Abstract
Migraine patients frequently report cognitive difficulties in the proximity and during migraine attacks. We performed an exploratory comparison of executive functioning across the four stages of the migraine cycle. Consecutive patients with episodic migraine undertook cognitive tests for attention, processing speed, set-shifting, and inhibitory control. Performance was compared between patients in different migraine stages, controlling for attack frequency and prophylactic medication. One hundred forty-three patients (142 women, average age 36.2 ± 9.9 years) were included, 28 preictal (≤48 h before the attack), 21 ictal (during the attack), 18 postictal (≤24 h after attack), and 76 interictal. Test performance (age and literacy adjusted z-scores) was not significantly different across migraine phases, despite a tendency for a decline before the attack. This negative study shows that cognitive performance fluctuates as patients approach the attack. To control for individual variability, this comparison needs to be better characterized longitudinally with a within-patient design.
Introduction
Migraine is a cyclic brain disorder characterized by recurring attacks of headache, intolerance to sensory stimuli and autonomic dysfunction. 1 The attacks are preceded and followed by brain changes, documented in functional brain imaging and neurophysiological measures, which involve sensory processing and autonomic and cognitive functioning. 2 –4 These phenomena have been conceptualized to cycle within four distinctive phases: premonitory (preictal), ictal, resolution (postictal), and interictal.
Cognitive difficulties are commonly reported in all active phases of migraine. During attacks, patients describe an inability to focus or think properly, they feel mentally slow and have difficulty in carrying more than one task simultaneously, which makes cognitive impairment to rank second, after pain, among the causes of attack-related disability. 5 Likewise, in the preictal and postictal phases, patients often report poor concentration, irritability, and difficulty in oral and written speech. 5 These descriptions suggest that migraine attacks are associated with a reversible disorder of attention and executive functions, which have been supported by neuropsychological evaluations performed during the attacks. 6 However, there are no systematic studies, assessing cognitive performance during the other phases of migraine. The aim of this study was to perform a prospective exploratory evaluation of the executive functioning of migraine patients observed in different phases of the attack. Our hypothesis was that patients would have a worse performance on the preictal and ictal phases compared to the interictal period.
Methods
This is a prospective, cross-sectional, observational study of patients with episodic migraine, assessed during different phases of migraine cycle. Patients were recruited in a Headache Outpatient Clinic of a University Hospital, during a scheduled medical appointment, after agreement and signing the informed consent.
Adult patients aged between 18 years and 60 years, with episodic migraine with or without aura fulling the International Classification of Headache Disorders-III criteria, 1 were invited to participate. Exclusion criteria were (a) a diagnosis of major depression or other psychiatric disorder; (b) illiteracy; (c) history of other neurologic disorders (e.g. stroke, head trauma, epilepsy, etc.) or developmental delay; and (d) pregnancy, in the case of female patients. The study protocol was approved by the Lisbon Academic Medical Center Ethics Committees.
Procedures
Demographic and clinical data were collected through a semistructured interview and the analysis of patients’ calendars. Data included present age, age at migraine onset, current attack frequency (<1, 1–3, and >3 attacks per month), average attack duration (<24 h, 24–48 h, and ≥48 h), average headache intensity (mild, moderate, and severe), pain type and location, current prophylactic treatment (in particular, antiepileptic medication), and a family history of migraine. Females in child-bearing age were asked if they were menstruating at the time of evaluation.
Patients undertook a brief cognitive evaluation performed by a psychologist or a trained medical student immediately after the appointment. This assessment comprised tests tackling sustained attention and processing speed (digit symbol subtest from the Wechsler Adult Intelligence Scale—III), 7 divided attention/set-shifting (trail making tests A, B, and B-A), 8 and selective attention/inhibitory control (stroop interference test). 9 Individual scores were converted to age and education adjusted z-scores according to the existing norms. 10,11
Patient’s migraine status
Date and time of cognitive testing and time since the last attack were recorded. Patients were contacted by telephone 48 h after the evaluation to check for new attacks. Time of assessment during the migraine cycle was then categorized into four stages: (a) preictal (assessment took place within 48 h before the onset of an attack), (b) ictal (during reported attack), (c) postictal (within the first 24 h following an attack), and (d) interictal (none of previous).
