Abstract
Background
Migraine is a disabling neurological disease adversely affecting many aspects of life. Most patients are still required to have failed several older oral preventive therapies before being reimbursed for a preventive, migraine-specific anti-calcitonin gene-related peptide treatment. In the 24-week placebo-controlled portion of DELIVER, eptinezumab was shown to reduce migraine frequency and resulted in higher migraine responder rates compared with placebo in patients with two to four previous preventive treatment failures. This subgroup analysis assessed if demographic or clinical characteristics were associated with differences in preventive benefits.
Methods
Migraine frequency reductions and responder rates (i.e., the proportion of patients reaching a ≥50% and ≥75% reduction in monthly migraine days relative to baseline) were determined in the total population and predefined subgroups by sex, age, migraine frequency (chronic migraine, episodic migraine, high-frequency episodic migraine, low-frequency episodic migraine), medication overuse, medication-overuse headache, and previous preventive treatment failures (2, >2). The primary endpoint was change from baseline in monthly migraine days over weeks 1–12.
Results
Eptinezumab 100 and 300 mg reduced monthly migraine days more than placebo over weeks 1–12 (−4.8 and −5.3 vs –2.1, respectively; p < 0.0001). In most subgroups, eptinezumab-treated patients demonstrated larger monthly migraine days reductions from baseline over weeks 1–12 than patients receiving placebo, with reductions maintained or increased over weeks 13–24. For ≥50% and ≥75% migraine responder rates, the odds ratios versus placebo all numerically favored eptinezumab.
Conclusion
Eptinezumab had larger monthly migraine days reductions and higher responder rates than placebo across clinically relevant subgroups showing that, across different demographic populations and clinical characteristics, eptinezumab is effective in patients with migraine and prior preventive treatment failures.
Introduction
Eptinezumab (Vyepti™, Lundbeck Seattle BioPharmaceuticals, Inc., Bothell, WA, USA) is a humanized monoclonal antibody indicated for the preventive treatment of migraine in adults in the United States (1,2), the European Union, and other countries. Eptinezumab selectively binds to and inhibits the activity of calcitonin gene-related peptide (CGRP) (1), a neuropeptide that plays a central role in migraine (3). Administered intravenously (IV), eptinezumab provides immediate and sustained therapeutic levels in patients, with peak plasma concentrations recorded 30 minutes to 1 hour after infusion (4,5). The efficacy and safety of eptinezumab for the preventive treatment of migraine in adults were established via multiple large-scale clinical studies, which demonstrated both rapid onset and sustained duration of its preventive effect (6–13).
DELIVER is a phase 3b clinical study designed to evaluate the efficacy and safety of eptinezumab for migraine prevention in patients with two to four prior preventive treatment failures. The primary data from the placebo-controlled portion of DELIVER illustrated the preventive effects of eptinezumab in this difficult-to-treat population, reducing migraine and headache frequency and severity and acute medication use relative to placebo (12). The objective of this subgroup analysis was to determine if any demographic or baseline clinical characteristics were associated with differences in the preventive benefits of eptinezumab compared to placebo in the placebo-controlled period of the DELIVER study.
Methods
Study design
DELIVER was a multicenter, parallel-group, double-blind, randomized, placebo-controlled phase 3b clinical study conducted at 96 locations across Europe and the United States. A detailed description of the study design has been published, as well as the protocol and statistical analysis plan (12). The full study comprised a 28–30-day screening period, a 24-week placebo-controlled period, and a 48-week dose-blinded extension period. The placebo-controlled phase of the study began on 1 June 2020, and was completed 7 October 2021. The placebo-controlled period of DELIVER was conducted in accordance with the standards of Good Clinical Practice as defined by the International Conference on Harmonisation and all applicable federal and local regulations, as is the ongoing extension. All study documentation was approved by the local review board at each site, or by a central institutional review board or ethics committee. All patients provided written informed consent prior to any study procedures. The DELIVER study is registered on ClinicalTrials.gov (NCT04418765) and EudraCT (2019-004497-25).
Patients
DELIVER enrolled adults aged 18–75 years (inclusive) with onset of migraine at or before 50 years of age and with a history of migraine (International Classification of Headache Disorders 3rd edition (ICHD-3) diagnostic criteria (14)) for ≥12 months before screening. Eligible patients had to have documented evidence (medical record or physician’s confirmation) of two to four prior preventive treatment failures of different medications (i.e., propranolol/metoprolol, topiramate, amitriptyline, flunarizine, candesartan, valproate/divalproex, botulinum toxin A/B) due to inadequate efficacy (i.e., no clinically meaningful improvement at the locally recommended dose for ≥3 months), tolerability reasons (i.e., discontinuation due to adverse events), or contraindications (i.e., ineligibility due to medical reasons) in the past 10 years. Due to specific requirements in some countries for access to anti-CGRP treatments, criteria were applied to the order and history of previous preventive medications. Specifically, at least two of the treatment failures had to have been propranolol/metoprolol, topiramate, amitriptyline, flunarizine, or candesartan, with ≥1 due to inadequate efficacy. Failure to succeed on valproate/divalproex or botulinum toxin A/B was considered only if the medication was regarded as the latest preventive prior to study inclusion. In addition, during the screening period, patients with chronic migraine (CM) had to report headache occurring on ≥14 days, of which migraine occurred on ≥8 days, and patients with episodic migraine (EM) had to report headache occurring on <14 days, of which migraine occurred on >4 days.
