Abstract
Introduction
Burning Mouth Syndrome is characterized by variable symptoms that include pain, burning and paraguesia in an otherwise healthy-appearing oral mucosa. Although the etiopathogenesis of Burning Mouth Syndrome is unknown, some studies provide evidence of subclinical inflammation leading to disrupted cytokine levels.
Aim
To investigate the expression of cytokines and role in the etiopathogenesis of Burning Mouth Syndrome.
Methods
Online databases (MEDLINE and EMBASE) were searched from November 1986 to November 2018 for case control/cross-sectional studies comparing the levels of cytokines in patients with Burning Mouth Syndrome and healthy controls.
Results
A total of eight studies were included in the current review. Four studies were of high and four studies were of moderate quality. Seven studies evaluated IL-6, out of which four showed comparable results, two showed higher levels and one study reported lower levels in Burning Mouth Syndrome patients compared to controls. Four studies assessed IL-2, out of which two reported comparable results whereas one study reported higher levels and one study reported lower levels in Burning Mouth Syndrome patients compared to controls. IL-10 levels were measured in three studies that reported no significant differences in the levels between Burning Mouth Syndrome and healthy controls.
Discussion and conclusion
The etiopathogenesis of Burning Mouth Syndrome is multifactorial. Studies have provided scientific evidence that inflammation plays a key role in Burning Mouth Syndrome pathogenesis. However, whether up-regulation or down-regulation of specific cytokines contribute to the etiopathogenesis of Burning Mouth Syndrome remains debatable. Further high-quality studies with larger sample size and assessing a wider array of cytokines are warranted in order to obtain strong conclusions.
Introduction
“Burning Mouth Syndrome (BMS) is the intra-oral burning or dysesthetic sensation, recurring daily for more than 2 hours/day for more than 3 months, without any clinically evident causative lesion” (1). The common symptoms include pain, burning and altered taste sensation that occur bilaterally in the oral cavity and predominantly involve the anterior two-thirds of the tongue (2,3). Despite the extensive research, the mystery of diagnosing and thereby treating BMS remains unsolved (4). Patients presenting with BMS pose one of the biggest challenges to dentists and oral health care professionals (5).
Cytokines are a group of proteins secreted by immune cells responsible for regulation of host responses to infection, immune modulation, inflammation, and trauma. Pro-inflammatory cytokines worsen the disease, whereas anti-inflammatory cytokines contain the inflammatory process and promote healing. Stress is a major determinant of cytokine levels in the body since cytokines are associated with major neurotransmitters (6). Cytokines play a major role in damaged tissue repair, in the development and spread of cancer, in the control of cell replication and apoptosis, and in the fine-tuning of immune reactions (7). Cytokine receptor activation and hence alteration in the cytokine balance has been proposed to play a major role in neuronal dysfunction and etiopathogenesis of stress (8).
The etiopathology of BMS is ambiguous. However, it has been proposed in studies that an interplay of local, systemic and psychological factors contribute to pathogenesis of BMS (9,10). In their study, Suh et al. reported that BMS is a sub-clinical inflammation that results in a change in cytokine levels, causing symptoms such as burning and pain (11). Cytokines act as potential neuromodulators in the processing of nociceptive signals causing neuropathic pain and/or hyperalgesia. They mediate their functions through glia proliferation, axonal regeneration and proinflammation (12). Galli et al. quantitatively assessed the role of psychological factors in BMS (13). However, to date, to the best of our knowledge there is no published systematic review that has evaluated the levels and role of cytokines in the etiopathogenesis of BMS. Recent cross-sectional studies highlight the significance of cytokines in BMS (12,14). For instance, in their study De Souza et al. concluded that some of the proinflammatory cytokines such as the levels of interleukin (IL)-6 are higher and those of tumour necrosis factor alpha (TNFα) are lower in patients with BMS when compared with healthy controls (HC) (14). However, Chen et al. proposed that serum levels of IL-6 in BMS are lower when compared with HC (12). On the other hand, Barry et al. concluded that levels of TNFα and IL-6 are comparable between BMS and HC (15). The findings with regards to cytokine profile in BMS and HC are conflicting, and a systematic review is deemed necessary to compare the levels of cytokines and to discuss their role in the etiopathogenesis of BMS.
Therefore, the aim of the current systematic review is to evaluate and compare the overall inflammatory burden assessed in terms of cytokine levels in adult patients suffering from BMS and healthy controls.
