Abstract
Background
Persistent idiopathic facial pain (PIFP) is a chronic disorder recurring daily for more than two hours per day over more than three months, in the absence of clinical neurological deficit. PIFP is the current terminology for Atypical Facial Pain and is characterized by daily or near daily pain that is initially confined but may subsequently spread. Pain cannot be attributed to any pathological process, although traumatic neuropathic mechanisms are suspected. When present intraorally, PIFP has been termed ‘Atypical Odontalgia’, and this entity is discussed in a separate article in this special issue. PIFP is often a difficult but important differential diagnosis among chronic facial pain syndromes.
Aim
To summarize current knowledge on diagnostic criteria, differential diagnosis, pathophysiology and management of PIFP.
Methods
We present a narrative review reporting current literature and personal experience. Additionally, we discuss and differentiate the common differential diagnoses associated with PIFP including traumatic trigeminal neuropathies, regional myofascial pain, atypical neurovascular pains and atypical trigeminal neuropathic pains.
Results and conclusion
The underlying pathophysiology in PIFP is still enigmatic, however neuropathic mechanisms may be relevant. PIFP needs interdisciplinary collaboration to rule out and manage secondary causes, psychiatric comorbidities and other facial pain syndromes, particularly trigeminal neuralgia. Burden of disease and psychiatric comorbidity screening is recommended at an early stage of disease, and should be addressed in the management plan. Future research is needed to establish clear diagnostic criteria and treatment strategies based on clinical findings and individual pathophysiology.
Definition
The International Classification of Headache Disorders (ICHD, version 3) published by the International Headache Society (IHS) describes persistent idiopathic facial pain (PIFP) as ‘persistent facial and/or oral pain, with varying presentations but recurring daily for more than 2 hours per day over more than 3 months, in the absence of clinical neurological deficit’ (1). PIFP is the current diagnostic terminology that historically would have been considered under the name of atypical facial pain (AFP).
Diagnostic criteria for Persistent Idiopathic Facial Pain (Section 13.12). With permission of the International Headache Society.
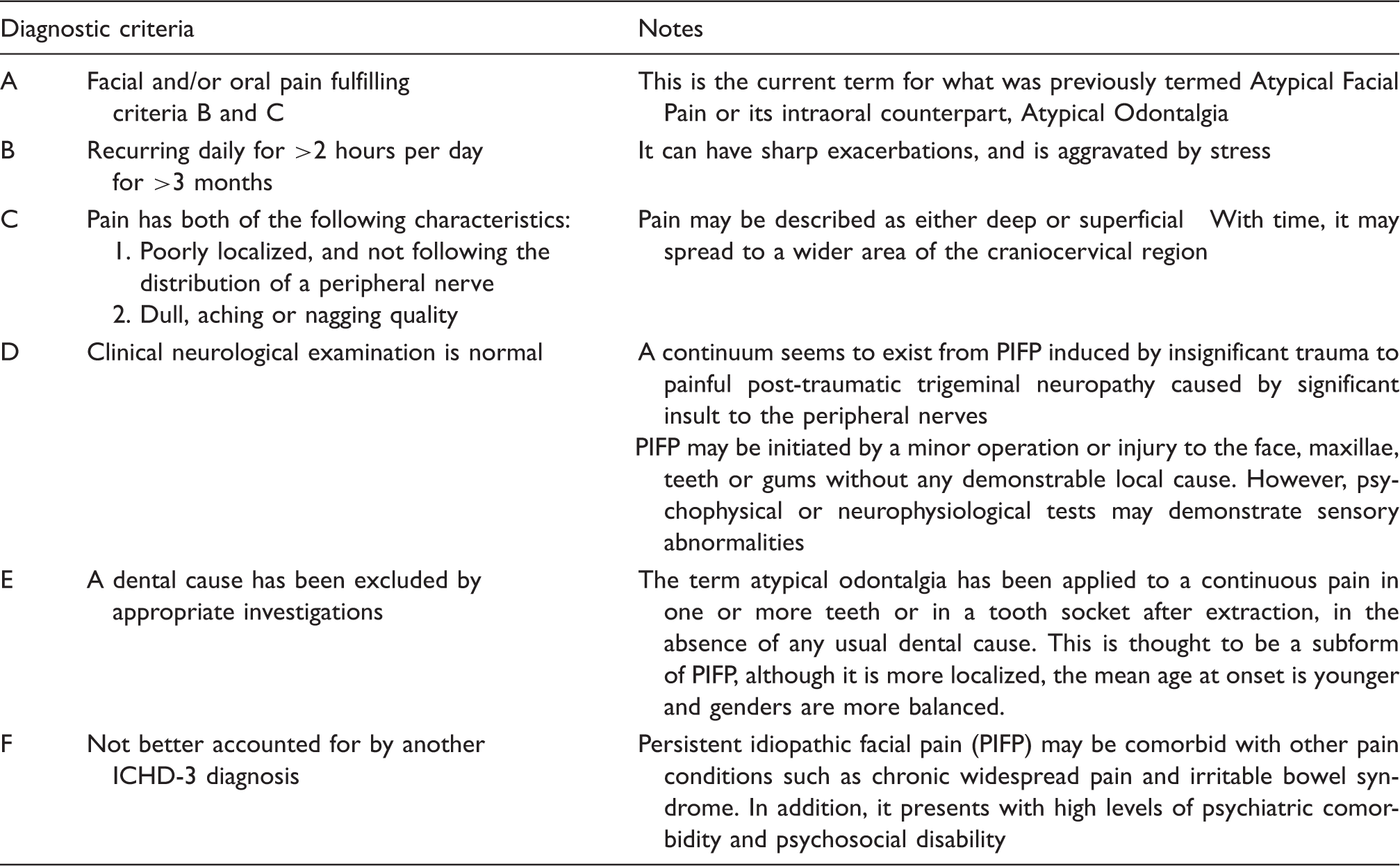
