs syndrome (RLS) have reported having BMS, too. 100 RLS has abnormal blink reflex too and has been associated with Parkinson’s disease. 101 There were significantly decreased in RLS patients’ DR2 in the putamen and increased in tyrosine hydroxylase in the substantia nigra. 102 The increase in tyrosine hydroxylase may represent the iron deficiency and should be interpreted with care as iron deficiency present oral burning symptoms and not a true BMS. Adding together the overlapping features, BMS subgroup phenotype that is related with RLS and Parkinson disease and not responding to conventional BMS therapies, may benefit from dopaminergic drug treatment. 100,103
Functional magnetic resonance imaging (fMRI) has demonstrated that BMS patients have decreased brain activation in the whole brain, especially in the thalamus. They have lower grey matter volume (GMV) in the bilateral ventromedial prefrontal cortex (VMPFC) and increased functional connectivity between this region and the bilateral amygdala. 104 The functional connectivity between the bilateral VMPFC and the amygdala correlated with the years of BMS illness. fMRI studies revealed that noxious heat stimulation applied at the perioral region evoked higher activity in the anterior cingulate, medial, prefrontal and insula cortices, which belong to the medial pain system, in BMS patients than in control. 105,106 Repeated thermal stimulation on the lower lip and not on the palm shows BMS patients have temporal suppression to continuous painful stimuli and lack of pain inhibition against repeated noxious stimuli compared with controls. 106 Thus, it explains the reasons for BMS patients to express a reduction in pain tolerance but no reduction in pain threshold or perception. 68 Overall, BMS patients displayed brain activation patterns similar to patients with other chronic neuropathic pain conditions. These findings suggest that brain hypoactivity may be an essential feature in the pathophysiology of BMS.
Endogenous descending inhibitory pain modulation facilitates or inhibits chronic pain in human. 107,108 A disruption in the descending pain modulation pathway are more likely to facilitate 109 and contribute to the development of chronic pain such as fibromyalgia, temporomandibular joint disorders, osteoarthritis and irritable bowel syndrome. 110 –113 Temporal summation (TS) and conditioned pain modulation (CPM) are two psychophysical tests that have been used to investigate central nervous system pain modulation. TS evaluates the ascending pain facilitatory pathways. CPM is dynamic testing on the descending pain inhibitory control and was developed from the diffuse noxious inhibitory control (DNIC) mechanism in animal studies and based on the model’ pain inhibit pain’. 114 It has been reported there was non-significant effect of both mechanical TS and thermal CPM test in a reduction of BMS pain score, 115 which suggests a dysfunction in BMS endogenous inhibitory pain system. 80,116 It is unsure if the pain modulation mechanism is disrupted before or after the presence of pain. Central sensitisation may develop from the continuous pain inhibition mechanisms process, responding to the chronic pain. 108 Similar results of BMS mechanical TS are shown in a study using intraepidermal pinprick on the chin between mouth angle and the midface (adjacent to the pain region), which imply impaired inhibitory pain control pathway. 117 The BMS TS scores were significantly increased after repetitively noxious stimulation. However, there was no significant difference between BMS and the healthy control group. The mean TS did not reveal a significant reduction upon receiving conditioned stimuli at 40°C and 47°C on the non-dominant hand.
