Abstract
Objective
To review the clinical entity of primary burning mouth syndrome (BMS), its pathophysiological mechanisms, accurate new diagnostic methods and evidence-based treatment options, and to describe novel lines for future research regarding aetiology, pathophysiology, and new therapeutic strategies.
Description
Primary BMS is a chronic neuropathic intraoral pain condition that despite typical symptoms lacks clear clinical signs of neuropathic involvement. With advanced diagnostic methods, such as quantitative sensory testing of small somatosensory and taste afferents, neurophysiological recordings of the trigeminal system, and peripheral nerve blocks, most BMS patients can be classified into the peripheral or central type of neuropathic pain. These two types differ regarding pathophysiological mechanisms, efficacy of available treatments, and psychiatric comorbidity. The two types may overlap in individual patients. BMS is most frequent in postmenopausal women, with general population prevalence of around 1%. Treatment of BMS is difficult; best evidence exists for efficacy of topical and systemic clonazepam. Hormonal substitution, dopaminergic medications, and therapeutic non-invasive neuromodulation may provide efficient mechanism-based treatments for BMS in the future.
Conclusion
We present a novel comprehensive hypothesis of primary BMS, gathering the hormonal, neuropathic, and genetic factors presumably required in the genesis of the condition. This will aid in future research on pathophysiology and risk factors of BMS, and boost treatment trials taking into account individual mechanism profiles and subgroup-clusters.
Definition
The International Headache Society (1) defines burning mouth syndrome (BMS) as an “intraoral burning or dysaesthetic sensation, recurring daily for more than 2 hours per day over more than 3 months, without clinically evident causative lesions”. This definition does not regard BMS as a psychogenic pain, as it was generally held to be until two decades ago (2,3), but on the contrary lists it under the heading “painful cranial neuropathies” in the IHS classification (1) and includes “burning or dysaesthetic sensation”, clearly referring to neuropathic mechanisms. This newer view has been substantiated by recent histological, neurophysiologic, brain imaging and quantitative sensory testing (QST) data, as will be described in detail further on.
Classification
The International Association for the Study of Pain (IASP) first recognized BMS in 1994 as an identified entity, labelled “glossodynia and sore mouth” (4). The initial proposal that BMS belonged to a group of idiopathic orofacial conditions, also including atypical odontalgia, atypical facial pain and some of the temporo-mandibular joint and masticatory muscle pains (5,6) was later confirmed when cluster analysis was applied to all types of chronic orofacial pain states in a prospective multicentre study (7). The analysis showed three main clusters, representing neuralgic, vascular, and idiopathic groups of orofacial pains. Within the idiopathic group, later called the dysfunctional group, BMS emerged as the most distinct subgroup with a homogeneous set of symptoms.
In 2013, IHS defined the following diagnostic criteria for primary BMS (1): (a) Oral pain fulfilling criteria B and C; (b) recurring daily for more than 2 hours per day for more than 3 months; (c) pain has both of the following characteristics: 1) Burning quality, 2) felt superficially in the oral mucosa; (d) oral mucosa is of normal appearance and clinical examination including (clinical) sensory testing is normal; (e) not better accounted for by another ICHD-3 diagnosis. The reliability and validity of these novel criteria have yet to be tested.
BMS and other conditions with a burning sensation in the mouth
When proposing BMS as an entity in 1994, IASP included oral candidiasis, erosive lichen planus, geographic tongue and several systemic causes of intraoral burning pain in the differential diagnoses of primary BMS (4). Pain of the oral mucosa that could be explained by a local or systemic pathology was therefore excluded from the entity of primary BMS, in which no systemic or local causes or visible pathology can be identified in careful clinical examination. The pain or burning sensation of the mucosa caused by a known disease process has been recognized as secondary BMS (8–10). Factors thought to be able to cause such secondary intraoral burning symptoms include deficiencies in iron, ferritin, zinc, vitamin B12, or folic acid, together with certain medications, Sjögren’s syndrome, diabetes, mucosal erosive lesions, candida infection, ill-fitting dentures, parafunctional habits such as tongue thrusting, and denture-induced allergy. These have not been found to play any role in the pathophysiology of primary BMS (1,11,12). The secondary burning mouth symptoms can often be successfully managed with an aetiological treatment (8) as shown, for example, for candidiasis (13) and allergy (14).
Terminology
Over the years many other terms have been proposed to emphasize one aspect or another of the disease, such as glossodynia, idiopathic glossodynia, sore tongue, burning tongue, stomatopyresis, and glossopyresis (8,15–17).
The term stomatodynia excludes “secondary BMS” and purposely avoids the term “syndrome”, since the condition may clinically be considered a homogeneous disease with various symptom combinations (e.g. burning pain, dry mouth, and dysgeusia including taste phantoms). The terms “sore mouth” and “oral dysaesthesia” offer the advantage of including those cases in which neuropathic signs without burning are the main symptoms, but are rather ill-defined and deny the condition the status of a disease or syndrome. Finally, the term BMS, though debatable, will be used here, as it is the current officially recognized term (1).
Symptomatology
More precise knowledge of the symptoms characteristic of primary BMS has recently been obtained by the use of diaries, which avoids the distortion induced by memory-based evaluation in transversal or retrospective studies, and a more accurate definition of BMS through better discrimination between primary and secondary BMS (18–20).
Localisation
The most frequently affected area is the tongue, especially its tip and anterior two thirds. The tongue is the only affected area in about half of patients, followed by the anterior palate and gingivae, the lower lips and the pharynx (3). In typical cases, symptoms are bilateral and symmetrical (21). While unilateral localisation is possible, it is always independent of the anatomy of the peripheral nerves.
Temporal features
In general, pain tends to worsen during the day (20–23), patients being sometimes pain-free during the night and on waking, but constant diurnal pain may also occur (18,19). Most patients describe a long-lasting but intermittent daily pain. In one study, total mean pain duration in 24 hours was 8.8 h ± 4.1 h (18). In the current definition of primary BMS, the pain should be daily (1).
In about one half of patients the onset is spontaneous, although one third associate it with a dental procedure or other medical triggering factor (11). BMS is generally present over a number of years (11,24), but there may be periods of remission, sometimes enduring (24). The natural evolution of the symptoms has not been evaluated in prospective studies. One recent five-year follow-up study suggested that resolution, either spontaneous or due to treatment, occurs in less than five percent of cases (25,26). In our clinical experience, the outcome may be more favourable at longer follow-up.
Interaction with sleep
Reports on sleep disturbance in primary BMS are conflicting. Some studies have reported high incidence of sleep interaction with BMS pain, whereas others have found that up to 70% of patients are not disturbed by pain when falling asleep (11,18,27,28). Sleep disturbance may actually be recorded as a parallel phenomenon (21,29), occurring as frequently as in other dysfunctional pain conditions, but with no causal relationship with pain, loss of sleep being rarely due to the presence of pain (22,29,30). On the other hand, shared vulnerability to BMS pain and depression or anxiety may also be associated with sleep disturbances in BMS (31).
