Abstract
GLADIATOR was a prospective, randomized, open-label, phase 3 study of lasmiditan 100 mg or 200 mg dosed intermittently for up to 1 year in patients with episodic migraine. Most patients had completed one of two single-attack studies before participation. A total of 2030 patients received ≥1 lasmiditan dose and 19,879 migraine attacks were treated. Safety results were similar to the previously reported interim analysis. The most frequently reported treatment-emergent adverse events (TEAEs) included dizziness (18.5%), somnolence (8.5%), and paresthesia (6.8%), with frequency of adverse events appearing to decrease with subsequently treated attacks. At 2 h post-dose, 26.7% and 32.2% of all attacks treated with lasmiditan 100 mg and 200 mg, respectively, were pain free. This pattern was generally consistent across study quarters and treated attacks. In conclusion, during a 1-year treatment period, intermittent lasmiditan for episodic migraine treatment was associated with generally decreasing TEAEs and consistent efficacy.
Introduction
Lasmiditan is a centrally penetrant, highly selective 5-HT1F receptor agonist for the acute treatment of migraine that lacks the vasoconstrictor activity of triptans. 1 –3 Lasmiditan was efficacious and generally well tolerated in two placebo-controlled, phase 3, single-attack studies. 4,5 In 2019, we published results of a planned interim analysis of GLADIATOR, a prospective, randomized, open-label, phase 3 study of lasmiditan 100 mg or 200 mg dosed intermittently on an as-needed basis for up to 1 year in patients with episodic migraine who had completed one of the single-attack studies. 6 In the interim analysis (N = 1978 patients, 19,058 attacks), no new safety findings were observed relative to the placebo-controlled studies and the frequency of treatment-emergent adverse events (TEAEs) appeared to decrease with repeated dosing. Additionally, consistent results for freedom from pain and most bothersome symptom (MBS) of migraine attacks across study quarters and treated attacks were observed. Here, we provide the final results for key safety (primary) and efficacy (secondary) endpoints from the recently completed study.
Methods
The GLADIATOR study evaluated the intermittent use of lasmiditan for the acute treatment of migraine for up to 1 year, with the primary objective to assess long-term safety and tolerability (adverse events (AEs) and changes from baseline in physical and laboratory examinations, electrocardiograms (ECGs), and vital signs) and the secondary objective to evaluate long-term efficacy (pain freedom and MBS freedom). Eligible patients met all of the following requirements: (1) had completed one of the two phase 3 single-attack lasmiditan studies (SAMURAI or SPARTAN) 4,5 or were lasmiditan naïve; (2) met the International Headache Society (IHS) criteria for migraine with or without aura (1.1 or 1.2.1 per ICHD, 2nd edition) 7 ; (3) had at least moderate migraine disability 8 ; and (4) had episodic migraine, which was defined as three to eight migraine attacks per month and <15 headache days per month (see Supplemental Materials for complete study inclusion and exclusion criteria). Patients were enrolled at 199 study sites in the United States, United Kingdom, and Germany. The study was conducted in compliance with the International Council for Harmonisation principles of Good Clinical Practice, ethics review boards approved the study protocol, and all participants provided written informed consent. The final database lock occurred on September 16, 2019 (ClinicalTrials.gov identifier: NCT02565186, https://clinicaltrials.gov/ct2/show/NCT02565186).
Regardless of previous treatment assignment to lasmiditan or placebo, patients were randomly assigned (1:1) to receive lasmiditan 100 mg or 200 mg (stratified by use of concomitant migraine preventive medications) and were instructed to use lasmiditan as the first treatment for each new migraine attack of moderate or severe pain intensity within 4 h of pain onset (Supplemental Figure 1). The requirement of at least moderate pain severity was included in order to ensure that the headaches were truly migraine attacks, in accordance with IHS guidelines for controlled trials of acute treatment of migraine. 9 The statistical analysis plan prespecified that all attacks, regardless of severity, would be included in the analyses on the basis of the intent-to-treat principle. If the pain did not respond at 2 h or pain recurred after becoming pain free, lasmiditan at the same dose or an alternative migraine medication was permitted between 2 and 24 h after study medication administration (except triptans, ergots, opioids, or barbiturates). No study medication dose adjustments were allowed.
