Abstract
The subarachnoidal cerebral blood vessels of the rat are innervated by nerve fibers containing different neuropeptides, e.g. pituitary adenylatecyclase activating polypeptide (PACAP). PACAP dilates brain arterioles and immunohistochemical studies of the rat have indicated that PACAP binds to a VPAC1-receptor in the cerebral vasculature of this species. We have investigated the perikaryal origin of the nerve fibers innervating the subarachnoidal blood vessels of the rat by combined retrograde tracing with Fluorogold and immunohistochemistry. The in vivo neuronal retrograde tracings were done by injection of 2% Fluorogold in water into the subarachnoidal space in the area of the middle cerebral artery. The retrograde transported tracer was detected by use of an antibody against Fluorogold. One week after the injections, the animals were vascularly perfused with Stephanini's fixative and labeled perikarya were found bilaterally in the trigeminal, sphenopalatine, and otic ganglia. The retrograde Fluorogold tracings were combined with immunohistochemistry for PACAP using a mouse monoclonal antibody and the biotinylated tyramide amplification system. Double labeled perikarya containing both Fluoro-gold and PACAP were found predominantly in the trigeminal ganglion, and only rarely in the otic and sphenopalatine ganglion. Summarizing, our retrograde tracings combined with immunohistochemistry indicate that the perikarya in the trigeminal ganglion are the main origin of PACAPergic nerve fibers projecting to the cerebral vasculature of the rat.
Different extracerebral sympathetic and parasympathetic ganglia innervate the subarachnoidal blood vessels of the brain. Thus, sympathetic nerve fibers with origin in the superior cervical ganglion and the stellate ganglion (Handa et al., 1990) project along the carotid arteries to the intracranial brain vasculature. Parasympathetic fibers reach the blood vessels from the sphenopalatine (in man called pterygopalatine), otic, and internal carotid artery ganglia (Suzuki et al., 1988, 1989; Edvinsson et al., 1989; Hara et al., 1989, 1993; Shimizu, 1994). In addition to the classical sympathetic and parasympathetic nerve fibers, intracranial blood vessels are also innervated by sensory nerve fibers with perikaryal origin in the trigeminal ganglion, and the first and second cervical spinal ganglion (Arbab et al., 1986; Suzuki et al., 1989). Surgical denervation of the trigeminal ganglion results in the loss of substance P and CGRP-positive nerve fibers in the cerebral circulation as studied by immunohistochemistry and quantitative RIA (Uddman et al., 1985).
The nerve fibers projecting to the cerebral vasculature contain a large variety of neurotransmitters. The adrenergic sympathetic fibers contain, in addition to norepinephrine, neuropeptide Y (NPY) (Gulbenkian et al., 2001; Zhang et al., 1991). The parasympathetic cholinergic nerve fibers contain vasointestinal polypeptide (VIP)–Fahrenkrug et al., 2000), and nitric oxide (NO)–(Edvinsson et al., 2001).
In the sensory nerve fibers originating from the trigeminal ganglion substance P, neurokinin A, calcitonin gene-related peptide (CGRP), cholecystokinin (CCK), somatostatin, dynorphin, NO (Edvinsson et al., 1988; Hardebo et al., 1994; Gulbenkian et al., 2001), and galanin (Suzuki et al., 1989; Lazarov, 2002) are present.
Pituitary adenylate cyclase-activating polypeptide (PACAP) is a neuropeptide belonging to the secretin, glucagon, and VIP-family (Arimura, 1998) and occurs in two molecular forms, PACAP-38 and the C-terminally truncated PACAP-27. PACAP together with VIP mediates its effects through 3 G-protein coupled receptors, PAC1, VPAC1 and VPAC2 (Harmar et al., 1998). PACAP dilates cerebral blood vessels in a concentration dependent manner (Seki et al., 1995; Uddman et al., 1993; Huang et al., 1993).
Nerve fibers containing PACAP have been demonstrated in the cerebral arteries of several species, e.g. rat (Kausz et al., 1998; Edvinsson et al., 2001) and cat (Uddman et al., 1993). By use of immunohistochemistry, the VPAC1 receptor has recently been demonstrated in the cerebral blood vessels of the rat (Fahrenkrug et al., 2000).
PACAP-immunoreactive perikarya expressing mRNA encoding PACAP are present in peripheral parasympathetic ganglia, but also in sensory ganglia, such as the dorsal root ganglion and the trigeminal ganglion (Sundler et al., 1996; Nielsen et al., 1998; Tajti et al., 1999).
The origin of the PACAP-containing nerve fibers of the subarachnoidal blood vessels has never been determined by use of the retrograde tracer Fluorogold. Therefore, in this study, we have mapped the location of the PACAP-immunoreactive perikarya projecting to the blood vessels of the brain. By combining in-vivo retrograde Fluorogold tracing with immunohistochemistry, we have traced the majority of the PACAPergic nerve fibers back to the trigeminal ganglion.
