Abstract
Background:
The otic ganglion (OG) is a cranial parasympathetic ganglion located in the infratemporal fossa under the foramen ovale (FO) and adjacent to the medial part of the mandibular nerve. Parasympathetic innervation of intracranial vessels from the OG has been shown both in animal and human models and evidence suggests that the OG plays an important role in the cranial vasomotor response. We review the evidence that positions the OG as a viable target for headache disorders. The OG is a small structure and not detectable on medical imaging. The FO is easily identifiable on CT scans and the mandibular nerve on MRI, hence, the position of the OG may be predicted if the mean distance from the FO is known.
Objective:
The objective is to describe the average distance between the FO and the OG in a sample of 18 infratemporal fossae from 21 cadavers.
Methods:
A total of 21 high definition photographs of 21 infratemporal fossae from 18 cadavers were analyzed. The distance between the inferior edge of the medial part of the FO to the OG was measured.
Results:
Four photographs of infratemporal fossae of four cadavers were excluded due to the inability to localize the inferior edge of the FO. A total of 15 infratemporal fossae from 17 cadavers were measured. The mean distance from the FO to the OG was 4.5 mm (SD 1.7), range 2.1–7.7 mm.
Conclusions:
We have described the average distance from the OG to an easily identifiable anatomical landmark that is visible in CT scans, the FO. This anatomical study may aid in the development of strategies to localize the OG in order to explore its role as a therapeutic target for headache disorders.
Introduction
Patients with trigeminal autonomic cephalalgias (TACs) and other headache disorders often experience cranial autonomic symptoms. Cluster headache is a prototypical example and patients often experience symptoms, such as miosis, conjunctival injection, ptosis, eyelid edema, epiphora, nasal congestion, and rhinorrhea. 1 Cranial autonomic symptoms in migraine are also common and have most likely been underestimated. 2 For instance, Riesco et al. found in a series of 100 patients with chronic migraine lacrimation in 49%, conjunctival injection in 44%, eyelid edema in 39% and nasal congestion in 20%. 3 Autonomic symptoms can be unilateral in 26.9% of patients with migraine. 2
The sphenopalatine ganglion (SPG) is thought to be involved in the pathophysiology of TACs and other headache disorders, including migraine. 4 The SPG has been a target for the treatment of primary headache disorders for more than a century. 5 It receives its preganglionic parasympathetic fibers via the vidian nerve and the postganglionic fibers travel with the trigeminal nerve branches to innervate the mucous membrane of the nose, palate, tonsils, uvula, pharynx, lacrimal gland, and meningeal vessels. 4 Different approaches and several drugs have been used to block the SPG in a broad range of conditions. 4,6 –8 A positive feedback loop from the trigeminocervical complex to the dural blood vessels involving the SPG has been described. 9
In addition to the SPG, there are three other major parasympathetic ganglia in the cranium: the ciliary ganglion, the OG, and the submandibular ganglion. 10 The OG has received little attention from clinicians, and thus far, there have not been any therapeutic attempts at targeting this structure for the treatment of primary or secondary headache disorders. 10 Frey’s syndrome may be the only known clinical entity related to the OG. The OG is a small structure (about 4 mm long, 3 mm wide, and 1.5 mm thick) located in the infratemporal fossa 11 (Figure 1). Its location and relationship to adjacent structures in humans have been described by Senger et al. 10 (Figure 2). In the inferior salivatory nucleus, preganglionic parasympathetic fibers exit the brain stem and travel through the glossopharyngeal nerve, the tympanic nerve, and the lesser petrosal nerve to reach the OG. The external sphenoidal nerve exits the OG with postganglionic fibers projecting toward the trigeminal ganglion and ganglia of the cavernous sinus. 10 This parasympathetic innervation of intracranial vessels from the OG has been shown in different animal models 12 –14 and in humans. 15,16 In addition, the OG has been shown to be involved in the cranial vasomotor response. 17

Location of the otic ganglion in the left infratemporal fossa. The OG is situated directly medial to the mandibular nerve under the foramen ovale. OG: otic ganglion.

Illustration of the roots and branches of the otic ganglion, with permission from Senger et al. 10
The role of the cranial parasympathetic system in primary headache disorders positions the OG as an interesting and potentially viable therapeutic target for the treatment of TACs and other headache disorders. To develop therapeutic strategies targeting the OG, it is important to understand its relationship with other structures that are easy to identify, such as the foramen ovale (FO), which can be easily localized on routine CT head scans. We have measured the distance from the FO to the OG in a series of photographs of anatomic preparations. This distance, not previously described in the literature, might be helpful for developing navigation-based therapies toward the OG.