Patients used an electronic diary (e-diary) to record the details of each migraine attack and treatment, including their response to lasmiditan and if they felt anything unusual after taking lasmiditan. An e-diary entry indicating that the patient felt something unusual after taking lasmiditan triggered a follow-up phone call from the study site to determine if an AE had occurred. AEs were coded using the Medical Dictionary for Regulatory Activities (MedDRA; Version 21.0), and those that started or worsened within 48 h after a lasmiditan dose were considered treatment-emergent. Physical and laboratory examinations (including vital signs) and ECGs were conducted during clinic visits. Safety analyses were performed on all randomized patients who took ≥1 lasmiditan dose (safety population).
To evaluate efficacy, a 4-point IHS pain severity rating scale (none, mild pain, moderate pain, and severe pain) 10 was employed at baseline and at 0.5, 1, 2, 4, 24, and 48 h post-dose for each migraine attack. Migraine attacks were considered pain free if pain severity was reduced from mild, moderate, or severe at baseline to none post-dose. Migraine attacks were considered MBS free if the MBS (identified at baseline as nausea, phonophobia, or photophobia) was absent post-dose. As prespecified, the pain freedom and MBS freedom analyses were conducted on the modified intent-to-treat population (all migraine attacks treated within 4 h of onset that had ≥1 post-dose pain severity or symptom assessment). Change from baseline in Migraine Disability Assessment (MIDAS) total score (0 = no disability to ≥21 = severe disability)8 and number of headache days over the past 3 months were also analyzed.
Analyses of TEAEs and efficacy measures were conducted for all patients, and, to assess a population without attrition, for migraine attacks 1 to 5 in a subset of patients who treated ≥5 migraine attacks. Fisher’s exact test was used to compare TEAEs among patients with and without cardiovascular (CV) risk factors (hypertension, hypercholesterolemia, smoking, obesity, diabetes mellitus, family history of coronary artery disease (CAD), men aged >40 years, and postmenopausal women). A mixed model repeated measures analysis was used to model MIDAS score changes. All analyses were performed with SAS (Version 9.4 or higher; SAS Institute, Cary, North Carolina, USA).
Results
Of the 2171 randomized patients, 2030 patients took ≥1 dose of lasmiditan 100 mg (n = 991) or 200 mg (n = 1039) (Figure 1). The treatment groups generally had well-balanced demographic and baseline disease characteristics. The majority of patients were women (85.3%) and were White (78.5%); mean age was 43.3 years (range, 18–79 years). Eighty-two percent of patients had ≥1 CV risk factor and 29.6% had a family history of CAD. A migraine preventive medication was used by 22.1% of patients. Characteristics of treated migraine attacks with ≥1 post-dose efficacy assessment in the final analysis (n = 17,878) are shown in Supplemental Table 1 and were similar to those at the interim analysis.6 Baseline pain intensity was severe for 33.3% of attacks and moderate for 65.1%. A small number of patients treated attacks (n = 286/17,878 (1.6%)) when pain was mild or none despite the instruction to treat when pain was of at least moderate severity. A second dose of lasmiditan was taken for recurrence for 6.2% of the attacks that were pain free at 2 h post-dose.

Flow of participants in the GLADIATOR study. aFifty-one patients in the final safety population were enrolled through an addendum, which included lasmiditan-naïve patients and allowed dosing of mild headaches up to 8 h after onset. bCommon adverse events leading to discontinuation (>1%) in the safety population were as follows: dizziness (28 patients; 2.8%) and somnolence (14 patients; 1.4%) in the lasmiditan 100 mg group; and dizziness (44 patients; 4.2%), paresthesia (19 patients; 1.8%), fatigue (17 patients; 1.6%), nausea (14 patients; 1.3%), and somnolence (11 patients; 1.1%) in the 200 mg group.
