Abstract
Objective:
The aim of this study is to critically analyze the evidence of the efficacy and safety of greater occipital nerve (GON) block for the preventive treatment of chronic migraine (CM).
Background:
A rigorous scientific assessment of efficacy and safety of the GON block for preventive treatment in CM is not available. This critical review was undertaken for this purpose.
Methods:
References for this review were identified by searches of articles published in the English language in PubMed between 1969 and April 15, 2020 using “greater occipital nerve block,” “chronic migraine,” “migraine,” “headache,” and “treatment” as keywords.
Results:
Out of potential 532 articles, 9 open-label and 4 placebo-controlled trials that studied the role of GON block for prevention of CM were identified and reviewed. Open-label trials reported a reduction of headache severity and frequency in 35–68% of patients. The beneficial effect of a single block lasted up to 4 weeks. Randomized controlled trials (RCTs) used varied methodology and techniques of GON block and the outcomes were reported at different time points. A single RCT showed a beneficial effect of the GON block at 1 week. However, the GON block was found to be safe and well tolerated.
Conclusion:
Long-term efficacy of GON block in CM shall need further well-designed RCTs using standardized methodology. This study, in addition, reviewed the limitations and uncertainties regarding the technique and methods of use of GON block in CM.
Introduction
Chronic migraine (CM) is defined as a headache occurring on ≥15 days/month for more than 3 months, having features of migraine headaches for at least 8 days/month. 1 The prevalence of CM is approximately 2% in the general population and is about 8% among migraine patients. 2 CM is a highly disabling condition. 2,3 Patients with CM invariably require prophylactic treatment and are difficult to treat. 4,5 Coexistence of overuse of acute migraine medications, probably the most important risk factor for chronification, makes the treatment of CM even more challenging. 6 A recent study has shown that multiple barriers exist for the correct diagnosis and treatment of CM. 7 Major share of the economic burden due to migraine in terms of direct and indirect cost is attributable to CM. 8
Pharmacological options for the prevention of CM are limited. 5 Most of the established drugs that are used for the preventive treatment of episodic migraine (EM) have not been found to be effective in CM. 9 Currently, only two drugs have a good evidence base for the preventive therapy of CM, namely oral topiramate and botulinum toxin A injection. 4,5 However, both these therapies have their limitations. Based on Phase 3 Research Evaluating Migraine Prophylaxis Therapy data and subsequent open-label extension studies, it has been found that the response to botulinum toxin varies among patients, that is, some may experience headache relief within few weeks after the beginning of treatment and some may take longer. 10 –13 Further, multiple site injections over the face, head, and neck (totaling at least 31 in number) and high cost are downsides of this treatment. Topiramate, based on two pivotal trials in the United States and Europe, has shown to be effective in CM. 14,15 However, again the benefit takes time and may require 4 weeks or more to be clinically evident. Slow titration, multiple adverse events (although mild), and noncompliance are additional downsides. 16 In fact, studies have shown that oral preventive therapies including topiramate in CM have a very poor adherence rate. 17 Thus, the treatment of CM is far from satisfactory. Recent studies using monoclonal antibodies against calcitonin gene-related peptide receptor or ligand for preventive treatment in CM have shown positive results and are well tolerated although cost is a barrier for many patients. 18 –21
One preventive treatment option which has emerged in the recent past for CM is greater occipital nerve (GON) block. 22 GON is the main sensory nerve of the occipital area and derives most of its fibers from the cervical (C2) dorsal root. The rationale for using the GON block for acute and preventive treatment in various headache disorders comes from the proximity of sensory neurons in the upper cervical spinal cord to trigeminal nucleus caudalis (TNC) neurons and the convergence of sensory input to TNC neurons from both cervical and trigeminal fibers. 23 –25 GON block can modulate the excitability of second-order neurons receiving input from both trigeminal and cervical afferents when either of them is stimulated. 26,27 This anatomical and functional continuum between trigeminal and cervical fibers in the so-called trigemino-cervical complex (TCC) underscores the importance of targeting GON for the modulation of central pain in migraine. 28 GON block has additional advantages of being safe, cheap, and having little potential for the drug-to-drug interactions. Despite lacking robust evidence base and a standardized method of its use, GON block is practiced in many headache centers as a preventive treatment for CM. 29 Therefore, it will be worthwhile to critically analyze the evidence for its efficacy and safety and find out the limitations of our current understanding of this treatment.
A recent review by Inan et al. in 2019 evaluated the role of GON block in migraine prophylaxis but did not focus on CM only. 30 They reviewed 14 trials, 7 of which were randomized controlled trials (RCTs). Of these, 5 had placebo-controlled arms and 2 studied the effects of addition of steroids to GON block. Two studies included patients of both EM and CM that did not analyze the results of efficacy and safety separately and therefore the inferences drawn may not be applicable to CM patients alone. While one open-label study included patients of other types of headaches, two others used peripheral nerve blocks (PNBs) in addition to GON block, further complicating the interpretation. A recent meta-analysis and systematic review on the role of GON block in CM also included other headache patients (e.g. cervicogenic headache and medication overuse headache). 31 In the present review, therefore, we shall critically evaluate the role of GON block (without any other PNBs) for the preventive treatment of CM by analyzing the studies that either involved only the CM patients or those that involved both EM and CM patients but the results were available separately for CM patients. Few new studies since the publication of the review by Inan et al. shall also be included. We will also analyze the available literature regarding the utility of adding steroids to local anesthetic (LA) and issues concerning standardization of the technique of GON block which may have a bearing on the outcome.
Materials and methods
We identified potentially eligible studies by searching in PubMed till April 15, 2020, for RCTs, systematic reviews, meta-analysis, observational studies, open-label studies, and case series on GON block for the treatment of CM in accordance with Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines. The keywords used were “greater occipital nerve block,” “chronic migraine,” “migraine,” “headache,” and “treatment.” The search terms were used as keywords and in combination as MeSH terms to maximize the output from literature findings.