Interestingly, the BMS group has reduced CPM scores during the low-density conditioning noxious stimuli (40°C) but an increased pain rating during a higher density conditioning stimuli (47°C). There was no difference noted between BMS and control group at 40°C. The differences in BMS patient’s CPM response may be due to distraction. Distraction may induce a mild but not effective pain inhibition if the conditioning stimuli is of weak strength. 117,118 However, there was a study stated CPM to be independent of distraction. 119 Both studies 80,117 on BMS TS and CPM demonstrate a suppressed descending inhibitory pain mechanism in BMS as seen in persistent post-endodontic pain study 120 and the lack pain habituation which is associated with impaired C fibres. 106 As CPM test and condition stimuli were, commonly performed in the upper or lower limbs and not in the orofacial region, it may suggest that BMS could be a generalised, widespread disorder and is not limited to the tongue or oral cavity. 79,116
Adrenal and gonadal steroid hormones, which are synthesised in the central and peripheral nervous system, act as important precursors for the synthesis of neurosteroids. Chronic anxiety, depression, or post-traumatic stress disorder, and menopause or gonadal hormonal imbalance lead to the changes of adrenal and gonadal steroid production. These neurosteroids provide neuroprotective against nervous system injuries and diseases, facilitate nerve regeneration, and synthesise neurotransmitters. Evidence on the involvement of neurosteroid in localisation of orofacial pain came after a report of decreased morning salivary dehyroepiandrosterone (DHEA) concentration in BMS. 121,122
Diagnosis
ICOP has established distinct unambiguous definitions and diagnostic criteria for oral-facial pain disorders/diseases. BMS is identified as an idiopathic orofacial pain and is diagnosed based on the clinical examination and investigation exclusion of local or systemic causative lesions. 12 The characteristic BMS features are superficial intraoral mucosa pain and recurrence of the pain daily for more than 2 hours per day and more than 3 months. Using quantitative and/or qualitative somatosensory testing, BMS is further subgroup to without and with somatosensory changes. This classification will further aid the clinical and research diagnosis of the possibility of BMS as neuropathic pain.
The consistency among studies strongly suggested that BMS remains under-recognised and under-appreciated by both dental and medical professionals. 1 It took an average of 12 to 13 months from the onset of symptoms until a definitive diagnosis of BMS was achieved. 1,6 Hence, it is crucial that health practitioners differentiate BMS from ‘oral burning symptoms’ by the exclusion of possible related local and systemic factors (Tables 3 and 4).