Overall, patients treated 19,879 migraine attacks. A total of 413 (21.4%) patients treated a maximum of five or more migraine attacks in any 30-day period. A total of 970 (47.8%) patients completed all 12 months of the study, with a median study duration of 345 days (100 mg, 338 days; 200 mg, 346 days). The most common reasons for discontinuation were patient request (21.8%), AEs (12.9%), and lost to follow-up (9.7%). Details were provided for 67.0% of the patients who discontinued due to patient request; reasons given were categorized as scheduling conflicts (16.5%), lack of efficacy (13.1%), e-diary requirements (12.4%), relocation (10.4%), other (9.0%), dislike of investigational product (5.9%), lack of/reduced frequency of migraine attacks (3.6%), and the initiation of prohibited or preventive medication (1.6%).
Safety
Overall, the percentage of patients reporting ≥1 TEAE was lower in the lasmiditan 100 mg group (45.1%) than in the 200 mg group (52.5%). Most TEAEs were mild-to-moderate in severity. Dizziness was the most frequently reported TEAE (Table 1). As observed in the interim analysis,6 the incidence of TEAEs decreased across treated attacks 1 to 5 in patients who treated ≥5 attacks (Figure 2). Overall, 11 patients reported 14 treatment-emergent serious AEs (Supplemental Table 2); none was considered treatment-related by the investigator and no deaths were reported. There were no CV TEAEs potentially due to vasoconstriction (e.g. angina pectoris, uncontrolled hypertension, and ischemic stroke), no difference in the incidence or type of TEAEs among patients with or without CV risk factors, no reports of any central nervous system TEAEs leading to accidents or injuries, and no pattern of baseline-to-endpoint changes in laboratory parameters, vital signs, or ECGs that were considered clinically meaningful (data not shown).
Common TEAEs (occurring in ≥2% of patients) in either treatment group by percentage of patients and percentage of attacks (safety population).