MATERIALS AND METHODS
Animals
Adult male Wistar rats, weighing 250–300 gr, were used in this investigation. The animals were kept in a 12:12 h light: dark cycle (light on at 6 a.m.) with food and water ad libitum.
All experiments were conducted in accord with the guidelines of European Communities Council Directive of November 1986 (86/609/EEC) and were approved by the Animal Experimentation Inspectorate of the Danish Ministry of Justice.
Injection of tracer
Sixteen rats were anesthetized during the light period with tribromethanol (50 mg/100g body weight, i.p.) and placed in a Kopf stereotaxic apparatus. An incision was made in the mid-line on the dorsal part of the skull, and the periost removed. A small circular bone piece (about 5 mm in diameter), to the right or left of the sagittal suture corresponding to the area supplied by the middle cerebral artery, was removed by using a small drill to expose the subarachnoidal space with its vessels on the surface of the brain. The dura mater was incised to gain access to the subarachnoidal space around the middle cerebral artery. A Hamilton syringe containing a solution of 2% Fluorogold in water was inserted into the subarachnoidal space and 1–3 μl of the solution were injected into the subarachnoidal space around the brain vessels. The Hamilton syringe was left in situ for another 10 minutes to avoid leaking of the tracer from the subarachnoidal space. The animals were allowed to survive for 7–10 days after the injection.
Perfusion fixation of animals
The animals were re-anesthetized with tribromethanol as described above and vascularly perfused through the heart with ice cold heparinized (15,000 IU/l) phosphate buffered saline (pH 7.4, PBS) for 2 min followed by Stephanini's fixative (2% paraformaldehyde, 0.2% picric acid in 0.1 M sodium phosphate buffer, pH 7.2) for 10 min. The brains together with the trigeminal ganglia, sphenopalatine, and otic ganglia were removed and postfixed in the same fixative for 2 days and afterwards transferred to PBS. The brains and ganglia were then cryoprotected for 1–2 days in 20% sucrose, frozen in crushed dry ice, sectioned in a cryostat at a thickness of 18 μm and collected on gelatinized glass slides.
Immunohistochemistry
Single immunohistochemistry.
The sections were processed for immunohistochemistry by using a well-characterized mouse monoclonal anti-PACAP antibody (code no. JH6f/10) (Hannibal et al., 1995). The monoclonal PACAP antibody displays equal affinity for PACAP-38 and PACAP-27 recognizing an epitope between amino acid 6-16 of PACAP, but showed no affinity for VIP or other structurally related peptides (Hannibal et al., 1995).
The sections were washed 3 × 5 min in PBS and then incubated in 5% normal swine serum in PBS with 1% BSA and 0.3% Triton X-100 (PBS-T). This was followed by incubation for 12-18 hours in the monoclonal mouse anti-PACAP serum (supernatant diluted 1:5) at 4°C. Afterwards, the sections were washed 3 × 10 min in 0.25% bovine serum albumin + 0.1% Triton X-100 (PBS-BT) followed by incubation for 60 min at room temperature in biotinylated rabbit anti-mouse IgG diluted 1:500 in PBS-BT. The biotinylated antiserum was further intensified using the principles described by Berghorn et al. (1994) in the following way: Firstly, the sections were washed 3 × 5 min in PBS-BT and incubated for 30 min at room temperature in ABC-streptavidin horseradish peroxidase complex (Vectastain, Vector, Burlingame; USA) diluted 1:100. After washing 3 × 5 min in PBS-BT, the sections were incubated in biotinylated tyramide (TSA indirect; NEN Life Science Products) diluted 1:100 in PBS + 0.005% H2O2, then washed 3 × 5 min in PBS-T (PBS with 0.1% Triton X-100) and incubated in ABC-streptavidin horseradish peroxidase complex (diluted 1:100) for 30 min. After washing in 2 × 5 min in PBS-T and 10 min in 0.05 M TRIS (pH 7.6) the sections were incubated for peroxidase activity in a solution of 0.05% diaminobenzidine 7.6) (DAB) and 0.01% H2O2 in 0.05 M Tris/HCl buffer (pH = for 15 min, and the reaction was terminated by washing the sections in excessive amounts of distilled water. Finally, the sections were mounted on gelatinized slides, dried and embedded in Depex®.
Double immunohistochemistry for Fluorogold and PACAP.
Firstly, the sections were washed in phosphate buffered saline (PBS) for 2–3 min. The sections were then incubated in 5% normal swine serum in PBS with 1% BSA and 0.3% Triton X-100 (PBS-T). This was followed by incubation in the primary antibodies, rabbit anti-Fluorogold 1/500–1/1,000 (code nr. AB153, Chemicon, Temecula, CA) and mouse monoclonal anti-PACAP diluted 1/5 in PBT-BT over night at 4°C. The sections were then washed in washing buffer (PBS with 0.25% BSA and 0.1% Triton X-100) for 3 × 10 min and incubated with biotinylated swine anti-mouse IgG (Dako, Copenhagen, code.nr.E464) diluted 1/600 in PBS-BT. After 2 × 10 min wash in the washing buffer, the sections were reacted with streptavidin-ABC in PBS-T for 45 min. After 3 × 5 min washing with PBS with 0.1% Triton X-100, the sections were incubated with biotinylated tyramide (TSA indirect; NEN Life Science Products) diluted 1:50 in PBS containing 0.005% H2O2 for 10 min followed by incubation in steptavidin-FITC and donkey anti-rabbit IgG labeled with Alexa Fluor 568 (Molecular Probes, Leiden, The Netherlands). After washing in PBS 3×5 min, the slides were dipped in distilled water and cover slipped in Aquamount®.