Possible local factors that may cause an oral burning sensation in the mouth.

Possible systemic factors that may cause an oral burning sensation in the mouth.

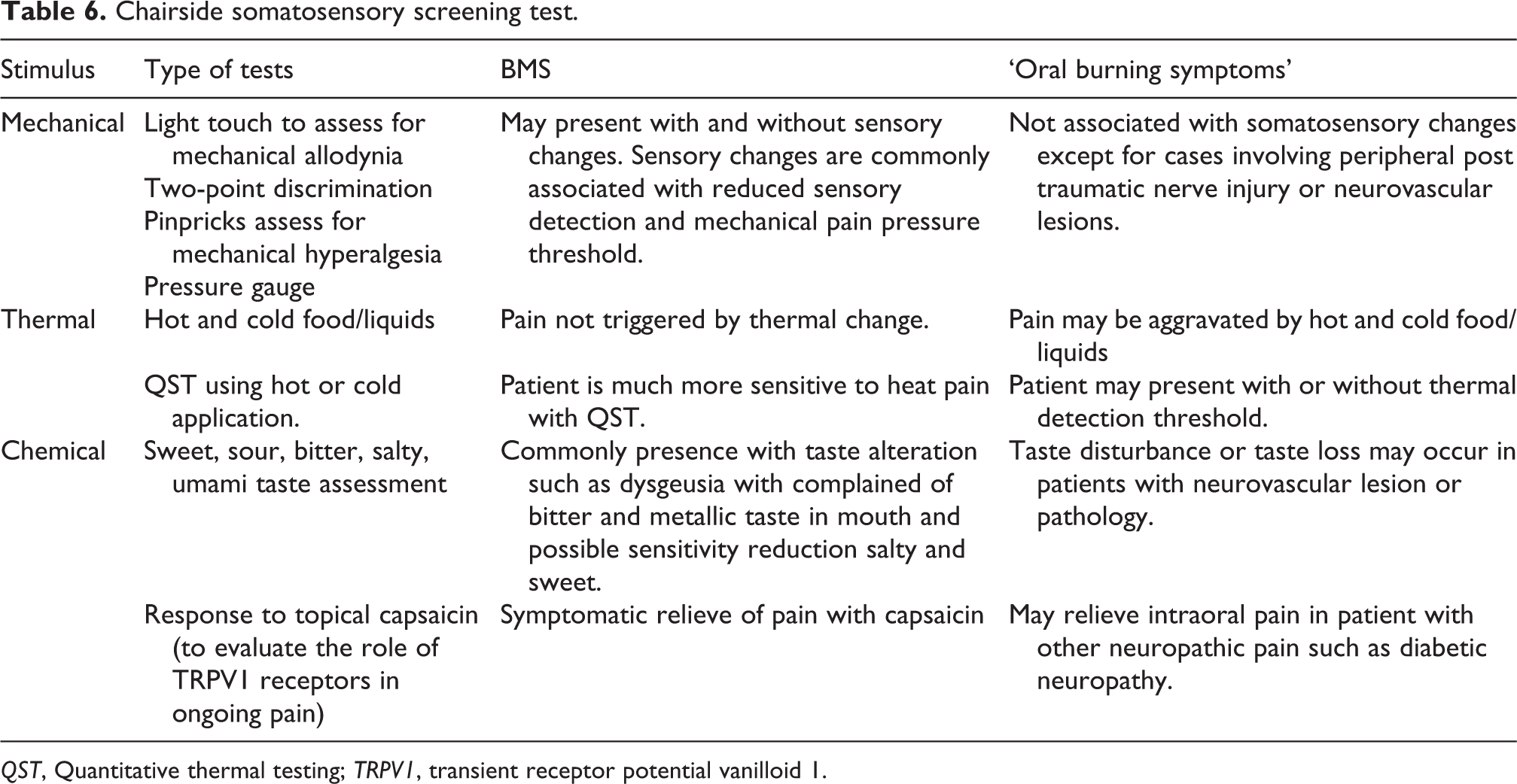
Information collected from the patients’ pain and medical histories, clinical examinations, and laboratory investigations are summarised to evaluate any possible local or systemic aetiology to the oral burning pain. A comprehensive and systemic methodology diagnostic workup is of particularly important (Table 5) along with somatosensory test (quantitative or qualitative) in diagnosing BMS. Simple chair sides somatosensory screenings on mechanical, thermal, and chemical can demonstrate the possible association of peripheral nerve fibres in BMS (Table 6). 123 Despite the increased burden in the health care financial and human resource, it is important to distinguish the difference between BMS and ‘oral burning symptoms’. Hence, the extensive clinical and laboratory investigation that exclude any possible contributable organic diseases and the assessment of sensory nerve dysfunction in BMS with quantitative sensory testing is vital to aid BMS diagnosis and the most effective treatment.
Burning mouth syndrome diagnostic workup.

Chairside somatosensory screening test.
Treatment
As there is still no consensus on the aetiopathogenesis of primary burning mouth syndrome, thus the management of BMS is a challenge. The proposed pharmacological and non-pharmacological treatments did not give consistent and effective outcomes. There is no standardised curative treatment for BMS, but purely symptomatic. Holistic assessment and care, including an empathetic and supportive of the patients’ complaints approach, are essential. 124 Patients should be reassured that the symptoms are not imaginary and not related to any form of cancer and to give a realistic expectation on the outcome of BMS treatment. 2,83,125 This encouraging support will reduce the patient’s anxiety, depression, fear, and frustration from multiple unsuccessful treatments. 26
BMS patients could take some simple measures upon themselves in easing the pain and the perceived dry mouth. Patients are advised to avoid substances that may irritate the oral mucosa such as highly acidic food or beverage (citrus fruits and juice), mint, cinnamon, tobacco, hot and spicy food, alcoholic beverages, mouthwashes that contain alcohol and toothpaste that contained sodium lauryl sulfate surfactant agent or abrasive substance such as calcium carbonate. 3,26 Sucking crushed ice and chew sugarless gum or pastille can stimulate saliva secretions and regular sips of cold beverage will help to relieve the dry mouth. 3,83 These home measures may relieve their oral symptoms and avoid escalation of pain score.
