Abstract
Background:
Deep brain stimulation of the posterior hypothalamic area is one of the neuromodulation treatments used for chronic cluster headache, but the number of published patients remains low.
Aim:
The aim of this article was to present the retrospective results of 12 consecutive chronic cluster headache patients treated with deep brain stimulation at Helsinki University Hospital.
Materials and Methods:
All chronic cluster headache patients treated with deep brain stimulation between 2004 and 2012 were included in the study. Patients were interviewed and their hospital files analyzed. Treatment effect was classified as good, partial, or no effect.
Results:
Of the 12 patients, four had a good treatment effect, five had partial, and three had no effect of deep brain stimulation. In contrast to previous studies, our patients reported an almost immediate benefit after the onset of stimulation.
Conclusions:
Deep brain stimulation provides clinically meaningful benefit to a subgroup of chronic cluster headache patients.
Introduction
Chronic cluster headache (CCH) is one of the most disabling disorders known to man. Current medical therapy is often inadequate, and thus invasive neuromodulatory strategies have been attempted. 1 These strategies have not led to a major breakthrough and the number of published CCH series remains low. This is especially true for the most invasive option, that is, deep brain stimulation (DBS) of the posterior hypothalamic area.
At the department of neurosurgery in Helsinki University Hospital, 12 CCH patients were treated with DBS between 2004 and 2012. Our aim was to report our experience and analyze the results of surgery. Our patients add significant volume to the previously published small case series, the largest of which has been 19 patients. 2
Methods
Patients
Twelve patients with incapacitating CCH were selected for hypothalamic DBS, which was offered as an experimental and off-label option, because conservative therapy was ineffective or caused side effects. Two patients had had femoral caput necrosis from long-term use of oral corticosteroids. All patients could be identified from the surgical records. The study was approved by institutional ethics review board of Helsinki University Hospital (117/13/02/2015)
Surgery and follow-up
Anterior and posterior commissures were localized from brain computer tomography (CT) (n = 4) or magnetic resonance imaging (MRI) (n = 8) and merged into CT with Leksell stereotactic frame (Elekta, Stockholm, Sweden). Electrode implantation coordinates were 2 mm lateral, 3 mm posterior, and 5 mm below the mid-commissural point (MCP), ipsilateral to the pain as previously described. 3 In a single session, electrode implantation was conducted with local anesthesia, and implantable pulse generator (IPG) implantation occurred under general anesthesia. Electrode location was verified intraoperatively by fluoroscopy and postoperatively by a CT scan. Stimulation was started 1–2 days postoperatively. Medical management, DBS programming, and postoperative follow-up was performed by neurosurgeons and neurologists experienced with CCH and DBS. The mean follow-up period was 8 years (range 4–13 years).
Study data were acquired retrospectively from all patients by reviewing hospital records; additional information was gained by phone interviews from consenting patients. Two headache specialists analyzed the data, confirmed the correct CCH diagnosis, graded treatment efficacy, and recorded treatment-related adverse effects. Because of the retrospective nature of data, the primary end point was chosen to be the subjective treatment effect reported by the patient—good, partial, or no effect. Good effect was defined as a significant 50–100% decrease in attack frequency or intensity with a long-lasting effect (more than 1 year); additionally, in one case, a good effect was scored because the duration of attacks shortened over 75% on average. Partial effect was scored if attacks decreased 30–50% or if the good effect was short term (months) corresponding to an overall below-optimum treatment effect; no effect was scored if the response was below 30%, very short term, or the patient did not benefit at all.
Electrode location analysis
Electrode locations were analyzed with Brainlab Stereotaxy software (version 3.0.5, Münich, Germany) from postoperative CT scans fused to preoperative MRIs, if available. The location of active electrodes was measured in relation to MCP.
Statistical analysis
Statistical analysis was done with SPSS 24.0 software (IBM Corp., Armonk, NY, USA). Mann–Whitney U test was used for the electrode location analysis. Significance level was set at p < 0.05.
Results
Effect on cluster headache
Of the 12 patients, 10 patients were interviewed by phone, patients 2 and 7 were analyzed only from patient records. Four patients (33%) had a consistently good effect, three from the beginning, and one after reprogramming at 1.5 years. Additionally, one patient had an initial good effect with over 50% reduction in attacks, but after 7 months, the attack frequency was reduced only by 30–50% as compared with baseline. However, the attacks were significantly milder and of shorter duration (over 75% reduction in both), and the treatment effect was scored as good. In three patients, an initial good effect was lost despite reprogramming, with attack frequency reduction of 30–50% after the initial good effect. One patient had a partial (but consistent) effect, and three (25%) had no effect at all. Two of the non-responders engaged in chronic corticosteroid use with caput necrosis of the hip, and in retrospective analysis, one was considered to be a very atypical case for chronic cluster. One patient died during follow-up due to unrelated reasons.