Only the studies published in English that examined the role of GON block for preventive treatment of CM (defined by the International Classification of Headache Disorders—(ICHD) criteria) were included. Both open-label studies and RCTs reporting the efficacy and safety of GON block for the treatment of CM were screened. Only those trials that either involved exclusively the CM patients or those that involved both EM and CM patients but the results were available separately for CM patients were included for further analysis. Among the RCTs, only placebo-controlled studies were analyzed for efficacy and safety. RCTs without a placebo arm but comparing add-on medications to GON block such as steroids or comparing different techniques were analyzed under separate headings. Studies that employed multiple PNBs in addition to GON block, single case reports, editorials, and commentaries were excluded. The modified Jadad scale was adapted to check the quality of the placebo-controlled RCTs by analyzing the following parameters, namely method of randomization, method of concealment of allocation to active and placebo arms, method of blinding, a priori sample size and power calculation, clear mention of inclusion and exclusion criteria, clear mention of outcome parameters and their reporting, mention of withdrawals and dropouts, statistical methods applied for adjusting the withdrawals and dropouts, and the adverse effects described for both the active and placebo arms.
Results
PRISMA flowchart showing the selection of articles is shown in Figure 1. A total of 532 articles were found. Both the authors independently read the titles and the abstracts and excluded the articles which prima facie did not address our review question regarding the efficacy and safety of the GON block in CM. Thirteen open-label studies 29,32 –43 and 10 placebo-controlled trials 44 –53 were read in detail. Of these, nine open-label studies 29,34 –36,38 –41,43 and four placebo-controlled studies 47 –50 were found suitable for the present review meeting the inclusion and exclusion criteria. Four open-label 32,33,37,42 and six placebo-controlled studies 44 –46,51 –53 that were excluded for efficacy and safety analysis have been briefly described separately (Appendix 1) to highlight the reasons for their exclusion.

PRISMA flow diagram showing the progression of article selection and the numbers of articles at each step. PRISMA: Preferred Reporting Items for Systematic Reviews and Meta-Analyses.
Evidence of efficacy of GON block for the preventive treatment of CM
Nonrandomized, uncontrolled, open-label studies
Although not considered useful for a true assessment of efficacy because of biases, uncontrolled open-label studies often pave the way for the proper conduct of RCTs if some beneficial effects are observed and the hypothesis is generated. Salient features of nine open-label studies 29,34 –36,38 –41,43 that have been included in this review are outlined in Table 1. These are briefly discussed below.
Uncontrolled open-label observational studies of GON block for preventive treatment of CM.
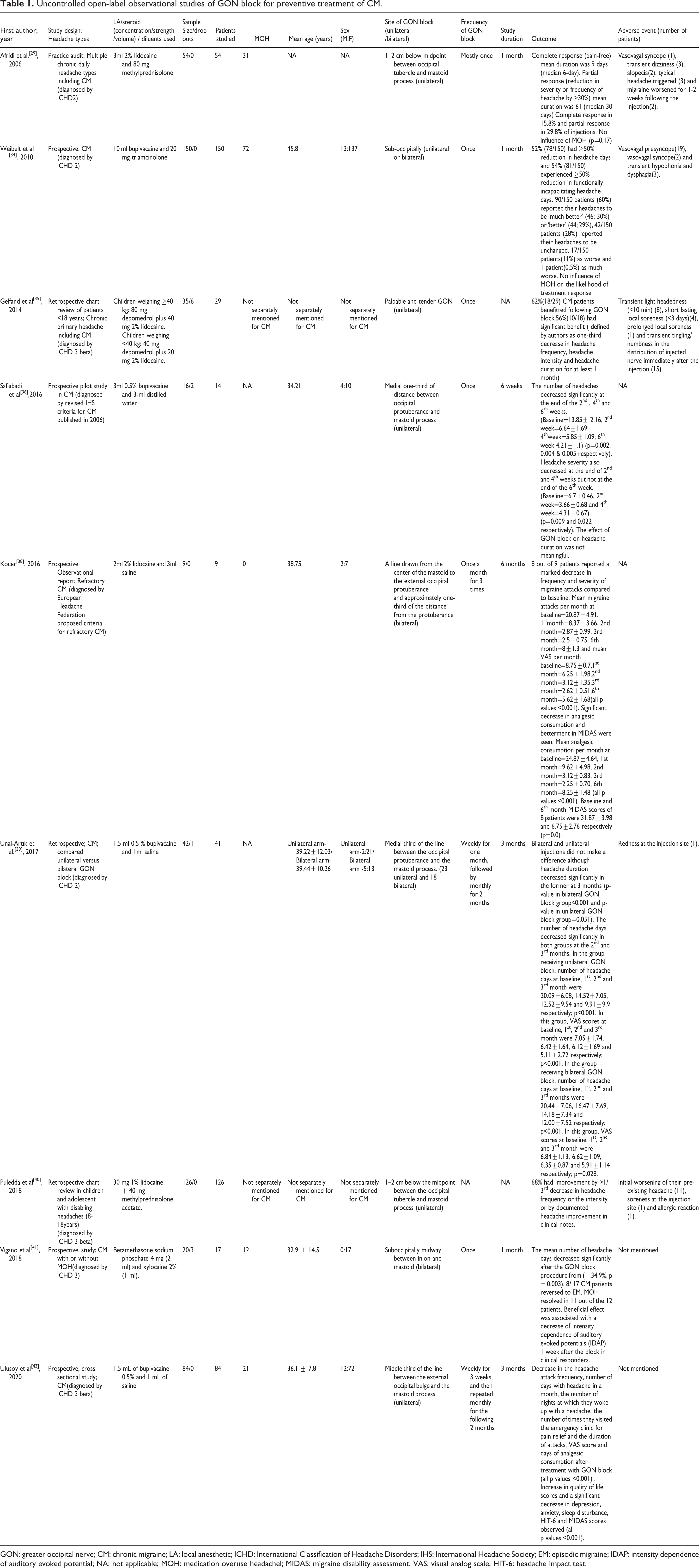
GON: greater occipital nerve; CM: chronic migraine; LA: local anesthetic; ICHD: International Classification of Headache Disorders; IHS: International Headache Society; EM: episodic migraine; IDAP: intensity dependence of auditory evoked potential; NA: not applicable; MOH: medication overuse headachel; MIDAS: migraine disability assessment; VAS: visual analog scale; HIT-6: headache impact test.
