Abstract
Background
Traditional Thai herbal medicine represents an alternative and complementary approach for managing hyperlipidemia, according to its low toxicity and advantageous benefits. However, there is limited clinical data for Thai herbal medicine formulations for hyperlipidemia.
Objectives
The present study aimed to evaluate the biological and clinical efficacy of Thai herbal formulation for lowering blood lipids in patients with borderline hyperlipidemia.
Materials and methods
The chemical profile, in vitro antioxidant activity, pancreatic lipase inhibition, and cytotoxicity tests were evaluated in the aqueous and ethanol extracts of Thai herbal formulation. The clinical efficacy and safety of Thai herbal formulation for reducing blood lipids were assessed in patients with borderline hyperlipidemia during a 12-week period.
Results
The main chemical constituents of Thai herbal formulation, including (+)-chebulic acid, 4-glucogallic acid, gallic acid, ellagic acid, piscidic acid, and 6-gingerol, demonstrated anti-oxidant and pancreatic lipase inhibitory effects. In cytotoxicity testing, the aqueous extracts exhibited no inhibitory effect on L929 cells and Vero cells (IC50 values above 80 μg/mL), whereas the ethanol extracts presented IC50 values of 18.59 ± 0.58 µg/mL in Vero cells and 39.86 ± 0.02 µg/mL in L929 cells. The clinical investigation revealed that the Thai herbal formulation significantly decreased total cholesterol and low-density lipoprotein levels (P < 0.05), and additionally reduced triglycerides and elevated high-density lipoprotein cholesterol, in comparison to the control group after 12 weeks, thereby demonstrating its efficacy in hyperlipidemia management.
Conclusion
The findings indicated that the Thai herbal formulation exhibited potential as a herbal therapeutic approach for borderline hyperlipidemia management.
Keywords
Introduction
Hyperlipidemia is the most common type of dyslipidemia, with an estimated global prevalence of dyslipidemia in adults reaching 80%. Dyslipidemia characterized by elevated blood levels of total cholesterol (TC), triglycerides (TG), low-density lipoprotein (LDL-C), and a lowered level of high-density lipoprotein (HDL-C), representing a major risk factor for cardiovascular disorders (CVD). 1 The disorder is mostly associated with behavioral variables including urbanization, consuming behaviors, obesity, smoking, and physical inactivity, whereas the present socioeconomic crisis has adversely affected healthcare services and public health. 2 Most of recommendations or guidelines for dyslipidemia therapy emphasize LDL cholesterol as the primary goal for lipid management. Statins are employed as the primary treatment for reducing LDL levels by inhibiting 3-hydroxy-3-methylglutaryl-coenzyme A (HMG-CoA) reductase. However, statins exhibit adverse effects, such as muscular myopathy and hepatic function abnormalities.3,4 Consequently, there is an urgent need for the development of safe alternative hypolipidemic medicines for the treatment of hyperlipidemia.
Traditional Thai herbal medicine represents an alternative and complementary approach for regulating hyperlipidemia due to its low toxicity and beneficial effects.5,6 The major advantage of multicomponent traditional Thai herbal therapy was the capacity to target multiple pathways and biological processes for managing complex problems.5–8
The Thai herbal formulation9,10 consisting of Allium sativum,11,12 Curcuma zedoaria, 13 Derris scandens,14–16 Garcinia hanburyi, 17 Phyllanthus emblica,18,19 Terminalia arjuna,20,21 Terminalia bellirica,22,23 Terminalia chebula, 24 Terminalia citrina,25–27 and Zingiber officinale 28 was established in traditional Thai medicine principles for the reduction of blood lipids. The main properties of the herbal medicine composition were to clear phlegm, eliminate intestinal mucus, circulate wind, and nourish the elements to restore physical balance.9,10 However, the evidence of improvements in the blood lipid profile among patients with hyperlipidemia was limited. The prescribing standards for LDL and TC in hyperlipidemia are based on the Royal College of Physicians of Thailand (RCPT) clinical practice guidelines; however, the present study aims to evaluate the biological and clinical efficacy of a Thai herbal formulation in patients with borderline hyperlipidemia for the first time.
Materials and Methods
Preparation of Thai Herbal Formulation and Ten Botanical Extracts
A novel formulation comprising Allium sativum, Curcuma zedoaria, Derris scandens, Garcinia hanburyi, Phyllanthus emblica, Terminalia arjuna, Terminalia bellirica, Terminalia chebula, Terminalia citrina, and Zingiber officinale was developed based on traditional Thai medicinal principles to lower blood lipids. The primary attributes of the herbal medicine formulation were to expel phlegm, eradicate intestinal mucus, promote circulation, and replenish the ingredients to reestablish bodily equilibrium.9,10
Ten medicinal plants used in the study (Table 1) were acquired from a Triburi herbal store in Songkla, Thailand. The herbs were cleaned, dried in an oven at 60 °C for 18 h, chopped into small pieces, pounded into a fine powder, and filtered through a sieve. All medicinal herbs were collected in a desiccator. The physico-chemical parameters of the herbs, including foreign matter, loss on drying, total ash, acid-insoluble ash, and extractive values (water-soluble extractive, ethanol-soluble extractive, 50% ethanol-soluble extractive, and 70% ethanol-soluble extractive), were assessed in triplicate according to the methodologies described in the Thai Herbal Pharmacopeia and the Ayurvedic Pharmacopeia of India.29,30
Thai Herbal Formulation for Hyperlipidemia.

The powder samples (200 g) were prepared in bottles. All the herbs were combined for the formulation. Each herb and the formulation were soaked in 95% ethanol or distilled water and shaken using an incubator shaker at 70 rpm at a temperature of 50 °C for 48 h, repeated three times. Subsequently, each infusion was filtered using Whatman No.1 paper. The filtrate was concentrated using a rotary evaporator at 45 °C under decreased pressure for ethanol extracts or freeze-dried for water extracts. 38 All extracts were stored in desiccators at 4 °C for later usage. The percentage yield of the extract was determined by the dry weight of the extract (a) and the weight of the starting dry material (b) using the formula: Percentage yield (%) = a/b × 100.
Investigation of Chemical Constituents in Thai Herbal Formulation Using Liquid Chromatograph-Quadrupole Time-of-Flight Mass Spectrometer (LC-QTOF MS) Analysis
The Thai herbal formulation extract was analyzed using a liquid chromatograph-quadrupole time-of-flight mass spectrometry (LC-QTOF MS) instrument (1290 Infinity II LC-6545 Quadrupole-TOF, Agilent Technologies, USA), use of a Zorbax Eclipse Plus C18 Rapid Resolution HD column (150 mm length × 2.1 mm inner diameter, particle size 1.8 μm). 39 Briefly, The mobile phase consisted of 0.1% formic acid in water (v/v, A) and methanol (B). The programed gradient was as follows: 10% B from 0 to 5 min, 10%-30% B from 5 to 10 min, 30%-85% B from 10 to 15 min, 85%-100% B from 15 to 20 min, 100% B from 20 to 23 min, and a return to 10% B from 23 to 30 min, at a flow rate of 0.2 mL/min. A post-run period of 5 min was observed, with an injection volume of 2 μL and a column temperature maintained at 40 °C. Dual electrospray ionization (ESI) source functioning in both negative and positive ionization modes. The extracts were examined using auto MS/MS mode within the m/z range of 100 to 1200. The Mass Hunter Workstation software (version B 08.00 Qualitative Analysis Workflows and Qualitative Analysis Navigator, Agilent Technologies, USA) was used for data analysis. The MassHunter METLIN PCD (Personal Compound Database) and PCDL (Personal Compound Database and Library) were used to discover the bioactive chemicals.