Fluorescence microscopy and filter combinations
The double fluorescent labeled sections were analyzed and photographed in a Zeiss Axioplan microscope equipped with and epifluorescence system and different Zeiss filter combinations. For viewing FITC, a band pass (BP) filter (transmitting between 450–490 nm) and a long pass emission filter (LP) 515 were used (Zeiss Filter set #09). For visualization of Alexa 568 a BP filter (maximum transmission around 530 nm) and the BP emission filter 615 were used (Zeiss filter set #00). For visualization of the double stained neurons with yellow fluorescence the Zeiss filter set #24 was used. In this set the excitation filter is a double BP filter with two excitation maxima (485 and 578 nm) and the emission filter site is a combined BP transmission filter (515–540 nm) and a LP filter (610 nm).
Quantification of the density of PACAP-immunoreactive neurons in ganglia: the number of PACAP-immunoreactive neurons and the total number of neurons were counted in four rats (Fig. 3). The counting was performed with a fluorescence microscope in sections where PACAP was visualized with bio-tinylated tyramide amplification and streptavidin-FITC. Four sections of each rat were used for counting.
RESULTS
Morphology of the trigeminal, sphenopalatine, and otic ganglia
The trigeminal ganglion of the rat is an elongated structure. The mandibular part of the nerve leaves the ganglion (Fig. 1A) before it terminates rostrally as the ophthalmic and maxillary nerves.
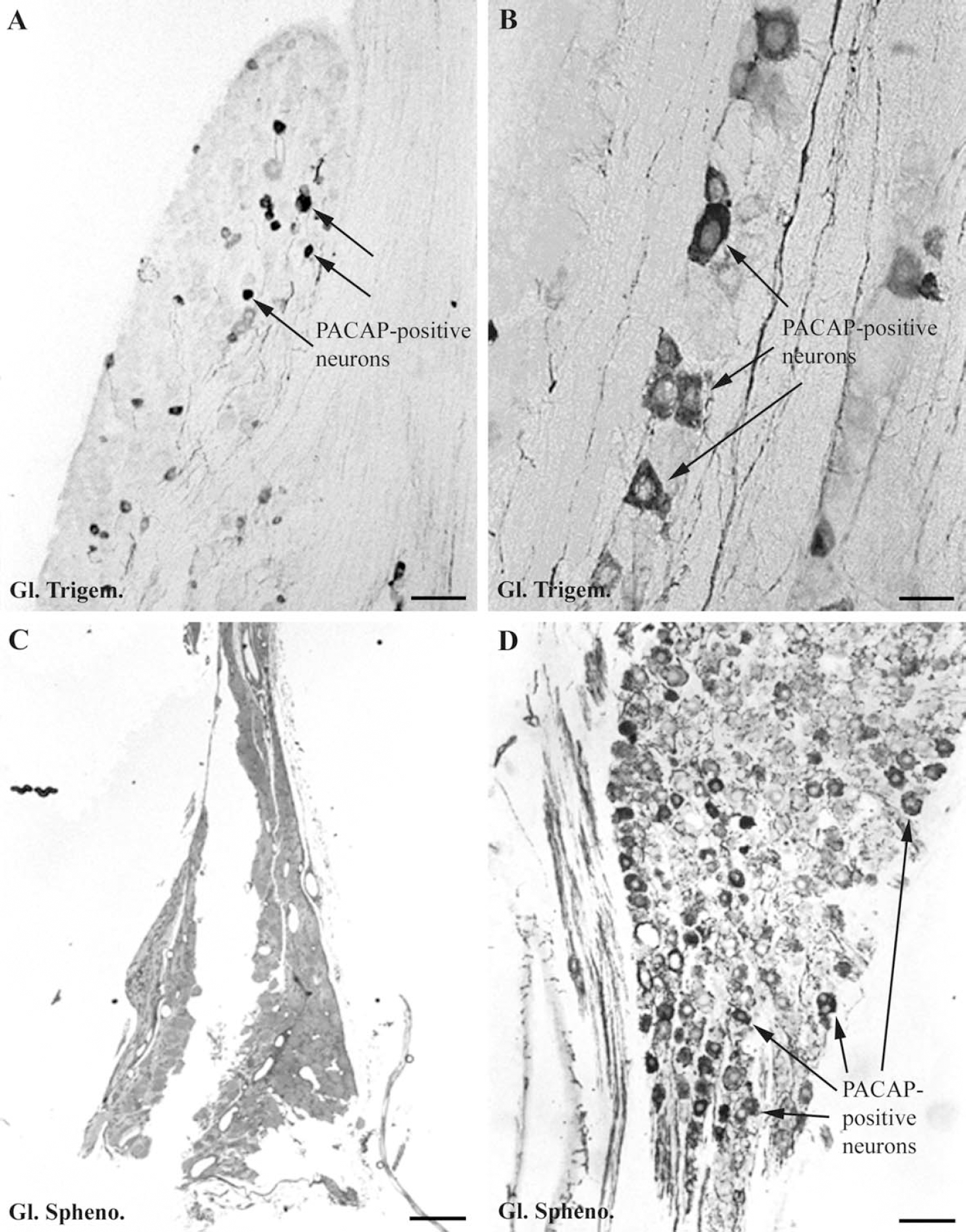
The sphenopalatine ganglion is located in the spheno-palatine fossa of the orbit between the medial surface of the maxillary nerve and the nasal orbital wall. The ganglion (Fig. 1C) is elongated in rostrocaudal direction and endowed with a rostral (anterior) and caudal (posterior) triangular swelling connected by a thinner middle part. The caudal part of the ganglion is connected to the Vidian nerve and via thin nerve fibers also to a fine nerve plexus of the sinus cavernosus.