Afridi et al. in a practice audit studied the role of GON block (3 ml 2% lidocaine and 80 mg methylprednisolone) in 54 patients of CM who have failed at least three classes of preventive medications before the intervention. 29 Patients were assessed at 4 weeks. It was found that 9 out of 57 injections (15.8%) yielded complete response and 17 out of 57 (29.8%) yielded a partial response. CM patients experienced 9 days of complete pain-free state (median 6 days) and 61 days (median 30 days) of partial pain-free state (defined as a reduction in severity or frequency of headache by >30%) with the intervention. Tenderness over the GON was strongly predictive of outcome, although local anesthesia after the injection was not. The presence or absence of medication overuse did not predict the outcome.
Weibelt et al. studied 150 patients of CM with a prominent cervicogenic component in a single treatment arm of GON block (10 ml bupivacaine and 20 mg triamcinolone) and evaluated the efficacy at the end of 1 month. 34 Stable prophylactic doses of oral medicines were continued. Thirty-seven patients received unilateral and 113 received bilateral GON block. Seventy-eight (52%) patients experienced ≥50% reduction in headache days at 1 month compared to baseline. Eighty-one (54%) patients experienced ≥50% reduction in functionally incapacitating headache days (functionally incapacitated by headache for at least 1 h) at 1 month compared to baseline. Patient’s impression about their headache state was also captured by a Likert-type scale. Sixty percent reported their headaches to be “much better” or “better.” No influence of MOH on the likelihood of treatment response was seen. A total of eight (5%) patients reported adverse events within the ensuing 72 h, and three (2%) experienced adverse events that reversed spontaneously but required emergent evaluation and management. Besides the efficacy, the authors emphasized the other benefits of GON block, namely the ease of use, very low occurrence of significant adverse events, and much lower cost of therapy.
Safiabadi et al. did a prospective pilot study involving 16 patients of CM to determine the efficacy of GON block (3 ml 0.5% bupivacaine and 3 ml distilled water). 36 Assessments were done at 2 weekly intervals till 6 weeks after the intervention. Two patients dropped out and 14 patients were followed up. A unilateral GON block was given. In those with unilateral symptoms, the symptomatic side was chosen. In those with bilateral headache, more severe symptomatic side was chosen. In case the patient was symptomatic equally on both sides, the choice was left to the patient. Headache frequency significantly decreased at fortnightly assessments compared to baseline. Headache severity also decreased significantly in the first and second fortnight of follow-up but not in the third fortnight. The effect of the GON block on the duration of the headache was not meaningful.
In a single-center, single-author, an observational study of nine cases of refractory CM (defined as patients nonresponsive to three preventive drugs in adequate doses for 3 months each for a total duration of at least 1 year) by Koçer, GON block (2 ml 2% lidocaine + 3 ml saline) was performed using once a month schedule for 3 months bilaterally. 38 Eight out of nine patients reported a marked decrease in the frequency and severity of migraine attacks compared to baseline. Disability measured by MIDAS was significantly less at 6 months as compared to baseline. This study showed a remarkable success with the GON block for refractory CM patients and that the GON block can result in rapid relief of pain with the effects lasting for perhaps several months.
In a retrospective study by Ünal-Artık et al., 23 CM patients received unilateral and 18 received bilateral GON block (1.5 ml 0.5% bupivacaine and 1 ml saline). 39 GON blocks were performed every week for 4 weeks followed by monthly injections for 2 months. Headache days, duration, and VAS did not show any difference between the two groups. However, no details about the laterality of the headaches or laterality of the occipital nerve tenderness were mentioned.
Two open-label studies 35,40 have specifically studied the utility of GON block in children and adolescents with CM. In a retrospective study by Gelfand et al., 35 pediatric patients with CM were analyzed who received GON block (lidocaine and methylprednisolone injection) unilaterally. 35 Sixty-two percent of patients benefited, out of which 56% reported significant benefit (more than at least one-third decrease in headache frequency, severity, and duration at 1 month) following the GON block. Medication overuse, sex, age, and sensory change in the distribution of the infiltrated nerve all did not independently predict benefit. Adolescents fared better than children.
In a recent service evaluation paper by Puledda et al., GON block was performed unilaterally with 30 mg 1% lidocaine and 40 mg methylprednisolone, to treat disabling headache disorders in 159 children and adolescents who received 380 injections. 40 Seventy-nine percent of patients had CM of which 68% reported more than one-third decrease in headache frequency or intensity following the GON block. Interestingly, after a single injection, the beneficial effect in the majority of the patients lasted for more than 3 weeks.
In another paper by Viganò et al., neurophysiological correlates of clinical improvement after GON block in CM were studied in 17 women of whom 12 had MOH. 41 GON block (lidocaine and betamethasone) significantly reduced the number of total headache days by 35% per month. Eight out of 17 CM patients reverted to EM and medication overuse resolved in 11 out of 12 patients following the GON block. It was also found that the neurophysiological correlates like visual evoked potential habituation and intensity dependence of auditory evoked potential (IDAP) improved after GON block when compared to 19 healthy controls. Therefore, it was suggested that IDAP might be potentially useful as an early predictor of GON block efficacy.
Ulusoy and Bolattürk did a prospective cross-sectional study to determine the effect of GON block on the quality of life, disability and comorbid depression, anxiety, and sleep disturbance in 84 patients with CM. 43 GON block was administered weekly for 3 weeks, and then monthly for the following 2 months. The VAS and HIT-6 scores were significantly reduced after GON block treatment. Also, there was a decrease in the headache attack frequency, number of days with headache in a month, the number of nights at which they woke up with a headache, the number of times they visited the emergency clinic for pain relief, and the duration of attacks. The disability stages were significantly lower after GON block treatment as reflected by lower MIDAS scores after GON block treatment compared to pretreatment values. Anxiety, depression, and sleep quality index also improved with GON block.
Randomized placebo-controlled trials
The salient features of four RCTs 47 –50 have been summarized in Table 2. Outcomes were evaluated at different time points in these studies. One RCT evaluated the short-term outcome at 1 week, 49 two RCTs at 1 month, 47,48 and one RCT at 1, 2, and 3 months 50 using variable injection protocols. A quality assessment of these four RCTs showed significant differences (Table 3). Briefly, these studies are discussed below.