Symptom quality and intensity
The most common term used to describe BMS pain is “burning” (3). Burning pain is also the most common pain quality in neuropathic pain; a further similarity between neuropathic and BMS pain is the frequent occurrence of dysaesthesia. BMS patients may use many additional pain descriptors, some relating to pain quality such as “stinging”, “pricking”, “throbbing”, others to dysaesthetic disturbances reported as “troublesome”, “numbness”, “annoying” or “pain evoked by brushing”, “scalding”, “tingling, sand or thread sensation” (16,19). There is no major paroxysmal component.
The use of a diary has confirmed that pain intensity varies widely among patients and during the day, ranging from a slight irritation to the worst pain imaginable. In prospective series, the mean pain intensity ranges between 3.1 and 5.1/10 on different pain scales (18–20), somewhat lower than reported in retrospective studies.
Modulating factors (triggering, enhancing, alleviating)
Pain is generally spontaneous, but in some patients it may be triggered by certain foods, particularly spicy or acidic ones (3,21,23). Stress and tiredness have also been mentioned as enhancing factors (18). In other patients, a cold drink may alleviate pain (3,23). Often, stimulation of the large fibres by eating (32), chewing gum, or sucking dried fruit alleviates pain, and may be resorted to by patients for that purpose.
Accompanying factors: Xerostomia
Dry mouth (xerostomia) is a frequent complaint, its prevalence ranging from 46% to 70% in primary BMS (8,33). It was first thought that xerostomia was only a subjective impression in BMS patients, possibly due to sensory impairment, influence of medication, or psychological problems, and not caused by a real decrease in salivary flow (11,23,28,32,34,35). Despite a probably unchanged stimulated salivary flow in patients with BMS compared with controls (36), it has recently been found that unstimulated salivary flow is nearly always decreased compared with controls (37–40). In addition, the composition of the saliva is different in BMS patients and controls. Saliva in patients shows decreased concentrations of albumin, immunoglobulins, zinc and lysozyme, but a higher viscosity, and higher concentration of electrolytes and alpha amylase, a marker of chronic stress (36,40–43). The changes in salivary flow or composition do not differ in subgroups of BMS patients characterized by symptoms of dry mouth, dysgeusia, or pain alone (42).
Accompanying factors: Dysgeusia
Dysgeusia (altered taste perception, including taste phantoms) is reported by more than two thirds of BMS patients (3,21,23,32,44). The nature of dysgeusia varies widely among primary BMS patients. It may involve all taste modalities, but a “bitter”, “metallic” or “foul” taste is most frequently described (8,32,45). Distorted taste sensations can be blocked by topical anaesthesia of the tongue in some BMS patients, indicating peripherally altered gustatory function (46). Quantitative evaluation of the taste system has confirmed the dysfunction underlying dysgueusia, but the multiplicity of available techniques of stimulation and sensation recording prevents any general conclusions on which modality is most impaired, or on the degree of dysgeusia. The whole mouth and sipping methods tend to show less impairment (40,45) than more local stimulation made with either drops of chemicals (43) or electrical stimulation in electrogustatometry (47,48,49).
Like the changes observed with salivary flow and composition, alterations in taste threshold are independent of the symptoms of dry mouth, subjective taste alteration, or pain alone (42).
Accompanying factors: Comorbidity
Patients with BMS complain more often than controls of dysfunctional illnesses such as gastrointestinal symptoms and chronic fatigue (50–52,). In addition, frequent comorbid conditions include depression and anxiety (3,12,21,30,31,53). BMS also commonly occurs simultaneously or consecutively with other orofacial (54,55) or more widespread chronic pain conditions (18,31). Association with vulvodynia is rare (56).
Accompanying factors: Psychological symptoms
Before its neuropathic nature was discovered, BMS had long been classified as a psychogenic pain. This mis-assignment was prompted by high scores in psychometric scales, in particular for depression and anxiety (3,12,21,30,34,48,57). One recent study suggests that anxiety may be more important than depression (50), linked to the high prevalence of parafunctional habits (58). A thorough analysis using structured psychiatric interviews (SCID axis I and II) more recently confirmed that anxiety (47.5%) and major depression (35%) were the most common lifetime disorders in BMS patients (31). Several studies have explored personality traits and related alterations in BMS (22,27,31,34,59–62). All except one (63) found higher scores in several characteristics related to type C personality disorders (fearful and neurotic). Neuroticism, i.e. overreaction to trivial stress stimulations, has been most frequently noted. Harm avoidance and perceived stress are also frequent. Reward dependence, obsessive-compulsive and schizotypal personality, and lower levels of novelty seeking and self-directedness have also been reported (8,12,31). In addition, many patients with BMS are cancerophobic (17,23,50), a feature that may be particularly marked when there is a family history of head and neck cancer (8). Regarding the pathophysiology of BMS (cf. Section 4), it is of interest that most psychiatric and personality disorders associated with BMS have in common low brain dopamine tone (31,64).
Although a relationship between psychological factors and BMS obviously exists, a strict causal relationship, specific to this condition, is unlikely. Several studies have shown that in any given sample of patients with BMS, some have high psychiatric scores and others do not (3,5). Rojo et al. (53) found that their patients with BMS could be divided into two groups of equal size. The first presented more symptoms of anxiety, depression, obsession, somatization and hostility than the control group, while the second was not distinguishable from controls. This shows that although anxiety and depression are frequently encountered in this population, they are by no means present in all cases. The correlation between psychological factors and BMS is no more pronounced than it is in any other chronic pain entities, including other idiopathic orofacial pain (22,31,51,65–70). Kim et al. (71) observed no difference in the psychological profiles between male, young premenopausal women and older postmenopausal women with BMS, although they observed significant symptom differences between these subgroups. In most cases, the presence of a psychiatric disorder can be considered as a consequence or a comorbid condition, and not the cause of BMS pain (31,72). Some authors have suggested that a psychogenic pain mechanism might be triggered by a preceding adverse life event (6,54,57,73), but no such association was found in BMS in a controlled study (30). A positive response to treatment with tricyclic antidepressants had earlier been taken as an argument for a psychogenic aetiology (74). It is, however, well known that the analgesic activity of antidepressants is largely independent of their antidepressant activity (75,76). These drugs have been shown to be most efficient in neuropathic pain (77), and also active in non-depressed patients (78,79). Similarly, the efficacy of behavioural and cognitive therapy may be more closely related to improvement in coping than to an aetiological cure (11).
In summary, psychological and emotional factors, personality characteristics or life events cannot be considered directly causal of BMS. However, psychosocial factors may predispose certain individuals to such pain, and may also perpetuate the condition once symptoms have become established (80). The observation of an onset of psychological disorders preceding the onset of pain symptoms might be a good indication for some kind of predisposing factor in the aetiology or mechanism of BMS. Such a sequence may exist for BMS (30,31).