Materials and methods
Protocol registration
This review was registered at the National Institute for Health Research PROSPERO, International Prospective Register of Systematic Reviews (http://www.crd.york.ac.uk/PROSPERO, registration number CRD42018114525 on 5 December 2018. The Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines (17) were followed. The following focused PECO question was formulated:
In adult patients (Patients) suffering from BMS (Exposure), what is the overall inflammatory burden assessed in terms of cytokine levels (Outcome) when compared with healthy controls (Control)?
Search strategy
The authors (JK and ZA) searched the PubMed (National Institutes of Health, Bethesda) and EMBASE from November 1986 to November 2018 for appropriate articles addressing the focused question. Literature was thoroughly searched for identification of papers that compared the levels of cytokines between BMS and healthy controls. The electronic database search was carried out using varying combinations of the following terminology: (a) “burning mouth syndrome” (b) “oral dysesthesia” (c) “glossodynia” (d) “stomatopyrosis” (e) 1 or 2 or 3 or 4 (f) “Cytokines” (g) “Interleukins” (h) 6 or 7, biomarkers.
Selection criteria
Screening and assessment of articles was independently carried out by two reviewers (JK and SM). Any disagreement among the authors regarding study selection or exclusion was resolved through discussion and/or by consulting a third reviewer (ZA). The eligibility criteria included: i) Cross sectional, case-control and/or retrospective studies in human adult patients; ii) study samples clinically diagnosed with BMS; iii) evaluation of cytokines in patients with BMS in comparison with HC using any type of assay method; and iv) articles published in the English language only. The exclusion criteria included animal studies, studies involving treatment intervention, in-vitro analyses, case reports, case series, abstracts, unpublished articles and letters to the editor.
Data extraction
Data from the included studies was extracted individually by two reviewers (JK and ZA). The information from the included studies was tabulated according to the following sections: Study designs, subject demographics, gender distribution, additional clinical parameters assessed, cytokines studied, sample and assay type and main outcomes (Table 1). All the information obtained was based on the focused question outlined for the present systematic review. The reviewers cross-checked all extracted data. Any differences in opinion were resolved by discussion until consensus was reached.
Quality assessment
The Newcastle-Ottawa Quality Assessment scale (NOS), a validated tool for convenient assessment of the quality of observational and nonrandomized studies was used to evaluate the quality of the included studies (18). According to this scale, the studies were evaluated under three broad perspectives: The selection of the study groups; the comparability of the groups; and the ascertainment of either the exposure or outcome of interest for included studies respectively (19).
Data analysis
Meta-analyses were not possible in the present study due to significant heterogeneity in the methods and presentation of the data.
Results
Study selection
Out of a total of 1160 search results, 17 were duplicate studies and were removed (K = 0.71). Out of 1143 studies, 1125 were excluded as they were found to be irrelevant to the focused question. Eighteen studies were selected for full text review following initial screening of titles and abstracts. A full text review resulted in the exclusion of another 10 studies that did not meet the inclusion criteria and finally a total of eight studies (6,11,12,14,15,20–22) were included in the current review (K = 0.92). All the studies in this review were carried out at registered medical facilities (i.e. clinical/healthcare setups or universities). Figure 1 reports the PRISMA flow diagram of the selection process and excluded articles with reasons.
PRISMA flow diagram for studies retrieved through the searching and selection process.