Placebo-controlled RCTs of GON block for preventive treatment of CM.
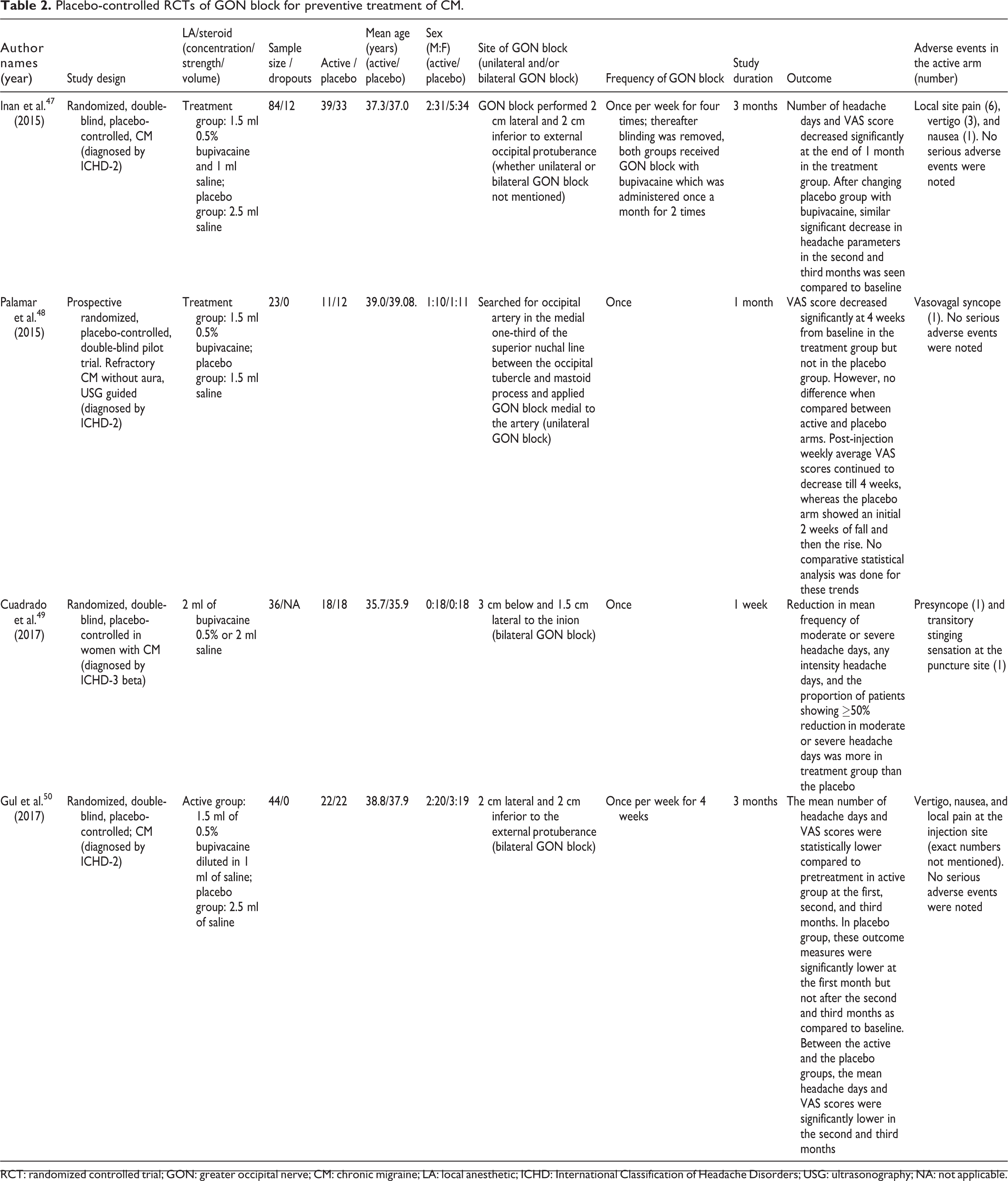
RCT: randomized controlled trial; GON: greater occipital nerve; CM: chronic migraine; LA: local anesthetic; ICHD: International Classification of Headache Disorders; USG: ultrasonography; NA: not applicable.
Quality assessment of the placebo-controlled, double-blind RCTs studying effectiveness of GON block in CM.