Intraorally, PIFP (or AFP) has been approached under the name of atypical odontalgia (AO). According to the ICHD-3 (1), AO is included as a subtype of PIFP and not classified as a separate headache disorder. In a similar fashion to PIFP, most researchers have concluded that AO is a neuropathic syndrome and it has been widely referred to as ‘phantom toothache’ (3–5). With recent developments in the classification of traumatic neuropathy due to dental treatment or other regional injuries, the term AO should not be used. This is discussed in the paper by Baad-Hansen and Benoliel in this issue.
Taking all the above together, the differential diagnosis of PIFP and the diagnostic work up, as discussed below, are often the most challenging processes in these patients.
Historical perspective
As stated, PIFP is the current term replacing the historical AFP. The term AFP was originally described as the atypical counterpart to trigeminal neuralgia (6). Since then, the statement by Frazier and Russel “when consulted by a patient with neuralgia of the face, the first question is: Has the patient major trigeminal neuralgia, or has he not?”, is still one of the important points to consider while taking the pain history. Regarding the differential diagnosis of PIFP, this would be pertinent if the presenting pain has an accompanying persistent, continuous quality to it (6).
AFP continued to be useful as a diagnosis for many years vis-à-vis the two historically major orofacial and head pain diagnoses; mainly trigeminal neuralgia but also migraine. Everything else was regarded as ‘atypical’ (7). The inherent contradictions of diagnostic criteria with a name that included ‘atypical’ and the possible dangers associated with this were recognized (8). This was followed by the interesting concept of a ‘typical’ AFP (8–10) that rapidly progressed to the understanding that essentially this disorder was ‘facial pain of unknown origin’ (11), and thus established the basis for the introduction of a change in terminology from AFP to PIFP.
Because PIFP is essentially employed when no other diagnosis is feasible it has tended to include a heterogeneous group of patients. For example, some PIFP patients responded partially to triptans, suggesting neurovascular mechanisms, whilst in another study they were ineffective (12,13). It seems, therefore, that past studies have probably included a heterogenous group of patients that may be unrelated.
This confusion in study homogeneity and the tendency to employ PIFP as a diagnosis of exclusion have greatly hampered our understanding. Despite this, the clinical reality of this group of patients remains, and we are therefore obliged to continue our endeavors to elucidate the pathophysiology, outline a clear clinical phenotype and establish evidence-based treatment recommendations.