Five patients with unsatisfactory effects from DBS had later additional neuromodulation, three with occipital nerve stimulation (ONS), and two with sphenopalatine ganglion stimulation (SPG). One patient had simultaneously ONS and DBS with moderate additional improvement over DBS alone (Table 1).
Individual patient characteristics.a

CCH: chronic cluster headache; V: volts; M: male; F: female; FU: maximal follow-up duration post-deep brain stimulation; DBS: deep brain stimulation; ONS: occipital nerve stimulation; SPG: sphenopalatine ganglion stimulation.
aGood effect: significant 50–100% decrease in attack frequency with a long-lasting effect during the whole observation period. Partial effect: attacks decreased 30–50% or the effect was not enduring.
Electrode location
The average active electrode location in relation to MCP was 1.8 mm lateral (SD 1.3 mm), 3.5 mm (SD 1.6 mm) posterior, and 2.7 mm (SD 2.4 mm) inferior. In two cases, the electrode tip was in the interpeduncular fossa, and in one case, the active electrode was inside the third ventricle. The location of the electrode was identical between groups, in the anterior–posterior and dorsoventral direction (Mann–Whitney U test p = 0.693–0.868); however, there was a statistically non-significant difference, suggesting that patients with a good treatment effect had a more medial location of the active electrode (p = 0.135, 0.2 mm vs. 2.0 mm from midline). An example of electrode location is shown in the Figure 1.
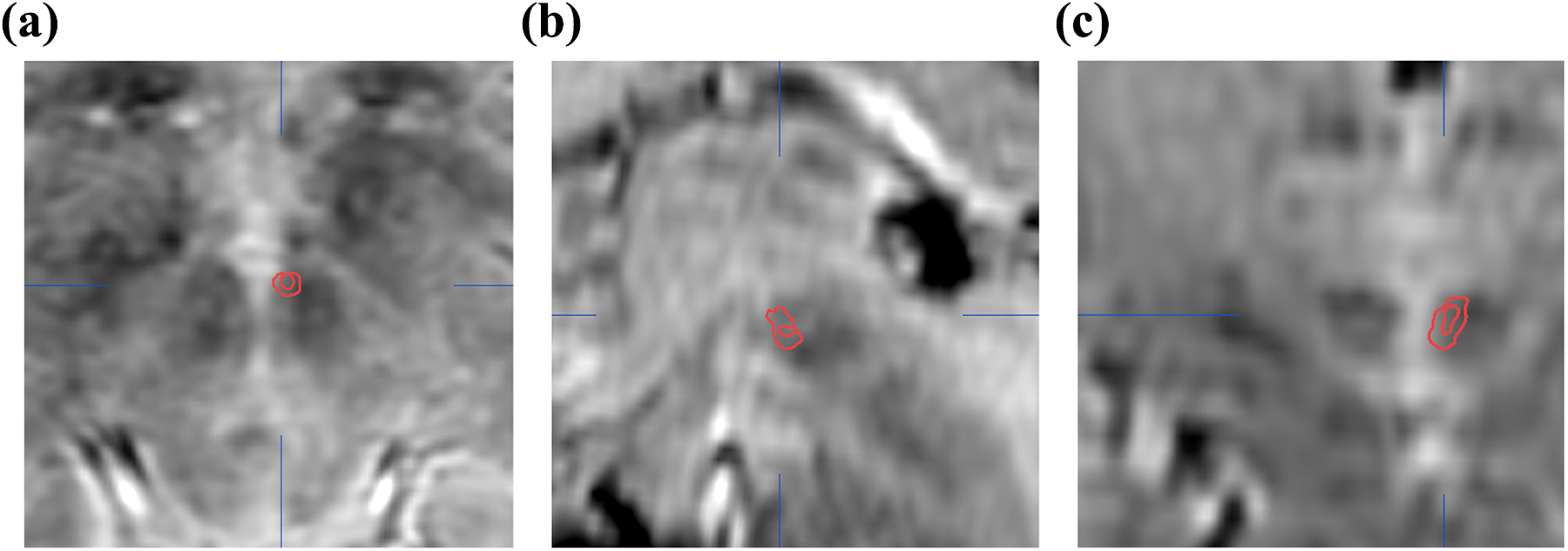
Example of location of an electrode from postoperative CT fused to preoperative MRI in (a) axial, (b) sagittal, and (c) coronal planes. Red circle indicates electrode segmentation from CT superimposed on preoperative MRI.