Determination of Total Phenolic Compound Content
The total phenolic compound content in plant extracts was investigated according to a previous study with minor modifications. 40 Briefly, 20 mg of the extract were dissolved in 10 ml of DMSO or ethanol. 25 µL of sample extract was combined with 100 µL of 25% Folin-Ciocalteau reagent in water (v/v). Subsequently, 75 µL of 10% Na2CO3 was added into the mixture and placed at room temperature. After 2 h, the absorbance was measured at 750 nm. Gallic acid was used as the standard. The results were presented as milligrams of gallic acid equivalents (GAE) per gram of extract. All determinations were performed in triplicate.
Determination of Total Flavonoid Content
The total flavonoid content in plant extracts were determined based on a previous study with minor modifications. 41 Briefly, 50 µl of extracts or standard solution (quercetin) were combined with 10 µL of 10% aluminum chloride solution, followed by 150 µL of methanol. Subsequently, 10 µL of 1 M sodium acetate was added to the mixture on a 96-well plate. All reagents were combined and incubated for 40 min at ambient temperature, covered from light. The absorbance was measured at 415 nm using a microplate reader. Total flavonoid concentration was calculated in milligrams of quercetin equivalents (QE) per gram of plant extract. All determinations were performed in triplicate.
Determination of Total Antioxidant Capacity of Thai Herbal Formulation
The total antioxidant capacity of Thai herbal formulation was evaluated using three assays using the organic synthetic radical DPPH, the ABTS radical cation, and the FRAP method.
DPPH Radical-Scavenging Assay
The free radical scavenging activity was measured according to a previous study with minor modifications. 40 Briefly, The DPPH working solution was prepared by diluting the DPPH stock solution (50 µM) in analytical-grade methanol to achieve an absorbance of 1 ± 0.02 at 515 nm. 30 µL of plant extract was combined with 170 µL of DPPH working solution. The experiment plate was subsequently incubated in darkness at room temperature for 30 min, after that the absorbance was measured at 515 nm. Ascorbic acid was employed as the positive standard, and the % inhibition of DPPH in the test samples was calculated by the following formula: DPPH inhibition (%) = [(Ac - As) / Ac] × 100. Ac represents the absorbance of the control (without sample), while As is the absorbance of the sample solution. All determinations were performed in triplicate.
ABTS Radical-Scavenging Assay
ABTS radical-scavenging assay was determined according to a previous study with minor modifications. 42 Briefly, The ABTS stock solution (7 mM) was prepared by combining 7 mM ABTS with 2.45 mM potassium persulfate as the oxidizing agent in a 1:1 volume ratio. The operational solution of ABTS+· was achieved by diluting the stock solution in analytical grade ethanol to provide an absorbance of 0.70 ± 0.02 at 734 nm. Then, 30 µL of sample extract were combined with 195 µL of ABTS+· solution. The reaction mixture was incubated in darkness at room temperature for 30 min, and absorbance was measured at 734 nm using a microplate reader. Ascorbic acid was used as a positive standard. The percentage inhibition of ABTS+· in the test samples was determined using the formula: ABTS inhibition (%) = [(Ac−As)/Ac] × 100. Ac represents the absorbance of ABTS radical in ethanol, while As is the absorbance of ABTS radical solution mixed with sample extract/standard. All determinations were performed in triplicate.
Ferric Reducing Antioxidant Power (FRAP) Assay
FRAP assay was carried out according to a previous study with minor modifications. 40 Briefly, the Frap reagent was prepared by mixing 10 mM TPTZ (24,6-tripyridyl-striazine, in 40 mM HCl), 20 mM FeCl3, and 300 mM acetate buffer (pH 3.6) in a 1:1:10 v/v ratio at 37 °C for 30 min before use. Then, 100 µL of sample extract was combined with 2000µL of Frap reagent, and absorbance measurements at 593 nm were recorded at 37 °C approximately 20 min after first mixing. The concentration was determined using quercetin as a standard. The results were presented as milligrams of quercetin equivalents (QE) per gram of extract. All determinations were performed in triplicate.
Pancreatic Lipase Inhibitory Activity of Thai Herbal Formulation
Porcine pancreatic lipase inhibitory activity was investigated in a previous study with minor modifications. 43 The pancreatic lipase and 4-methylumbelliferyl oleate samples were prepared in a Tris-HCl buffer solution (13 mM Tris-HCl, 150 mM NaCl, 1.3 mM CaCl2, pH 8.0). Then, 25 μL of sample solution (at different concentrations), or 25 μL of orlistat, in addition to 25 μL of pancreatic lipase solution (1 mg/ml), were added onto a 96-well plate. After a pre-incubation at 37 °C for 10 min, the reaction started by the addition of 50 μL of 1 mM 4-methylumbelliferyl oleate solution. Subsequently, incubation at 37 °C for 20 min was followed by a stop of the reaction with the addition of 100 μL of 0.1 M citrate buffer solution (pH 4.2). The quantity of 4-methylumbelliferone produced by pancreatic lipase was determined fluorometrically at an excitation wavelength of 355 nm and an emission wavelength of 460 nm. The control sample was prepared by adding a buffer solution in place of the testing sample. Orlistat was used as the positive control. All of the experiments were carried out on three occasions. The inhibition of pancreatic lipase activity was assessed using the following formula: Inhibition (%) = [1 - A1/A0] × 100. A1 represents the absorbance of the test sample, while A0 represents the absorbance of the blank (without samples).
Evaluation of Cytotoxic Activity of Thai Herbal Formulation
The cytotoxic activity was evaluated using the MTT method with normal cell lines: L929 cells (mouse fibroblast cell line) and Vero cells (African green monkey kidney cell line). 44 L929 cells were cultivated in Dulbecco's Modified Eagle Medium (DMEM) at a density of 8 × 10³ cells per well, whereas Vero cells were cultured in RPMI 1640 Medium at a density of 1 × 10⁴ cells per well in 96-well plates. The cell lines were exposed to extracts at doses of 5, 10, 20, 40, and 80 μg/ml, respectively. Doxorubicin at doses of 0.625, 1.25, 2.5, 5, and 10 μM was used as positive controls for a duration of 72 h. Subsequently, the cells were rinsed with 1x PBS and incubated in 100 μL of 0.5 mg/mL MTT at 37 °C for 30 min. After that, 100 μL of DMSO was added to dissolve the formazan produced in the cells, and the mixture was incubated at 37 °C for an additional 30 min at room temperature. Absorbance was measured at 570 and 650 nm using a microplate reader. All experiments were performed in triplicate.