Symptomatology
Pain onset in PIFP is often associated with minor surgical or other invasive dental or otolaryngologic procedures (14); these may be reported as the initiating event or as as an attempt to manage the pain (7,15). However, many patients cannot reliably recall the sequence of events. Is their pain the result of treatment or was pain present before treatment was initiated and subsequently worsened?
Although there should be no clinically evident neurosensory deficits in PIFP (1), hypoesthesia has been reported in studies using quantitative sensory testing (QST) (16–18). Patients with a neuropathic type pain following surgery or other trauma with neurosensory changes should be diagnosed as painful traumatic trigeminal neuropathy (PTTN), as defined by the IHS (1,19). These changes in inclusion criteria, both in classification systems and in published cases and case series, must be taken into account when interpreting the data on the clinical profile and indeed management protocols, see for example (16–18,20,21).
Pain in PIFP is usually deep but can be superficial as well (1). It is poorly localized, radiating and mostly unilateral, although up to 40% of cases may describe bilateral pain (20). PIFP is commonly described as aching, burning, throbbing and often stabbing (1,16–18,20–22). Severity, usually mild to severe (rated 7 on an 11-point VAS), may be aggravated by emotional stress (1,20). PIFP patients with severe pain often demonstrate a disparity between their apparent calm emotional and physical state and the reported pain severity. Most PIFP patients report persistent, long lasting (years) daily pain (20) that tends to spread, in a non dermatomal pattern, with time (1). Typically, pain characteristics, location and associated features change over time. Rarely, some PIFP patients report pain free or remission periods (20). Often PIFP may coexist with other chronic orofacial pain or headache syndromes (20).
Psychiatric and psychosocial disability have often been associated with PIFP (23), although one study on 14 PIFP patients found no significant comorbid psychiatric disorders compared to controls (22). Increased scores for anxiety and depression are common, especially in PIFP patients reporting higher pain intensity (24), indicating that a psychiatric screening should be performed (25). Systematic screening revealed that 41.3% of patients suffering from PIFP or burning mouth syndrome (BMS) had an axis I disorder (major depression was the most frequent) before the onset of orofacial pain. Therefore, the authors conclude “that psychiatric morbidity, and comorbidity to other chronic pain conditions, in chronic idiopathic orofacial pain can be best understood in terms of shared vulnerability to both chronic pain and specific psychiatric disorders” (23). We conclude that an interdisciplinary approach is needed for the diagnosis and management of PIFP (26).
Epidemiology
On the basis of PIFP’s rather loose criteria, there is difficulty in interpreting epidemiologic data. However, based on existing studies, the estimated lifetime prevalence of PIFP is around 0.03% (27) and the incidence rate is 4.4 per 100,000 person years (28). These data clearly indicate that that PIFP is a rare disorder.
In orofacial pain clinics, PIFP may account for around 10–21% of the patient population (29,30). In a neurological tertiary care centre that studied patients referred for facial pain, about 21–27% had persistent idiopathic facial pain (31), while only 3% of patients with side-locked unilateral headache and facial pain presenting to a neurology outpatient clinic were diagnosed with PIFP (32). Most patients seen are female and mean age of onset is in the mid 40s (20).
Pathophysiology
Several studies have now established that there is no evidence for a pathophysiologic role for neurovascular compression (NVC) of the trigeminal dorsal root entry zone in PIFP (20,33,34).
The large number of PIFP patients presenting with a history of mild trauma and subclinical sensory changes has led to the suggestion that PIFP and PTTN may represent extremes of a spectrum of clinical presentations. As such, PIFP would therefore be considered a neuropathic pain syndrome. In support, studies of PIFP patients reveal increased neuronal excitability at the brainstem level (18,22,35), disturbed inhibitory function of the prefrontal cortex (36), and alterations in the dopamine systems associated with either/both pain transmission and its modulation (37). Additionally, sensory changes consistent with a neuropathy or neuropathic pain have been shown employing QST in patients with PIFP (16–18). The data seem to indicate that PIFP is indeed a neuropathic pain syndrome. Forssell et al. (18) highlighted the need for detailed neurophysiologic and quantitative sensory examinations as a consequence of their study comparing neurophysiological findings in PIFP and trigeminal neuropathic pain. They were the initial proponents that PIFP might be a heterogeneous entity representing one extreme of a continuum that ranges from definitive neuropathic pain syndromes to idiopathic pains with an unclear ‘neuropathic’ involvement (18).