Epidemiology
The prevalence of BMS has been reported to range hugely, from 0.01 % (16) to 40 % (81). This very wide range of prevalence figures for primary BMS may be due to several factors. Rather loose diagnostic criteria applied in earlier studies have obviously resulted in heterogeneous patient populations including both primary and secondary BMS, resulting in misleadingly high prevalence figures (17). The highest prevalence has been reported in studies taking all forms of pain or burning sensation in the oral mucosa into account, particularly in epidemiological studies undertaken using postal questionnaires (41,82–84). These factors could explain why a prevalence as high as 15% has been reported in the general population (41). The studies of the Toronto group also demonstrate this effect: The initial prevalence of BMS was 4.5% in their general population sample following a postal questionnaire (85), but when these results were followed up by a telephone interview, and a more precise diagnosis was made, the prevalence fell to 1.5% (24). Even according to the authors this figure is probably unrealistically high (32).
Choice of sample is another poorly controlled source of variation in many epidemiological studies, leading to study groups less representative of the general population. This is emphasized in a study in which three different populations were sampled for comparison. It was found that the prevalence figures in patients attending a menopause, diabetic, or general dental clinic were 26%, 10% and 2.6%, respectively (83). The highest value of 40% so far reported was for elderly women attending a menopause clinic (81). Bergdahl and Bergdahl (34) found that the prevalence of primary BMS increased with age, with the highest prevalence (12%) in women aged 60–69 years. Other studies have also reported rather high prevalence in older age groups, especially in postmenopausal women (18%) (11).
All studies agree that women are much more frequently affected than men. The relative proportion is 3–20 women for one man, depending on the study (28). These figures may be biased by the fact that women are more likely to seek medical assistance than men (82), but this does not fully explain the constant female preponderance in all studies of primary BMS. Women affected are most often menopausal or post-menopausal, with an average age of approximately 60 years (17,28,86). Men with BMS have been reported to be either younger (41) or older (86) than female patients.
In summary, the more reliable prevalence figures as indicated by well-controlled studies range from 3.7% in an adult population (34) to values around 1% as shown in a recent retrospective study (87), or even as low as 0.01% according to one population-based study (86) and one review (16).
Pathophysiology
The pathophysiology of primary BMS has remained largely unknown until recently. However, during the last decade, after the introduction of the 2004 IHS classification criteria defining primary and secondary BMS (IHS 2004)(88), systematic and detailed neurophysiological, psychophysical, neuropathological and brain imaging studies using modern diagnostic criteria have brought great advances in the understanding of the pathophysiology of primary BMS. They have provided consistent evidence for neuropathic alterations in primary BMS as reviewed, for example, by Jääskeläinen (64), Forssell et al. (89), Kolkka-Palomaa et al. (90), and Woda et al. (91). The current data illustrates that similar clinical BMS conditions can result from various nervous system lesions along the trigeminal neuraxis, as well as from low dopaminergic tone within the nigrostriatal pathway, which exerts descending inhibitory control on the medullary trigeminal complex (64). Despite growing evidence for neurogenic mechanisms in most primary BMS patients, there is a small subgroup of primary BMS patients (≈15%) showing no abnormalities in neurophysiologic and QST measurements.
As discussed above, in earlier studies, psychiatric disorders were often causally linked to the “wastebasket diagnosis” of BMS (30). More recently, these comorbidities, while recognized in BMS (31,48), have been suggested to arise from shared vulnerability reflected in similar neurotransmitter pathology, i.e. low brain dopamine in both BMS and depression (31,64), or as a secondary phenomenon common for chronic pain conditions in general (92).
Role of peripheral neuropathology in primary BMS
Initial neurophysiological evidence for trigeminal system neuropathology in BMS came from brainstem reflex recordings made in patients clinically diagnosed as having primary BMS (93,94). The blink reflex (BR) tested with supraorbital, mental and lingual nerve stimulations has revealed focal or more widespread trigeminal system pathology in approximately 20% of the primary BMS patients either within the peripheral trigeminal distributions (lingual, mandibular or trigeminal nerves) or within brainstem connections (93–95). In addition, the electrical thresholds to elicit BR have been found to be higher in primary BMS patients than in control subjects, indicating hypofunction of the trigeminal tactile Aβ fibres, even within the non-symptomatic supraorbital nerve distribution. These neurophysiological measures of large fibre function suggest that subclinical trigeminal neuropathy forms one subgroup of primary BMS, when diagnosed using the current criteria (1,88). The neurophysiological signs of peripheral trigeminal neuropathic involvement are in line with the finding that 50% of primary BMS patients show significant pain relief after anaesthesia of the lingual nerve with lidocaine block, indicating a peripheral origin of the pain (48). The finding of subclinical trigeminal neuropathy in primary BMS is unsurprising, as neurophysiological measurements have repeatedly been shown to outperform clinical examination in detecting peripheral neuropathies (96–103).
Since small fibre damage is considered a prerequisite for the development of neuropathic pain (104), recent research interest in primary BMS has focused on the role of small myelinated Aδ and umyelinated C fibres. Small fibre function can be assessed with either psychophysical quantitative sensory testing (QST) of thermal and nociceptive modalities, or neurophysiological recording of thermal and pain-evoked potentials (EP) and nociceptive reflexes (102,103,105–107). Neuropathological investigation of epithelial small fibre endings from skin or mucosal biopsies provides anatomical information on the peripheral small fibre system (108,109).
The first study on thermal quantitative sensory testing (tQST) in BMS (22) found decreased heat pain tolerance only at the tip of the tongue in BMS patients compared with control subjects, whereas the thermal detection thresholds were normal with the large thermode used. Later studies applying more adequately sized stimuli with laser (110) or small thermodes (48,94,95,111,112) enabling stimulation of the small trigeminal distributions have revealed profound alterations in thermal detection thresholds at tongue mucosa of primary BMS patients. Most often found are negative signs, i.e. hypoaesthesia or anaesthesia to innocuous thermal stimuli and, slightly less often, to heat pain (hypoalgesia, analgesia). Later, these negative signs were found to occur in 76% (94) of primary BMS patients, giving the first clear evidence for somatosensory small fibre pathology in primary BMS, and indicating neuropathic involvement in these patients. Loss of function in innocuous thermal modalities reflecting small fibre system pathology can be considered a hallmark of neuropathic pain, as recently reported in a large multicentre QST study (113). More specifically, in trigeminal neuropathies due to peripheral lesions of the nerve and presenting with persistent burning pain, multimodal EP recordings have demonstrated specific sparing of C fibre responses in the absence of Aδ responses (114). This indicates that the remaining C fibres, which in physiological conditions mediate innocuous warming and slow second pain sensations, are responsible for the ongoing painful burning sensation in these patients. In addition, Fabry disease (hereditary lysosomal storage disease) is associated with peripheral small fibre neuropathy with predominant Aδ fibre damage and very typical severe burning pain symptoms (115). Analogically, QST profiles with more frequent and pronounced Aδ than C fibre damage have been noted in primary BMS (94,95). Furthermore, the clinical representation of BMS recalls the burning, dysaesthetic pain sensation evoked by the thermal grill illusion (116). In this illusion, distorted activity of the Aδ cool afferents (which normally inhibit polymodal C fibre nociceptors) results in disinhibition of the system, and a consequent burning pain sensation (117). Thus, decreased Aδ channel signalling with relative preservation of C fibre function could be responsible for burning pain sensations in primary BMS (64).