MedDRA: Medical Dictionary for Regulatory Activities; TEAE: treatment-emergent adverse event.
a TEAEs were coded using MedDRA, Version 21.0.
b A TEAE was defined as an adverse event that started or worsened within 48 h after the last dose (either the first or the second dose) of lasmiditan.
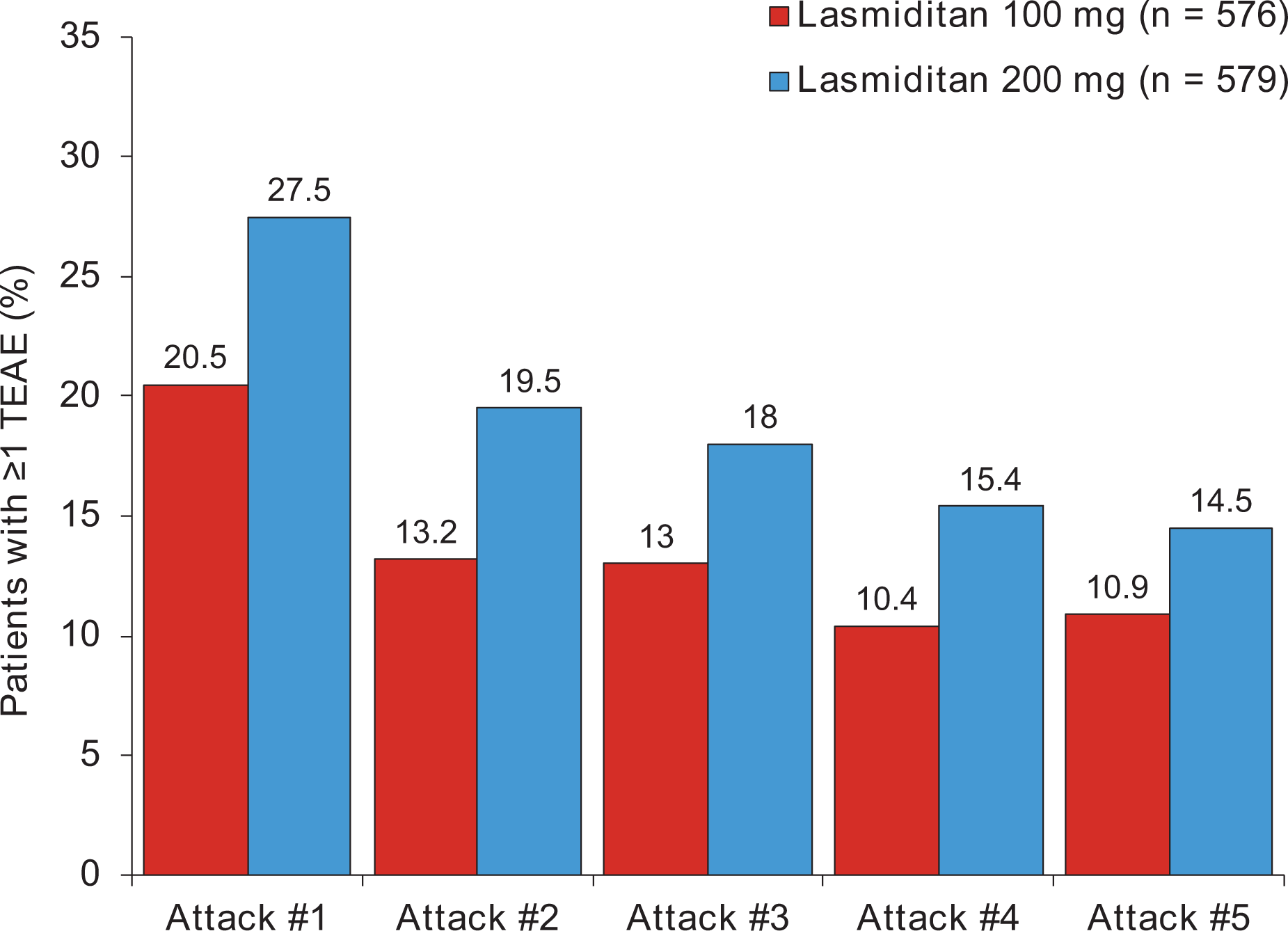
Percentage of patients who treated ≥ 5 attacks who had ≥ 1TEAE during migraine attacks 1 to 5 (safety population). TEAE: treatment-emergent adverse event.
Efficacy
Overall, at 2 h post-dose, 29.4% of attacks were pain free (100 mg, 26.7%; 200 mg, 32.2%) and 38.8% were MBS free (100 mg, 37.2%; 200 mg, 40.5%). Results were similar for all patients across all four quarters of the study (range across quarters; pain free: 100 mg, 23–30%; 200 mg, 31–33%; MBS free: 100 mg, 33–39%; 200 mg, 38–42%) and for the first five consecutive attacks in patients who treated ≥5 attacks (range across attacks; pain free: 100 mg, 27–33%; 200 mg, 32–38%; MBS free: 100 mg, 38–44%; 200 mg, 42–47%). Significant decreases were observed in mean change (±standard deviation) from baseline to 12 months for MIDAS scores (27.0 ± 19.73 to 14.35 ± 17.10; p < 0.001) and number of headache days in the past 3 months (14.37 ± 10.07 to 8.49 ± 9.85; p < 0.001). A full report of interim MIDAS results from this study has been published. 11
Discussion
The final analysis results of GLADIATOR are consistent with the interim analysis results6 and provide support for the safety and efficacy results observed in the placebo-controlled, phase 3, single-attack lasmiditan studies. 4,5 No new safety issues were identified. Moreover, these long-term safety data suggest that the incidence of TEAEs decreases over time with subsequent lasmiditan-treated migraine attacks. To address the potential explanation that the decreasing TEAEs over time could be due to patient attrition, we conducted additional analyses of patients who treated ≥5 attacks. These analyses, which were not subject to attrition, also showed a reduction in TEAEs during ongoing treatment.