However, unchanged somatotopy of the somatosensory cortex (by magnetoencephalography) and inconsistent changes in the blink reflex in a group of PIFP patients indicate no significant alterations in the trigeminal somatosensory pathways, suggesting that PIFP may not always be a neuropathic pain syndrome (22). In these same patients, the QST profile, other than thresholds for warm and heat pain, was not significantly different to that in controls. These findings, if duplicated, suggest we may have subtypes of PIFP; neuropathic and “other”.
Further thoughts on the pathophysiology of PIFP
The history of a varied clinical result to injuries in the trigeminal region, ranging from mild to severe, and resulting in clinical phenotypes ranging from PIFP to PTTN, is reminiscent of Complex Regional Pain Syndromes (CRPSs).
CRPS is a chronic, painful neuropathic disorder that develops as a disproportionate consequence to injury (38) and is distinguished by significant autonomic, trophic and motor changes. CRPS is characteristically observed in the extremities but rarely in the trigeminal region, possibly due to differences in the structure of the autonomic nervous system and the reaction of the trigeminal nerve to injury (see article by Baad-Hansen and Benoliel in this special issue).
Two main subtypes have been defined: CRPS I (previously reflex sympathetic dystrophy) and CRPS II (previously causalgia) (39). Both these entities present with spontaneous pain accompanied by allodynia and hyperalgesia not limited to dermatomal regions (40), and by other motor and autonomic phenomena (41). CRPS I may develop as a consequence of remote or relatively minor local trauma with a minor or unidentifiable nerve lesion, and would be comparable to PIFP. The most common causes are surgery, fractures, crush injuries and sprains (42). Even injections, local infection and burns have been implicated (43). Some believe, however, that it is the subsequent surgical attempts at treatment of the injury that may contribute more to establishing pain than the original injury (43,44). Again, this is reminiscent of the typical story of some PIFP patients.
The less frequent form, CRPS II, is characterized by a substantiated injury to a major nerve, most often following high velocity missile trauma or surgery and therefore similar to PTTN. It is often difficult to distinguish clinically between CRPS I and II, as is sometimes the case for PIFP and PTTN. Moreover, small fiber pathology has been shown in CRPS-I (45), so that some have questioned the need to subdivide the CRPSs (43). Eventually we may also view PIFP and PTTN as related syndromes on different ends of a clinical spectrum resulting from nerve injury.
Following a separate line of thought, the clinical presentation of PIFP is often difficult to distinguish from a chronic myofascial pain or chronic tension type headache (CTTH) with no pericranial muscle tenderness. Could some PIFP patients be an atypical type of CTTH with no pericranial muscle tenderness, and share pathophysiologic elements?
Clearly, the pathophysiology of PIFP remains elusive. However, at this point in time, based on available data, one may postulate that PIFP may involve a disproportionate response to mild injury. This would therefore incriminate established mechanisms in typical traumatic neuropathy and other factors, as in CRPS.
Diagnostic considerations
PIFP is often diagnosed when the clinician has exhausted all possible alternatives that are within his knowledge base. Depending on the clinician’s knowledge and experience, this may include a number of less recognized regional pain syndromes. Therefore, we suspect that in the past, some of these less recognized regional pain syndromes were misdiagnosed as PIFP, see Figure 1. As knowledge and diagnostic skills accumulated, many PIFP patients were diagnosed as having chronic myofascial pain, pre or atypical trigeminal neuralgia, orofacial/facial migraine or atypical trigeminal autonomic cephalgias (TACs). This continued improvement in diagnosis will aid in establishing a homogenous PIFP diagnosis that may be studied. Table 2 summarizes the salient features across the entities discussed below.