Patients were excluded from study participation if they had documented evidence of failure on a previous treatment targeting the CGRP pathway; history or diagnosis of other headache types; history of clinically significant cardiovascular disease; use of any traditional migraine preventive medication (beta-blocker, anticonvulsant, tricyclic, calcium channel blocker, angiotensin II receptor antagonist, or other locally approved migraine preventive) within one week prior to the screening visit; use of oral anti-CGRP for acute treatment <4 weeks prior to the screening visit; use of botulinum toxin <16 weeks prior to the screening visit; or use of eptinezumab or other monoclonal antibody targeting the CGRP pathway <24 weeks prior to the screening visit.
Patients using acute treatments for migraine were allowed to participate in the study, as were those with concurrent diagnosis of medication-overuse headache (MOH), which was prospectively confirmed by study investigators at screening using ICHD-3 criteria (14). Individuals with opioid-overuse headache were excluded from the study given that opioid use was only allowed providing its use did not exceed four days per month.
For a more detailed description of eligibility criteria, please refer to Ashina et al. (12).
Randomization and treatment
Patients were randomized (1:1:1) via a centralized randomization system to eptinezumab 100 mg, eptinezumab 300 mg, or placebo, which was stratified by monthly headache days (MHDs) at baseline (≤14 MHDs or >14 MHDs) and by country. Study drug was administered by IV infusion at baseline (day 0) and week 12. Treatments were administered by blinded personnel over a period of 30 minutes (plus ≤15 additional minutes, per protocol, if required), and patients were monitored for ≥1 hour after infusion completion.
Data collection and outcomes
At screening, patients were instructed to complete an electronic diary (eDiary) daily from the screening visit until the completion/withdrawal visit. The eDiary consisted of applications and reports which were used to derive the migraine and headache endpoints. The eDiary included an evening report (completed daily regardless of whether the patient had a headache) and a headache report (completed for each headache). Headache and migraine items were assessed with a yes/no response; and severity was rated as mild, moderate, or severe. For each headache reported, the start and stop date and time was collected. In addition to capturing headache episodes and migraine attacks, the eDiary captured information regarding headache characteristics and the intake of acute medications.
There were eight scheduled visits during the placebo-controlled period. Four were physical visits (screening, baseline, and at the end of weeks 12 and 24) and four were phone contact visits (at the end of weeks 4, 8, 16, and 20). Patients were contacted via phone every four weeks between infusion visits for eDiary compliance checks, to ensure that select patient-reported outcome measures had been completed, and for collection of relevant information such as adverse events and concomitant medication.
The primary efficacy endpoint was the change from baseline in the number of monthly migraine days (MMDs) following the first infusion (weeks 1–12). A migraine day was defined using the International Headache Society criteria (15). Key secondary endpoints included the percentages of patients with ≥50% reduction from baseline MMDs (i.e., ≥50% migraine responder rate (MRR); weeks 1–12) and ≥75% reduction from baseline MMDs (i.e., ≥75% MRR; weeks 1–12), as well as the change from baseline in the number of MMDs following the second infusion (weeks 13–24). Additional efficacy endpoints included the ≥50% and ≥75% MRRs following the second infusion (weeks 13–24). Safety endpoints analyzed during the placebo-controlled period, already reported (12), included treatment-emergent adverse events; absolute values and changes from baseline in clinical safety laboratory test values, vital signs, weight, and ECG parameter values; potentially clinically significant clinical safety laboratory test values, vital signs, weight changes, and ECG parameter values; and the Columbia–Suicide Severity Rating Scale score.
Statistical analyses
As analyses involved were both exploratory and post hoc, no sample size calculations were made specifically for these analyses. In the DELIVER primary study, based on simulations, the power was calculated to be at least 90% for the primary endpoints and at least 68% for individual key secondary endpoints (12). Data were analyzed for the following prespecified subpopulations defined at baseline: men, women, patients ≤35 years, patients >35 years, patients with chronic migraine (CM; >14 MHDs including ≥8 MMDs), patients with episodic migraine (EM; ≤14 MHDs including ≥4 MMDs), patients with high-frequency episodic migraine (HFEM; ≤14 MHDs including 8–14 MMDs), patients with low-frequency episodic migraine (LFEM; ≤14 MHDs including 4–7 MMDs), patients with an MOH diagnosis, patients with two prior preventive treatment failures, and patients with >2 prior preventive treatment failures.
For the primary endpoint, data were analyzed using the mixed model for repeated measures used for the primary analysis, described in detail in the statistical analysis plan (12). The model included effects for four-week periods from weeks 1–4 to 21–24, country, stratum (baseline MHDs: ≤14/>14), treatment as factors, baseline MMDs as a continuous covariate, treatment-by-month interaction, baseline score-by-month interaction, and stratum-by-month interaction. For subgroups EM/CM and LFEM/HFEM, terms with strata were not included.
The MRRs for ≥50% and ≥75% reduction from baseline in MMDs were analyzed using logistic regression. The model included baseline MMDs as a continuous covariate, and treatment and stratification (MHDs at baseline: ≤14/>14) as factors. The model was fitted using the maximum likelihood method and the logit link function. The odds ratios for eptinezumab 100 mg and eptinezumab 300 mg compared to placebo were estimated from the model and presented with p-values based on the likelihood ratio test and 95% confidence intervals (CIs) based on the profile likelihood.