In line with the considerations of the role of somatosensory Aδ fibre deafferentation in the pathophysiology of primary BMS and neuropathic pain, a hypothesis of primary BMS has been developed with the assumption that damage to Aδ taste afferents travelling in the chorda tympani nerve can lead to primary BMS (118,119). Following this hypothesis, the burning pain phantom is caused by a reduction in the inhibitory control normally exerted by taste fibres on somatic afferents, acting either peripherally or centrally (120). Psychophysical assessment of taste thresholds to electrical stimuli can be used to investigate taste deficits or alterations in different oral areas (121). This electrogustatometry has indeed revealed elevation of taste detection thresholds of the tongue mucosa in primary BMS patients, indicating hypofunction of Aδ fibres of the chorda tympani nerve mediating taste sensations (47,48). More specifically, a high taste-to-tingling detection threshold ratio in primary BMS seems to differentiate these patients from both healthy control subjects and patients with secondary BMS (47).
Further confirmatory evidence for focal small fibre damage of the tongue mucosa as an important pathogenetic factor in primary BMS has come from neuropathological investigation of oral mucosal biopsies (95,108,122,123). The structural studies have consistently shown significant loss of epithelial small diameter nerve fibres in the tongue mucosa of BMS patients. Results of these small studies (
Specific immunohistochemical stainings of the tongue mucosal biopsies have demonstrated significant increases in the expression of NGF, TRPV1 ion channels, and P2X3 receptors within the surviving subepithelial nerve fibres of BMS patients (122,123). Up-regulation of these factors is associated with hypersensitivity and neuropathic pain symptoms, and so may also be associated with the pathogenesis of BMS symptoms (122,123).
In addition to negative signs in QST, some primary BMS patients may show positive sensory signs: Decreased heat pain tolerance (22,112), heat allodynia (94) or decreased heat pain detection thresholds, i.e. increased sensitivity to heat pain on the group level comparison (48). With capsaicin applied to the oral mucosa, similarly incongruent results have been reported in primary BMS patients. They may either show capsaicin hypersensitivity with higher pain ratings and longer persistence of pain than controls (110) or increased capsaicin pain thresholds, i.e. less sensitivity to capsaicin-evoked pain than controls (49). Relative capsaicin insensitivity has been associated with the duration of BMS: The longer the disease duration, the more insensitive the patients are. Thermal hyperaesthesia and heat allodynia seem to be rather infrequent, originally reported to be present in 4% of primary BMS patients (94). On the other hand, prevalence figures as high as 40% have also been published (48). The latter study defined normal range with ± 1 SD instead of the more common laboratory practice of using ± 2 SD (or even ± 2.5 SD) to define the reference limits, which may partly explain the higher percentage of heat allodynia. Compatible with signs of gain of function in QST, oral mucosal vasoreactivity to dry ice stimulation has been found to be higher at the hard palate, and the overall variability in blood flow responses over time greater in BMS patients than in control subjects; the disturbed blood flow reactivity is specifically related to BMS symptoms (125).
The fact that primary BMS patients may show opposite types of thermal QST abnormalities (hypoaesthesia or hyperaesthesia, and sometimes hypoaesthesia combined with hyper phenomena, which also occurs in other neuropathic pain conditions) renders group level comparisons of QST findings between patients and control subjects susceptible to random effects that may explain conflicting earlier reports. In addition, as the duration of the BMS condition may influence the results of pain threshold measurements (49), the heterogeneity of patient groups in this regard may lead to further discrepancies between studies. Profiling of QST abnormalities on individual patient level against laboratory-specific reference values may offer a more informative approach to the study of small fibre systems in BMS (94,107,126).
There are two reports on possibly more widespread peripheral nervous system pathology in BMS patients in the form of generalized peripheral small fibre neuropathy (127) or peripheral neuropathy with both large and small fibre involvement (95), but these pilot reports need further confirmation in larger patient groups with properly set diagnosis of primary BMS.
Role of the central nervous system in primary BMS
Functional brain imaging with fMRI has verified that primary BMS patients have patterns of decreased brain activation in the whole brain, and especially in the thalamus, similar to those in patients with neuropathic pain with other origins (128). Brainstem reflex recordings have also shown that some BMS patients may in fact have subclinical focal brainstem pathology in the trigeminal pathways, causing the clinically typical and indistinguishable primary BMS (93,94). Peripheral lidocaine block of the lingual nerve relieves pain in only 50% of primary BMS patients, the other half either remaining at the same level of pain or worsening after the peripheral anaesthesia (48). While in the first half, the pathophysiological process is probably within the peripheral nervous system, in the second half, central mechanisms likely play a major role. In line with this, psychiatric comorbidities such as depression and anxiety also occur more often in the central group (48).
Further evidence for central nervous system pathology in primary BMS comes from neurophysiological recordings. Habituation of the blink reflex is deficient in a subgroup of primary BMS patients (20–30%; 64,93,94). As this deficiency in blink reflex habituation is similar to what is routinely found in Parkinson’s disease, a series of neurotransmitter positron emission tomography (PET) studies were conducted to investigate potential striatal dopamine system dysfunction in primary BMS (64,129,130). These showed depletion of endogenous dopamine in the putamen, prompting the hypothesis that poor efficacy of the endogenous pain inhibitory control via the brain dopamine-opioid system might predispose individual patients to chronic neuropathic pain (64,131). In line with this, it has been shown in experimental studies that lesions of the nigrostriatal pathway (132) and depletion of dopamine (133) within the pathway induce allodynia within the trigeminal distribution. Low brain dopamine tone found by PET imaging in primary BMS patients (64,129) is similar to PET findings in early Parkinson’s disease (134). This is compatible with the view that Parkinson’s disease can predispose patients to BMS and other types of central neuropathic pain, because of the hypofunction of the basal ganglia dopamine system (135,136). In addition, primary BMS patients often experience similar benefit from sleep as do Parkinson’s disease patients, whose condition is best at awakening, owing to an increase in brain dopamine tone during night sleep, while symptoms increase in severity during the day, like in most primary BMS patients (64). Low brain dopamine tone has also been associated with many of the personality traits and psychiatric disorders reported in BMS patients (31). Thus, low endogenous brain dopamine activity might be a shared pathway of vulnerability to both chronic pain and depression, giving an explanation for the frequent comorbid occurrence of these conditions.