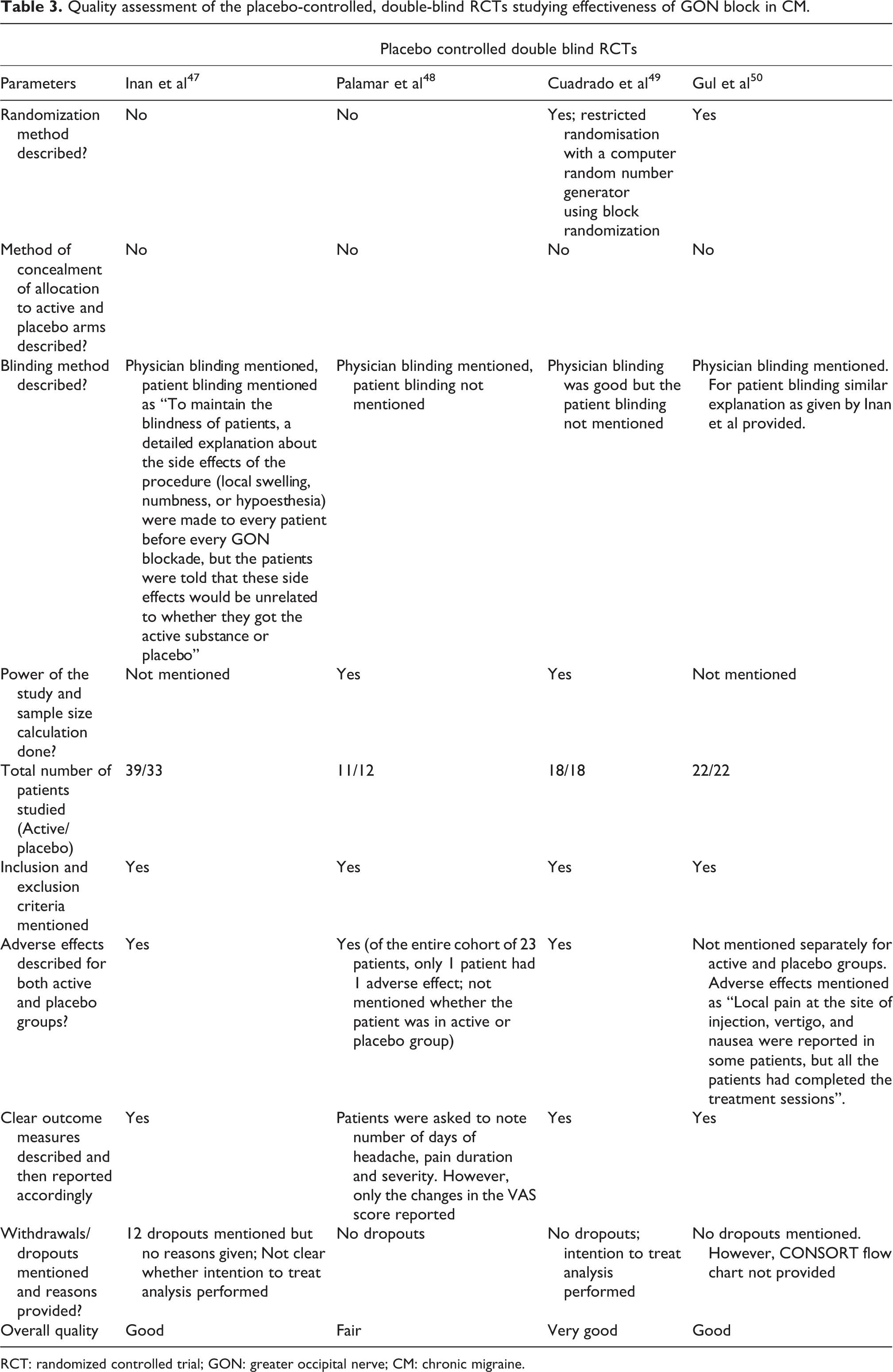
RCT: randomized controlled trial; GON: greater occipital nerve; CM: chronic migraine.
Inan et al. studied the efficacy of GON block (1.5 ml 0.5% bupivacaine versus saline injection) in CM using an interesting protocol. 47 GON block was administered once per week for four times; thereafter blinding was removed and both groups received GON block with bupivacaine, administered once a month for 2 months. Seventy-two out of 84 patients completed the study and 14.3% dropped out. Reasons for dropout were not provided. After 1 month of treatment, the number of headache days and headache severity decreased significantly in the bupivacaine group. After changing the placebo group with bupivacaine, both the groups showed a similar significant decrease in outcome parameters in the second and third months. This study showed a very high effect size in the treatment arm at the end of the first month producing huge difference with the placebo arm which continued even in the second month when the placebo arm was also receiving the active drug. This can possibly be accounted for high-frequency intervention in the beginning, that is, weekly GON blocks over first month. Further, adequate patient blinding might not have been achieved. Nevertheless, the study showed that GON block with bupivacaine was superior to placebo and is an effective, safe, and cost-effective treatment for prevention in CM.
In a prospective randomized, placebo-controlled, double-blind pilot trial by Palamar et al., ultrasound-guided GON block using 1.5 ml of bupivacaine 0.5% versus saline injection was studied. 48 Twenty-three patients of refractory CM were randomized to receive unilateral GON block. Interestingly, the authors chose to analyze VAS score changes separately for two sides of the cranium, one ipsilateral to the injection and other contralateral to the injection. VAS scores showed a significant decrease in the active arm when compared to baseline (within the group comparison). However, when compared between the groups (active versus placebo), the VAS scores did not differ. There was no change when VAS scores were analyzed for the side that did not receive the injections. VAS score trends were also plotted that showed a decrease until 2 weeks following the intervention in both the groups but thereafter the VAS decreased in the active group only.
Cuadrado et al. studied the short-term efficacy of single injection of bilateral GON block (2 ml of bupivacaine 0.5% versus saline injection) in 36 women with CM in a well-designed, randomized, double-blind, placebo-controlled study. 49 They also analyzed the effect of the GON block on pressure pain thresholds (PPTs) in different territories. The assessment was done 1 week before and 1 h and 1 week after the GON block. The absolute reduction in mean frequency of moderate or severe headache days and the reduction of any intensity headache days were significantly greater in the active group. The reduction in the number of days of acute medication consumption was not significant. The proportion of patients showing ≥50% reduction in moderate or severe headache days was also higher in the active group. Overall, PPTs increased after the LA blockade and decreased after the placebo. This study concluded that the GON block was effective in the short-term treatment of CM.
Gul et al. assessed the efficacy of GON block (1.5 ml of 0.5% bupivacaine versus saline injection) in patients of CM in a randomized, double-blind, placebo-controlled study involving 44 patients. 50 The assessment was done once per month for 3 months post-intervention. The mean number of headache days and VAS scores were significantly lower as compared to pretreatment in the active group at the first, second, and third months, whereas in the control group, they became nonsignificant after the second and third months. This study concluded that the GON block with bupivacaine not only was superior to placebo in the treatment of CM but also had long-lasting effects of up to 3 months. Between the active and the placebo groups, the mean headache days and VAS scores were significantly lower in the second and the third month.
Adverse effects of GON block
Overall, GON block use in CM has been found to be very safe. In open-label studies, although the reported adverse events frequency have ranged from 2.4% to 99.6%, mostly these were mild and transient. Two open-label studies 36,38 did not report any adverse events. Three open-label 35,36,39 studies reported dropouts but only one 39 mentioned the reason for discontinuation of GON block (due to redness at the injection site). The adverse events in RCTs were fewer and similar to open-label studies. Only one RCT reported dropouts but the reasons were not provided. 47 Three consistent adverse events that have been reported are local site pain (15.4%), vasovagal syncopal attacks (9.1%), and vertigo (7.7%). The adverse effects seen in the active and placebo arms in the RCTs are compared in Table 4. Other adverse events seen in open-label studies included transient dizziness or light-headedness, initial worsening of migraine, transient hypophonia and dysphagia, local site soreness, transient tingling/numbness in the distribution of injected nerve immediately after the injection, allergic reaction, local hematoma, redness at the injection site, nausea, pseudoseizures, and headache.