Analysis of the primary endpoint (change from baseline in MMDs over weeks 1–12) in prespecified subgroups was a predefined exploratory analysis. Post hoc analyses were conducted evaluating secondary endpoints of change from baseline in MMDs (weeks 13–24), ≥50% MRR (weeks 1–12, weeks 13–24), and ≥75% MRR (weeks 1–12, weeks 13–24). In addition, the subgroup of patients who, based on eDiary data during the baseline period, reported acute medication use at or above medication overuse (MO) thresholds (per ICHD-3 criteria (14)) was also analyzed for the primary and secondary endpoints. MO thresholds were ≥10 days/month of triptans, ergots, opioids, or combination analgesics or ≥15 days/month of simple analgesics, or using two of the following types: simple analgesics, triptans, ergotamines, combination analgesics or opioid on ≥10 days/month.
Rules for missing data were previously published (12). In brief, for the primary endpoint (MMDs), prorating was used to calculate MMDs if the eDiary was completed on at least 14 of the 28 days of each four-week period, with MMDs classified as “missing” if the eDiary was completed on less than 14 of the 28 days. A missed day was defined as one in which the patient did not complete the evening report and did not report a headache. Prorated imputed missing data by four-week periods using the number of migraine days within the four-week period divided by the number of days with observations for the four-week period × 28 was used to obtain a monthly score for the four-week period. If the MMD value was missing for a given month, the responder status across a set of months including this, was derived based on the months with available values. Overall, there was a high level of compliance with the eDiary. The mean rate of missing eDiary data was ≤10% for all four-week intervals during the placebo-controlled period; the proportion of patients with ≥14 or ≥21 days of compliance was >96% and >90%, respectively, for all the treatment groups across intervals.
The following 3-way interaction tests were performed, all of which returned p-values above the usual cut-off value of 0.1: sex (p = 0.5103), EM/CM (p = 0.5091), age group (p = 0.4116), MOH (p = 0.5618), number of previous failed treatments (p = 0.1777), and LFEM/HFEM/CM (p = 0.8818). The models were fitted using maximum likelihood and compared via a likelihood ratio test. The tests showed no statistically significant interactions between treatment and any of the subgroups.
All p-values were based on two-sided tests; the CIs are two-sided and, as an exploratory analysis, all p-values generated for the subgroup analyses were not controlled for multiplicity. All statistical analyses were conducted using SAS software (SAS Institute, Inc., Cary, NC, USA) v9.4 or later.
Results
The full analysis set comprised 299, 293, and 298 patients receiving eptinezumab 100 mg, eptinezumab 300 mg, and placebo, respectively. In total, patients were predominantly female (n = 800/890 (90%)) and white (n = 854/890 (96%)). The majority of patients (n = 697/890 (78%)) were older than 35 years. There were slightly more patients with EM (n = 484) than CM (n = 405), and most patients with EM had HFEM (n = 372/484 (77%)). At screening, 12% of patients were diagnosed with MOH (n = 110/890); the post hoc analysis revealed that 49% of patients (n = 438/890) reported MO levels of acute medication during the 28–30-day baseline period. Most patients (n = 550/890 (62%)) had experienced two prior preventive treatment failures; 38% (n = 337/890) had experienced three or four. Nearly all patients had experienced ≥1 failure attributable to lack of efficacy (n = 889/890 (99.9%)), and more than half had ≥1 failure due to tolerability issues (n = 494/890 (56%)); only 28/890 (3.1%) had a failure attributed to contraindication.
Baseline MMD frequency varied across subgroups: means of 14.6 in men and 13.7 in women, and 14.4 in patients ≤35 years of age and 13.6 in patients >35 years (Table 1). Mean baseline MMDs were 6.4, 10.7, and 18.7 in patients with LFEM, HFEM, and CM, respectively. Patients with MOH and MO experienced an average of 17.3 and 17.1 MMDs at baseline, respectively. Baseline MMDs were higher in patients with >2 prior preventive treatment failures (14.4) than in those with just 2 (13.5).
Number of patients and baseline MMDs by subgroup and treatment.

aDerived using the eDiary reports of MMDs and monthly headache days during the 28–30-day screening period and missing data rules in the statistical analysis plan (12). A total of 292 patients received eptinezumab 300 mg. b298 (100 mg), 292 (300 mg), and 297 (placebo) patients were included within the treatment subgroups. cThree patients had <2 prior preventive treatment failures and were not included.
CM, chronic migraine; HFEM, high-frequency episodic migraine; LFEM, low-frequency episodic migraine; MMDs, monthly migraine days; MOH, medication-overuse headache; NC, not calculated.
In the total population, eptinezumab 100 mg and 300 mg reduced MMDs over weeks 1–12 by 4.8 and 5.3 days, respectively, compared with 2.1 days with placebo (p < 0.0001) (Figure 1(a)). Across all subgroups except men treated with eptinezumab 100 mg (n = 22) and patients with LFEM treated with eptinezumab 100 mg (n = 40), both of which were smaller subgroups, eptinezumab-treated patients demonstrated larger reductions from baseline in MMDs over weeks 1–12 than did patients receiving placebo (p < 0.05; Online Supplementary Figure 1(a)). The 95% CIs for the mean differences from placebo in change from baseline did not cross 0 for any subgroups except men and patients with LFEM (both of which had ≤40 patients per treatment arm).