Another line of evidence for a role of low brain dopamine tone in the pathogenesis of BMS comes from genetic and brain stimulation studies. Dopamine D2 receptor polymorphism C957T has an effect on the function of striatal D2 receptors, influencing the synaptic concentration of endogenous dopamine. Subjects homozygous for T allele have the lowest striatal dopamine tone, and seem to be overrepresented (50%) in neuropathic orofacial pain patients, including primary BMS (131). In addition, repetitive transcranial magnetic stimulation (rTMS), which releases endogenous dopamine in the putamen (137), holds promise as a novel treatment for intractable neuropathic orofacial pain (138,139).
Diagnostic considerations
Diagnosis of primary BMS is clinical and relies on subjective symptoms according to the current diagnostic criteria (1). Primary BMS is a diagnosis of exclusion, based on the careful ruling out of secondary BMS (8,89,140). A thorough dental examination and detailed history form the basis of the diagnostic work-up, and neurological examination can be helpful in differential diagnosis. When major psychiatric comorbidities are suspected, psychiatric consultation is necessary. In primary BMS, oral mucosa is intact in visual inspection, and orofacial palpation does not reveal any signs of intraoral structural pathology. Careful intraoral sensory testing may help to elucidate somatosensory abnormalities caused by underlying trigeminal system pathology (107). In addition, local and systemic factors leading to treatable secondary BMS should be looked for (for the detailed list, see Section 1).
Tests currently available for the diagnostic work-up of burning mouth (BMS) patients. With appropriate combinations, diagnosis and grading of subclinical trigeminal neuropathies and brainstem pathology can be done. In addition, multiple tests profile the pathophysiological processes on individual patient level, enabling a rational search for more specific aetiologies and mechanism-based treatment approaches.

Clinical neurophysiology with brainstem reflex recordings aids in topographic level diagnostics of focal trigeminal system pathology, allowing accurate diagnosis of subclinical trigeminal pathology. It also aids in the assessment of descending inhibition via the brain dopamine system. The frequently occurring focal intraoral small fibre neuropathy in BMS can be diagnosed with either thermal QST or mucosal biopsies for epithelial nerve fibre density assessment. Even electrogustatometry using both taste and tingling detection thresholds has been suggested as a convenient tool for common diagnostic purposes and differential diagnosis between primary and secondary BMS (47). In one study, local lingual nerve block has also been suggested to be useful in differentiating between the two major subtypes of primary BMS, peripheral and central (48), which requires further confirmatory studies.
Treatment
Secondary BMS can often be treated and cured according to its specific aetiology, after careful diagnosis (see Sections 1 and 5).
As primary BMS displays no objective clinical signs, the pain may be thought psychogenic or psychosomatic in nature. If they feel their suffering is dismissed by their family and medical personnel, patients may lose self-confidence. Faced with a patient’s distress, the doctor’s empathy, listening and reassurance that there is a physiological basis for their symptoms, and that the condition does not imply any threatening evolution, are very important. Patients should be informed afterwards that there may be no treatment that achieves a complete cure, and that only partial relief for the time of the treatment can be expected.
The treatment of BMS is difficult, as it is for all neuropathic pain conditions: On average only 40% of patients benefit from the neuropathic pain medications available (77). Choice among treatment proposals must be made in reference to the quality of the evidence. High-quality RCTs giving positive results, i.e. showing a significant difference from controls, are rare for primary BMS. The following overview is based mainly on RCT studies and recent reviews (33,140–148). Open studies and expert advice will be evoked as weaker evidence.
A recent meta-analysis concluded that clonazepam was effective in inducing symptom remission in patients with BMS (147). It encompasses two modes of use: Topical and systemic. Topical clonazepam was evaluated in two RCTs (149,150). A 1 mg tablet three times daily, sucked for three minutes before spitting, significantly alleviated pain intensity compared with placebo (2 points mean improvement in VAS). The benefit was still present after six months (150). The results were similar in these two studies, and in several other open studies. One third of patients enjoyed total or almost total relief, one third found partial improvement, and one third experienced no improvement (48,149–151,). Although a small concentration can be detected in blood after topical administration, the activity is local, as shown by the kinetics of the effects (149,150). A topical application of clonazepam by mouthwash has been proposed in an open study (152), but the results await confirmation in a controlled study.
Systemic clonazepam has been evaluated in only one small RCT (
In a recent retrospective study, 80% of patients obtained a more than 50% reduction of pain with a combination of topical and systemic clonazepam (average decrease in pain score was 4.7/10 points). Patients kept the clonazepam tablet in their mouths until it had dissolved, before swallowing (156). This retrospective pilot study requires confirmation, especially since data could not be obtained from half of the initially selected patients, but it opens up the possibility of an improved efficiency of clonazepam with the summed effect of two pharmacodynamic modes, as topical and systemic clonazepam would act respectively through peripheral and central mechanisms.
As discussed above in Section 4, there is neuropathic involvement at several levels of the somatosensory system in primary BMS pain. Accordingly, a pure peripheral small fibre neuropathy would best be controlled with topical clonazepam, whereas a central mechanism might better benefit from systemic clonazepam. Interestingly, the hypothesis of an action of clonazepam on GABAA receptors located in the nerve fibres of the oral mucosa (149) has recently been demonstrated in the rat (157). Symmetrically, it can be argued that systemic clonazepam acts on the central component of BMS by exerting an anxiolytic effect, as suggested in a retrospective study reporting that anxiety and depression levels and the presence of dysgeusia or xerostomia were associated with better systemic clonazepam efficiency (158). Higher incidence of taste alterations is reportedly linked with high depression and anxiety scores (159). Diazepam, another benzodiazepine with anxiolytic activity, is also effective for BMS, though less so than clonazepam (155).
Clonazepam, like all benzodiazepines, may induce side effects, mostly drowsiness (154) and when used for long time, impairment of memory and cognitive functions. Side effects have not been reported when it is given topically, despite a very small systemic absorption. After 15 days of topical administration three times daily, the blood concentration of clonazepam was 8 µg/l, well below the minimal therapeutic concentration for anti-epileptic activity (20 µg/l) (149). As an antiepileptic, it is usually used at 4–8 mg per day. Besides unfavourable adverse effects with long-term use, the fact that systemic clonazepam can lead to dependence (140,160) favours topical administration. The availability of clonazepam tablets varies between countries. This difficulty could be overcome by using liquid clonazepam for i.v. or oral use as drops applied directly on the tongue or on a support such as blotting paper or a lump of sugar.
