Abstract
Patients with demyelinating diseases (DDs) such as multiple sclerosis have a 20-fold higher risk of developing trigeminal neuralgia (TN). DD-related TN is more frequently refractory to the usual medical and surgical treatment. We report the case of a 57-year-old man presenting to our neurology outpatient clinic with a 12-year history of medical and surgical refractory TN associated with demyelinating lesions on magnetic resonance imaging. After a new failure of pharmacological treatment with oxcarbazepine, pregabalin, baclofen, and duloxetine, motor cortex stimulation (MCS) was performed, and the patient remained mostly pain-free, without any pharmacological treatment during the 3.5 years of follow-up. MCS may be a useful approach for DD-related refractory TN, and further studies can clarify its role in TN management.
Introduction
Trigeminal neuralgia (TN) is an uncommon cause of facial pain affecting less than 0.1% of the general population. 1 However, patients with demyelinating diseases (DDs) such as multiple sclerosis (MS) have a 20-fold higher risk of developing TN. 2 TN can occur either in classical form, mostly related to a neurovascular contact, or in symptomatic form like the one related to DDs. 3 However, the symptomatic form could be even more complicated—that is, a high proportion of patients with DD-related TN were found to have simultaneously a pontine plaque coexisting with a neurovascular compression. This led to the proposal of a double-crush mechanism, where increased neuronal susceptibility to ectopic excitation due to demyelination coexists with a vascular compression. 4 Probably this contributes to DD-related TN being harder to manage pharmacologically and surgically, with lower response rates than classical TN. 5
It becomes, therefore, necessary to consider different treatment for patients with refractory DD-related TN. We report the case of a refractory TN in a patient with a clinically isolated syndrome (CIS) that was treated with motor cortex stimulation (MCS) with a good outcome (3.5 years of follow-up). Informed consent was obtained for the case report.
Case report
A 57-year-old man presented to our neurology outpatient clinic with a 12-year history of paroxysmal and jolt-like episodes of right facial pain (affecting the territories of V2 and V3) associated with face movement, facial touch, and masticatory movements.
His magnetic resonance imaging (MRI) disclosed spinal, infratentorial, juxtacortical, and periventricular hyperintense T2 lesions suggestive of a primary DD. Besides the TN, there were no other clinical episodes suggesting MS (per Polman definition) and the lesions presented no radiological progression up to 5 years from the original MRI (Figure 1(a) to (e)). A diagnosis of CIS was done at the time, and no MS disease-modifying drug was initiated. Complementary immunological testing was negative. No lumbar puncture was performed at the time (No clinical relapses were documented nor imagiological progression. According to the MS criteria used at the time - McDonald 2010 - paraclinical findings as positive cerebrospinal fluid would not change the diagnosis).