Misdiagnosed persistent idiopathic facial pain (PIFP) as a result of not recognizing atypical or rare orofacial pain syndromes such as atypical neurovascular pains, regional myofascial pain and rarer or other neuropathic pain syndromes. Salient demographic, clinical and therapeutic characteristics in persistent idiopathic facial pain and entities in its differential diagnosis. Key: PIFP: persistent idiopathic facial pain; L-CH: lower cluster headache; OF-migraine: orofacial migraine i.e. atypically located in the lower face; PTTN: painful post traumatic trigeminal neuropathy; TN: trigeminal neuralgia; RMP: regional myofascial pain; *: onset age differs between sexes; ?: unknown or unclear data; +: mild, ++: moderate, +++: severe; I, II, III: first, second or third trigeminal dermatomes; m: minutes, h: hours, d: days, w: weeks, m: months, yrs: years; Mult: multiple attacks; B: burning; Th: throbbing; St: stabbing; E: electrical; Par: paroxysmal; Preg: pregnancy; Men: menopause; REM: rapid eye movement; NS: neurosensory; -: none; #: allodynia associated with migraine; Systemic signs: nausea, vomiting, photo/phonophobia; Menst: menstruation; **: usually allodynic area but triggering may occur; Tr: triptan, AED: antiepileptic drugs; Ver: verapamil; TCAs: tricyclic antidepressant; SNRI: serotonin noradrenaline reuptake inhibitors antidepressant; CBZ: carbamazepine; Mod: moderate

Although rare, “PIFP-like” syndromes may be associated with significant underlying pathology (46,47), and the clinician must exclude these by thorough clinical examination, follow up and imaging if there are indications. There is a need for a multidisciplinary diagnostic approach to exclude relevant differential diagnoses. Usually the treating specialist (neurologist or orofacial pain) needs a definitive statement from the treating dentist that clinical examination and dental radiography have excluded dental pathology. Depending on location, similar statements may be required from otolaryngologists.
In the following sections, we briefly review some of the more common disorders that may be confused with PIFP
Traumatic trigeminal neuropathies
In general, injuries to the trigeminal nerve largely result in either no residual neurological deficit or in a non-painful neuropathy. A minority, ranging from 3–5%, develops a painful neuropathy and it is suspected that sex, previous pain experiences, deficiencies in pain modulation and genetics are involved in this predisposition (48–51). The initiating factors include macrotrauma, as in altercations and motor vehicle accidents (52). Iatrogenic injuries to nerves may result from facial, otolaryngological or neuro-surgery (53,54). Dental interventions (extractions, root canal therapy, implants) are common invasive procedures that may pose the risk of neuropathy secondary to direct or indirect neuronal trauma (55–58). Injury from dental nerve blocks has also been implicated in PTTN (59–61).
Pain is unilateral and may be precisely located to the dermatome of the affected nerve with demonstrable sensory dysfunction, particularly if a major nerve branch has been injured. These are major differentiators as compared to PIFP. Over time, PTTN pain may however become diffuse and spread across dermatomes. Pain intensity is moderate to severe (VAS 5–8) and quality is usually burning or shooting (62–65), as is typical of a neuropathic pain syndrome. Cases may report excruciating, spreading and distant pain on light touch; a triggering-like mechanism, but these are relatively rare and have no latency or refractory period as in trigeminal neuralgia (19,65). More often there is clinically severe allodynia, hyperalgesia or negative neurosensory signs (19,63,66), which should be absent in PIFP. Hyperalgesia and other sensory changes may be found in extratrigeminal sites of PTTN patients, suggesting more extensive changes in central somatosensory processing (67–69). Pain is continuous, lasting most of the day, and present on most days of any month. Patients may complain of a feeling of swelling, foreign body, hot or cold, local redness or flushing (19,65). Both gross clinical and advanced neurophysiologic techniques may be used to detect, quantify and monitor sensory deficits or changes (54,70–74).
Patients are characterized by a history of multiple treatment modalities aimed at eliminating pain, often including pharmacotherapy, dental adjustments and a variety of oral, otolaryngological, and neurosurgical procedures (63).