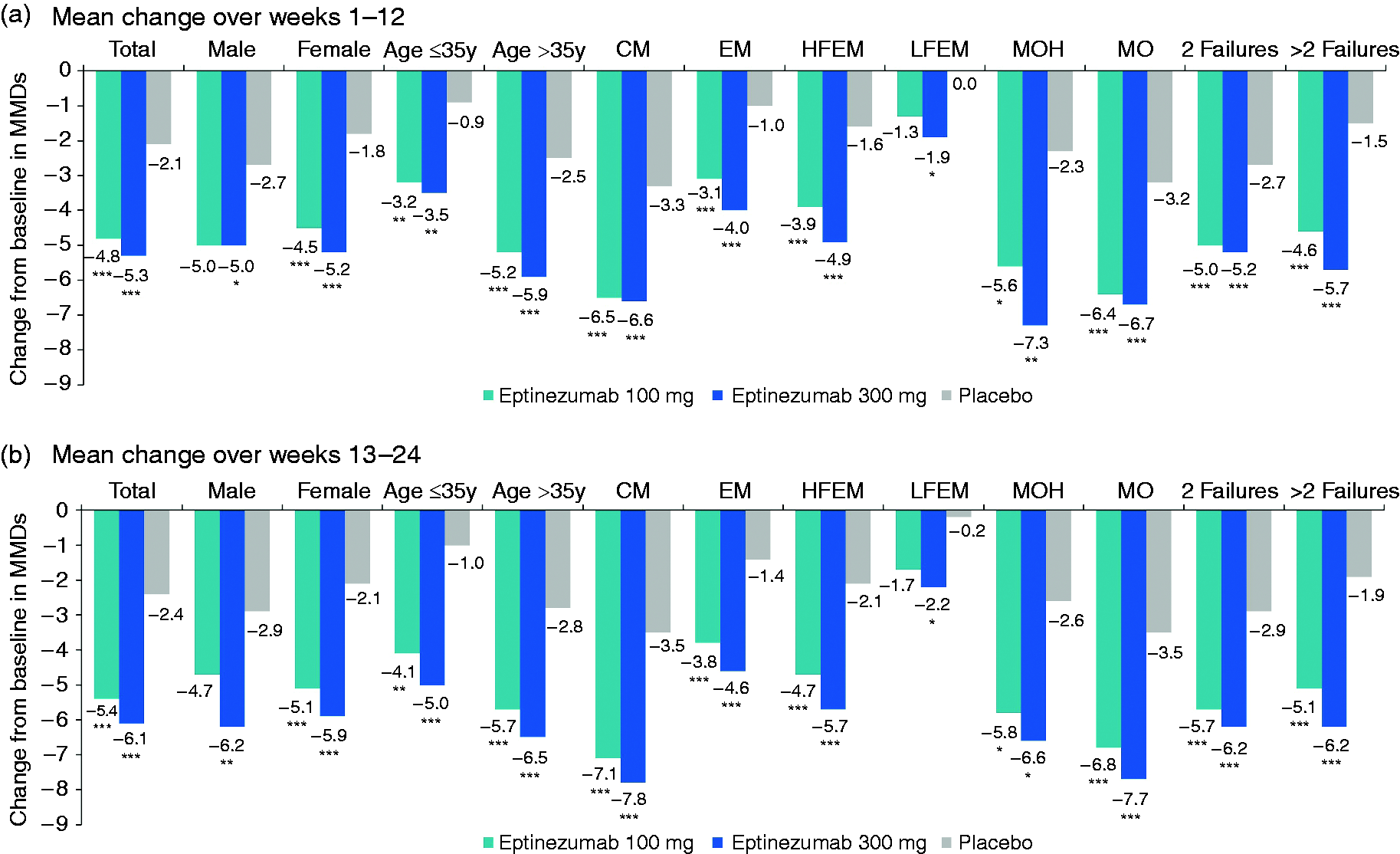
Mean change from baseline in monthly migraine days: (a) During weeks 1–12 and (b) During weeks 13–24.
Change from baseline MMDs with eptinezumab treatment over weeks 1–12 was similar in men and women (100 mg, –5.0 and –4.5; 300 mg, –5.0 and –5.2; placebo, –2.7 and –1.8 MMDs, respectively). Change from baseline MMDs was numerically larger in patients >35 years of age (100 mg, –5.2; 300 mg, –5.9; placebo, –2.5) than in younger patients (100 mg, –3.2; 300 mg, –3.5; placebo, –0.9). In the subgroups designated by migraine frequency, reductions in MMDs were largest in patients with CM (100 mg, –6.5; 300 mg, –6.6; placebo, –3.3), followed by HFEM (100 mg, –3.9; 300 mg, –4.9; placebo, –1.6) then LFEM (100 mg, –1.3; 300 mg, –1.9; placebo, 0.0). This is consistent with baseline differences across the subgroups, with baseline MMDs highest in CM, followed by HFEM then LFEM. Reductions from baseline in MMDs with eptinezumab did not appear to be numerically different depending on the number of previous preventive treatment failures (100 mg, –5.0; 300 mg, –5.2; placebo, –2.7 for patients with two previous treatment failures vs 100 mg, –4.6; 300 mg, –5.7; placebo, –1.5 for patients with >2 previous treatment failures). A diagnosis of MOH, a subgroup that demonstrated high baseline MMDs, was associated with greater eptinezumab-associated MMD reductions relative to the total population (100 mg, –5.6; 300 mg, –7.3; placebo, –2.3 vs 100 mg, –4.8; 300 mg, –5.3; placebo, –2.1, respectively). In the post hoc analysis in patients with MO during baseline, changes from baseline were at least twice as large with eptinezumab as placebo (100 mg, –6.4; 300 mg, –6.7; placebo, –3.2).