Clinical Efficacy and Safety of Thai Herbal Formulation for Patients with Borderline Hyperlipidemia
Preparation of Thai Herbal Formulation Capsules
The capsules containing Thai herbal formulation powder and corn starch (Grade USP 42) as the placebo were prepared similarly in form, size, and color by a Thai traditional medicine practitioner using a semi-automatic capsule filling machine. Each capsule contained 500 mg. Then, the quality control of the capsules included appearance features, weight variation, and disintegration time, in accordance with the methodologies established in USP 41-NF 36 and THP 2018.45,46 Both Thai herbal formulation capsule and the placebo were contained in sealed plastic bottles. All containers were labeled with labels (A or B) on the outside of the packaging to maintain participant blinding.
Study Design and Interventions
A randomized, double-blind, clinical trial was performed at two Traditional Thai Medical Clinics in Hat Yai Hospitals, Thailand including the Thai Traditional Medicine Hospital of Prince of Songkla University and Kho Hong Health Promoting Hospital. Prior to participation, all subjects provided written informed permission for the protocol approved by the Human Research Ethics Committee of Faculty of Traditional Thai medicine, Prince of Songkla University (reference number: EC.63/TTM.01-001). The protocol was also registered in Thai Clinical Trials Registry (registration ID: TCTR20241111003). Study-related procedures were conducted in accordance with the accepted guidelines and ethical standards established in the Helsinki Declaration.
The enrolled participants were randomly assigned to two groups: the intervention group (Thai herbal formulation group) and the control group. Participants in both the Thai herbal formulation group and the control group were given the instructions to consume 2 capsules before breakfast and 2 capsules before dinner for a continuous duration of 12 weeks. The recommended daily dosage of the Thai herbal formulation and placebo capsules was 2000 mg per day, based on traditional Thai medicinal practices.
All participants were advised to maintain their usual dietary and physical routines and to report any adverse effects throughout the intervention. The participant's compliance was evaluated using the pill count technique, and those who used over 80% of the treatments were included into the study.
Randomization and Blinding
The patients were randomly assigned to either the Thai herbal formulation group or the control group using the random number approach (a 1:1 ratio employing SAS 9.2 software with a block size of 4). All participants and researchers, protect the data analysts, were oblivious to group allocation. The shape, color, and scent of Thai herbal medication were indistinguishable to preserve masking, and the packaging remained uniform. The random assignment was concealed behind sequentially numbered, opaque, sealed envelopes. The allocation group was kept confidential until an investigator provided data for a fully eligible patient, so preserving the integrity of the randomization process. Both the patients and the investigators assessing results were oblivious to the random allocation to the treatment group (double-blind). Blinding remained oblivious to the generated allocation codes throughout the experiment. The blinding was to be maintained (unless in emergencies) until the data was collected and verified for analysis.
Inclusion Criteria and Exclusion Criteria
Participants with borderline hyperlipidemia of both genders aged 25 to 55 years had at least one of the following indices: Total serum cholesterol ranged from 200 to 239 mg/dl; triglycerides ranged from 150 to 199 mg/dl; LDL ranged from 130 to 159 mg/dl; and HDL was less than 50 mg/dl. Patients using drugs or supplements that affect serum lipids, steroids; participants with coronary vascular disease; those with renal and hepatic dysfunction; hematological disorders; uncontrolled hypertension; uncontrolled diabetes; pregnant or breastfeeding women; and patients at risk of experiencing adverse complications during the study, such as headache, vertigo, nausea, participants unable to adhere with the prescribed capsule regimen, or allergic reactions to the herb or any capsule component, were considered not eligible based on the exclusion criteria.
Outcome Measurements
Demographic characteristics including age, weight, height, gender, medical and drug history, family history, physical activity, consuming high-fat foods, and others were assessed and recorded through interview with the patients.
Measurements including body weight (kg), body mass index (kg/m2), and blood testing [fasting serum concentrations of lipid parameters (total cholesterol, triglycerides, low-density lipoprotein, high-density lipoprotein), complete blood count and toxicity tests (blood urea nitrogen, creatinine, alanine transaminase, and aspartate aminotransferase)] were measured at baseline and end of the intervention. Furthermore, in the follow-up of the Helsinki Heart Study, medication adherence was assessed in all patients using capsule counting, supplemented by physical activity questionnaires, dietary intake forms, and physical examinations at 4, 8, and 12 weeks.
Safety Assessment
The safety of the clinical trial was evaluated by tracking adverse events throughout the investigation. This study defined major adverse effects as mortality, life-threatening conditions, hospitalization or prolonged hospitalization, and permanent or severe impairment.
Sample Size
In the context of two distinct parallel groups with a 1:1 allocation, an effect size of 0.8, a Type I error rate of 5%, and a Type II error rate of 20%, the sample size calculated using G*Power 3.1.9.2 Software was 52 participants, with 26 allocated to each group. Cohen characterizes effect size as the quotient of the difference in means and the pooled standard deviation, asserting that d = 0.8 signifies a large effect in a pilot clinical study, suggesting a significant difference between the groups for comparison. Anticipating a 25% attrition rate, 70 volunteers with borderline hyperlipidemia were recruited and randomized into two groups.
Statistical Analysis
Descriptive analyses for categorical variables were expressed as frequency and percentage, while continuous variables were reported as mean and median/interquartile range. Inferential statistics were used for a comparative analysis of the baseline data between the intervention and control groups. Categorical data were analyzed using the Chi-Square Test of Independence, whereas continuous variables were assessed using Welch's t-test (for normal distribution) or the Wilcoxon rank sum test. A two-way mixed analysis of variance (ANOVA) was used to investigate the interaction effects between time (pre/post) and group (intervention/control), using repeated measurements on the last factor. Bonferroni's pairwise comparisons were used for post hoc analysis. The impact of these characteristics on dietary habits was evaluated using a three-way mixed ANOVA (dietary habits/group/time) and the Bonferroni comparison test. P-values < 0.05 were considered to indicate statistically significant results.
Results
Physicochemical Characteristics of Thai Herbal Medicines
The physicochemical characteristics such as foreign matter, loss of drying, total ash, acid-insoluble ash, ethanol-soluble extractive, and water-soluble extractive are important parameters in studying the physicochemical compositions of Thai herbal medicines.
The physicochemical specification of herbal components was shown in Table 2.
Physicochemical Specification (% by Weight) of Herbal Components.

Allium sativum, AS; Curcuma zedoaria, CZ;
ND, not detectable; NA, not applicable; a The parameters were tested according to Ayurvedic herbal pharmacopeia; b The parameters were tested according to Thai herbal pharmacopeia; c The parameters were tested according to Myanmar herbal pharmacopeia; d A Manual of material medica and pharmacology.