Adverse events reported by RCTs in active versus placebo arms.

RCT: randomized controlled trial.
Efficacy of addition of steroid to LA in GON block for CM patients
Open-label studies using methylprednisolone 29,35,40 and triamcinolone 34 along with LA have shown that these are effective in preventing headaches in CM patients. However, because of the lack of a comparator arm, it is uncertain whether the beneficial effect was due to LA or the combination. Two RCTs have tried to answer this question. Ashkenazi et al. did a randomized, single-blind study in 37 patients of transformed migraine. 44 One group received a GON block with LA and triamcinolone and the other group received a GON block with LA and saline. LA used was 4.5 ml 2% lidocaine and 4.5 ml 0.5% bupivacaine. GON block significantly decreased the headache severity, increased the mean duration of headache-free days, and reduced the mean analgesic consumption in either group, but none of these outcomes differed significantly between the groups. Hence, it was concluded that the addition of steroids to LA in GON block did not offer any extra benefit. Kashipazha et al. studied 52 patients of migraine in an RCT of which 42 patients completed the study using a similar protocol. 45 No difference was observed in the outcome between the two groups.
Effect of coexistent MOH on GON block efficacy in CM patients
Two observational studies 29,34 found no evidence that coexistent MOH affects the efficacy of GON block in CM. None of the RCTs included MOH patients.
Other predictive factors for a favorable outcome
Predictive factors for GON block in CM patients have not been analyzed by RCTs. Only one RCT found that patients with GON sensitivity respond 50% less than those without (although it was a nonsignificant difference). 49 One open-label study found that GON tenderness was predictive for favorable outcome, 29 whereas another failed to find so. 34 The level of anesthesia following the GON block did not predict the outcome. 29 The number of prophylactic therapies previously used or failed did not affect the outcome. 34 In the pediatric and adolescent groups, sex, age, and sensory changes in the distribution of the nerve did not predict the outcome. 35 In another study involving a similar population, however, older age, female gender, increased number of past preventive use, medication overuse, and developing side effects with preventive medications were all associated with an increased likelihood of positive treatment outcome. 40 Continuation of conventional preventive medications did not affect the outcome in another open-label study. 38
Blocking techniques
Studies have used various landmarks for locating the GON block injection site. Some studies have used the most tender point located at the junction of medial one-third and lateral two-third of an imaginary line drawn from the external occipital protuberance to the tip of the mastoid process as the site of GON block injection. 36,38,39 Other sites of GON block injections that were used were 2 cm lateral and 2 cm inferior to external occipital protuberance, 47,50 3 cm below and 1.5 cm lateral to the inion, 49 and 1–2 cm below the midpoint between external occipital protuberance and the mastoid process. 29,40 Gelfand et al. palpated and located the tender GON and then performed the GON block injection. 35 Some have just injected suboccipitally. 34 Some researchers have also used ultrasonography (USG) to localize the occipital artery and administer the GON block medial to it. 48 Flamer et al. in a recent study compared two USG-guided GON block techniques in a double-blind RCT. 53 A “proximal or central” technique targeting the GON at the level of the second cervical vertebra or a “distal or peripheral” technique targeting the GON at the level of the superior nuchal line was employed. Numerical Rating Score (NRS) for pain was the primary outcome. NRS pain scores were significantly reduced at 24 h and at 1 week post-procedure in both cohorts. However, at 1 and 3 months, NRS pain scores decreased significantly only in the proximal group. There was no significant difference in NRS pain scores between the two cohorts at any of the follow-up time points.
There is also some uncertainty regarding the single-injection technique versus fanning technique involving three or four directions for GON block injection. Single-injection technique was used by most of the researchers, 35,39,40,47 –50 whereas the fanning technique was used by two groups, 34,36 and one researcher performed GON block by injecting at two target points around the GON. 38 Some have observed that by employing the fanning technique, the complication such as local bleeding occurs more commonly. 34
Laterality of GON block: Unilateral or bilateral?
Ünal-Artık et al. specifically looked at this question in a retrospective study. 39 They found that unilateral or bilateral GON block did not differ in terms of outcome such as reduction of headache days and VAS score at the end of 3 months. One open-label study 34 used both unilateral and bilateral blocks. Four other open-label studies 29,35,36,40 and one RCT 48 employed unilateral and one open-label 38 and two RCTs used bilateral GON block. 49,50 One RCT did not mention the laterality of injection. 47
LA type, concentration, dose, and combinations
Lidocaine and bupivacaine have been used for the GON block in CM. Concentrations of lidocaine used in various studies included in this review were 2% and bupivacaine 0.5%. The volume of lidocaine has ranged from 2 ml to 3 ml and that of bupivacaine from 1.5 ml to 3 ml. One study used 10 ml of bupivacaine but concentration was not mentioned. 34 A few open-label studies have used saline or distilled water as diluents. 36,38,39 Two RCTs have also used saline as diluents in the active arm. 47,50
Needle size
RCTs have used needle gauge sizes varying from 22 G to 30 G.
Patient position during and after the block
Two RCTs and two open-label studies used sitting position 47,50 and one RCT used prone position. 48 Rest did not mention the patient’s position during the GON block. None mentioned the patient’s position after the block.