Reductions in MMDs were maintained or further improved over weeks 13–24 across subgroups (Figure 1(b) and Online Supplementary Figure 1(b)).
Across subgroups, more patients in each of the eptinezumab treatment groups than in the placebo group achieved ≥50% reduction from baseline in MMDs during weeks 1–12 (Figure 2) and weeks 13–24 (Online Supplementary Figure 2). The odds ratios of achieving ≥50% reduction in MMDs favored eptinezumab over placebo (odds ratio >1; p < 0.05) for the majority of subgroups over each dosing interval (Online Supplementary Figure 3). There was numerical benefit with eptinezumab versus placebo across all subgroups; however, over weeks 13–24, p-values were >0.05 for men (100 mg, p = 0.1035) and those diagnosed with MOH (100 mg, p = 0.2316; 300 mg, p = 0.0578).
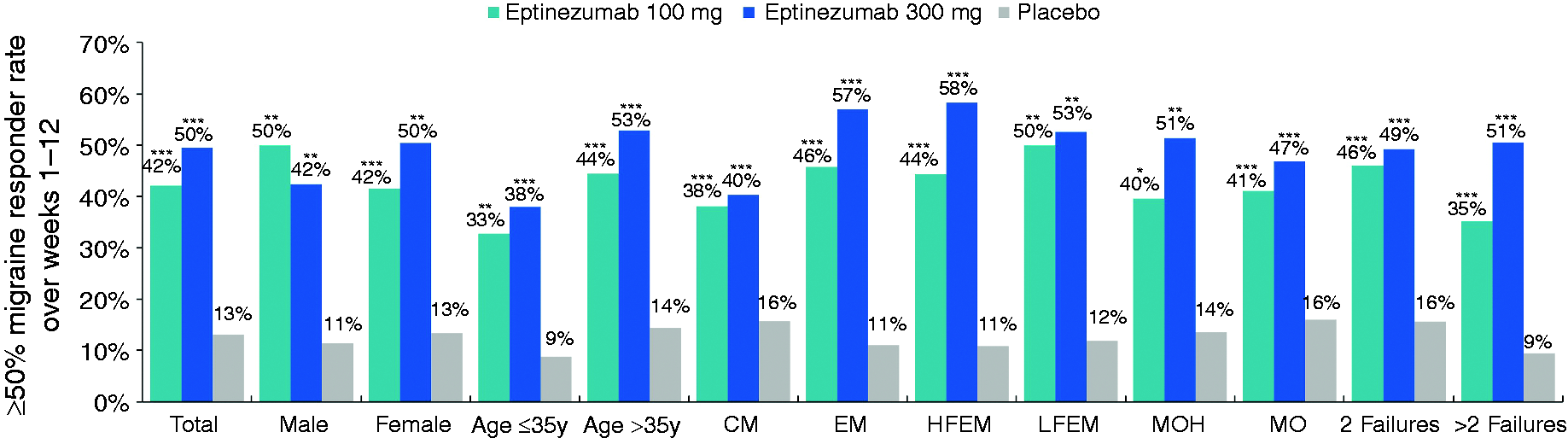
≥50% Migraine responder rates over weeks 1–12.
Across subgroups, more patients in each of the eptinezumab treatment groups than in the placebo group achieved ≥75% reduction from baseline in MMDs during weeks 1–12 (Figure 3) and weeks 13–24 (Online Supplementary Figure 4). The odds ratios of achieving ≥75% reduction in MMDs favored eptinezumab over placebo (odds ratio >1; p < 0.05) for most subgroups for which odds ratios could be calculated (Online Supplementary Figure 5). Despite numerical benefit with eptinezumab versus placebo across all subgroups, over weeks 1–12, the p-value was >0.05 for patients with LFEM (100 mg, p = 0.1080); over weeks 13–24, p-values were >0.05 for men (100 mg, p = 0.9697), patients ≤35 years of age (100 mg, p = 0.0744), and those with LFEM (100 mg, p = 0.0747), and those diagnosed with MOH (100 mg, p = 0.5590; 300 mg, p = 0.2141).

≥75% migraine responder rates over weeks 1–12.