Most medicinal plants, including C. zedoaria, Z. officinale, P. emblica, T. bellirica, T. arjuna, T. chebula, and G. hanburyi met standards for foreign matter, moisture loss, total ash, acid-insoluble ash, ethanol-soluble extractives, and water-soluble extractives. A. sativum exhibited a total ash concentration of 2.64 ± 0.14, above the standards established by the Thai herbal pharmacopeia (< 2.5%), whereas D. scandens revealed a moisture content of 7.89 ± 0.03, again beyond the required parameters (< 7.0%). The aqueous extract concentration of T. citrina, quantified at 34.15 ± 1.40, was marginally below the standards established in the Myanmar herbal pharmacopeia (> 35.2%).
Percentage Yield of Aqueous and Ethanol Crude Extracts of Herbal Components and Thai Herbal Formulations
The ten herbal medicines and herbal formulations, approximately 200 grams, were extracted by a 95% ethanol solvent and subsequently with distilled water. The results of the study indicated the percentage of coarse extract content (Figure 1 and Table 5). The ethanol crude extract yield of Thai herbal formulation was 14.32%. In the ethanol extraction, G. hanburyi extract exhibited the greatest percentage of coarse extract at 38.87%, followed by T. arjuna extract at 33.24% and T. citrina extract at 32.16%, respectively. The aqueous extraction of Thai herbal formulation produced a coarse extract content of 27.26%, whereas the water extract of A. sativum demonstrated the highest coarse extract content at 71.31%, followed by T. citrina extract at 28.52% and P. emblica extract at 28.28%, respectively.