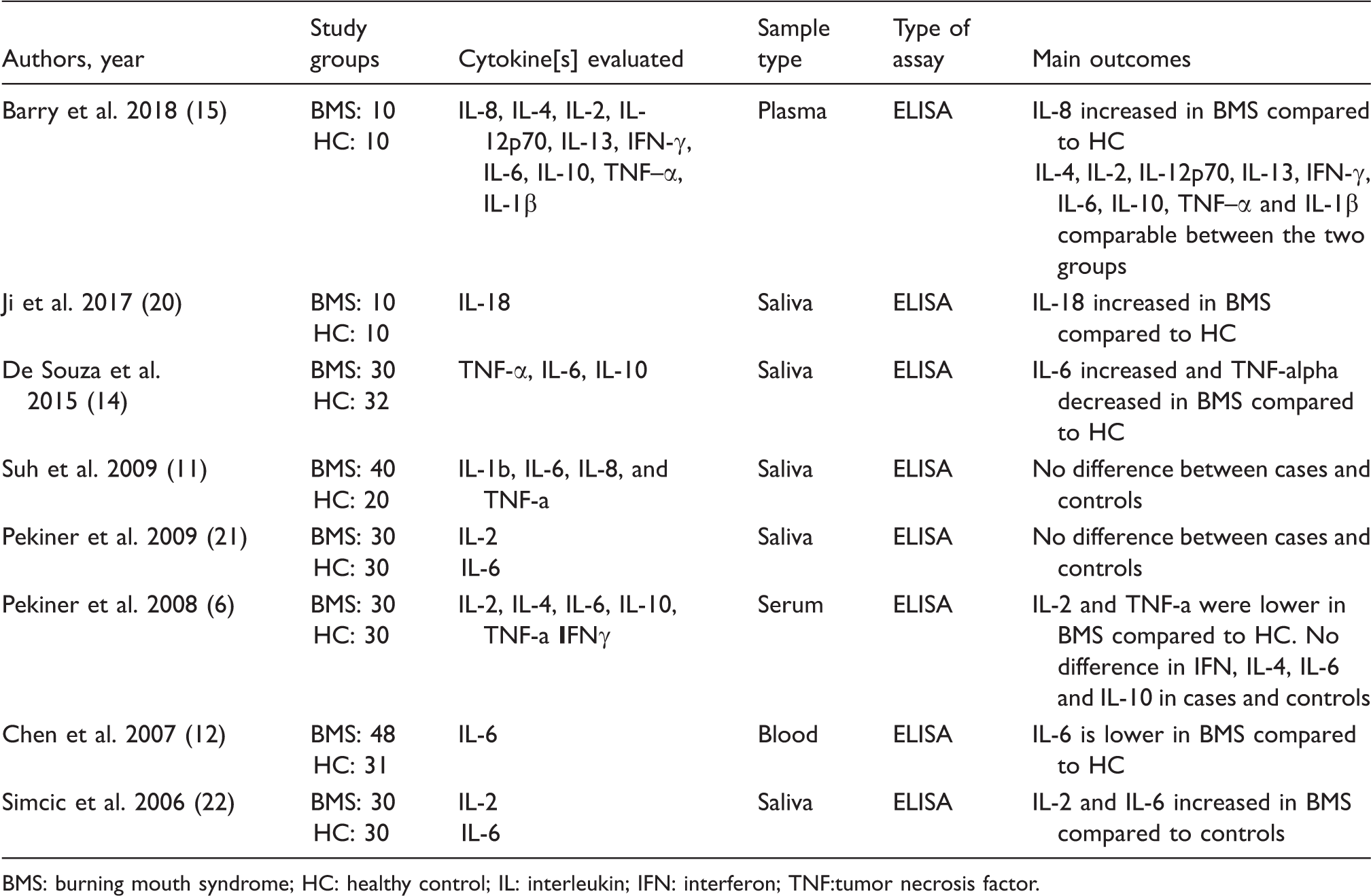
Characteristics of the selected publications
Cytokine assessment of study groups.

BMS: burning mouth syndrome; HC: healthy control; IL: interleukin; IFN: interferon; TNF:tumor necrosis factor.
Cytokine levels among BMS patients
A total of eight cytokines including IL-2, IL-4, IL-6, IL-8, IL-10, IL-1ß, TNF-α and gamma interferon showed differential expression when their levels were evaluated and compared in BMS patients and healthy controls (6,11,12,14,15,20–22). Seven out of eight studies evaluated IL-6 (6,11,12,14,15,21,22) while four studies quantified IL-2 (6,15,21,22) and three studies evaluated TNF-α (6,11,14), respectively. Three out of eight studies measured IL-10 (6,11,14,15), whereas two studies evaluated IL-4 (6,15), IL-8 (11,15), IL-1ß (11,15) and gamma interferon (11,15) levels, respectively.
Main outcomes of the study
The data of the present systematic review could not be quantitatively analyzed due to different presentations of the results, including graphs (15,20), or because the quantities were expressed in mode, median and/or mean without standard deviation (14,21). Therefore, the findings of the present study are reported in narrative manner.