End points and statistical analysis
Statistical analysis was performed with the SPSS 25.0. Descriptive statistics of mean and standard deviation were obtained for continuous variables, and counts and frequencies for categorical variables. Group differences were tested using a one-way analysis of variance for continuous variables or χ 2 test for categorical variables. A multivariate analysis of covariance (ANCOVA) was performed with cognitive performance as the dependent variable and migraine cycle phase as the independent variable. Attack frequency and the use of prophylactics were entered in the model as covariates. Statistical significant differences between groups were analyzed with Bonferroni post hoc test. Results were considered statistically significant at p < 0.05. The sample size was calculated for primary outcome (Stroop words, trail making test (TMT) A) based on a report on cognitive performance during and between attacks, 6 assuming a level of significance of 95%, an 80% power, and an allocation ratio of 3/2. For Stroop Words, we estimated a sample size of 73 (44 in interictal and 29 in perictal) to detect a difference of 13 words between the two groups. For TMT A, we estimated a sample size of 163 (98 in interictal and 65 in perictal) to detect a difference of 6.8 s between the two groups.
Results
A total of 143 patients were included, 142 (99.3%) were women, with an age average of 36.2 years (±9.9 years, ranging between 18 years and 56 years). Migraine features are depicted in Table 1. Since there were some missing data that could not be collected or did not apply to all subjects (for instance, the menstrual period), Table 1 also presents the number of subjects that were eligible for the analyses of each variable. Patients were more frequently observed in the interictal phase (53.7%), followed by the preictal (19.6%), ictal (14.7%), and the postictal phases (12.6%). Most patients were under prophylactic treatment, 16 taking antiepileptic medication. There were no significant differences in patients’ demographic or clinical features between phases. Twelve patients were observed during menstruation with a similar frequency across the four phases.
Sample’s demographic, clinical, and cognitive data.

m: months; SD: standard deviation; stroop C: stroop color naming; stroop CW: stroop interference; stroop W: stroop reading; TMT A: trail making test part A; TMT B: trail making test part B; ANOVA: analysis of variance.
aOne-way ANOVA (continuous variables) or χ 2 test (categorical variables); significance set at p < 0.05.
b Eligible cases for analyses are indicated for each variable: n = 143.
c Eligible cases for analyses are indicated for each variable: n = 118.
d Eligible cases for analyses are indicated for each variable: n = 137.
e Eligible cases for analyses are indicated for each variable: n = 141.
f Eligible cases for analyses are indicated for each variable: n = 142.
g Eligible cases for analyses are indicated for each variable: n = 40.
h Eligible cases for analyses are indicated for each variable: n = 91.
Patients’ test scores were, on average, within normal range (i.e. between −0.5 and +0.5 on age and education adjusted z-scores; Table 1). Cognitive performance was not statistically different between phases, although some trends could be identified (Figure 1), namely a preictal decline on Stroop Color Naming and interference and TMT A and B (measures of inhibitory control and divided attention), a worse performance on TMT B−A on the postictal phase (measuring attention shift), and a decline on Digit Symbol in the ictal phase (measuring processing speed). No significant differences were found when the interictal scores were compared with all other migraine phases. Moreover, no differences were found on the multivariate ANCOVA controlling for attack frequency and prophylactic medication (Table 2).

Cognitive performance across migraine cycle phases.