Extraction Yields of Thai Herbal Formulation and its Herbal Components.
Chemical Composition of Thai Herbal Formulation Using LC-QTOF MS
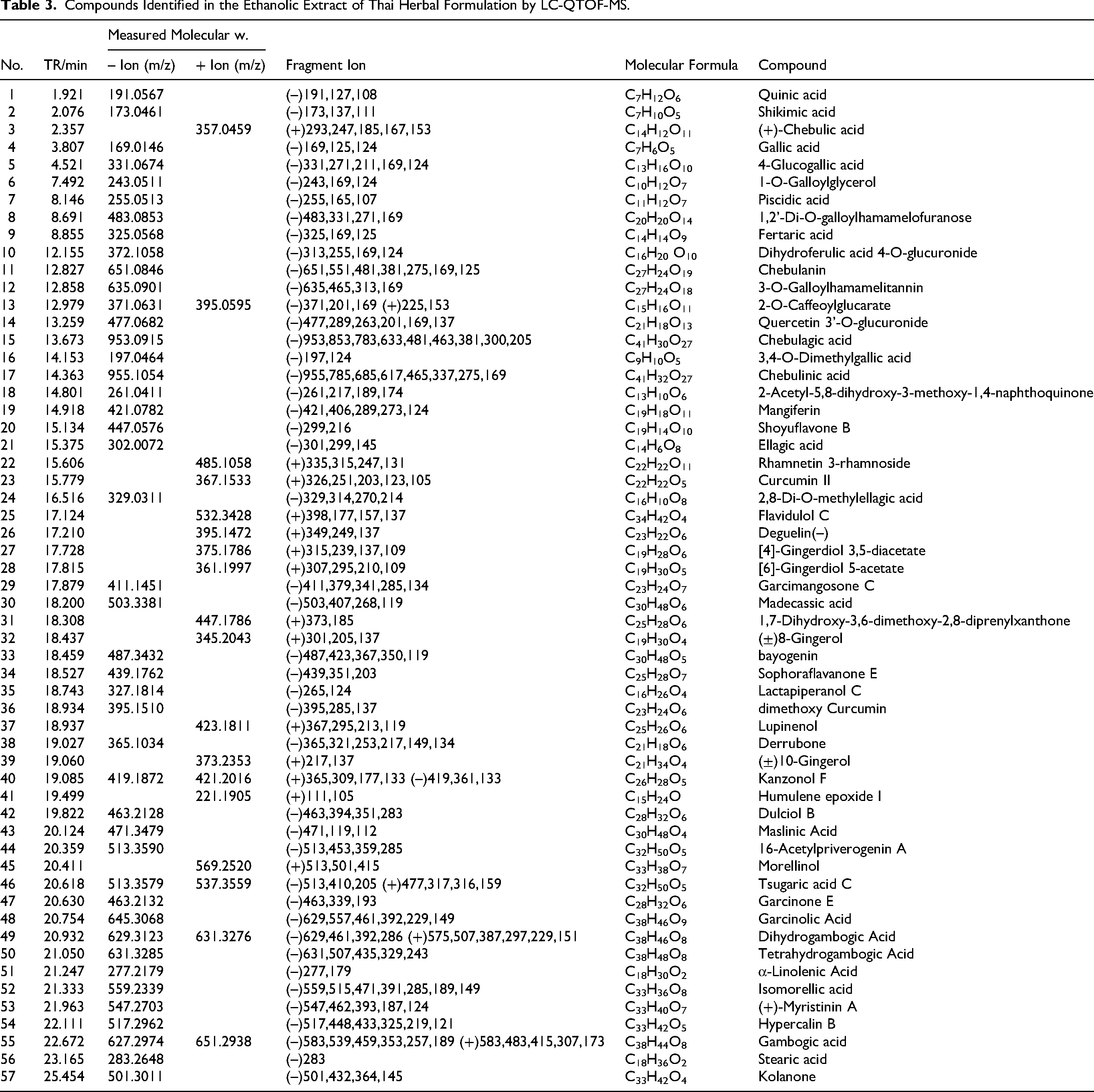
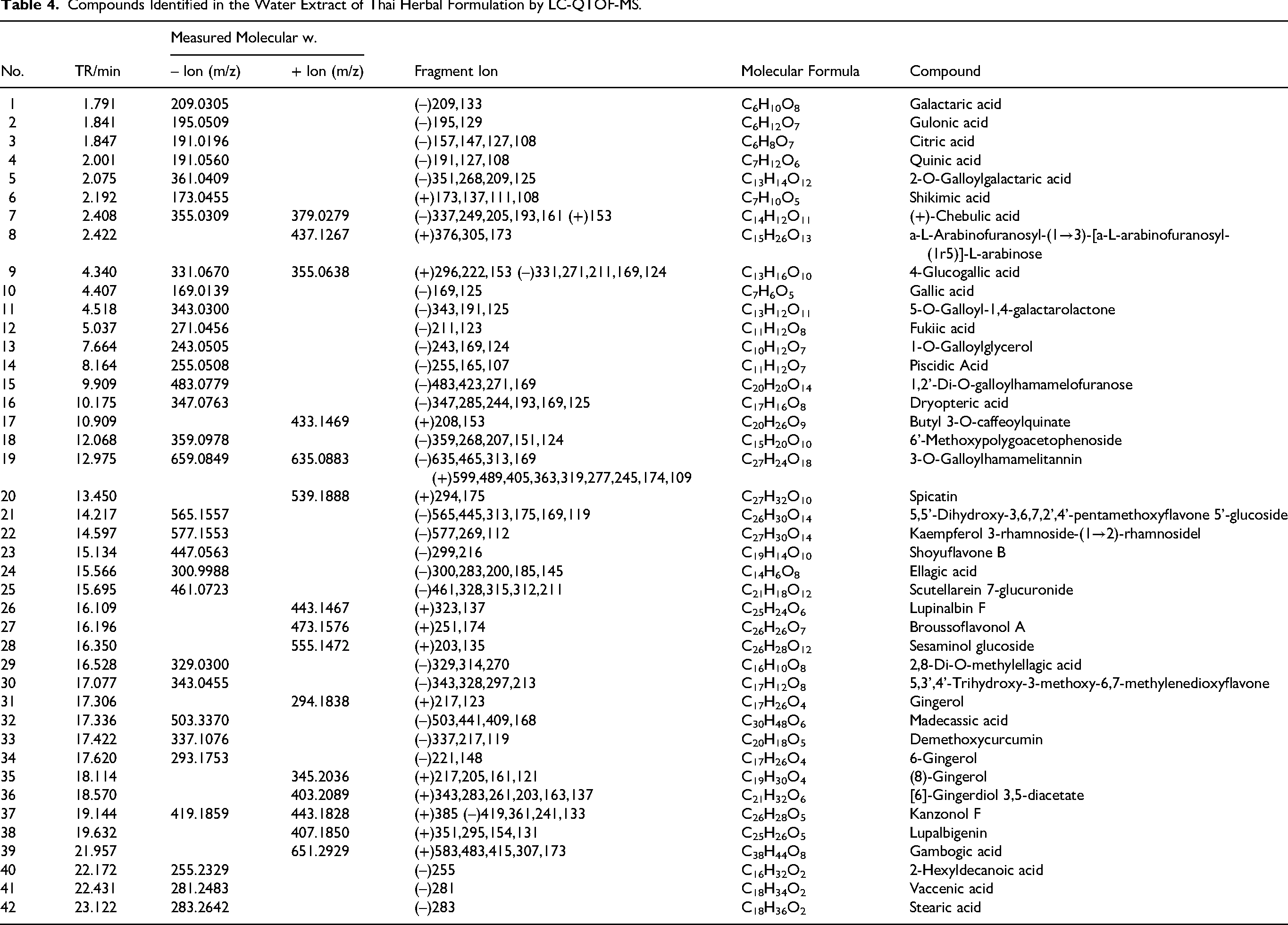
Evaluation of the chemical composition of ethanol and aqueous extracts from Thai herbal formulation using LC-QTOF MS, specifically the 1290 Infinity II LC-6545 Quadrupole-TOF, utilizing an Electrospray Ionization (ESI) source in both negative and positive modes, in relation to chromatograms (Figure 2). The ethanol extract of Thai herbal formulation identified 57 identified compounds (Table 3), whereas the water extracts of Thai herbal formulation included 42 identified components (Table 4). The main chemical constituents of aqueous and ethanol extracts from Thai herbal formulation, demonstrating antioxidant properties and lipid-lowering effects, including (+)-chebulic acid, 4-glucogallic acid, gallic acid, ellagic acid, piscidic acid, and 6-gingerol (Figure 3).
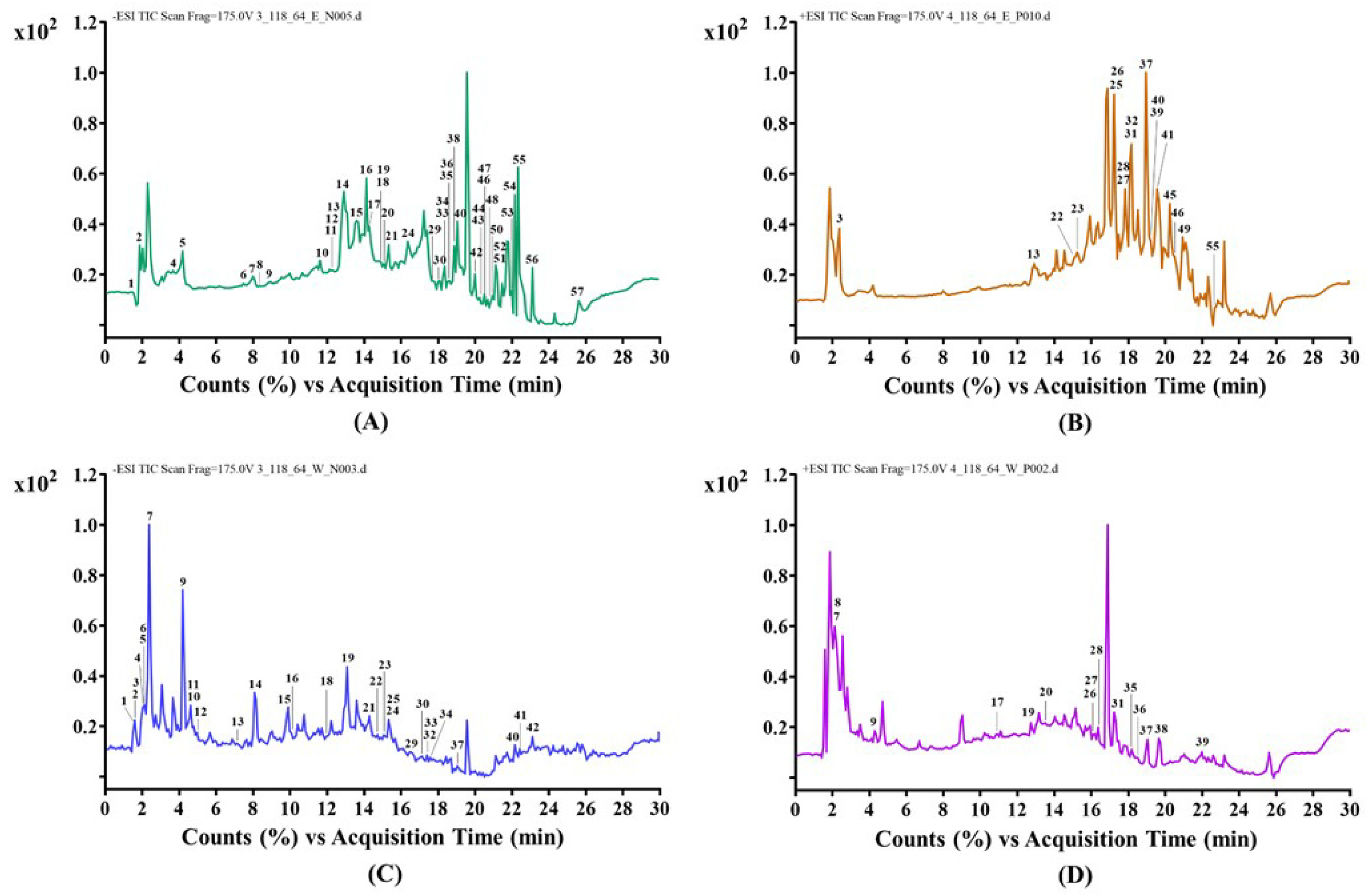
LC-QTOF-MS Chromatogram of Bioactive Compounds from Thai Herbal Formulation; Ethanol Negative Mode (A); Ethanol Positive Mode (B); Water Negative Mode (C); Water Positive Mode (D).

Chemical Structure of Bioactive Compounds from Thai Herbal Formulation Tentatively Identified by LC-QTOF-MS.