Discussion
In this analysis of data from the placebo-controlled portion of DELIVER, the efficacy of eptinezumab for migraine prevention in patients with two to four prior preventive treatment failures met the primary endpoint and demonstrated a greater change from baseline in MMDs (weeks 1–12) for eptinezumab than for placebo in the total population (12), as well as most subgroups. The results for MMD reductions over weeks 13–24 were consistent with the primary endpoint, and results for ≥50% and ≥75% MRRs demonstrated numerical benefit with both eptinezumab doses during weeks 1–12 and 13–24 for all subgroups. Results demonstrate that at least one-third of eptinezumab-treated patients in any subgroup experienced a clinically relevant reduction in migraine frequency (i.e., ≥50% MRR) after a single dose. Most subgroups demonstrated additional benefit (i.e., larger reductions and/or higher MRRs) after a second dose. Together, these exploratory analyses indicate that a single dose or an additional dose of eptinezumab is similarly effective in reducing migraine frequency across clinically relevant patient subgroups, with no one subgroup suggesting a substantially different impact than the total population, showing that eptinezumab is efficacious across patients with different clinical and disease characteristics.
In the subgroups designated by migraine frequency, reductions in MMDs over weeks 1–12 were numerically greater in patients with CM, followed by HFEM, then LFEM. This was consistent with average baseline MMDs; that is, patients who started with a higher migraine frequency (e.g., patients with CM) at baseline had more MMDs to reduce when compared to those with a lesser amount of MMDs at baseline (e.g., patients with LFEM). This finding is of importance as it shows that eptinezumab can reduce MMDs in patients with a varying frequency and severity of migraine.
Similarly to the general migraine population in the PROMISE studies (16), more than half of all eptinezumab-treated patients in the HFEM and LFEM subgroups achieved a ≥50% reduction from baseline MMDs during weeks 1–12, as did 39% of patients in the CM subgroup (16). Unlike the PROMISE studies, the DELIVER study only examined patients with 2 or more previous treatment failures; therefore, although a comparison suggests that a higher proportion of patients in the PROMISE studies had a robust response, the DELIVER study represented a subpopulation of patients with migraine that may be harder to treat based on their history of treatment failure. In addition, patients with CM in DELIVER had a higher number of baseline MMDs than patients with CM in PROMISE-2 (18.7 vs 16.1, respectively) (8). Despite a seemingly more difficult-to-treat population, DELIVER succeeded in reducing MMDs in patients with multiple previous preventive treatment failures and across various subpopulations.
Consistent with findings in the general PROMISE population (16), eptinezumab-associated reductions in MMDs were similar in males and females across weeks 1–12 and 13–24. The male subgroups were smaller in both DELIVER (n = 90 (10.1%)) and PROMISE (n = 231 (13.3%)) (16), a reflection of the epidemiology of migraine (i.e., more common in females than in men (17)). While these data suggest similar efficacy in males and females, it is difficult to draw conclusions based on a small number of males. Historically, males with migraine have been underrepresented in clinical trials (6,7,9–12,18–20). Future clinical trials including more male participants are needed to demonstrate equal treatment efficacy across genders.
The ≥50% MRRs in the current analysis were greater in patients >35 versus patients ≤35 years of age, contrasting with the PROMISE studies which had similar responder rates of ≥50% reduction from baseline MMDs across different age groups (16). It should be noted, however, that the number of patients in the ≤35 years of age subgroup was relatively small and that mean baseline MMDs was higher than for the subgroup >35 years of age. Interestingly, during weeks 1–12 of the FOCUS study, which examined the efficacy and safety of fremanezumab in a similar preventive-treatment–resistant population, reductions in migraine frequency were numerically greater in younger patients (19). Further studies are needed to understand the impact of patient age on treatment efficacy given these contrasting results.
Changes in MMDs following eptinezumab treatment did not appear to be associated with the number of previous preventive treatment failures; however, the subgroup with >2 previous treatment failures had a lower placebo MMD response than the subgroup with two previous treatment failures. The placebo response was also lower in the overall cohort of DELIVER patients, who were only eligible for study inclusion if they had previous treatment failures, in comparison to PROMISE-1 (7) and PROMISE-2 (8) studies that included patients regardless of previous treatment failures. The efficacy of CGRP inhibitors was similarly demonstrated in a severe subpopulation of the CONQUER study, for which galcanezumab effectively reduced migraine frequency versus placebo in patients with three to four previous preventive therapy failures (20). Similarly, the FOCUS study examining the effects of fremanezumab in patients with two, three or four previous therapy failures observed a greater effect size with increasing number of previous treatment failures (21).
The proportion of patients with MOH—a potentially disabling secondary diagnosis for patients with migraine (22,23)—in DELIVER was unexpectedly low (12%) for a population composed mostly of patients classified as high-frequency EM and CM (12). This proportion is very different from those with MO in the FOCUS and CONQUER studies and likely reflects differences in how overuse was defined. The MOH subgroup in DELIVER comprised patients diagnosed with MOH at screening per ICHD-3 criteria; FOCUS and CONQUER reported MO during the baseline period (i.e., not fulfilling the MOH diagnostic criteria of a minimum of three months of medication overuse) (24,25). In addition to evaluating the subgroup with diagnosed MOH, we conducted a post hoc analysis of each endpoint in patients with MO—that is, those who were not necessarily prospectively diagnosed in the DELIVER study but reported acute medication use during a 28–30-day baseline period that was at or above thresholds used for MOH diagnosis (14). The analysis found that 49% of patients reported MO levels of acute medication use, and efficacy analyses indicated a preventive effect comparable to that in the population diagnosed with MOH. This indicates that preventive treatment with eptinezumab can be used to help break the cycle of MOH as well as preventing patients with MO progressing to chronic migraine and MOH. Another study is in progress to evaluate the efficacy and safety of eptinezumab as add-on to patient education in treating patients with CM and MOH (26) and further research is needed to determine the impact of eptinezumab on preventing migraine progression towards more severe migraine and MOH.