Observations on stimulation cessation
Discontinuing stimulation for various reasons (patient 3 experienced loss of effect; patients 4 and 6 experienced battery depletion; patient 7 had side effects; and patient 8 had an occupational accident) resulted in worsening of symptoms. Resuming stimulation resulted in a good effect in patient 6; some benefit in patients 4, 7, and 8; and no effect in patient 3. Patient 6 also experienced second battery depletion; however, implanting a new rechargeable IPG did not restore the therapeutic effect. In patient 3, ONS was added to DBS resulting in a benefit.
Postoperative medication
At the last follow-up, seven patients did not have any prophylactic medication for CCH. In two patients, this was due to a good effect of DBS, and in five patients, it resulted from lack of medication benefit. Five patients continued prophylactic medication after DBS, with verapamil being the most common drug (used by four).
Adverse effects
The most common stimulation-related adverse effect was double vision or nystagmus (three patients), fatigue (two patients), and affective symptoms (one patient). One patient reported pain at the IPG site and ipsilateral arm. Treatment-related fatigue (one patient) and double vision (two patients) were reported to limit treatment efficacy. One patient had an occupational accident where external forces broke the stimulation equipment. There were no removals due to infection, and there were no surgery-related side effects.
Discussion
We presented a retrospective qualitative analysis of 12 patients treated with DBS of the posterior hypothalamic area for CCH. Five patients had a good, long-lasting, and clinically significant effect, and an additional three patients had a transient good effect with a longer lasting partial effect. In five patients (two good and three partial responders), there was stopping or discontinuing stimulation for various reasons resulting in worsening of the clinical situation with only partial or no effect after resuming.
The first use of DBS for the treatment of CCH was published in 2000, and the current review reports data on 79 patients, 4 with an additional seven patients published recently. 5 Typically, case series have been small (1–19 patients), and average follow-up varied (from 1–8.7 years). On average 30.4% of patients became pain-free, and 39.5% had at least 50% improvement in headache frequency and/or intensity. Overall our results are in line with previous studies, particularly when the small sample size and the retrospective nature of our data are taken into account. Based on our experience, long-term use of steroids, psychiatric comorbidity, and atypical CCH are markers for bad prognosis. Three of our patients with these characteristics (two with osteonecrosis of the hip due to steroids and one with major psychiatric problems with atypical symptoms) did not benefit at all. In our series, stimulation effect was limited in three patients due to side effects.
Contrary to previous reports, most of our patients reported an almost immediate benefit after electrode implantation. Thus, it seems that DBS might have immediate effects for CCH along with previously suggested slow and gradual functional reorganization of the brain pain circuits. 1 The immediate effect might also be due to a microlesion effect, especially because some patients later lost their initial benefit.
The posterior hypothalamic area has been chosen as a DBS target, because it is activated during pharmacologically provoked cluster headache-like attacks. 6 In addition, posteromedial hypothalamotomy is reported to improve intractable facial pain in cancer patients. 7 However, a recent study suggests that the ventral tegmental area and specifically the trigeminohypothalamic tract could be the real targets. 5 The location of electrodes in our series was similar, but 2 mm superior, compared with previous studies. In agreement with previous reports, 3 we found no clear correlation of electrode location and a good therapeutic effect, although the number of patients is low for statistical analysis. Additionally, the accuracy of electrode location analysis is limited in our study, because the first four patients had only CT scans available for localization of anatomical landmarks.
Our study supports the beneficial effects of DBS of the posterior hypothalamic area in a subgroup of CCH patients. Long-term use of steroids, psychiatric comorbidity, and atypical symptoms could be markers for treatment failure. The fact that two patients with a partial response to DBS experienced additional benefit from ONS and SPG provides some support that patients failing with DBS therapy might still benefit from other neuromodulation modalities. New therapeutic options, along with the reports of all previous experiences, are clearly indicated to better understand CCH and to make the lives of patients less intolerable.
Clinical implications
DBS of the posterior hypothalamic area can alleviate CCH. The addition of ONS or SPG might improve the treatment effects of DBS in some patients. Our experience suggests that CCH patients with excessive chronic steroid use, psychiatric comorbidity, or atypical symptoms might not benefit from DBS.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