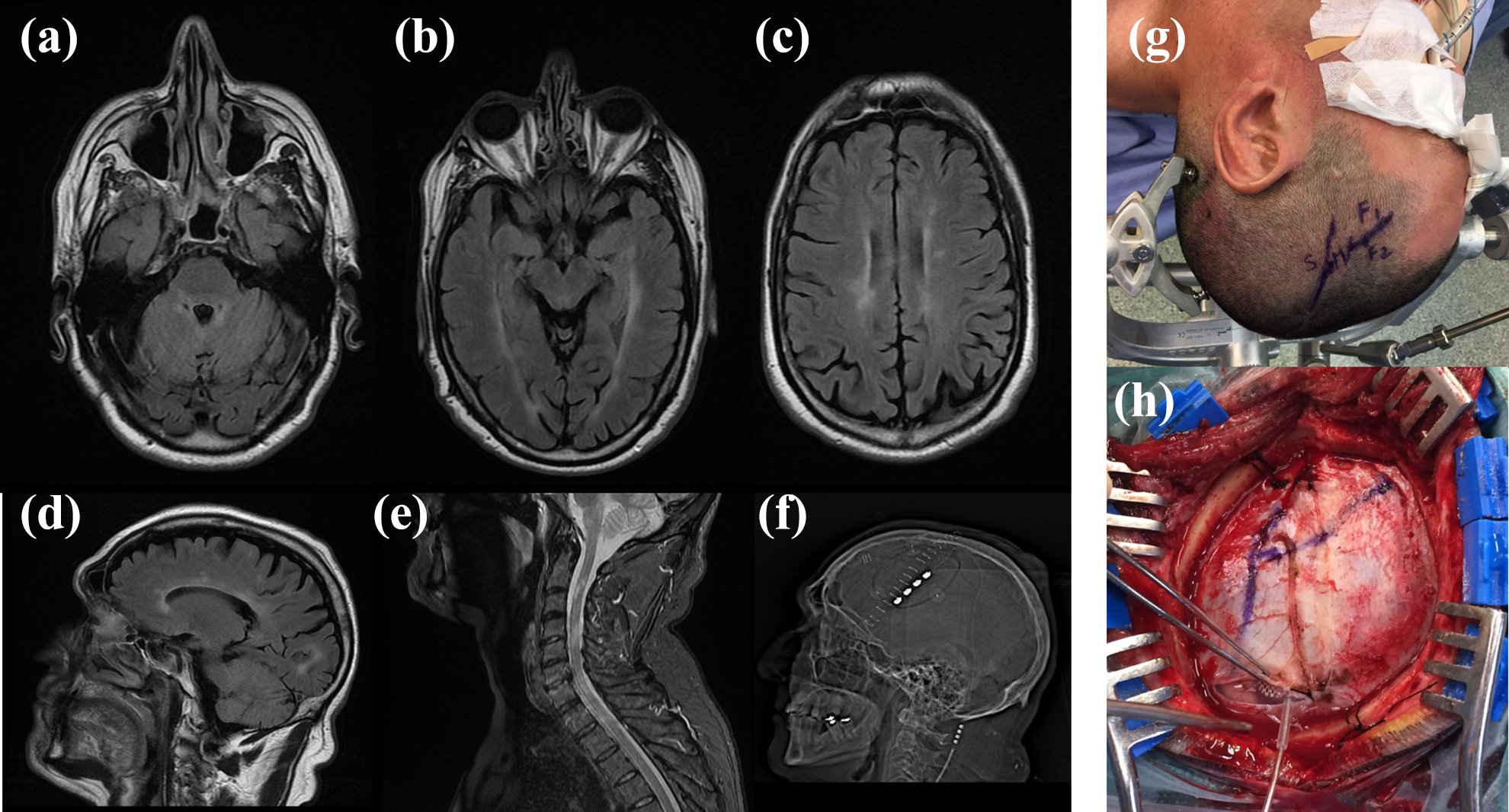
Brain and cervical MRI performed before the first assessment in the neurology clinic. Axial (a)–(c) and sagittal (d) T2 flair sequences disclosing hyperintense lesions in the right middle cerebellar peduncle and pons (ipsilateral to the pain side), juxta-ependymal to the lateral ventricles, and corona radiata typical of MS. No gadolinium enhancement was noted at any time (e) T2-weighted cervical MRI disclosing multiple cervical spinal cord lesions. (f) Scout view of postoperative CT scan indicating the position of four-contact MCS electrode in the patient. (g) Identification of the central sulcus (with the postcentral (S) gyrus posteriorly and the precentral (M) anteriorly) and the frontal inferior sulcus (separating the frontal inferior gyrus from the middle gyrus (F2)). (h) Same identification as previously described but in the dural plane. The electrode was implanted in the subdural space over the precentral gyrus (M) centered in the inferior and middle gyrus). MRI: magnetic resonance imaging; CT: computerized tomography; MCS: motor cortex stimulation; MS: multiple sclerosis.
After original pharmacological treatment with carbamazepine (not tolerated due to side effects), pregabalin, baclofen, and duloxetine, the patient was proposed for a vascular decompression of a neurovascular contact 7 years after symptom onset (Figure 2). There was a slight, but transient, improvement that motivated referral for stereotactic radiosurgery 1 year after the original surgery. Gamma Knife radiosurgery was performed with 85 Gy of maximum dosage. There was an improvement with full remission of paroxysms and complete withdrawal of oral treatment. After this procedure, the patient reported consistent hypesthesia of the right V3 territory. Two years after the radiosurgery, there was a resurgence of painful paroxysms that motivated reinitiation of pregabalin (450 mg/day), duloxetine (60 mg/day), oxcarbazepine (2400 mg/day), and baclofen (40 mg/day). Lamotrigine was avoided as a therapeutic option due to a concomitant diagnosis of psoriasis. At this time, the facial pain was classified, according to Burchiel and colleagues, as a mixed facial pain (symptomatic DD-related TN possibly complicated by a trigeminal deafferentation pain). 6 During a 1-year period, there was no significant reduction on the intensity, frequency, or severity of the episodes, and an MCS procedure was proposed.