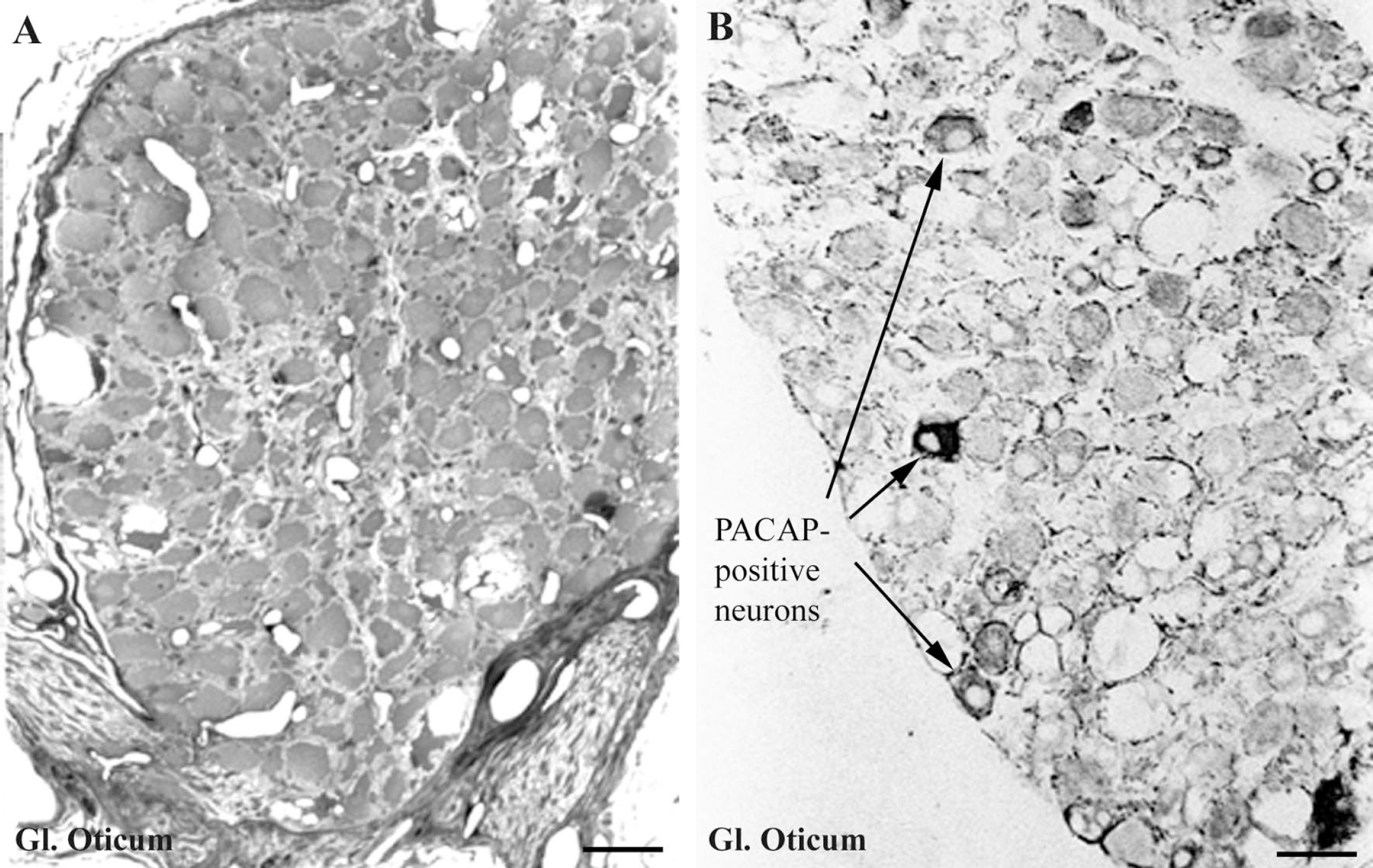
The otic ganglion is a small oval or triangular structure (Fig. 2A) located just beneath the sphenoid bone, close to the foramen ovale. Two or three thin nerve fibers connect the ganglion with the mandibular nerve.