Alphalipoid acid (ALA), an antioxidant sold in health food shops, has been tested in at least nine controlled trials of ranging quality. The most positive results came from the first papers published by Femiano et al. (161–163). Their results have been replicated in one RCT (164) and refuted in several others (165–168). Although some short-term results may be obtained, the results are too heterogeneous between studies to support the efficacy of ALA for the treatment of primary BMS (140,146). Although little or no effect was found in a good open study (169), gabapentin and other anticonvulsivants such as pregabalin have been routinely proposed. A recent RCT showed a positive outcome with gabapentin, with the best results obtained when combined with ALA (164). Lafutidine, a histamine H2-receptor antagonist, has been advocated in one RCT of intermediate quality (170). The large percentage improvement (>50%) warrants a confirmatory study.
Many drugs have been proposed based on open-label studies, expert opinion and case reports. They frequently echo therapy used for other neuropathic pain conditions. For example, tricyclic antidepressants, e.g. amytriptyline and clomipramine, have been tried for BMS, with a rather poor reported outcome of only 19% of patients improving. In one controlled trial, trazodone failed to relieve BMS (171). One reason for poor efficacy of tricyclic antidepressants in BMS may be their adverse effect profile, including dry mouth. Antidepressants belonging to the specific serotonin reuptake inhibitor (SSRI) group have also been proposed (172). Tramadol is frequently used in some countries, although there is little literature to support its use in BMS. Anxiolytic drugs such as chlodiazepoxide (5–10 mg 3/day) or diazepam (6–15 mg/day) have been advocated. Finally, several topical treatments such as salivary substitute (173), tongue protectors or capsaicin (0.01%) mixed in xylocaine gel (2%) twice a day for one month may be useful when a peripheral mechanism is suspected. Figure 1 presents a summary proposal for the pharmacotherapy of primary BMS. Many other recommendations can be found in the recent literature (140,142,145,146,174).
Proposed pathway for pharmacological management of burning mouth syndrome.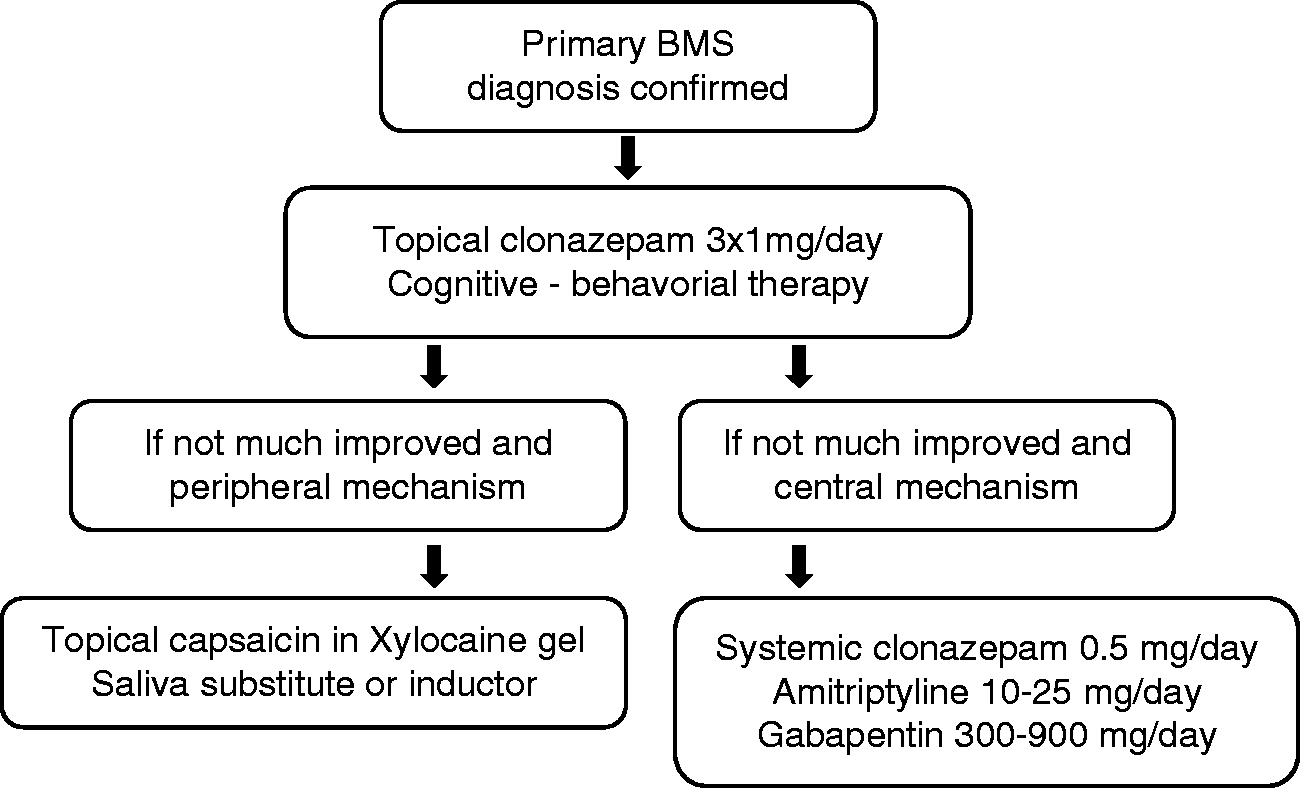
Cognitive behavioural therapy (175) and group psychotherapy (176) have been tested in two RCTs once a week for three or four months with a reduction of pain intensity in most patients. These treatments are non-invasive, and can be indicated although there is no long-term follow-up data after the intervention. Another question arises about the availability of psychotherapeutic treatments. As in other chronic pain conditions, the impact of these therapies may be more on mood and coping than on primary pain mechanisms (140).
Brain stimulation with repetitive transcranial magnetic stimulation (rTMS) has been tested for BMS in a recent well-designed but small RCT (
In summary, there is little evidence at present for an efficient therapy for primary BMS apart from topical clonazepam, and possibly cognitive behavioural therapy. These are completely efficient in only one third of patients. This explains the widespread feeling in the medical community that primary BMS outcome is poor. Novel neuromodulation techniques with non-invasive brain stimulation may in time provide efficient, safe and clinically valid new treatment options for orofacial pain, including BMS.
Expert opinion: Open questions and burning desires
The neuropathic character of BMS is well established, but a question remains: Why do central and peripheral nervous systems undergo pathological changes? Causes must be clearly differentiated from the cascades of mechanisms leading to oral small fibre neuropathy and dopamine system dysfunction and then to symptoms. The pathophysiological cascade itself needs to be better understood. Figure 2 proposes a hypothesis that addresses the four key features of BMS symptomatology and epidemiology: (i) the over-representation of menopausal women; (ii) the high prevalence of anxiety/depressive disorders among BMS patients; (iii) the strictly oral location of the symptoms (91); and (iv) the recent finding that there are peripheral and central neuropathic signs in most primary BMS patients when assessed with more sophisticated diagnostic methods than clinical examination (64).