Regional myofascial pain (RMP)
Patients with RMP complain of a deep, dull pain felt diffusely throughout the face and temples (29,75–77), clearly overlapping with the PIFP phenotype. Pain is usually unilateral but may be bilateral in 30–40% of cases (29,78). Patients may report dysfunction associated with chewing foods and a limited range of mandibular movements (29,79).
Characteristically the pericranial, masticatory and cervical muscles are painful to moderate manual pressure and some may display the classical “trigger point” phenomenon in response to muscle manipulation (77,80). This phenomenon includes referral of pain to distant sites such as the eye, jaws and teeth (81,82). These findings should be absent in PIFP patients.
Atypical neurovascular pains
Migraines and TACs are classically located around the ocular and frontal regions (1,83). They present with usually classical symptomatology, making their diagnosis straightforward. However, cases of isolated oral and facial pain with neurovascular features have been reported with features suggesting that these are “facial” or “orofacial migraines” (29,84–88). In cases in which the patient has a history of migraines, the term “relocated migraines” has been used. However, while facial pain is not unusual in migraine (8.9%), isolated facial migraine is exceptionally rare (0.2%) (89). Notwithstanding, these atypical presentations have caused extensive misdiagnosis with dental (as above) and maxillary sinus (90–96) pathology leading to misguided interventions.
Similarly, ‘lower’, ‘orofacial’ and ‘upper’ subtypes of cluster headache (CH) have been reported (97–100). Pain in ‘lower CH’ is ocular, temporal, and suboccipital with radiation to the teeth, jaws and neck (97,98). Intra/perioral radiation of pain includes the jaws (37%), teeth (maxillary: 50%, mandibular: 32%) and the cheeks (45%). This atypical presentation and referral pattern is also present in other trigeminal autonomic cephalgias, similarly causing diagnostic difficulties (101–106). However, it is now recognized that pain may occur throughout the trigeminal system (107), including the mid and lower face, so that in its chronic forms short-lasting unilateral neuralgiform headache attacks (SUNHA) may cause confusion for the clinician.
Therapeutic responses to triptan or indomethacin treatments as indicated, and to recommended prophylactic treatments may confirm the diagnosis of those migraines and TACs presenting in more “atypical” locations. Furthermore, frequency and duration patterns are different from most cases of PIFP. Together these should aid in diagnosis.
Atypical neuropathic pain
Two presentations of trigeminal neuralgia (TN) may cause diagnostic difficulties: TN with concomitant persistent background pain (108,109) (previously termed atypical) and a rare form called pre-TN (110). Between 35–49% of CTN (classical trigeminal neuralgia) patients describe two types of pain: paroxysmal attacks of short sharp pain and a dull background pain of varying duration. Background pain may be described as dull, throbbing and burning, and is of varying intensity with a mean VAS of 4.6 (111,112). It is this persistent pain that may cause diagnostic difficulty with PIFP.
An early form of TN termed “pre-trigeminal neuralgia” (PreTN) has been reported in 18% of CTN patients, and is characterized by a dull continuous pain in the jaws that lasts from days to years (110) with no particularly striking clinical characteristics. Certainly, this is a strong candidate for potential misdiagnosis with PIFP. However, as the PreTN process continues, the pain becomes more typical with characteristic flashes of pain. Thermal stimuli may cause triggering at a relatively higher rate, and a throbbing quality to PreTN pain is sometimes present. PreTN is however highly responsive to anticonvulsant therapy. The lack of clear and consistent diagnostic criteria makes this a problematic entity to recognize; it is usually diagnosed when all other possibilities are exhausted or in retrospect once CTN develops.
A common pathway to a difficult diagnosis
The above entities are indeed relatively unknown and, in the past, may have been mistaken for AFP or PIFP. However, at times these entities are being misdiagnosed as pain due to dental or otolaryngologic pathology. These patients subsequently undergo invasive treatments aimed at managing their pain. The result in some of these is a painful post-traumatic trigeminal neuropathy (PTTN) induced by the same interventions aimed at relieving this pain. As discussed there seems to be a continuum between PIFP and PTTN (see Table 3), so a subset of these patients may develop PIFP. In these misdiagnosed and mistreated patients, we may be faced with two comorbid clinical phenotypes: the original and still undiagnosed pain, and either PIFP or PTTN, see Figure 2 and Table 3. In our experience, this complex combination is often subsequently diagnosed as PIFP.