IL-2
Out of the four studies (6,15,21,22) evaluating IL-2 levels, Pekiner et al. (6) reported that subjects in the control group had significantly higher mean values for IL-2 compared with BMS patients. On the contrary, Simcic et al. (22) reported that the average value of IL-2 concentration was significantly higher in the group of patients with BMS compared to the control group. The other two authors concluded that no statistical difference in the IL-2 levels was observed between the two groups (15,21).
IL-6
Four out of seven studies evaluating IL-6 reported comparable results between BMS and healthy controls (6,11,15,21). De Souza et al. and Simcic et al. concluded that levels of IL-6 are higher in BMS (14,22), whereas Chen et al. revealed that the levels were significantly lower in BMS compared to healthy controls (12).
TNF-α
Barry et al. and Suh et al. reported comparable levels of TNF-α between BMS and controls (11,15). On the contrary, De Souza et al. and Pekiner et al. reported lower levels of TNF-α in patients with BMS (6,14).
IL-8
Barry et al. (15) reported IL-8 to be significantly raised in BMS, whereas Suh et al. (11) reported comparable results between BMS and controls.
IL-18
Ji et al. reported a statistically significant increase in the levels of IL-18 in BMS patients compared with healthy controls (20).
IL-4, IL-10, IL-1ß, Gamma interferon
All studies reported no significant difference in the levels of IL-4, IL-10, IL-1ß and gamma interferon between BMS and controls (6,11,12,14,15,20–22).
Risk of bias across studies
General description of the included studies.
BMS: burning mouth syndrome; HC: healthy control; F/M: female/male; IL: interleukin; IFN: interferon; TNF: tumor necrosis factor; UWS: unstimulated whole saliva; SWS: stimulated whole saliva; NA: not applicable; ELISA: enzyme-linked immunosorbent assay; VAS: visual analog scale; HRSD: Hamilton Rating Scale for Depression; QIDS-SR16: Quick Inventory of Depressive Symptomatology-Self-Report.
Quality of studies assessed by Newcastle Ottawa Scale.

Discussion
This review aimed to systematically evaluate the levels of cytokines among patients suffering from BMS and healthy controls, and to further conclude whether cytokines play a part in the etiopathogenesis of BMS. To the best of our knowledge, this is the first systematic review that has evaluated cytokines and their contribution to BMS pathogenesis. However, the outcomes of the present systematic review should be interpreted with extreme caution for several reasons.
There are various studies that postulate the underlying etiopathogenesis of BMS as an amalgamation of inflammation, psychological disturbance and neuropathic derangement (5,23). However, few of the authors have suggested only one of these factors to be solely responsible for the pathogenesis of BMS. Research suggests that conditions altering the psychological well-being of patients, including depression, stress and chronic anxiety, cause an alteration in the production of adrenal steroids (24). This is turn causes dysregulation in the production of a few precursors required in the synthesis of neuroactive steroids that are present in skin, mucosa, and the nervous system (25). The outcome is permanent neurodegenerative alterations in small nerve fibers of the oral mucosa and/or some brain areas involved in oral somatic sensations, resulting in irreversible neuropathic change and hence the symptoms of pain, burning, dysgeusia, and xerostomia associated with BMS (26). Several studies have highlighted the fact that neuropathy is concomitant with BMS (27–29). By providing evidence of diffuse degeneration of epithelial and sub-papillary nerve fibers in the anterior two-thirds of the tongue, Lauria et al. demonstrated that BMS is associated with small-fiber sensory neuropathy (30). Another study reported hypofunction of Aδ fibers in the distribution of the lingual nerve of individuals diagnosed with BMS in addition to hypofunction of C-fibers, proving BMS represents a generalized neuropathy (31).
The etiopathogenesis of BMS has been widely attributed to subclinical inflammation, which has been measured via different cytokine expression levels (14,15,22). However, whether disrupted cytokine levels are a cause or an effect of the disease process in BMS is still undecided. A disruption in the balance between proinflammatory and anti-inflammatory cytokines can either enhance or dampen inflammatory reaction and immune response (32). IL-2 stimulates secretion of proinflammatory cytokines such as IL-1, TNF-α and TNF-β, inducing infiltration and activation of immune cells that result in recruitment of greater numbers of macrophages, mast cells and T-lymphocytes, creating a vicious cycle that makes the inflammatory state constant and chronic. Proinflammatory cytokines are associated with nociceptive signaling and are usually increased in neuropathic pain disorders (33) including BMS (15).