Overall, when analyzing smaller subgroups of the participants in the DELIVER clinical trial the results of this study demonstrate that eptinezumab’s efficacy is generally similar across different subpopulations of patients. This is of importance, as this may show that in real-world situations, eptinezumab can be effective across diverse populations.
Limitations
The population of DELIVER was not demographically diverse, comprising predominantly white females over the age of 35 (12). Our findings, therefore, may not be indicative of effects in other demographic segments including patients of other ethnic and/or racial backgrounds (27). Demographic representation in migraine related studies is important and future clinical trials will attempt to improve sample representativeness, particularly regarding race and ethnicity. Several approaches—such as developing a diverse pool of investigators and staff at clinical trial sites, engaging the community, developing patient-friendly resources that make it easier to identify and enroll in relevant clinical trials, and using alternative recruitment techniques—can be used to increase diversity in studies. Ensuring care equity by showing that preventive treatment is effective in all patient populations can help increase quality of life for all of those who suffer from migraine. For several of the subpopulations, sample sizes were small—the male subgroup had only 90 patients (10% of the total population) and the ≤35-years age group had 193 (22% of the total population). Only 112 (13%) had LFEM and 110 (12%) had MOH. DELIVER was not powered for these subgroup analyses, making it difficult to demonstrate separation in these patient subgroups. Comorbidities and life events may influence disease activity and while this study was placebo-controlled and did control for levels of disease activity, the study did not control for factors such as mood disorders, insomnia, caffeine use, or stressful life events (28). The study also did not examine effects in patients with a history of anti-CGRP therapy failure and thus is only demonstrative of effects in patients with a history of failures of traditional preventive medications.
Conclusion
In adults with migraine and documented evidence of two to four previous preventive treatment failures, eptinezumab-treated patients demonstrated larger reductions from baseline in MMDs over weeks 1–12 than did patients receiving placebo. Reductions in MMDs across demographic subgroups, migraine classification, medication overuse, or number of prior preventive treatment failures also demonstrated larger reductions in MMDs from baseline than did patients receiving placebo.
Article highlights
In patients with migraine and multiple previous preventive treatment failures, eptinezumab resulted in greater MMD reductions than placebo during weeks 1–12 and 13–24 across subgroups defined by demographic and clinical characteristics. Rates of ≥50% and ≥75% MMD reductions were consistently higher with eptinezumab than with placebo across all subgroups.
Supplemental Material
sj-jpg-1-cep-10.1177_03331024231170807 - Supplemental material for Efficacy and safety of eptinezumab for migraine prevention in patients with prior preventive treatment failures: subgroup analysis of the randomized, placebo-controlled DELIVER study
Supplemental material, sj-jpg-1-cep-10.1177_03331024231170807 for Efficacy and safety of eptinezumab for migraine prevention in patients with prior preventive treatment failures: subgroup analysis of the randomized, placebo-controlled DELIVER study by Messoud Ashina, Michel Lanteri-Minet, Anders Ettrup, Cecilie Laurberg Christoffersen, Mette Krog Josiassen, Ravinder Phul, Bjørn Sperling and Patricia Pozo-Rosich in Cephalalgia
Supplemental Material
sj-jpg-2-cep-10.1177_03331024231170807 - Supplemental material for Efficacy and safety of eptinezumab for migraine prevention in patients with prior preventive treatment failures: subgroup analysis of the randomized, placebo-controlled DELIVER study
Supplemental material, sj-jpg-2-cep-10.1177_03331024231170807 for Efficacy and safety of eptinezumab for migraine prevention in patients with prior preventive treatment failures: subgroup analysis of the randomized, placebo-controlled DELIVER study by Messoud Ashina, Michel Lanteri-Minet, Anders Ettrup, Cecilie Laurberg Christoffersen, Mette Krog Josiassen, Ravinder Phul, Bjørn Sperling and Patricia Pozo-Rosich in Cephalalgia
Supplemental Material
sj-jpg-3-cep-10.1177_03331024231170807 - Supplemental material for Efficacy and safety of eptinezumab for migraine prevention in patients with prior preventive treatment failures: subgroup analysis of the randomized, placebo-controlled DELIVER study
Supplemental material, sj-jpg-3-cep-10.1177_03331024231170807 for Efficacy and safety of eptinezumab for migraine prevention in patients with prior preventive treatment failures: subgroup analysis of the randomized, placebo-controlled DELIVER study by Messoud Ashina, Michel Lanteri-Minet, Anders Ettrup, Cecilie Laurberg Christoffersen, Mette Krog Josiassen, Ravinder Phul, Bjørn Sperling and Patricia Pozo-Rosich in Cephalalgia
Supplemental Material
sj-jpg-4-cep-10.1177_03331024231170807 - Supplemental material for Efficacy and safety of eptinezumab for migraine prevention in patients with prior preventive treatment failures: subgroup analysis of the randomized, placebo-controlled DELIVER study
Supplemental material, sj-jpg-4-cep-10.1177_03331024231170807 for Efficacy and safety of eptinezumab for migraine prevention in patients with prior preventive treatment failures: subgroup analysis of the randomized, placebo-controlled DELIVER study by Messoud Ashina, Michel Lanteri-Minet, Anders Ettrup, Cecilie Laurberg Christoffersen, Mette Krog Josiassen, Ravinder Phul, Bjørn Sperling and Patricia Pozo-Rosich in Cephalalgia
Supplemental Material
sj-jpg-5-cep-10.1177_03331024231170807 - Supplemental material for Efficacy and safety of eptinezumab for migraine prevention in patients with prior preventive treatment failures: subgroup analysis of the randomized, placebo-controlled DELIVER study
Supplemental material, sj-jpg-5-cep-10.1177_03331024231170807 for Efficacy and safety of eptinezumab for migraine prevention in patients with prior preventive treatment failures: subgroup analysis of the randomized, placebo-controlled DELIVER study by Messoud Ashina, Michel Lanteri-Minet, Anders Ettrup, Cecilie Laurberg Christoffersen, Mette Krog Josiassen, Ravinder Phul, Bjørn Sperling and Patricia Pozo-Rosich in Cephalalgia
Footnotes
Acknowledgements
The authors thank Mary Tom, PharmD, and Nicole Coolbaugh, CMPP, of The Medicine Group (New Hope, PA, USA), for providing medical writing support in accordance with Good Publication Practice guidelines, which was funded by H. Lundbeck A/S.