Persistent idiopathic facial pain (PIFP) established as a result of misdiagnosed orofacial pain entities (e.g. atypical neurovascular pains, regional myofascial pain and rarer or other neuropathic pain syndromes) that undergo invasive dental, otolaryngologic or neurosurgical interventions. In this scenario, PIFP may be the result of neuropathic mechanisms and on one end of a spectrum, with definitive painful traumatic trigeminal neuropathy at the other. From persistent idiopathic facial pain to painful post-traumatic trigeminal neuropathy: Continuum of diagnosis and pathophysiology.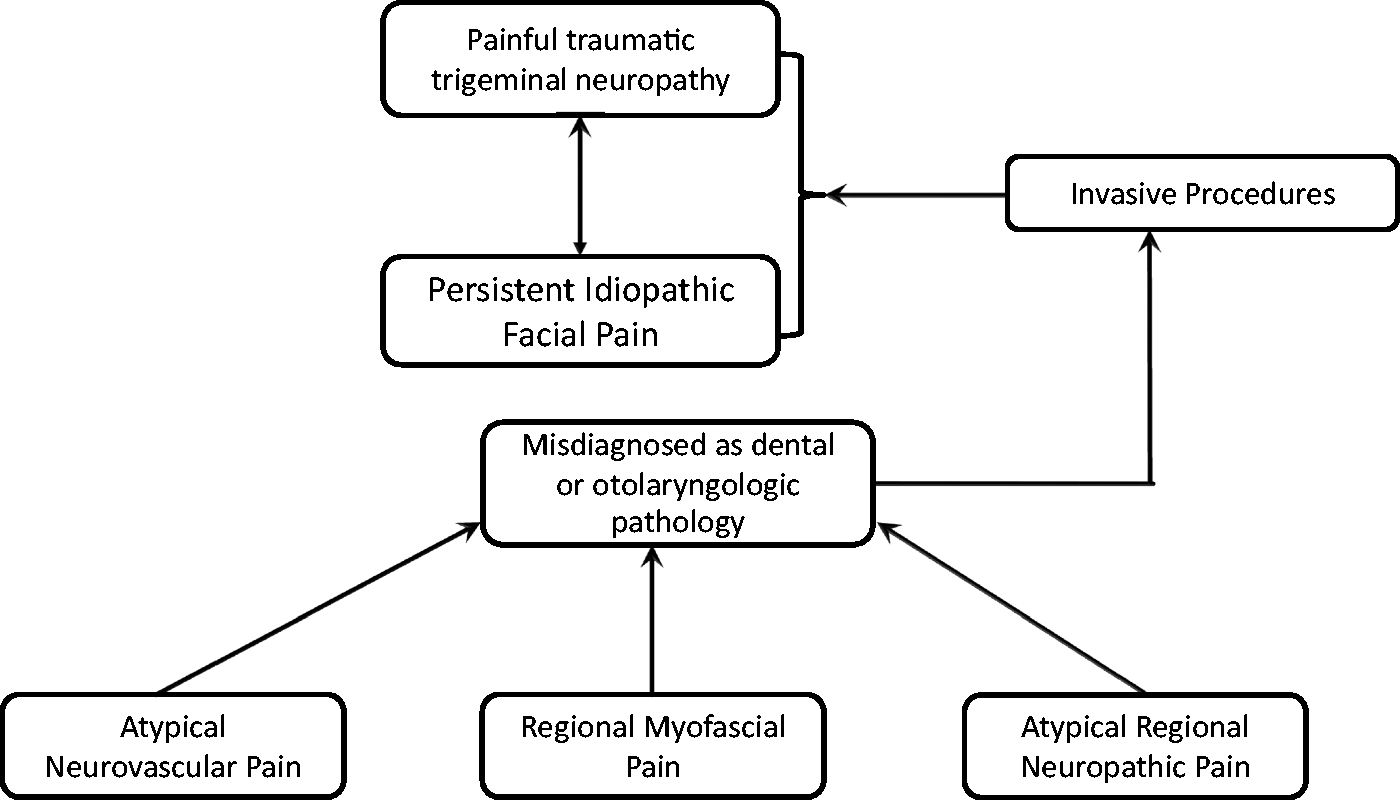

Treatment
The lack of a clear pathophysiological basis precludes the establishment of a treatment protocol. The approach to the management of PIFP patients should consider patients’ beliefs on pain and the consequences of the pain disorder on their personal lives (113). Considering the psychiatric comorbidity, chronic course of disease in many patients, and the lack of drug treatment RCTs, a multidisciplinary approach encompassing the comorbidities is suggested, comparable to treatment concepts in other chronic headaches (114). Considering the chronicity and resulting distress, behavioral interventions are indicated. Accumulating evidence suggesting that PIFP may be a type of painful neuropathy underlies the preferential use of medications known to have an effect in painful neuropathies, i.e. antidepressants and antiepileptic drugs. Patient education is needed to clarify the diagnosis, and certainly the patient should be discouraged from any further invasive interventions aimed at pain relief in the absence of clear associated pathology.