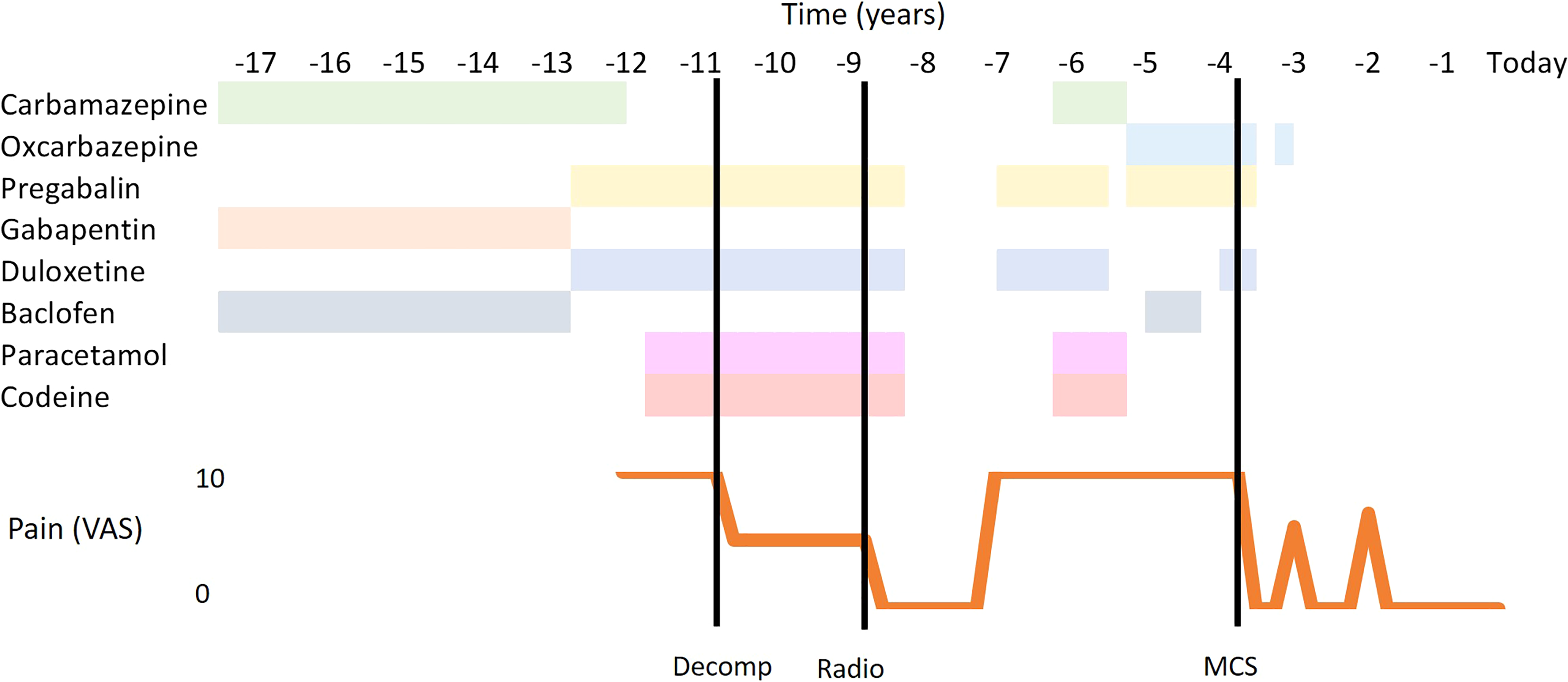
Schema of pharmacological and surgical treatments during the disease timecourse and patient-reported pain intensity. Maximal daily doses of drugs: oxcarbazepine 2400 mg, pregabalin 450 mg, baclofen 40 mg, paracetamol 2000 mg, codeine 60 mg, and duloxetine 60 mg. Carbamazepine unclear in the first period and 800 mg in the second period. Gabapentin is unclear. Decomp: decompression; radio: radiosurgery; MCS: motor cortex stimulation; VAS: visual analog scale.