Topical management
Capsaicin is an active component of chilli peppers plants, which produces a burning sensation when it encounters tissue. Capsaicin binds to the TRPV1 nociceptors, and continuous exposure to capsaicin caused desensitisation of afferent nociceptors (C fibres). Topical capsaicin concentration between 0.025% and 0.075%, is usually used and able to reduce the VAS burning pain score by two units. 126 Systemic capsaicin gave a similar result as a topical application, but epigastric pain has been reported as a major side effect. 127
Clonazepam is a benzodiazepine anticonvulsant that binds to the GABAA receptors and activates the descending pain inhibitory pathway in the spinal cord and peripheral nociceptors. 128 Administration of topical clonazepam decreased the afferent peripheral nerve excitability. 129 An in-vivo study found GABAA receptors on the of the rat’s tongue nerve fibres, and a 10 minutes topical application of aqueous GABA powder on the oral mucosa increased the mechanical threshold of the tongue. 128 GABA receptors are found in human oral mucosa, including cranial ganglia, mandible, palate and salivary gland. 130 Therefore, application of topical clonazepam may express its analgesic effect by activation of peripheral GABAA receptors and subsequently decrease the tongue afferent fibres sensitivity. 128,131 Also, GABAA receptors are found in the taste pathway, and the loss of taste due to dysfunction chorda tympani and/or the lingual nerve may produce an imbalance pain control that leads to the loss of central pain inhibition. 132
A meta-analysis 133 on two RCT studies 131,134 on the use of topical clonazepam in two-thirds of the in BMS patients exhibited a significant reduction of pain scores by 1.5 unit. 133 Both studies involved dissolving the clonazepam tablets with a daily dosage range of 0.5 mg to 3 mg in the saliva and withheld for 3 minutes and followed by expectorating it. 131,134 The sedative side effect of clonazepam is usually well tolerated as the dose used is well below the anticonvulsant therapeutic level. 131 Topical administration of clonazepam via mouth rinse, gel or by sucking the tablet and followed by expectorating the saliva, is a preferred treatment due to its fast onset of analgesia and short duration of action (4 to 5 hours) 135 ; to avoid the unwanted sedative, withdrawal effect and dependency. 83
Systemic medical approach
Several kinds of psychotropic agents, including tricyclic antidepressants, serotonin, and norepinephrine reuptake inhibitors (SNRIs), selective serotonin reuptake inhibitors (SSRIs), and benzodiazepines, have been used for BMS patients. 136 Tricyclic antidepressants (TCA) such as amitriptyline and nortriptyline are considered as the first-line analgesia used in chronic neuropathic pain. Low doses of amitriptyline 10–40 mg per day have been demonstrated to be beneficial in relieving the pain. 44 A retrospective study that evaluated the efficacy of clonazepam and amitriptyline in the treatment of BMS reported a significant reduction of pain score by 2.2 units and 1.4 units (after 6 weeks), and 2.7 units and 3.4 units (after 3 months) respectively. Clonazepam gave an immediate pain relief effect, but no statistical differences between both treatments in the reduction of pain. 137 Both clonazepam and amitriptyline were effective in the treatment of BMS. However, the undesirable side effects of amitriptyline, such as dry mouth and drowsiness, may be poorly tolerated by patients.