Compounds Identified in the Ethanolic Extract of Thai Herbal Formulation by LC-QTOF-MS.
Compounds Identified in the Water Extract of Thai Herbal Formulation by LC-QTOF-MS.
Phenolic and Flavonoid Contents and Antioxidant Capacity of Thai Herbal Formulation
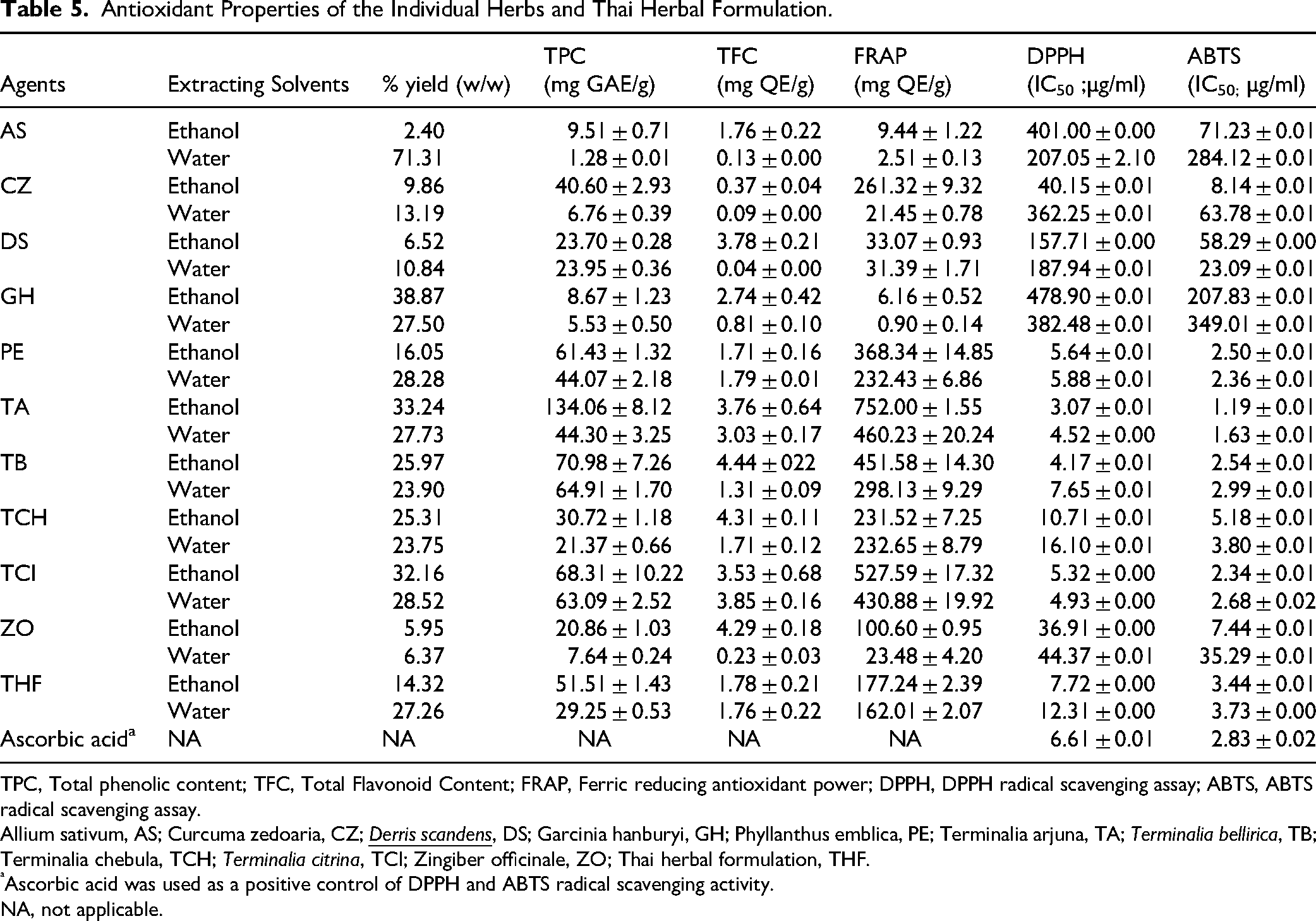
Total phenolic and flavonoid contents of the individual herbs and Thai herbal formulation were demonstrated in Table 5. The total phenolic content varied among different extracts, ranging from 1.28 to 134.06 mg GAE/g of dry material. The total phenolic content of the ethanol extract from the Thai herbal formulation was 51.51 ± 1.43 mg GAE/g. The highest total phenolic levels among the herbal components were observed in T. arjuna (134.06 ± 8.12 mg GAE/g), T. bellirica (70.98 ± 7.26 mg GAE/g), and T. citrina (68.31 ± 10.22 mg GAE/g), while the lowest was found in G. hanburyi (8.67 ± 1.23 mg GAE/g). The aqueous extract of Thai herbal formulation had 29.25 ± 0.53 mg GAE/g. T. bellirica exhibited the greatest total phenolic content at 64.91 ± 1.70 mg GAE/g, followed by T. citrina at 63.09 ± 2.52 mg GAE/g, and T. arjuna at 44.30 ± 3.25 mg GAE/g, while A. sativum demonstrated the lowest amount at 1.28 ± 0.01 mg GAE/g.
Antioxidant Properties of the Individual Herbs and Thai Herbal Formulation.
TPC, Total phenolic content; TFC, Total Flavonoid Content; FRAP, Ferric reducing antioxidant power; DPPH, DPPH radical scavenging assay; ABTS, ABTS radical scavenging assay.
Allium sativum, AS; Curcuma zedoaria, CZ;
Ascorbic acid was used as a positive control of DPPH and ABTS radical scavenging activity.
NA, not applicable.
The total flavonoid content of all extracts ranged from 0.04 to 4.44 mg QE/g of dry material. The ethanol extract of the Thai herbal formulation contained 1.78 ± 0.21 mg QE/g, whereas, T. bellirica exhibited the greatest total flavonoid content at 4.44 ± 0.22 mg QE/g, followed by T. chebula at 4.31 ± 0.11 mg QE/g and Z. officinale at 4.29 ± 0.18 mg QE/g, respectively. The herbal component in the formulation exhibiting the lowest flavonoid concentration was C. zedoaria, with an amount of 0.37 ± 0.04 mg QE/g. The aqueous extracts revealed that the Thai herbal formulation extracts contained a total flavonoid content of 1.76 ± 0.22 mg QE/g. T. citrina exhibited the highest flavonoid content at 3.85 ± 0.16 mg QE/g, followed by T. arjuna at 3.03 ± 0.17 mg QE/g and P. emblica at 1.79 ± 0.01 mg QE/g, while D. scandens had the lowest at 0.04 ± 0.00 mg QE/g.