Hypothesis for the cascade of causes and mechanisms leading to symptoms of primary burning mouth syndrome.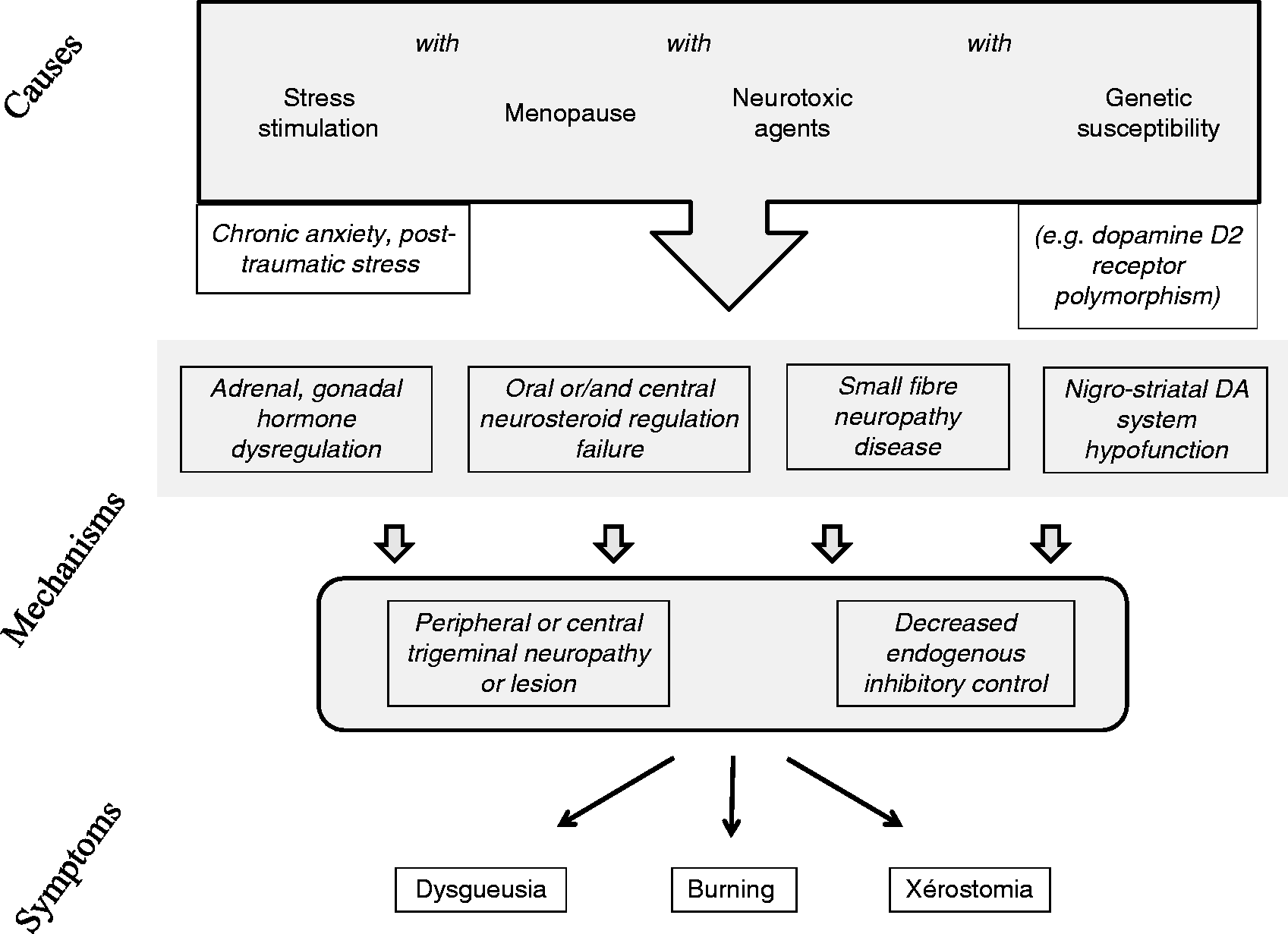
According to the hypothesis proposed in Figure 2, four conditions must be met to trigger BMS: Chronic anxiety or post-traumatic stress with alterations in adrenal steroid physiology and the large changes in gonadal steroid production due to menopause, both leading to a decrease in neuroprotective steroids, the presence of neurotoxic agents, and finally, genetic susceptibility.
The high prevalence of anxiety/depressive disorders found in BMS is associated with a history of post-traumatic chronic stress and/or major chronic anxiety (30,50), which leads to alteration of adrenal steroid production. Among the different modes of HPA dysfunction that have been described in dysfunctional pain conditions, BMS is characterized by basal hypercortisolism (181–183) unlike, for example, myofascial pain, which is characterized by hyperreactivity to ordinary stress events, or fibromyalgia, in which basal hypocortisolism is combined with hyporeactivity to ordinary stress stimuli (184).
The conspicuous preponderance of menopausal or postmenopausal women among the primary BMS patients has been underlined above. Although insufficiently documented, many of the young women or men with BMS also seem to display hormonal imbalance. For example, an over-representation of oophorectomized or hysterectomized women has been reported in two studies (44,50). Also, patients with anti-estrogenic treatment or anti-prostatic treatments, and very old men, can often be found among BMS patients (unpublished observation). Menopause is characterized by a sudden fall of estradiol, progesterone and ovarian androgens. Concomitantly, at that time of life, the gradual decrease in adrenal androgen production has reached 70–80%. Changes in adrenal and gonadal steroid hormones also gravely disturb the synthesis of neurosteroids. Many of the gonadal and adrenal steroids acting as circulating hormones can be synthesized locally in the brain or in peripheral tissues, and are important precursors for neurosteroid synthesis such as that of allopregnanolone or dehydroepiandrosterone (DHEA). The neurosteroids are synthesized in skin and mucosa by some neurons and glial cells in the central nervous system, and by Schwann cells in the peripheral nervous system. They activate the short-acting membrane receptors rather than the genomic receptors activated by the gonadic and adrenal hormones. Neurosteroids modify chloride conductance through GABA A receptors through their interaction with benzodiazepine receptor sites. They are synthesized locally, have a paracrine and autocrine mode of action, and their effects thus appear in restricted brain or somatic areas. Support for a special role of neurosteroids in the localization of pain in the orofacial area in BMS has come from the report of decreased morning salivary DHEA concentration in BMS (185) and from an experimental study on ovarectomized female rats showing increased sensitivity to nociceptive stimulation only of the orofacial region and not of the hind paw. Conversely, gonadectomized male rats showed increased sensitivity of the hind limb and not of the orofacial area. In addition, an increased number of oestrogen receptors in the trigeminal subnucleus caudalis were found in ovarectomized female rats (186).