Lasmiditan treatment showed consistent efficacy across the first five treated attacks and across quarters at the population level for up to 1 year. Although a relatively high dropout rate (52.2%) was observed compared to studies of other migraine treatments, 12,13 study design and burden might have contributed to this finding. First, the study was unique in that it required daily e-diary entries and intensive data collection via an e-diary during each attack (entries at baseline, 30 min, 60 min, 2 h, 4 h, 24 h, and 48 h) for each migraine treated for up to a year. During the study, site surveys were conducted, revealing that patients frequently complained to site staff about e-diary burden. Among patients who discontinued because of “patient request,” one of the most common cited reasons was e-diary requirements. Second, patients were advised to not drive or operate machinery for at least 12 h after taking lasmiditan. Third, many other long-term safety studies of acute migraine therapies allow patients to treat their migraine attacks when headache pain is of mild severity. The current study replicated many of the features of the parent (controlled) studies, including pain severity requirements, which were based on IHS guidelines for controlled studies of acute treatment of migraine. 9 The IHS guidelines further describe that headaches of mild intensity might be due to other etiologies, such as tension. To ensure that the attacks were indeed migraine attacks, the current study required treatment of attacks with headache pain of at least moderate severity.
In conclusion, in the acute treatment of migraine, final GLADIATOR results showed mostly mild-to-moderate AEs that generally decreased with subsequent treated attacks and consistent efficacy over time when lasmiditan was used intermittently for up to 1 year.
Clinical implications
The final results of the GLADIATOR study were consistent with the safety and efficacy results observed in the previously published placebo-controlled, phase 3, single-attack lasmiditan studies and the interim results from GLADIATOR.
For the treatment of episodic migraine attacks, intermittent, repeated administration of lasmiditan for up to 1 year was both well tolerated and showed consistent efficacy (at the population level, by quarter).
Supplemental material
Supplemental Material, Brandes_LasmiditanBrReport_CephalalgiaReports_SupplMaterials - Long-term safety and efficacy of lasmiditan for acute treatment of migraine: Final results of the GLADIATOR study
Supplemental Material, Brandes_LasmiditanBrReport_CephalalgiaReports_SupplMaterials for Long-term safety and efficacy of lasmiditan for acute treatment of migraine: Final results of the GLADIATOR study by Jan Lewis Brandes, Suzanne Klise, John H Krege, Michael Case, Rashna Khanna, Raghavendra Vasudeva, Joel Raskin and David Kudrow in Cephalalgia Reports
Footnotes
Authors’ note
Joel Raskin is a former employee of Eli Lilly and Company, Indianapolis, IN, USA. Writing support for the development of this manuscript was provided by Cindy C Taylor, PhD, at Synchrogenix LLC, a Certara company. Draft development and revisions were under the direction of the authors in accordance with the International Committee of Medical Journal Editors criteria for authorship.
Author contributions
All authors were involved in the analysis and interpretation of the data and critically revised manuscript drafts for intellectual content. In addition, Jan Lewis Brandes and David Kudrow conducted the study as principal investigators at their respective sites, and Michael Case conducted statistical analyses.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Suzanne Klise, John H Krege, Michael Case, Rashna Khanna, and Raghavendra Vasudeva are employees and minor shareholders of Eli Lilly and Company. Joel Raskin was an employee of Eli Lilly and Company when the study was performed. David Kudrow has received research grants from Eli Lilly and Company, Amgen, Allergan, Teva, Alder, Biohaven, Roche, and Zosano and has served on advisory boards for Amgen/Novartis, Eli Lilly and Company, Alder, and Biohaven. Jan Lewis Brandes has received research grants from Allergan, Teva, Amgen, Alder, Biohaven, Zosano, Colucid, and Eli Lilly and Company and has served on advisory boards/as a lecturer for Eli Lilly and Company, Teva, Amgen, Promius, Supernus, Valeant, and Avanir.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The GLADIATOR study was sponsored by Eli Lilly and Company. Professional writing and editorial support was provided by Synchrogenix LLC, a Certara Company.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