Effect of migraine cycle on the cognitive performance (multivariable linear regression analysis).a
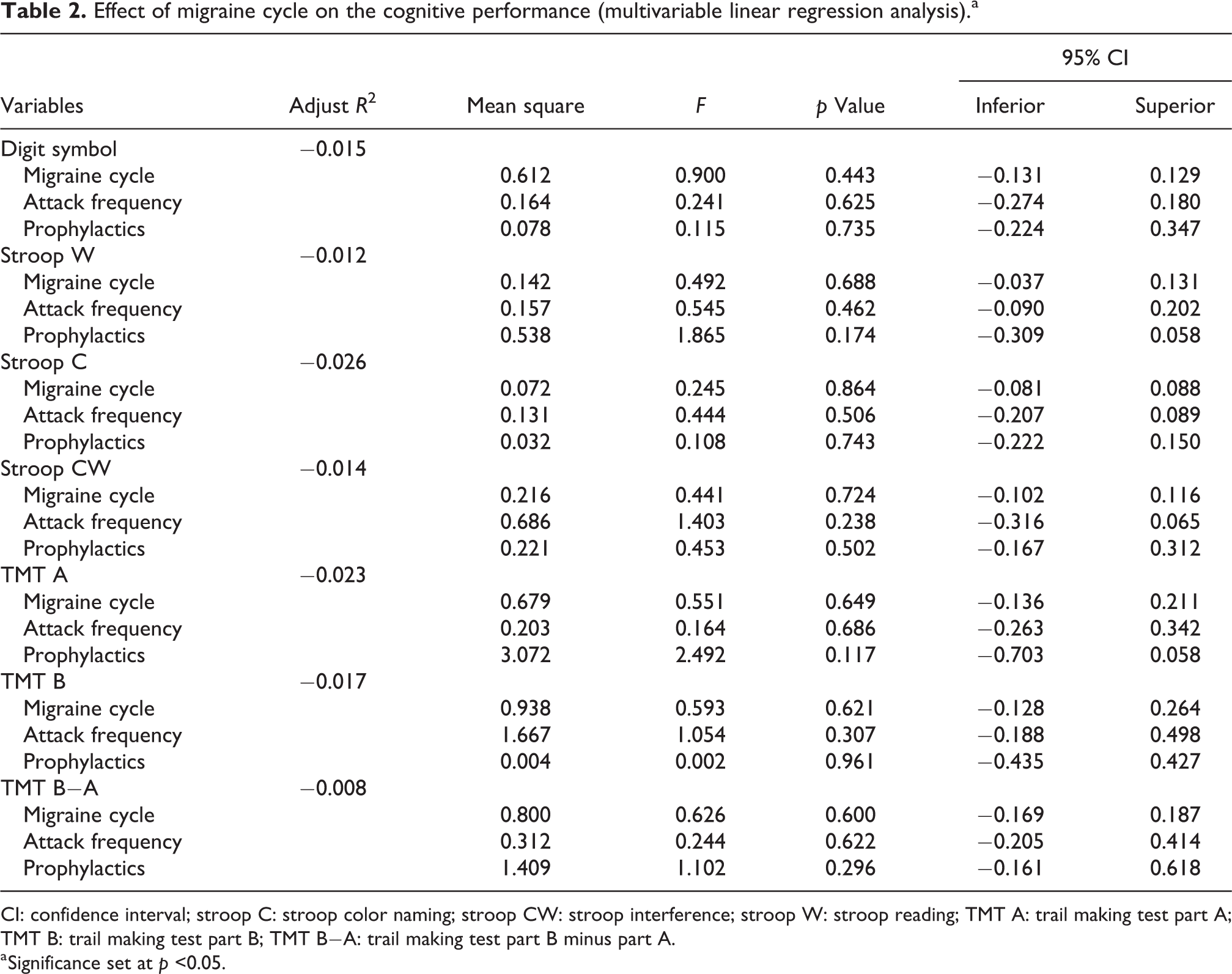
CI: confidence interval; stroop C: stroop color naming; stroop CW: stroop interference; stroop W: stroop reading; TMT A: trail making test part A; TMT B: trail making test part B; TMT B−A: trail making test part B minus part A.
a Significance set at p <0.05.