Immunohistochemistry for PACAP
A high number of perikarya and nerve fibers immunoreactive for PACAP were present in the trigeminal ganglion (Figs. 1A and 1B). In both the sphenopalatine (Fig. 1D) and otic ganglion (Fig. 2B), the number of PACAP-immunoreactive perikarya was lower than in the trigeminal ganglion, although the number of immunoreactive nerve fibers was fairly high. The labeled perikarya (Figs. 1A, 1B, 1D, and Fig. 2B) were about 20–30 μm in diameter, and in the trigeminal ganglion the cell bodies could not be distinguished morphological from the classical pseudounipolar sensory perikarya present in the ganglion.
Most of the immunoreactive nerve fibers were endowed with large (2 μm in diameter) axonal swellings. In all 3 ganglia, PACAP-immunoreactive nerve fibers were observed to surround many non-immunoreactive cell bodies (Fig. 2B).
Estimation of the number of PACAP-immunoreactive neurons in the trigeminal, sphenopalatine, and otic ganglia
As seen in Fig. 3, the density of PACAP-immuno-reactive neurons varied from the lowest, about 4.8%, in the otic ganglion to about 10% in the trigeminal ganglion.
Combined retrograde tracings with Fluorogold and PACAP immunohistochemistry
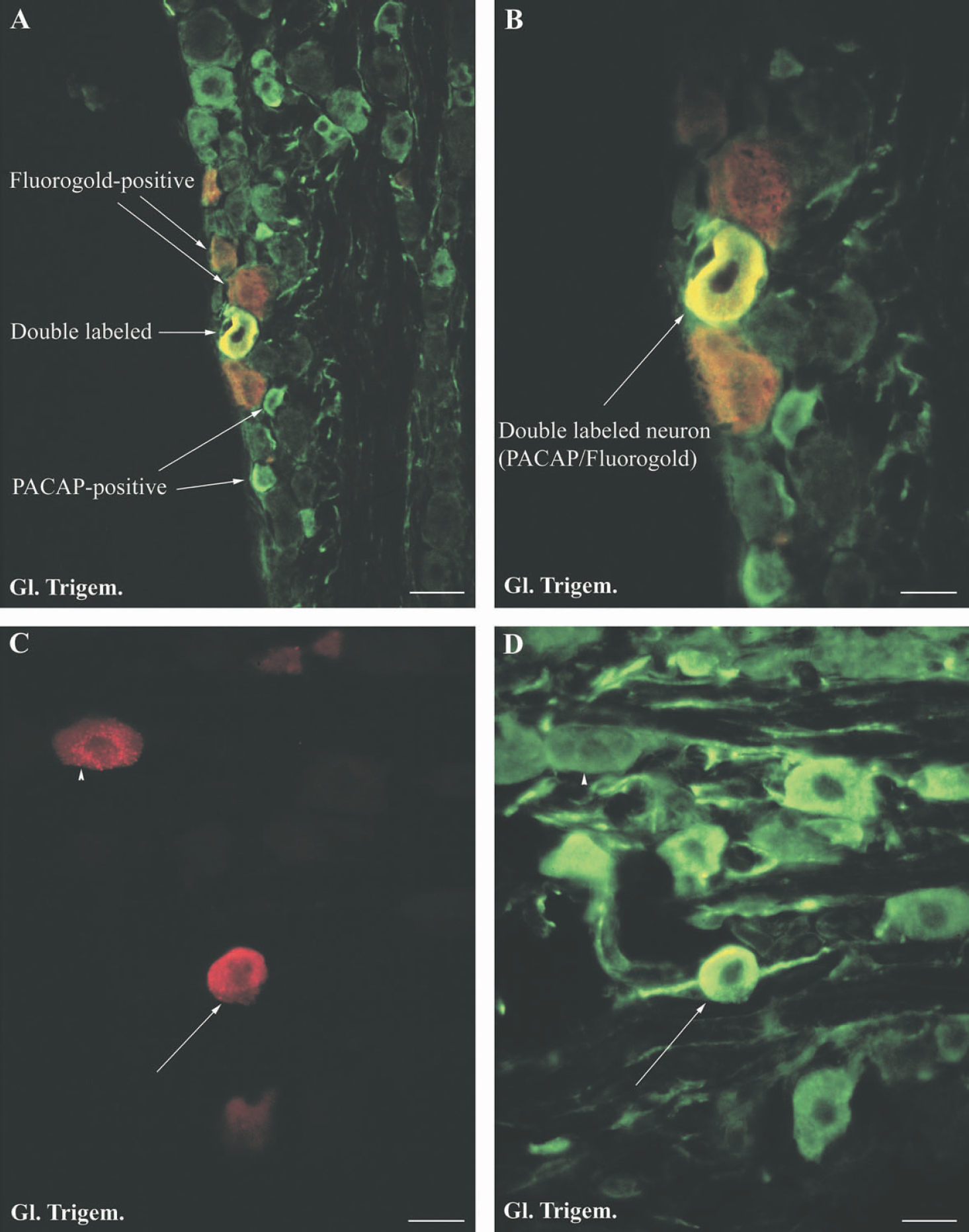
After injection of Fluorgold into the subarachnoidal space, retrograde labeled perikarya were observed in both the trigeminal, sphenopalatine, and otic ganglia. However, the number of retrogradely labeled neurons was higher in the trigeminal ganglion compared to the number in the sphenopalatine and especially the otic ganglion.