Methods
A total of 21 high-definition photographs of 21 infratemporal fossae from 18 cadavers were analyzed. The distance between the inferior edge of the medial part of the FO to the OG was measured.
An anatomical study using the same 21 halves of 18 human cadaveric heads was published by Senger et al. 10 In this study, the topography, syntopy, and morphology of the OG were described, though the distance from the FO to the OG was not documented.
The anatomical preparations had been used by students in the practical dissection course 2012/2013 at the Anatomical Institute of the University of Cologne (Germany). 10 All heads had been fixed in 10% aqueous formalin solution and sectioned in the midsagittal plane. The samples were not specified according to gender or age of the donor. The study by Senger et al. received authorization of the Ethics Committee of the Faculty of Medicine of the University of Cologne. 10 The cadaveric preparations were no longer available for analyses since they had been inhumed. For this reason, high-resolution photographs were used instead. These photographs were taken with a Nikon D50 and an Olympus DP21.
The approach used by Senger et al. to expose the OG in the preparations was by removing all structures that covered the OG from the medial side of the halved heads. 10 The structures removed from medial to lateral to reach the OG were the torus tubarius, the salpingopharyngeal muscle, the levator veli palatine, the tensor veli palatine, and the medial pterygoid muscle. 10
To measure the distance from the FO to the OG on the photographs, we used free downloadable software for Mac OS, RulerSwift Version 1.0. All photographs had a reference scale situated in the same plane of measurement (Figure 3(a)). The distance was measured from the inferior aspect of the FO in its central part to the center of the OG. All measurements were performed by two researchers (JC and DB) and the mean results of the measurements are given.

(a) Photograph showing an anatomic preparation of the left OG. (b) An enlargement of the same preparation with the measured distance between the inferior aspect of the FO and the OG (0.59 mm in this preparation). FO: foramen ovale; OG: otic ganglion.
Statistical analysis
Stata/MP 15.1 for Mac (64-bit Intel, Copyright 1985-2017 StataCorp LLC) was used in the data analyses. Results are given as mean ± standard deviation if not otherwise stated.
Ethics approval and consent to participate/consent for publication
This study used photographs of anatomical preparations obtained by Senger et al. Authorization for the use of these photographs was granted by the Ethics Committee of the Faculty of Medicine of the University of Cologne.
Results
Four photographs of infratemporal fossae of four cadavers were excluded due to the inability to perform the measurements (in these four cases, it was not possible to localize the inferior part of the FO by JC and DB).
A total of 15 infratemporal fossae from 17 cadavers were measured. The mean distance from the FO to the OG is presented in Table 1.
Results of the measurements between the inferior aspect of the foramen ovale and the otic ganglion.

FO: foramen ovale; OG: otic ganglion.
Figure 3(b) shows an example of the measured distance between the FO and the OG.
Discussion
The OG’s location and relationship to adjacent structures appear to be constant 10 (Figure 2). It is situated directly medial to the mandibular nerve (slightly ventral in some cases). 10 To develop therapeutic strategies targeting the OG, it is important to understand its relationship with other structures easy to identify in clinical practice. The mandibular nerve is easy to localize on MRI. 18 The relationship between the OG and an anatomical landmark, which would allow locating precisely the OG along the mandibular nerve and which is readily identifiable in clinical practice, has not been previously reported. In this study, we have seen that the OG is located directly caudally from the FO with an average distance of 4.5 mm. The FO is easy to localize in CT scans. The combination of these two anatomical landmarks (the mandibular nerve and the FO) might be of help when trying to predict the location of the OG.
A positive feedback “loop” from the trigeminocervical complex to the dural blood vessels has been described. 9 The efferent limb of the cranial parasympathetic system is activated either via a reflex arc from the trigeminal nucleus caudalis (from activated trigeminal nociceptors) or via descending modulatory influences from supraspinal and supratentorial structures, notably the hypothalamus. 19 This could lead to efferent activity, the release of vasoactive and inflammatory peptides at the level of the cranial vasculature and dura as well as the mucosal structures in the face. 19 This then, in turn, activates trigeminal afferents. A block of the SPG, therefore, affects efferent outflow and activation of the trigeminal sensory system peripherally (Figure 4).