Frequency of administration of GON block
Varied frequency of administration of GON block has been employed in studies of CM. Some have administered GON block once per week for 4 weeks 47 while others have followed it up by once per month for 2 months thereafter. 39 One RCT administered GON block and placebo weekly for 4 weeks followed by an open-label phase in which both the arms received active drug monthly for 2 months. 44 In one observational study, the frequency of GON block was once per month for 3 months. 38 Some of the RCTs and open-label studies have used single injections of GON block. 25,34 –36,45,46
Discussion
Although few reviews in the past have examined the role of GON block in various headache disorders 22,54 –57 including migraine, none have focused exclusively on CM. Two meta-analyses evaluated the role of GON block in reducing the severity and frequency of headache in migraine (but not specifically in CM). 58,59 A recent meta-analysis and systematic review on the role of GON block in CM included patients without CM (e.g. cervicogenic headache). 31
In general, open-label studies included in this review showed good efficacy of GON block for the preventive treatment of CM in adults, adolescents, and children. A total number of 456 patients were studied involving five prospective and four retrospective studies. Four studies used steroids along with LA. GON block reduced the headache severity and frequency in 35–68% of patients of CM. The study by Afridi et al. was particularly important because it showed that the duration of the response time (analgesic effect) far exceeded the local aesthetic effect of LA and the beneficial effect of a single GON block lasted up to 4 weeks. 29 Also, because of a quick response in terms of reduction in pain intensity and frequency, the GON block could provide interim relief till other preventive measures could be instituted for these patients.
It is difficult to make a composite outcome assessment based on the four RCTs because of inherent methodological heterogeneity. Further, they differed significantly in terms of quality (Table 3). Of the four RCTs, one study evaluated the short-term outcome at 1 week following a single GON block. 49 This was a well-conducted RCT that showed a significant reduction in moderate to severe headache days and any headache days in CM patients receiving GON block. Two other RCTs used identical frequency of initial GON block (once a week for 4 weeks). 47,50 Of these, only one study found a significant difference in favor of active treatment at the end of 1 month. 47 The other one did not find any significant difference as compared to placebo at 1 month although on repeat GON block monthly for the next 2 months, a significant reduction in VAS score and headache days in the active arm was found at the end of the second and third month. 50 One RCT reported no difference between active and placebo arms in terms of headache severity at 1 month. 48 Based on these results, the efficacy of the GON block in CM remains a suspect although there is a fair suggestion that GON block probably works well as a short-term preventive at 1 week. In both open-label and RCTs, the headache duration in CM did not decrease with the GON block. Further, not enough data are available regarding predictive factors for favorable outcomes following GON block in CM as open-label studies reported conflicting results.
Our review also suggests a possible role of GON block in children and adolescents with CM based on 2 open-label studies using unilateral GON block with methylprednisolone in 194 patients. 35,40 Both studies found significant efficacy of GON block in 58–68% of CM patients. However, the authors in both the papers chose a cutoff level of 33% decrease in headache frequency, headache intensity, and headache duration for at least 1 month to be considered as significant instead of the usual 50% response to assess the effectiveness of the procedure. Nonetheless, the treatment was well tolerated, and the effectiveness of a single GON block lasted for as long as 3 weeks. A robust placebo-controlled RCT of GON block for CM in this group may provide the much-needed evidence for such an intervention.
GON block was remarkably well tolerated by CM patients and the adverse effects were mild and transient. No serious adverse event was reported in RCTs. Two absolute contraindications for using GON block are known allergy to LA and open skull defect craniotomy. 60,61 Since local site bleeding is a known complication of GON block, patients who are on antiplatelets and antithrombotics should be kept under observation for a longer duration after GON block and the local site be pressed for 5–10 min. 56 Systemic toxicity has been extremely rare in the recommended doses. Care should be taken to avoid accidental intravascular injection by ensuring that the plunger is withdrawn before injecting the LA. 22
Besides efficacy and safety, some other issues regarding the use of the GON block in CM also merit our attention. First is the issue of adding steroids to LA in GON block. Two RCTs concluded that the addition of steroids to LA may not be beneficial 44,45 although the sample sizes were small in both the studies. Interestingly, in cluster headache patients, GON block with steroids has been found to be efficacious. 62,63 It has been proposed that nociceptive C-fibers in substantia gelatinosa are reversibly blocked by corticosteroids. 64 Further, the addition of corticosteroids in GON block is postulated to reduce dynamic mechanical allodynia in migraine patients. 65 Steroids may also prolong the effects of LA. 66 Whether GON block using steroids alone can help CM patients remains unanswered. Only a single study reported the administration of GON block in migraine patients using corticosteroid alone. 67 The addition of steroids has been associated with some specific adverse events such as alopecia, hypopigmentation, and cutaneous atrophy. 68 Recurrent GON block with corticosteroids should be avoided and patients must be warned of the risks of potential local and systemic adverse affects, 69 especially those with other medical conditions such as diabetes and glaucoma. 70 During pregnancy, corticosteroids, especially long-acting agents like dexamethasone and betamethasone, should be avoided. 56
The second issue concerns the laterality of the injections. One study suggested that unilateral block may be as good as bilateral blocks. 39 However, a small sample size (23 patients received unilateral and 18 bilateral blocks) and its retrospective nature makes it difficult to make meaningful inferences from this study. It has been demonstrated in experimental animals that convergent neurons in the TCC have inputs not only from ipsi- but also from contralateral cervical afferents and that bilateral activation of the TNC and cervical cord occurs by the same afferent sending bilateral projections to the TNC and lateral cervical nucleus (LCN). 71,72 Also, 40% of patients of migraine can have bilateral headaches 73 and bilateral GON tenderness and even those with unilateral headaches, around 44% may have bilateral GON tenderness. 74 There are also reports of change in laterality of pain following occipital nerve stimulation in cluster headache (CH). 75 Hence, we believe that bilateral GON block may be preferred in CM but this hypothesis needs to be proven by a good RCT.
Third issue is whether the coexistence of MOH in CM patients impacts the efficacy of the GON block. The question still remains unanswered as none of the RCTs included MOH patients although an RCT involving triptan overuse headache (TOH) patients exclusively found a beneficial effect of repeated GON block. 51 However, contrarily in a retrospective study, symptomatic medication overuse almost tripled the risk of GON block failure in a mixed group of headache patients including refractory migraine. 76 Few open-label studies have found that MOH does not affect the efficacy of the GON block. Because headache severity decreases quickly following the GON block, it may provide interim relief to MOH patients when their offending medications such as analgesics and triptans are withdrawn. Hence this issue is worth exploring further.