When immunohistochemistry for PACAP was used in combination with retrograde tracing with Fluorogold (Fig. 4), the majority of double-labeled cell bodies were observed in the trigeminal ganglion (Figs. 4B, 4C, and 4D). Few double-labeled perikarya were observed in the sphenopalatine ganglia and in our study no double-labeled perikarya were seen in the otic ganglion.
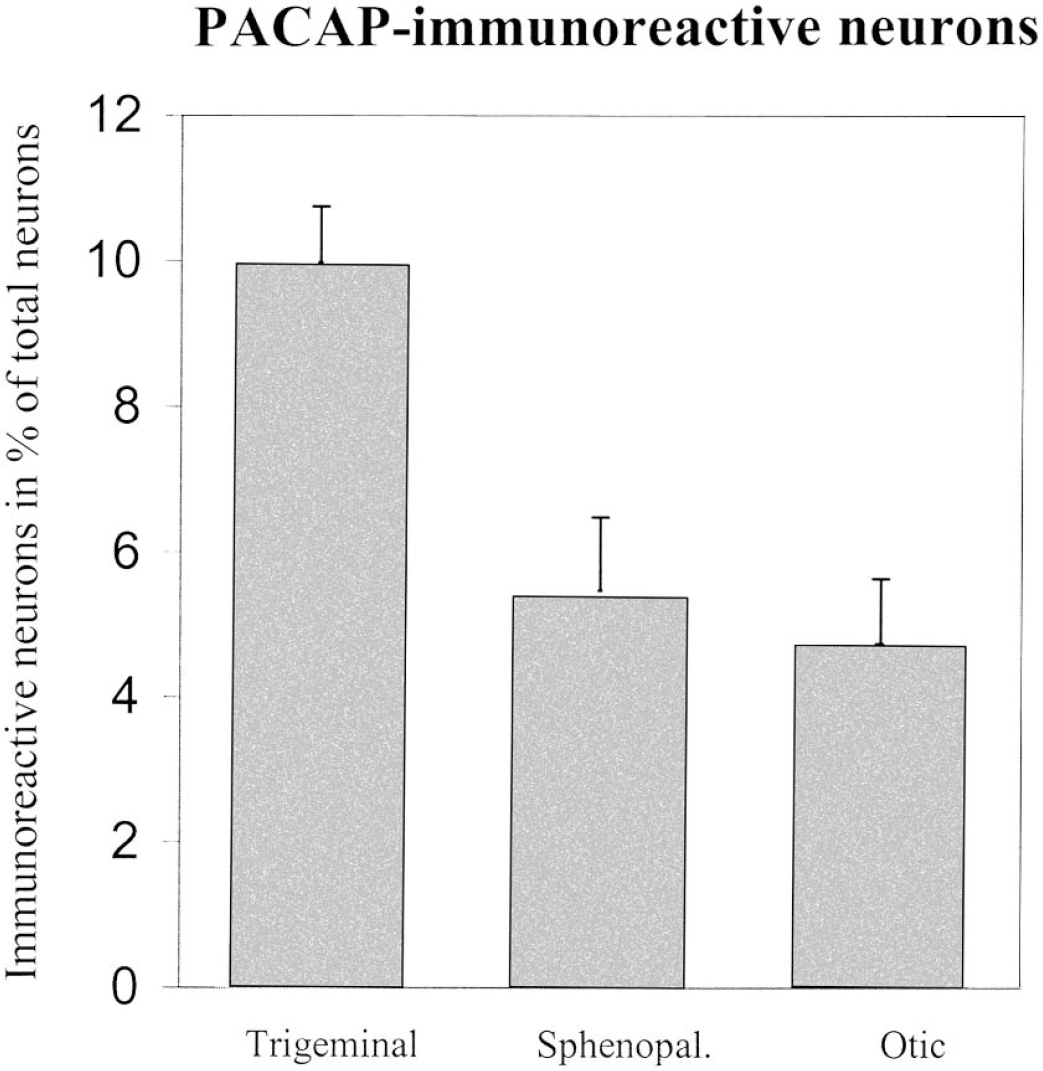
Bar graph showing the density of PACAP-immunoreactive neurons counted in the trigeminal, sphenopala-tine, and otic ganglia of the Wistar rat. Error bars = SEM. n = 4 animals.