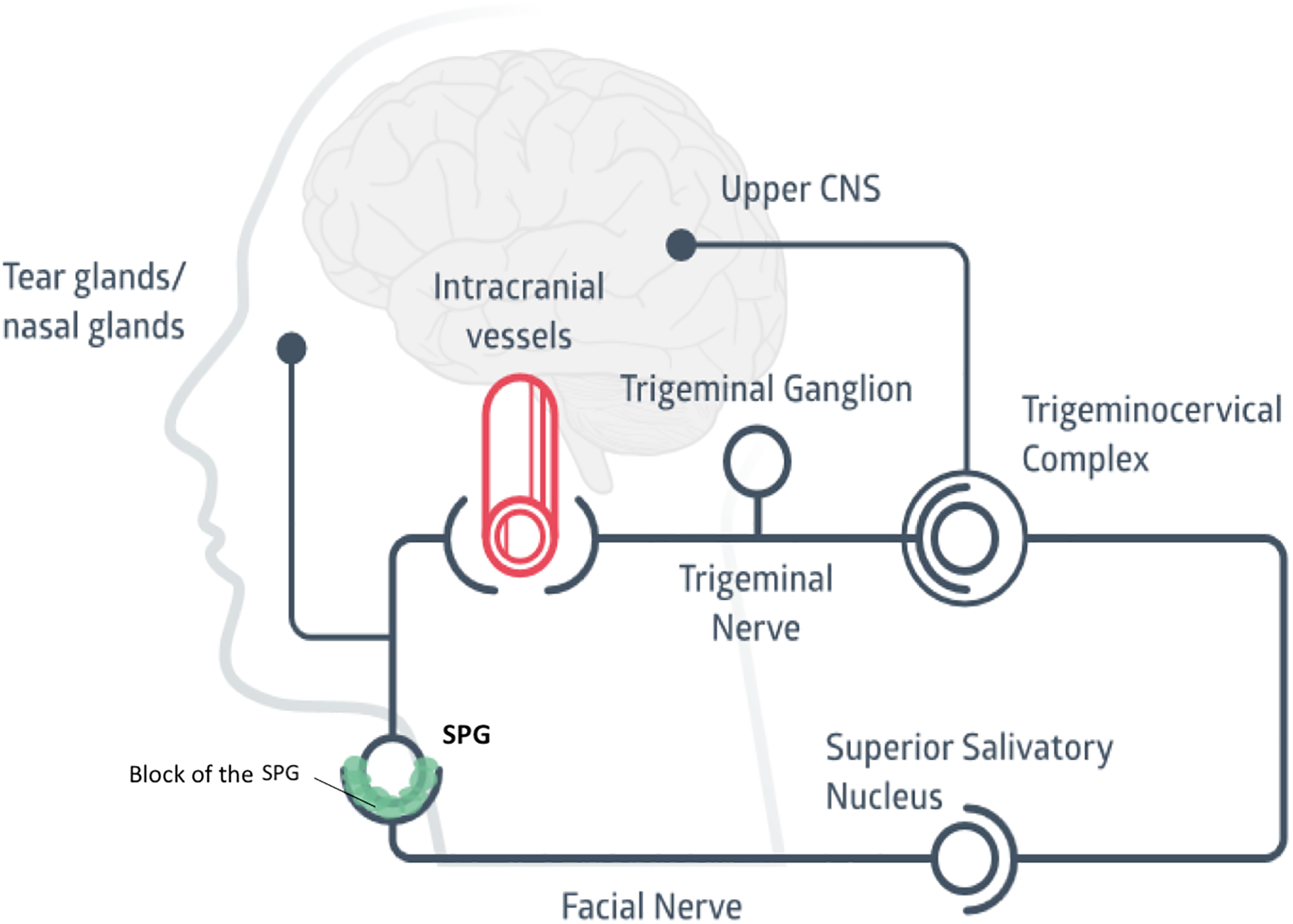
Illustration showing the assumed mechanism underlying how a block of the SPG works. SPG: sphenopalatine ganglion.