Clonazepam inhibits the central neuronal hyperexcitability by binding to the GABA receptors and elevates the serotonergic descending pain inhibition pathway. 138 Analysis of two retrospective studies 139,140 and a cohort study 141 reported a significant reduction of an average three units pain score with a mean daily clonazepam dosage of 0.5 mg to 1.5 mg. These similar findings were reported in a daily 0.5 mg clonazepam intake randomised control trial, 142 with an effective decrease of 2.5 units of pain score using daily 0.5 mg clonazepam. Amos et al. combined both topical and systemic effect of clonazepam by dissolving the tablet in the mouth before swallowing it and recorded 80% of the patients obtained a 50% reduction in pain score. 140 Clonazepam is generally well tolerated or has mild and/or transient side effects such as tiredness, dizziness, mood changes, forgetfulness, vivid dream and temporary unpleasant taste. 140,142 However, when a higher dose was used, the associated side effects were intolerable and caused discontinuation of the medication. 141 Clonazepam longer half-life may contribute to the lesser withdrawal effect upon discontinuation. 141 Barker et al. revealed a 70% reduction of pain in the clonazepam group compare to 55% of BMS patients using diazepam. 139
If patient unable to tolerate the side effect of clonazepam and TCA, other alternatives, such as gabapentin, alpha-lipoic acid, or cognitive-behavioural therapy, can be used. Anticonvulsants such as gabapentin and pregabalin are often used in neuropathic and somatoform pain, and patients are much more acceptable to the drugs side effects such as dizziness or fatigue. A randomised control double-blind study by Lopez-D’alessandro and Escovich 143 has reported that the combination used of alpha-lipoic acid (600 mg/day) and gabapentin (300 mg/day) for 2 months demonstrated a definite pain reduction in BMS patient than placebo or single drug used. However, the used of gabapentin in BMS are not well studied, with inconsistent results due to the limited numbers of prospective control trials and small study size. Supportive psychology care, in combination with medications, may improve BMS symptoms to better control and a positive impact on patient’s oral health quality.
Selective serotonin reuptake inhibitors (SSRI) and serotonin-norepinephrine reuptake inhibitors (SNRI) are increasingly used in the treatment of BMS. SSRI and SNRI are thought to behave less frequently and have milder adverse reactions compared to TCA. A non-control prospective study revealed that 80% of BMS patients achieved improvement in the BMS symptoms after receiving 12 weeks of 10–30 mg of paroxetine. 144 The use of SNRI has reduced 50% of VAS scores in the BMS patients after 12 weeks of treatment with milnacipran (30 to 60 mg). 145 Treatment with 20 mg to 30 mg per day of duloxetine for 12 weeks reported a desirable outcome in managing non-organic chronic neuropathic pain. 146
Psychological interventions
Chronic pain disorders may not be a fatal disease but affect markedly on the quality of life that may lead to psychiatric comorbidity with a 20% suicidal risk. 21 The clinician should always be vigilance on patients with suicidal behaviours risk, especially for a patient that has co-occurring of other pain disorders. There is a closed relationship between pain and psychology disorders, but whether they are independent of each, remains unknown. Cognitive behavioural therapy and group psychotherapy are non-pharmacological treatments that could be used alone or in combination with medications in BMS. 147 –151 It is non-invasive and has shown positive results in reducing pain intensity and anxiety in BMS patients. 147 –151 It is believed that cognition, emotion, and behaviour are interlinked, and alteration of this dysfunctional complex can rectify the irrational emotional responses to pain. 152 The behavioural and psychotherapy session aims to enable patients to understand the symptoms and their emotional distress; to be motivated and independent in exploring a more targeted personalised pain management.
Homoeopathic approach
Alpha-lipoic acid (ALA) is an antioxidant that able to eliminate free radicals and exert activity in nerve repair and can be found easily in health supplement shops. BMS, which is associated with stress, has been related to the production of toxic free radicals. A follow-up of 73% BMS patients at 12 months showed an improvement with a 2-month trial of 200 mg alpha-lipoic acid supplement pills. 153 However, the efficacy of ALA requires further research.