DISCUSSION
Our study confirms earlier observations of the presence of PACAP-immunoreactive perikarya in the trigeminal (Kausz et al., 1998; Møller et al., 1999), sphenopalatine, and otic ganglion of the rat (Uddman et al., 1999; Edvinsson et al., 2001). In our study we found 4.8% PACAP-immunoreactive perikarya in the otic ganglion, 5.5% in the sphenopalatine ganglion, and 10% in the trigeminal ganglion. In a combined in situ hybridization and immunohistochemical study of the sphenopalatine and otic ganglion of the rat (Mulder et al., 1995) it was reported that nearly all perikaya in the sphenopalatine and otic ganglia contained mRNA encoding PACAP and that the majority of cell bodies also expressed the protein. However, the same paper pointed out that the intensity of the immunofluorescence for the PACAP-protein in the sphenopalatine and otic ganglion varied considerably between the perikarya. This might indicate that not all mRNA encoding PACAP is translated in the neurons of these ganglia.
Our results showing a low number of PACAPergic neurons in the ganglia are also in accord with a study in the cat (Elsås et al., 1996) where the number of PACAPergic nerve cell bodies in the sphenopalatine ganglion was estimated to be about 10%. In the same study, the density on PACAP-containing perikarya in the trigeminal ganglion was estimated to 5%, a number lower than the 10% we have observed in the rat.
In this study, we detected the Fluorogold tracer by use of an antibody against the Fluorogold molecule. This technique is more sensitive than the traditional technique in which Fluorgold is excited with ultraviolet light. In addition, the Fluorogold-labeled neurons can be observed in confocal microscopes not supplied with a UV-laser. By using this sensitive detection technique, it was easy to distinguish the PACAP-containing neurons in the ganglion from non-labeled cell bodies and to observe double localization (Fig. 4B). A problem in retrograde tracing from nerve fibers located in the subarachnoidal space is unspecific diffusion of the tracer. We do not believe this to be a problem in our study: firstly, unspecific diffusion of the tracer would have resulted in a labeling of the peripheral located neurons in the ganglia as well as the glial cells of ganglia. Such labeling was never observed. Secondly, the otic ganglion is anatomically located close to the trigeminal ganglion, and the difference in the number of retrograde labeled cells in the two ganglia would not have occurred if the labeling was due to unspecific diffusion. The survival time of 7–10 days was determined in previous retrograde tracing studies (Møller and Baeres, 2003) and ensures maximum labeling of perikarya without loss of tracer from the perikarya. However, it must be kept in mind that the number of double labeled cells, seen in our study, depends on the sensitivity of the immunohistochemical PACAP-reaction and the variable number of nerve fibers, which take up the retrograde tracer. Some of the injected tracer might have diffused to the posterior cerebral artery and perhaps also the anterior cerebral artery, although this artery of the rat is located very rostral and covers little of the cerebral surface. We have in preliminary experiments determined the concentration and amount of Fluorogold that results in optimal retrograde labeling of perikarya in the trigeminal ganglion. A further in increase in the amount of injected Fluorogold did not result in a higher number of labeled perikayra.
To confine the subarachnoidal injected tracer to a space around the middle cerebral artery is not possible. However, when tracings have been performed from an extracerebral artery, e.g. the temporal artery, such a confinement has been successful.
Our combined retrograde tracings using Fluorogold and immunohistochemistry for PACAP showed that the majority of PACAPergic neurons innervating the blood vessels originated in the trigeminal ganglion with only a minor contribution from the sphenopalatine and otic ganglia. This origin is supported by the fact that after removal of the sphenopalatine ganglia, no reduction of the PACAPergic innervation of the brain vasculature is observed (Edvinsson et al., 2001). Further, in a study of the origin of nerve fibers innervating the temporomandibular joint, combined retrograde tracing and immunohistochemistry also indicated the trigeminal ganglion as the origin of the PACAP-immunoreactive nerve fibers (Uddman et al., 1998). The number of retrograde labeled neurons in the sphenopalatine, otic, and trigeminal ganglia obtained in this study was lower than the number previously reported (Edvinsson et al., 2001). This might be due to different tracing techniques in the two studies. In the study of Edvinsson et al., the tracer was applied via a trepanation between the anterior lacerated foramen and the foramen ovale, close to the origin of the middle cerebral artery. In the present study, the tracer was applied on the superolateral surface of the brain decreasing the risk of an unspecific diffusion of the tracer to the trigeminal and parasympathetic ganglia located on the ventral part of the skull.