Antioxidant properties including DPPH radical-scavenging assay, ABTS radical scavenging assay, and FRAP assay of the individual herbs and Thai herbal formulation were presented in Table 5. The DPPH radical scavenging activity of ethanol extracts from Thai herbal formulation exhibited an IC50 of 7.72 ± 0.00 μg/mL, which was higher than the IC50 value of 12.31 ± 0.00 μg/mL for aqueous extracts. For the ethanol and aqueous extracts of the medicinal plants, the results revealed that the ethanol extracts of T. arjuna, T. bellirica, and T. citrina exhibited the most effective DPPH radical scavenging, with IC50 values of 3.07 ± 0.01, 4.17 ± 0.01, and 5.32 ± 0.00 μg/mL, respectively. Whereas, the highest antioxidant potential was observed in the water extracts of T. arjuna (4.52 ± 0.00 μg/mL), followed by T. citrina (4.93 ± 0.00 μg/mL) and P. emblica (5.88 ± 0.01 μg/mL), in comparison to the standard ascorbic acid (6.61 ± 0.01 μg/mL).
The ABTS radical scavenging activity of ethanol extracts from the Thai herbal formulation demonstrated an IC50 of 3.44 ± 0.01 μg/mL, which was higher than the IC50 value of 3.73 ± 0.00 μg/mL for aqueous extracts. For the medicinal plant extracts, the results indicated that both ethanol and water extracts of T. arjuna (1.19 ± 0.01 and 1.63 ± 0.01 μg/mL) demonstrated higher antioxidant capacities relative to ascorbic acid (2.83 ± 0.02 μg/mL), followed by ethanol extracts of T. citrina (2.34 ± 0.01 μg/mL) and P. emblica (2.50 ± 0.01 μg/mL). In addition, the ABTS efficacy of ethanol extracts from P. emblica and T. citrina was comparable to that of the aqueous extract of P. emblica (2.36 ± 0.01 μg/mL) and T. citrina (2.68 ± 0.02 μg/mL), respectively.
The FRAP experiment demonstrated that the ethanol and aqueous extracts of Thai herbal formulation displayed ferric reduction effects comparable to the quercetin standard curve, measuring 177.24 ± 2.39 and 162.01 ± 2.07 mg/g dry extract, respectively. For the medicinal plant extracts, the results were noted that both the ethanol and aqueous extracts of T. arjuna exhibited the greatest FRAP values of 752.00 ± 1.55 and 460.23 ± 20.24 mg QE/g, respectively, followed by T. citrina with values of 527.59 ± 17.32 and 430.88 ± 19.92 mg QE/g, and T. bellirica with values of 451.58 ± 14.30 and 298.13 ± 9.29 mg QE/g. Notably, the ethanol and aqueous extracts of these three species yielded consistent results in this investigation.
Pancreatic Lipase Inhibitory Activity of Thai Herbal Formulation
The pancreatic lipase inhibitory activities of the herbal components and Thai herbal formulation extract were shown in Figure 4 and Table 6. All medicinal plant extracts demonstrated anti-lipase activity in comparison to the standard orlistat (IC50 = 0.0737 ± 0.0091 μg/ml). The ethanol extract of the Thai herbal formulation exhibited pancreatic lipase inhibition over 50%, estimated at 124.79 ± 2.92 μg/ml. The herbal component displaying the most significant pancreatic lipase inhibition was C. zedoaria, with an IC50 value of 39.44 ± 2.28 μg/ml, followed by Z. officinale at 39.79 ± 2.04 μg/ml and P. emblica at 63.34 ± 1.02 μg/ml, respectively. The aqueous extract revealed that the Thai herbal formulation extract presented an IC50 value of 371.18 ± 1.85 μg/ml. Regarding the herbal components in the formulation, G. hanburyi revealed an IC50 value of 81.21 ± 1.38 μg/ml, followed by P. emblica at 201.17 ± 3.18 μg/ml and T. arjuna at 208.75 ± 1.77 μg/ml, respectively.

Pancreatic Lipase Inhibitory Activity of Thai Herbal Formulation Ethanol Extract (A), Thai Herbal Formulation Aqueous Extract (B), and Orlistat (C).
Pancreatic Lipase Inhibitory Activity of the Individual Herbs and Thai Herbal Formulation.

Allium sativum, AS; Curcuma zedoaria, CZ;
Orlistat was used as a positive control of lipase inhibitory activity.
Cytotoxic Activity of Thai Herbal Formulation
The cytotoxic activity of herbal extracts was assessed by MTT test utilizing normal cell lines including L929 cells and Vero cells. The IC50 value was the concentration required to inhibit 50% of cell growth. The results indicated that the aqueous extracts exhibited no inhibitory effect on either cell line (IC50 values above 80 μg/mL), whereas the ethanol extracts presented IC50 values of 18.59 ± 0.58 µg/mL in Vero cells and 39.86 ± 0.02 µg/mL in L929 cells. Doxorubicin was used as a positive control, exhibiting an IC50 of 1.89 µg/mL in the Vero cell line and an IC50 of 0.41 µg/mL in the L929 cell line.
Clinical Efficacy and Safety of Thai Herbal Formulation for Patients with Borderline Hyperlipidemia
Quality Evaluation of Thai Herbal Formulation
The assessment of the exterior characteristics revealed no damage to the exterior of the herbal capsules. The weight variation test of herbal capsules revealed an average weight of 502.24 mg (USP 41-NF 36), with an acceptable range of 452.02 mg to 552.46 mg, and an average weight of 504.28 mg (THP 2018), with an acceptable range of 453.85 mg to 554.71 mg. The evaluated placebo capsules had an average weight of 493.16 mg (USP 41-NF 36), within an acceptable range of 443.84 mg to 542.48 mg, and an average weight of 490.24 mg (THP 2018), within an acceptable range of 441.22 mg to 539.26 mg. The disintegration test of the capsules indicated that disintegration occurred in 19.61 min for the Thai herbal formulation capsule and 13.05 min for the placebo capsule. Consequently, none of the examined capsules higher the permissible limits, and both the weight variation and disintegration time corresponded to the requirements established by USP 41-NF 36 and THP 2018.
Study Participants and Demographic Characteristics
According to the framework of the study, one hundred and five participants offered interest. Among the participants, only sixty-one achieved the inclusion requirements during the screening examination, and nine participants withdrew from the study. In the THF group, there was one for nausea, one for diarrhea, one for stomachache, and two for COVID-19. One due to an accident, two related to COVID-19, and one lost to follow-up in the placebo group. In the end, 61 participants completed the investigation (Figure 5). No serious adverse events were noted during this study.

Flow Chart of the Study.
The demographic characteristics of participants were presented in Table 7. In the THF group, there were 35 patients, including 17 females (56.7%) and 13 men (43.3%). The placebo group also consisted of 35 subjects, including 20 females (64.5%) and 11 males (35.5%). At baseline, there were no notable variations in demographic features and lipid profiles between the two groups.
General Characteristics and Blood Chemistry of Participants in Thai Herbal Formulation and Placebo Groups.

Values are mean ± SD; BMI, Body Mass Index; TC, total cholesterol; TG, triglyceride; LDL-C, low-density lipoprotein-cholesterol; HDL-C, high-density lipoprotein-cholesterol.