Discussion
In this exploratory study, we observed minor fluctuations in cognitive test performance between the four migraine phases, but we were unable to document significant differences across the stages even when the three peri-ictal phases were aggregated and compared with the interictal period. This is, therefore, a negative study that contrasts with previous reports, in which repeated measures of the same participants showed a significant decrease in processing speed and memory during the attacks, compared to interictal phase. 6
We interpret these negative results due to several factors. The first is related to individual variability both on baseline functioning (interictal phase) and attack-related impairment. Although we found no differences between groups on demographic and literacy variables (which are strong predictors of cognitive performance), we did not measure their cognitive background or IQ, which might have allowed to correct intersubject variability and reduce group differences. On the other hand, migraine is known as a highly individualized disorder with a variability of the attacks within and between subjects. Within-patient differences have been documented in cognitive, imaging, and neurophysiological studies with smaller samples of patients but using within-subjects comparisons. 2,3,6 A longitudinal study, with a within-patient design, is, therefore, more adequate to investigate this question controlling for individual variations.
Secondly, changes observed were minor since, on average, all the results obtained in the four migraine phases were within normal range. This may indicate that the tests used may not have sufficient sensitivity to document the changes that are perceived by the patients and may impair their performance in more complex and demanding activities of work or daily living. Thirdly, migraine affects mostly women, and there is some evidence that cognitive performance fluctuates along the menstrual cycle, with a worse performance in the luteal phase 12 and that brain connectivity changes in relation with the menstrual phase in natural cycles. 13 However, given that in this study, we included women on contraception, postmenopausal, and women in different phases of the cycle, and it is difficult to contemplate this information in the analysis. Nevertheless, we included the presence of menstrual period during assessment, an occasion where migraine attacks tend to cluster, but this had no impact on performance.
Finally, it is known that frequent attacks and chronic migraine are more frequently associated with cognitive changes 14,15 than episodic infrequent migraine, which could explain the lack of significant changes found on the present sample, in which most patients had one to three attacks per month.
We acknowledge other limitations and confounders that need to be addressed in future studies, in addition to the questions concerning the study design. One is the difference of sample sizes between groups of patients within each migraine phase. For instance, the higher interictal group size reported herein can be explained by the fact that episodic migraine diagnosis, by definition, includes patients that have migraine less than 15 days per month, which suggests that the odds of screening/recruiting patients in other phases is lower. Secondly, the necessary sample size varied among cognitive tests, which may have impaired significance on some of the measures used. Furthermore, we compared group averages, a method with less statistical power than within-subject comparisons.
Finally, and based on previous findings, we restricted the cognitive evaluation to our initial hypothesis of an expected decline in executive functioning and did not test for memory, fluency, vocabulary, or other cognitive functions. Besides, tests used were designed to diagnose cognitive impairment and may lack sensitivity when looking for mild fluctuations. Yet, all have a good test–retest reliability (ranging from 0.71 to 0.91). 16 –18 It is possible that more cognitively demanding tasks, such as those with computerized reaction time, sustained periods of attention, verbal fluency, or demanding memory tasks, more similar to the abilities required to work, are more significantly affected in the peri-ictal period.
Taken together, these factors may have contributed to the negative results obtained and limited their interpretation, underlining the need for longitudinal studies with a within-subject design.
Clinical implications
Patients with migraine experience cognitive difficulties during and on the vicinity of migraine attacks that contribute to attack-related disability.
We evaluated 143 patients in different phases of migraine with cognitive tests and could not find an objective decline in cognitive performance between the interictal phase and the ictal or peri-ictal periods, which can be due to patients’ and attack variability.
Longitudinal studies comparing cognitive performance within the same subject along the migraine cycle are warranted to clarify this question.
Footnotes
Acknowledgment
We would like to thank Dr Pedro Alves for his contribution to the calculation of the sample size.
Declaration of conflicting interests
The author(s) declared following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: IPM and RG-G have received honoraria from Allergan, Novartis, Teva, and Eli Lilly.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