In the sheep trigeminal ganglion, PACAPergic perikarya represent a population of neurons with a diameter smaller than the larger sensory trigeminal perikarya. Also in the dorsal root ganglion, PACAPergic perikarya are small (Mulder et al., 1994). However, in this study of the rat, the PACAPergic neurons did not differ in diameter from other perikarya of the trigeminal ganglion.
We have in our laboratory studied the innervation of the rat pineal gland and the blood vessels in the meninges surrounding the gland. PACAP is known to stimulate the melatonin secretion of the rat pinealocyte. The rat pineal gland possesses a fairly dense innervation with PACAP-containing nerve fibers (Møller et al., 1999). During these investigations we observed that the PACAPergic pineal nerve fibers also contained CGRP, whereas VIP did not co localize with PACAP (Møller et al., 1999). This indicated to us that the origin of the PACAPergic nerve fibers innervating the pineal and the pial blood vessels surrounding the gland, might originate from the trigeminal ganglion as this ganglion is know to contain neurons innervating the blood vessels of the brain containing immunoreactivity for substance P and CGRP (McCulloch et al., 1986; Uddman et al., 1989; Suzuki et al., 1989). We, therefore, lesioned bilaterally the ophthalmic branch of the trigeminal ganglion by a subtemporal approach (Liu and Møller, 2000) and studied degenerating nerve fibers in the rat pineal gland and in the pial arteries surrounding the gland. After 3 days of survival, we observed a nearly total disappearance of the number of PACAPergic nerve fibers in the pineal and a 70% reduction of nerve fibers in the pial blood vessels in the pineal area.
The pathway of the fibers from the trigeminal ganglion to the brain vasculature is described to follow the ophthalmic branch of the trigeminal nerve, and to continue into the ethmoidal nerve, which enters the brain cavity via the ethmoidal foramen in the nasal orbital wall (Hara and Weir, 1986, 1988, Suzuki et al., 1988, 1990). From there the fibers enter the dura mater of the rostral basal part of the skull to the internal ethmoidal artery, which is a branch of the anterior cerebral artery. However, experiments in our laboratory did not support this pathway for the PACAPergic innervation of brain vessels. Thus, lesions of the anterior ethmoidal nerve in the orbit of the rat did not decrease the number of PACAPergic nerve fibers of the pial blood vessels in the pineal area, although a lesion of the opthalmic and maxillary branch resulted in a reduction in the same area (Liu and Møller, 2000). This indicates that the PACAPergic nerve fibers to the blood vessels of the pineal area leave the ophthalmic and maxillary branch to enter the cranial cavity central to the ethmoidal nerve.
It is, therefore, of considerable interest that modern detailed analysis of the pathways from the trigeminal and sphenopalatine ganglia show a much more diverse and complicated anatomy (Bleys et al., 1996) compared to the classical description. Thus nerve fibers connecting the trigeminal ganglion with the sphenopalatine ganglion have been described. This indicates that sensory trigeminal fibers might originate in the distribution area of the sphenopalatine ganglion. Further, the caudal part of the sphenopalatine ganglion is connected with a plexus located in the cavernous sinus (Bleys, et al., 1996). From this plexus nerve fibers follow the trochlear and abducens nerves in caudal direction to the internal carotid artery and the meninges (Bleys et al., 1996, 2001). The presence of these newly detected pathways might explain why the PACAPergic innervation of brain blood vessels does not disappear after cutting of the ethmoidal nerve.
Both PACAP-38 and PACAP-27 are able to dilate brain arteries and arterioles of the cat (Uddman et al., 1993) and rat (Seebeck et al., 2002). PACAP exerts its biological functions by binding to 3 receptors, the PAC1-receptor that is a specific high affinity PACAP receptor, and VPAC1 and VPAC2 with similar affinity for both VIP and PACAP (Hashimoto et al., 1993). Unlike VPAC1 and VPAC2 receptors, the PAC1 receptors exist in several splice variants (Spengler et al., 1993). Both the VPAC1 and VPAC2 receptors are strongly coupled to adenylyl cyclase and stimulation of these receptors result in activation of cAMP (Kobayashi et al., 1994) and protein kinase A (Spengler et al., 1993). Contrarily, the PAC1 receptor stimulates the phospholipase C second messenger pathway (Spengler et al., 1993).
By use of RT-PCR, mRNAs encoding all three receptors have been detected in cerebral human blood vessels (Knutsson and Edvinsson, 2002). Further, the presence of the VPAC1 receptor has been immunohistochemically demonstrated in the brain vasculature of the rat (Fahrenkrug et al., 2000).
From a clinical point of view, the PACAPergic innervation of the brain vessels is of interest, because PACAP along with substance P and CGRP might be involved in pathological mechanisms behind migraine attacks.
Summarizing, our combined retrograde tracings and immunohistochemistry for PACAP indicates that the majority of PACAP-containing nerve fibers innervating the brain vessels originate from perikarya located in the trigeminal ganglion.
Footnotes
Acknowledgements
We wish to thank Ursula Rentzmann for expert technical assistance