The preganglionic parasympathetic fibers of the OG originate in the inferior salivatory nucleus and exit the brain stem via the glossopharyngeal nerve, then join the tympanic nerve, then the lesser petrosal nerve and synapse in the OG. The postganglionic fibers that are best documented are those exiting the ganglion toward the parotid gland through the auriculotemporal nerve. However, it is important to notice that other fibers leave the ganglion via the external sphenoidal nerve (in the literature also called dorsal rami, ganglionic cord, internal sphenoidal nerve, and rami communicantes cum sinus cavernosus). 10 These fibers reach the trigeminal ganglion and ganglia of the cavernous sinus. This parasympathetic innervation of intracranial vessels from the OG has been shown in different animal models 12 –14 and in humans. 15,16 Nociceptive fibers come in very close contact with parasympathetic and sympathetic fibers in the cavernous sinus. 20 This proximity of cranial parasympathetic fibers and trigeminal nociceptive fibers is relevant. 19 It has been documented that efferent fibers that innervate meningeal blood vessels and dura mater release neuropeptides, which directly and indirectly through inflammatory cascades activate nociceptive fibers and result in cephalic pain. 19 The cavernous sinus has been proposed to have a central role in cluster headache pathophysiology, 21 and cluster headache-like attacks have been reported in patients with lesions compressing or affecting the cavernous sinus. 22,23 In addition, it has been documented that roughly 50% of the cranial vasomotor response in cats is mediated by the OG and the other 50% by the SPG 17 (Figure 5).

This illustration intents to summarize the findings by Goadsby et al., where it was observed that approximately 50% of the cranial vasomotor response in cats is mediated by the otic ganglion and the other 50% by the sphenopalatine ganglion. 17
Considering the work described above, we believe that there is evidence, both from an anatomical and a physiological point of view, that the OG might play a role in the pathophysiology of TACs and other headache disorders. We hypothesize that the loop described previously (between the trigeminocervical complex and dural vessels, see Figure 4) may be more complex than previously thought. The efferent part of this loop, in addition to the projections from the SPG, may also involve another efferent pathway: fibers from the inferior salivatory nucleus, which project to the OG via the glossopharyngeal nerve (Figure 6). John et al. 24 removed the SPG (histologically verified) in 13 patients with cluster headache, 24 but with no or only modest clinical effect: 7 patients had no effect, 4 had incomplete relief and only 2 had complete relief over the next 12 months. One may speculate that parasympathetic efferent signaling through the OG may be sufficient in some patients to maintain the above-mentioned positive feedback system to activate trigeminal nociceptive afferents through the same mechanism as SPG efferents. This may explain the incomplete response even when the SPG is blocked, radiated (gamma knife) or resected.

Illustration showing a proposed model of how the otic ganglion might be involved in the generation of trigeminoautonomic headaches.
Despite the work described above, the SPG remains the only parasympathetic cranial ganglion targeted in headache. This might be due to anatomical differences, better-documented localization, and a general impression that the SPG is easier to target for interventions. 8
Limitations of the study
The main limitation of the study is that the measurements were not done in vivo but in cadavers and the measured distance may have changed due to postmortem desiccation or during the anatomic preparation.
The measurements of the distances between the FO and the OG were performed on photographs and not on the cadavers since they had been inhumed. These photographs might have a different angle on the trajectory of the mandibular nerve and this might affect the measurements of the distance between the FO and the OG. Four samples were excluded since we could not be sure that we had identified properly the inferior aspect of the FO in order to try to decrease the risk of error in our measurements.
Another limitation is that all cadavers were Caucasians and the measurements in other ethnicities may be different.
Conclusions
We have described the average distance from the OG to an easily identifiable anatomical landmark on CT-scans, the FO. Cluster headache is one of the most severe pains described in the medical literature with a big burden for patients suffering from it. 25 Migraine is the first cause of disability in people under 50 years of age. 26 Patients suffering from TACs and other headache disorders are in need of new and better treatments. The identification of new targets is pivotal for the development of new treatments for these patients. The OG may become a future target in headache disorders. This anatomical study might be of help when trying to develop strategies targeting the OG. The topography of the OG in living human beings has not been described. Further anatomoradiological studies might be necessary in order to increase the efficacy, reliability, and safety of therapies targeting the OG.
Future research is needed to establish the role of OG in headache disorders. Studies in animal models to determine the possible function of the OG in the pathophysiology of headache are warranted. The feasibility and safety of a block of the OG have to be assessed in future studies.
Clinical implications
The OG might become a new target in headache disorders. The OG appears to have a constant location, being situated 4.5 mm inferior of the FO and medial to the mandibular nerve. The FO is easily localized on CT scans and may be an interesting anatomical landmark when trying to develop navigation-based therapies towards the OG.
Footnotes
Authors’ contributions
JC, DB, and ET had the original idea for the manuscript. JC and DB analyzed the data. JC reviewed the literature for the introduction and drafted the manuscript. DB, DD, MM, and ET assisted for drafting the manuscript and revision of the text. MS and DA revised the manuscript. All authors read and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by a grant given by NTNU (Norwegian University of Science and Technology) and “The Liaison Committee for Education, Research and Innovation in Central Norway” (Samarbeidsorganet); grant number 46056923.