Therapeutic trials of PIFP (and AFP) have been reported as efficient, but the trials are not all randomized or controlled. Case series using tricyclic antidepressants (115), an open study on duloxetine (116), a randomized controlled trial on venlafaxine (117), and open studies on anticonvulsants (118,119) and low level laser (120) have all shown beneficial effects, but other than one study the level of evidence is clearly low. Similarly, based on the premise that PIFP is neuropathic, high-frequency repetitive transcranial magnetic stimulation (rTMS) on the right secondary somatosensory (S2) cortex in patients with neuropathic orofacial pain induced significant pain relief compared to stimulation of the sensorimotor (S1/M1) cortex and sham stimulation (121). The right S2 cortex is therefore a promising new target for the treatment of neuropathic orofacial pain and possibly PIFP using high-frequency rTMS.
Some authors claim success with treatments aimed at the regional musculoskeletal system, but no convincing evidence is available. Occipital nerve blocks have not been efficient in PIFP patients (122). Often the clinician is at a loss when all accepted treatments have failed.
There are no contraindications to non-interventional novel therapies (e.g. based on virtual reality (123)) or complementary and alternative medicine (124), and these may be beneficial. Hypnosis might be a promising approach for therapy (125). However, the evidence for psychosocial interventions is limited, due to the lack of controlled studies (126).When all treatment fails, some have suggested pulsed radiofrequency treatment of the sphenopalatine ganglion (127), but this is based on an open trial in a small number of patients and there is no high level evidence for this or any other neurosurgical type of intervention (128,129). We do not recommend invasive procedures, as these always carry the risk for inducing a traumatic neuropathy and therefore may end up increasing pain.
Available evidence for any of the treatments is limited, and randomized clinical trials are missing. We therefore recommend a conservative, multidisciplinary approach based on experiences with comparable chronic headache disorders including medications, relaxation training, psychological interventions and physiotherapy (114).
Expert opinion: Open questions and burning desires
It seems imperative to prospectively collect patients with features matching the PIFP criteria as published by the IHS. Certainly, some leeway should be allowed so as to closely examine “atypical” cases of PIFP. This may enlighten us as to the range of presentations that may be consistent with a diagnosis of PIFP. Based on these studies we should attempt to establish modified criteria, striving to make these positive inclusion criteria rather than a “diagnosis of exclusion”.
As has happened with many of the head and facial pain disorders, subsets of PIFP may be identified in this manner, and with beta-criteria they could be better studied and phenotyped.
Once this complex task is completed, there is no doubt we need functional imaging, QST and other neurophysiological tests in these in clinically well phenotyped PIFPs. At the same time, we will then be able to test management protocols and examine outcomes based on clinical parameters, imaging, QST and clinical neurophysiologic data.
Footnotes
Article highlights
PIFP is often a relatively featureless headache that may mimic other facial pain syndromes.
Often, PIFP seems to be initiated by minor trauma, suggesting a shared pathophysiology and a clinical spectrum with painful traumatic neuropathies.
Carefully interdisciplinary collaboration is needed to establish the diagnosis and management of persistent idiopathic facial pain (PIFP).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