Author contributions
All authors had full access to the study data and take responsibility for the integrity of the data and the accuracy of the data analysis. MA, MKJ, RP, and BS contributed to the conception or design of the study. AE, CLC, MKJ, RP, and BS contributed to obtaining funding. MA, MLM, and PPR were study investigators, contributing to the acquisition of data. CLC and MKJ conducted the statistical analyses, and all authors contributed to the interpretation of data. MA, AE, CLC, and RP were involved in drafting the manuscript, and all authors critically revised the manuscript for important intellectual content. All authors provided final approval of the manuscript content for submission and had final responsibility for the decision to submit for publication.
Availability of data and materials
In accordance with EFPIA’s and PhRMA’s “Principles for Responsible Clinical Trial Data Sharing” guidelines, Lundbeck is committed to responsible sharing of clinical trial data in a manner that is consistent with safeguarding the privacy of patients, respecting the integrity of national regulatory systems, and protecting the intellectual property of the sponsor. The protection of intellectual property ensures continued research and innovation in the pharmaceutical industry. Deidentified data are available to those whose request has been reviewed and approved through an application submitted to ![]() .
.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: MA reported receiving personal fees from AbbVie, Allergan, Amgen, Eli Lilly, Lundbeck, Novartis, and Teva Pharmaceuticals during the conduct of the study. MA reported serving as associate editor of Cephalalgia, associate editor of The Journal of Headache and Pain and associate editor of Brain.
MLM reports personal fees from AbbVie, Allergan, Amgen, Grunenthal, IPSEN, Pfizer, Reckitt-Benkiser, Salvia BioElectronics, Sanofi, Sun Pharmaceutical, UPSA, and Zambon; grants, personal fees from Eli Lilly, Lundbeck, Medtronic, Novartis, and Teva Pharmaceuticals. MLM reported serving as associate editor of The Journal of Headache and Pain and associate editor of Current Pain and Headache Reports.
AE, CLC, MKJ, RP, and BS are full-time employees of H. Lundbeck A/S or one of its subsidiary companies.
PPR reports honoraria as a consultant and participation in the last three years in advisory boards for Allergan/AbbVie, Almirall, Amgen, Biohaven, Chiesi, Eli Lilly, Lundbeck, Novartis, and Teva Pharmaceuticals; institutional research support from AbbVie, AGAUR, EraNet NEURON, Instituto Investigación Carlos III, International Headache Society, La Caixa Foundation, Novartis, PERIS, RIS3CAT FEDER, and Teva Pharmaceuticals; being a principle investigator for over 45 clinical trials (phases II, III, and IV) for the preventive treatment of migraine and other headaches; education projects with Allergan/AbbVie, Almirall, Chiesi, Eli Lilly, Lundbeck, Medlink, Medscape, Neurodiem, Novartis, and Teva Pharmaceuticals; participation in the Scientific Advisory Board of Migraine Research Foundation & Lilly Foundation Spain and Honorary Secretary of the International Headache Society; and being an associate editor for Cephalalgia, Headache, Neurologia, and Frontiers of Neurology, director for headache section of Revista de Neurologia, and editorial advisor for The Journal of Headache and Pain.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was sponsored and funded by H. Lundbeck A/S, including medical writing support for the development of the manuscript. In collaboration with the academic authors, the sponsor participated in the design and conduct of the study and in the collection, management, analysis, and interpretation of the data. The preparation, review, and approval of the manuscript was undertaken by all authors and by a professional medical writer and editor funded by the sponsor. All authors and H. Lundbeck A/S prepared, reviewed, and approved the final version of the manuscript and made the decision to submit the manuscript for publication. The sponsor did not have the right to veto publication or to control the decision regarding to which journal the manuscript was submitted.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