An impairment of the HPA axis associated with menopause may disturb the control and protection of the nervous tissues. Circulating estradiol and other gonadal steroids, such as testosterone and progesterone with its derivative allopregnanolone, offer protection against injuries and diseases of the nervous system. They also facilitate nerve regeneration (187–190). Neurosteroids including DHEA and DHEA sulphate have proved to be neuroprotective in animal models of, for example, Alzheimer's disease, Parkinson's disease, brain injury and ageing (188,190–192). The myelin sheath and Schwann cells seem to be special targets for the progesterone derivatives, since they have been shown to influence the proliferation of Schwann cells and increase the number of small myelinated fibres in the peripheral nervous system. This is compensated for by a decrease of similar magnitude in the number of unmyelinated axons (187,193). Altered concentration of cortisol, either increased or decreased, may be a major mark of the negative neuronal effects of stress (189,194). Importantly, the local production of the active metabolites of DHEA induces an anti-glucocorticoid effect via a paracrine mechanism (189,195,196).
According to our hypothesis, in addition to menopausal and anxiety-related changes in hormonal and local neuro-active steroids, external or internal factors toxic for the nervous system need to be present at the same time. The hypothesis is that there is a burden on both peripheral small fibres and central nervous system nuclei by environmental, pharmacological or systemic disease-related neurotoxic agents. As already emphasized, a decrease in inhibitory tone exerted by endogenous dopamine-opioid descending systems may be involved in painful BMS condition. Nigrostriatal dopamine neurons are particularly susceptible to environmental toxic agents, which has been speculated to be the reason for the increasing frequency of Parkinson’s disease in modern societies (197,198). Parkinson patients also have a high incidence of comorbid peripheral small fibre neuropathy (199,200). Furthermore, small fibre neuropathies are frequently caused by toxic agents: Most often reported are drugs, but in around 50% of generalized peripheral small fibre neuropathy cases, the causative agent cannot be found (201). These external and internal neurotoxic agents exert their effects simultaneously at various levels of the neuraxis, which may trigger the development of chronic neuropathic pain conditions such as BMS. As neuropathic pain only occurs in a proportion of the patients with neuropathic involvement (104,106), the subjects with genetic liability to neuropathic pain will be at highest risk of developing BMS. The finding of modified salivary function and taste could also be attributed to a regional small fibre neuropathy affecting oral sensation and salivary secretion.
In summary, changes in production of the three sources of steroids (gonads, adrenals and neurosteroids) converge with factors causing nervous system degeneration to trigger small fibre neuropathy and/or nigrostriatal neuronal injury, and finally, with or without genetic susceptibility, painful BMS. Initially, the neuropathic changes are functional and the symptoms may be reversible. Removal of a contraceptive patch, modification of an anti-prostatic treatment, or non-continuous oral contraception may be effective at this stage. Later, nervous system lesions become irreversible, and therapeutic hormonal adjustments will become inefficient.
Expert opinion: What new avenues?
The
Since BMS may be linked to anxiety, or changes in gonadal hormones and neurosteroids, a comprehensive study of plasma levels of several steroids and neurosteroid precursors should be carried out in BMS and matched with healthy controls. An extremely important point to consider is the relative concentrations of the different steroids, which seem to be controlled in a very subtle way (194,202,203). One drawback of these approaches might be highly fluctuating levels of cortico-steroids, and so repetitive cortisol assessments over time with non-invasive methods such as saliva samples or hair sample analyses (204) would be preferable. It is also important to explore the reactivity of the HPA axis. The cortisol awakening response or the dexamethasone/corticotropin-releasing hormone test could be carried out in patients with BMS to find out whether a permanent change in HPA axis reactivity has taken place.
In all research projects, careful clinical diagnosis according to the current criteria (1) should be adopted to rule out secondary BMS. In addition, it is recommended that the patients be accurately profiled with psychophysical and neurophysiological tools for somatosensory and taste functions in future scientific studies on the aetiology, pathophysiology and especially treatment of primary BMS. For current clinical practice, it is suggested that patients should be classified into the peripheral or central type of BMS, which would already at this stage offer a rational way to select the best option from the few treatments available. This could be done either with neurophysiologic and QST tests (Table 1) or by blocking the lingual nerve with a local anaesthetic injection. As it is currently evident that primary BMS mechanisms are not homogeneous, detailed profiling of the neurogenic functional alterations in each patient will allow individual tailoring of the treatment. Even combination therapies against both peripheral and central components may be warranted in the subgroup of primary BMS patients, in whom the investigations reveal signs of both peripheral and central nervous system dysfunction. Future trials should test whether recording of the BR in paired pulse paradigms, measuring habituation of nociceptive EPs, or more accurate testing of taste and smell functions (90) might offer novel, practical diagnostic tools for primary BMS.
A purely psychological approach would also be useful to seek a correlation between post-traumatic chronic stress and primary BMS. More generally, a better understanding of psychiatric comorbidities in BMS patients can be gained with large scale clinical studies adopting structured psychiatric interviews (e.g. SCID I and II) that separate trait and state-related features, i.e., present and past psychiatric disorders, and their occurrence in relation to the manifestation of BMS. These may then finally confirm the comorbid or causal relationships between these entities.
New treatment lines will hopefully emerge from the above hypothesis for the pathophysiology of primary BMS. These might go on to include tailored hormone replacement therapy at the early stages of the disease in menopausal or postmenopausal women, and in men with iatrogenic gonadal hormone dysfunction. As a better understanding of the causes initiating the disease will be gained, aetiological treatment may be expected. Avoidance of exposure to environmental neurotoxic agents such as pesticides may be sought in the future through rising individual and societal awareness. Similarly, rehabilitation of the normal responses from the steroid, sympathetic and immune systems to daily stressful life events could be proposed.
Another novel treatment line for BMS is opened up by the finding of decreased nigrostriatal dopaminergic tone. Medications with dopaminergic effects (e.g. levodopa, specific D2 receptor agonists, bupropion, and tricyclic antidepressants) should be systematically evaluated in RCTs on BMS patients, clustering the patients according to the specific profiles of findings (see Table 1), for example central
rTMS, which activates the endogenous descending dopamine pathway (137–139), also holds promise, and can be used to treat the comorbid psychiatric problems such as depression simultaneously (178). Novel non-invasive neuromodulation techniques are emerging for the same indications as rTMS. In the future, there may be patient-operated transcranial direct current stimulators (tDCS) designed for the home treatment of depression and neuropathic pain, including BMS. Meanwhile, properly controlled tDCS trials on carefully diagnosed primary BMS patients are required.
Article highlights
Primary burning mouth syndrome (BMS) is a chronic neuropathic intraoral pain condition. Primary BMS is most frequent in postmenopausal women, with general population prevalence around 1%. Treatment of primary BMS is difficult; best evidence exists for efficacy of topical and systemic clonazepam. With advanced diagnostic methods, primary BMS can be classified into a purely peripheral or central type or a combination of both types. These types seem to differ regarding pathophysiological mechanisms, efficacy of available treatments, and psychiatric comorbidity.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