The use of low-level laser therapy (LLLT) or photobiomodulation has shown to relieve the pain symptoms in BMS patients. However, the inconsistency in LLLT methodologies such as irradiation parameter, number of visits and mixed trial results, suggest further studies are required in determining the efficacy of LLLT treating BMS patients. 154 LLLT is a non-invasive chair-side clinical application treatment. It provides analgesics and anti-inflammatory effects by increasing the release of endogenous pain inhibitors such as serotonin and β-endorphins and decreased in the secretion of inflammatory mediators such as prostaglandins and bradykinin. 155 –157 It prevents the depolarisation of peripheral nerve C-fibre by reducing the action potential amplitude and decreased impulse conductivity velocity. 158 Eight randomised controlled trial using low-level laser therapy reported a significant reduction of the BMS patients’ VAS pain score at the end of the irradiation therapy. All six studies have different laser parameters with a range of wavelength between 630 nm to 980n, the average power density or fluence delivers between 1 J/cm2 to 200 J/cm2 and an exposure time of 4 seconds to 3 minutes and 51 seconds. The session frequency was reported from once weekly to three times weekly with a total minimum session of 4 and a maximum of 10 sessions. 159 –166 No significant difference in pain score reduction was reported between the types of laser used, LLLT or red laser, and between LLLT groups of different wavelength, fluence and therapy frequency. 159,166 All three methods showed significant pain score reduction in comparison to control. The sustainability of LLLT efficacy in pain control throughout 1 month and 3 months was reported in 3 studies. 161,162,164 A study comparing the efficacy of LLLT (980 nm, fluence 10 J/cm2, 10 second per point and twice a week for 5 weeks) and topical mouth rinse of 1 mg clonazepam (three times daily for 21 days) reported improvements in pain perception in both studies, but LLLT was statistically superior in VAS score reduction. Despite LLLT significantly reduced the pain at the end of therapy, the level of anxiety and depression or quality of life has no apparent changes. 162,164,165 However, Valenzuela and Lopez-Jornet reported a significant improved in the quality of health after 2 weeks and 4 weeks of LLLT regardless of the laser doses. 166 Contradictory, LLLT failed to achieve a significant improvement in the BMS patients’ pain 167 even with the most LLLT sessions; a total of 20 sessions in 4 weeks. This study also reported a significant decreased in whole salivary proinflammatory interleukins such as tumour necrosis factor-α (TNF-α) and IL-6. This reflects the reduction in the inflammatory process in the oral cavity and the potential of salivary inflammatory mediators as therapy marker in BMS.
Acupuncture is a traditional Chinese medicine technique, and its benefits have been popularised in Western countries as an alternative method for treating pain. A review control trial in Chinese articles that compared the treatment of BMS with acupuncture had presented a significant improvement in symptoms compared with the control. 168 The pain relief period from acupuncture may not be consistent for every patient and require multiple treatment visits.
A randomised control trial using repetitive transcranial magnetic stimulation (rTMS) for brain stimulation, published significant pain relief in BMS patients compared with controls after rTMS for 10 days. 169 The favourable outcome from rTMS allows us to confirm that BMS is a neuropathic pain involving the trigeminal nerve. It is non-invasive if the safety guidelines are applied, but further studies need to confirm the use of rTMS.
Conclusion
BMS is a diagnosis by exclusion, as yet a systematic and standardised BMS diagnosis protocol is yet to be established. In distinguishing BMS from ‘oral burning symptoms’, multiple strategies are required. A consensus in a stratified approach to assess and diagnose BMS using the agreed ICOP BMS classification should be sought and hopefully ascertain with the extent of centrally or peripherally driven neuropathic and/or nociplastic pain. This approach is required to improve the management of patients with BMS, which, to date, is unsatisfactory. Research must focus on genetic, environmental, and familial factors to better understand the susceptibility of individuals in developing BMS and preventing it where possible.
Article highlights
The relevance of burning mouth syndrome (BMS) classification as chronic idiopathic pain with its aetiopathogenesis (psychology, neuropathic, and endocrinology disorders), diagnosis, and treatment.
The relevance of the interdependent relationship between psychological factors, comorbidities or other chronic pain disorders, and the peripheral and central nervous systems in BMS.
BMS has significant functional and psychological impacts on the patient’s quality of life.
Efficacy of various treatment options concerning the pathophysiology and diagnosis of primary BMS, to provide holistic care to BMS patients.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: We would like to acknowledge our research grant funder 'WellcomeTrust' for the BMS project.