From the technique point of view, two important issues are the site of GON block injection and the choice and dose of LA used in the GON block. GON has considerable inter- and intraindividual variations. 77 Few cadaveric studies aimed to evaluate the most appropriate site for injection reported variable coordinates for GON localization. 78,79 This variability in choosing the site has also been seen in clinical studies. USG-guided GON block probably adds to better identification of the site but it is uncertain whether this increases the effectiveness of the GON block. A recent RCT that compared two USG-guided techniques of GON block in CM patients, however, found more sustained analgesic benefit by proximal injection at C2 level as compared to the standard site at the superior nuchal line. 53 Nonetheless, for wider applicability, tender point palpation with the use of standard anatomical landmarks may be a more feasible option.
Lidocaine and bupivacaine have been used for the GON block in CM. Both are amide LA agents and have their advantages and disadvantages. 22 They act by reversibly blocking sodium channels within unmyelinated C-fibers and myelinated nerve fibers. This causes blockage of depolarization within the nerve, thereby preventing the pain signal transmission. 80 Lidocaine (1–2%) has a rapid onset (4–8 min) after injection and shorter duration of action (1–2 h), whereas bupivacaine (0.25% or 0.5%) has a longer onset (8–12 min) and duration of action (4 and 8 h). However, bupivacaine is more cardiotoxic than lidocaine. Both are metabolized by the hepatic cytochrome P-450 3A enzyme systems. Mean effective volume and minimum effective anesthetic concentration are the two important concepts for LA use. The density of the block decreases if the volume of LA agents is increased to improve the diffusion of the drug, whereas the drug may not be able to penetrate the perineurium if the concentration of LA is decreased due to excessive dilution. Hence a balance between concentration and volume needs to be maintained. Further, if the volume of LA used is more than that is required for GON blockade, then, technically, it cannot be called as GON block but rather becomes suboccipital block or occipital injection. 81 American Headache Society Special Interest Section for PNBs and other Interventional Procedures (AHS-IPS) recommends lidocaine 1–2% (10–20 mg/ml) and/or bupivacaine 0.25–0.5% (2.5–5 mg/ml). 56 Volume of injection should be 1.5–3 ml per nerve. However, a recent cadaveric study comparing the injectate spread and nerve involvement between different injectate volumes for ultrasound-guided GON block at the C2 level showed that using 5 ml of dye solution consistently involved most major nerves in the suboccipital area and therefore seemed to be suitable for therapeutic purposes. 82 Concentrations of lidocaine above 2% have been discouraged because of possible greater adverse events although, in a retrospective small series of 13 patients of migraine and occipital neuralgia, 10% lidocaine produced prolonged relief in headaches lasting months. 83 Only one open-label study reviewed here used a combination of lidocaine and bupivacaine possibly to augment quicker onset and longer duration of action. However, the benefit of such a combination is questionable. A study has shown that the plasma level of the long-acting agent becomes less when the combination of LA is used. 84 If a combination of the two drugs is used, the recommended volume ratio (lidocaine:bupivacaine) is 1:1–1:3. 56 Role of additional diluents such as saline to LA remains questionable. AHS-IPS recommends using needle gauge sizes of 25, 27, or 30 G. 56,22 Typically a 3–5 ml syringe with a needle length of 0.5–1 inch is sufficient for the purpose of GON block.
The patient’s position during and after GON block may be important in terms of adverse effects and efficacy. The prone position is uncomfortable for the patients but is useful for those who have a history of vasovagal or syncopal attacks. 56 Assumption of supine position just after receiving the GON block in sitting position has been found to enhance its effectiveness in a recent study. 42
There is also uncertainty regarding the frequency of GON block repetition if long-term prevention is planned. Some researchers have used an intensive phase of weekly GON block followed by monthly injections. A previous study estimated the median duration of complete and partial response following single GON block in CM as 6 and 30 days, respectively. 29 AHS-IPS, therefore, recommends the frequency of treatment by GON block for CM once every 2–4 weeks. 56
Future research
Larger well-devised RCTs are needed keeping in mind the limitations and the uncertainties detailed above. Unblinding that occurs due to hypoesthesia produced by LA is a potential problem for performing double-blind trials with GON block. We agree with the proposal of Dilli et al. that future trials may employ the local application of anesthetic gel at the injection site in both the active and placebo groups before the intervention to prevent unblinding. 46 Further, assessment of change in migraine days (in addition to headache days) following GON block should also be incorporated as one of the primary endpoints as suggested by the International Headache Society (and not done in any of the previous studies reviewed here except in an open-label study by Koçer 38 ). 85 A uniform and standardized technique of GON block should be used as suggested by the expert consensus guidelines. 56
Conclusion
Our review finds that GON block may be useful in CM patients. All open-label studies reported positive results. Four randomized, placebo-controlled trials used heterogeneous methodology and techniques and assessed outcomes at various time points. One RCT found good evidence of the efficacy of GON block at 1 week, whereas the other three studies reported conflicting results at 1 month. Thus, the studies suggest that GON block appears to be effective for the short-term prevention of CM, although long-term efficacy is uncertain. Hence further robust randomized placebo-controlled trials using standardized methodology are needed to definitely answer about the long-term efficacy of GON block for the preventive treatment of CM. There still exists conflicting literature regarding the use of steroids as add-on to GON block and the efficacy of GON block in CM patients with MOH.
On the face of it, GON block appears to be very useful in CM because of many attractive features such as early effect in reducing the severity of pain (very useful in the context of CM with MOH), sustained effect following a single injection (very useful for spacing out the injection schedules every 2–4 weeks), easy technique (can be learned and practiced by physicians, specialist nurses, and neurologists), minimum invasiveness, minimum adverse effects, no drug-to-drug interactions (oral preventive(s) can continue if needed), and low cost. A single study with a small number of participants found GON block to be useful in treating CM during pregnancy. 86 However, more evidence is required in this regard. Finally, in many countries, especially the developing ones, GON block can provide an easy and cheap alternative to other costly preventive treatments of CM.
Key findings
As a short-term preventive at 1 week, single GON block injection decreases headache severity and frequency of headache days in CM.
More data are required to derive meaningful conclusions about its role for the long-term prevention at 1 month and beyond.
Our study showed that there are also uncertainties regarding the use of GON block in CM in terms of additional steroid use, use in coexistent MOH patients, use of unilateral against bilateral injections, and standardization of technique. These may be addressed by future RCTs.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