Left MCS was performed (PrimeAdvance, Medtronic, Minneapolis, Minnesota Figure 1(f) to (h)). The usage of this device was off-label, with a multidisciplinary group oversight. A strip with four electrodes was placed in the subdural space over the motor cortex, parallel to the central sulcus (Figure 1(f) to (h)). Surgery was performed under anesthesia without halogenate agents and without motor block. The precentral gyrus was identified and intraoperatively mapped using neuronavigation (vectorVision BrainLab, Munich, Germany). Regional anatomy of sulci and gyrus confirmed neuronavigation guidance. The final position of the electrode was confirmed with intraoperative electrical stimulation. The electrode grid was used to perform stimulation, and contralateral hemifacial spasm was monitored with orbicularis oralis electromyography (NIM Medtronic, Minneapolis, Minnesota). Stimulation was turned on with electrodes 0, 1, and 2 acting as a cathode (−) and electrode 3 as an anode (+); amplitude of 1.90 V, frequency of 60 Hz, and pulse duration of 210 ms, prompting a complete reduction of TN-related drugs. Symptoms recurred transiently in the first year after surgery, and this was managed with a voltage increase to 2.70 V with complete resolution. The patient was completely weaned off medication 9 months after surgery. Another recurrence of symptoms was noted 2 years after surgery, and this was managed with a voltage increase to 4.00 V and pulse duration increase to 300 ms. No further recurrence of symptoms was noted in the following 1.5 years, and the patient remains without any oral treatment. Dysarthria was present as a side effect but only with amplitudes above the therapeutic ones and with a progressively higher threshold across time.
Discussion
Pharmacological treatment of TN may be unsatisfactory, particularly when associated with DD. 3,5 Medically refractory DD-related TN has been estimated to affect 1.2 person/million/year with surgical failures and retreatment rates reaching up to 90%, with some patients requiring up to 12 surgical procedures. 5 .
Facial pain outcomes for common surgical approaches in the context of a DD are poor when compared to patients with idiopathic TN. 7,8 MCS can be an efficient approach for TN, 8 and we report its effectiveness in a patient with a highly refractory DD-related TN, after failure of previous procedures.
In DD, TN frequently has concomitant lesions besides vascular compression, making microvascular decompression to have a lower probability of effectiveness. 9 DD-related TN has been associated not only with the presence of lesions in the pontine trigeminal afferents but also with the presence of lesions in the contralateral insula (a region involved in pain processing and modulation) and hippocampus. 10 Connectivity- and plasticity-dependent changes may contribute to chronic pain maintenance with mechanisms independent of trigeminal inputs leading to a lower short- and long-term effectiveness of rhizotomy techniques. 7 While other pain management procedures could have been considered (repeat the gamma knife or performing a percutaneous glycerol rhizotomy), we believe that multiple mechanisms were involved in the pain suffered by this patient (peripheral lesion in relation to the history of contact and gamma knife surgery, central demyelination in the trigeminal tract, and upper-level plasticity demonstrated by the presence of allodynia). MCS antinociceptive effect seems to be related to the activation of the descending inhibitory pain pathway 11 —a mechanism that is not explored by most analgesic approaches and that could be a powerful alternative in pains with multiple neuropathic lesions (peripheral/root/supratentorial). Albeit the mechanisms remain unclarified, MCS is a possible approach for TN with evidence of long-term effectiveness. 12 The long-term loss of analgesic effect can be counterbalanced by changes in the stimulation parameters. Identifying good candidates for MCS remains a challenge, and like any other neuromodulation procedure, short- and long-term complications (infection, electrode migration, and loss of effect due to fibrosis) should be considered when this therapy is proposed to the patient. However, based on the possible effectiveness, MCS can be considered for patients with refractory DD-related TN. The development of registries and longitudinal studies of MCS in TN patients can clarify the role of this therapy in TN treatment.
Clinical implications
MCS may be effective in treating refractory DD-related TN.
Adjustment of stimulation parameters may help treating pain relapses.
Footnotes
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Marcelo D Mendonça has received travel fees from Medtronic Portugal.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