IL-6 is a pleotropic cytokine that influences the antigen-specific immune responses and inflammatory reactions, in addition to modulating pain and inducing hyperalgesia (22). IL-6, along with IL-1 and TNF-α, is a potent modulator of corticotropin-releasing hormone, which is responsible for increased hypothalamic-pituitary-adrenal axis activity leading to an increase in adrenocorticotropic hormone and cortisol, eventually resulting in a major depression event. Pain due to subclinical inflammation and depression is a major component of BMS. Persistently elevated cytokine levels cause a permanent alteration in neurotransmitter function, leading to neuropsychiatric dysfunction, particularly depression (34). On the other hand IL-18, a pro-inflammatory cytokine, acts as an inflammatory mediator by accelerating the immune response via recruitment of neutrophils, macrophages and further acting upon other cytokines like IL-8 and TNF-α (35). In their study, Ji et al. reported elevated levels of IL-18 as most likely contributing to the etiopathogenesis of BMS (20). However, some studies included in this review failed to find any association between deranged cytokine levels and BMS pathogenesis (11,15,21). Two studies reported a negative correlation between BMS and cytokine signature (6,12), implying that inflammation does not play a significant role in BMS etiopathogenesis. It may be hypothesized that psychological and neuropathic disorders could be held responsible as the precipitating factors in the disease initiation. These authors have in common drawn a conclusion that in order to assess BMS development a variety of causative factors need to be considered. The findings of these studies prove to be of little help in designating cytokines as biomarkers for the diagnosis of BMS.
Apart from biochemical analysis, clinical parameters like pain and depression were also assessed by means of calibrated scales and questionnaires. All the authors unanimously concluded that BMS is accompanied by pain of variable intensity. However, whether depression is a cause or an effect of BMS is yet to be affirmed. According to Barry et al. (15), cytokines are elevated in patients with depressive disorders and chemokines/cytokines play a vital role in the development of neuropsychiatric disorders via neural serotonin-catecholamine signaling. Suh et al. (11) measured the gingival index in order to assess the blood contamination of saliva and found no significant difference between patients and controls.
Like any other study, the present study has a few limitations that need to be considered. Firstly, in our present review we have included studies that were published in the English language, which in our view might have resulted in publication bias and loss of relevant studies published in other languages (36,37). The small number of studies eligible for the qualitative analysis may not be sufficient to give us clear results. The number of studies for the quantitative meta-analysis was found to be further reduced because we failed to obtain the means and standard deviations even after correspondence with the authors. In order to extract maximum data for meta-analysis, the corresponding authors of all the articles were contacted where the data was presented in graphical form or there was a lack of data. However, the response rate was 0%. Although IL-6 was the most commonly studied cytokine, a noteworthy fact was that all the included studies evaluated different cytokines. Moreover, the fact that half of the studies had moderate quality reflects a questionable outcome. Additionally, the sample type was not the same in all studies, which could have had a significant impact on the final outcome of the current review while interpreting the results. The different body fluids used in studies could have given skewed outcomes. The oral cavity represents a distinct environmental niche and immune biomarkers present in saliva are influenced by processes of local immunity, leading to a possible discrepancy in the cytokine levels in saliva compared to other body fluids (38,39).
The available studies on BMS and cytokine levels are sparse and variable. Therefore, further high-quality studies are warranted to address the interplay of psychological dimensions, neuropathic involvement and subclinical inflammation in BMS. Since BMS has a severe adverse psychological, physical and social impact on life, a collaborative effort by dentists, oral surgeons and clinical psychologists is needed for timely diagnosis and effective management of the condition.
Conclusion
The available studies on BMS and cytokine levels are sparse and variable. From the present review, it remains debatable whether increased or decreased cytokine levels contribute to the etiopathogenesis of BMS. Therefore, further high-quality studies are warranted to address the interplay of psychological dimensions, neuropathic involvement and subclinical inflammation in BMS.
Footnotes
Article highlights
The expression of cytokines and their role in the etiopathogenesis of BMS were investigated.
A total of eight studies were included in the current review.
Four studies were of high and four of moderate quality.
Whether up-regulation or down-regulation of specific cytokines contribute to the etiopathogenesis of BMS remains debatable.
Further cross-sectional studies with a larger sample size and assessing a wider array of cytokines are warranted in order to obtain strong conclusions.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