Chi-Square Test of Independence; b Wilcoxon Rank Sum Test; c Welch's t-test, d Fisher's Exact Test for Count Data.
Significant differences at P < 0.05.
Efficacy and Safety of Thai Herbal Formulation in Borderline Hyperlipidemia
The values of biochemical parameters in two groups at baseline and after 12 weeks of treatment are shown in Table 8. The results indicated that the THF group exhibited a significant decrease in total cholesterol and low-density lipoprotein cholesterol (P < 0.05), together with decreased triglycerides and increased high-density lipoprotein cholesterol. Conversely, the placebo group demonstrated an increase in total cholesterol, triglycerides, low-density lipoprotein cholesterol, and high-density lipoprotein cholesterol after 12 weeks of treatment. Notably, neither group had any significant changes on the blood levels of alanine transaminase, aspartate aminotransferase, blood urea nitrogen, creatinine, and complete blood count after 12 weeks. The Thai herbal formulation effectively reduced detrimental lipid levels, particularly low-density lipoprotein cholesterol and triglycerides, while increasing advantageous high-density lipoprotein cholesterol levels. Consequently, Thai herbal formulation may diminish the risk factors associated with cardiovascular disease.
Comparison of Variables Before and After the Intervention in Thai Herbal Formulation and Placebo Groups.
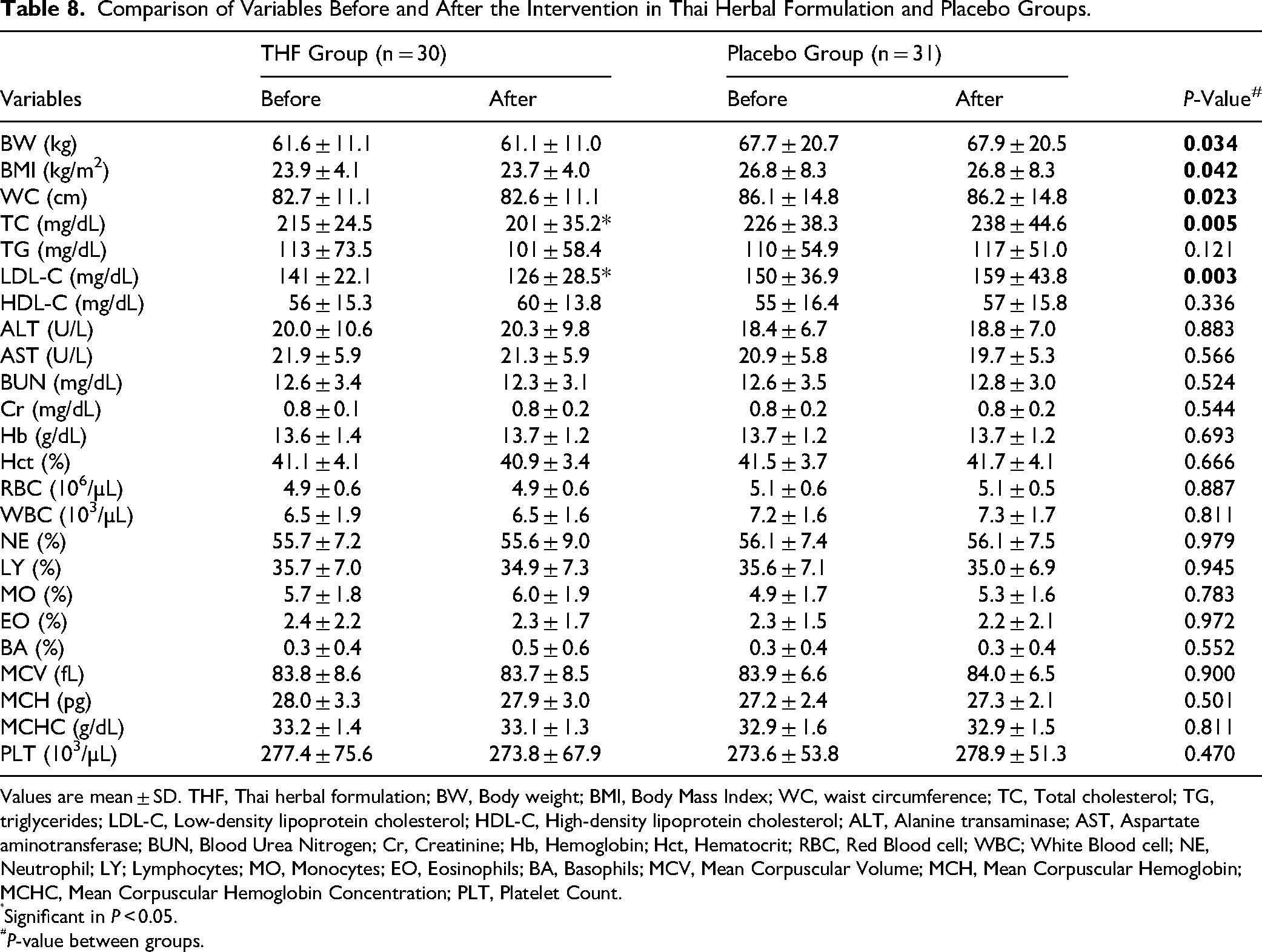
Values are mean ± SD. THF, Thai herbal formulation; BW, Body weight; BMI, Body Mass Index; WC, waist circumference; TC, Total cholesterol; TG, triglycerides; LDL-C, Low-density lipoprotein cholesterol; HDL-C, High-density lipoprotein cholesterol; ALT, Alanine transaminase; AST, Aspartate aminotransferase; BUN, Blood Urea Nitrogen; Cr, Creatinine; Hb, Hemoglobin; Hct, Hematocrit; RBC, Red Blood cell; WBC; White Blood cell; NE, Neutrophil; LY; Lymphocytes; MO, Monocytes; EO, Eosinophils; BA, Basophils; MCV, Mean Corpuscular Volume; MCH, Mean Corpuscular Hemoglobin; MCHC, Mean Corpuscular Hemoglobin Concentration; PLT, Platelet Count.
Significant in P < 0.05.
P-value between groups.
The findings indicated that the Thai herbal formulation was both efficient and safe in lowering cholesterol, low-density lipoprotein cholesterol, and triglycerides, while increasing high-density lipoprotein levels, hence demonstrating anti-hyperlipidemic properties in borderline hyperlipidemia.
Effectiveness of Thai Herbal Formulation in Participants Consuming High-fat Diets
The effect of high-fat food consumption on total cholesterol and low-density lipoprotein cholesterol levels, both pre- and post-intervention, in the THF and placebo groups was shown in Table 9. The THF group indicated decreases in total cholesterol and low-density lipoprotein cholesterol levels in participants with high-fat dietary habits, while the control group exhibited statistically significant increases in both total cholesterol and low-density lipoprotein cholesterol levels compared to baseline measurements before treatment (Figure 6 and Table 9). The findings demonstrated that Thai herbal formulation successfully exhibited anti-hyperlipidemic properties by reducing total cholesterol and low-density lipoprotein levels in patients with borderline hyperlipidemia.

Trends in Change in Total Cholesterol (A) and LDL Cholesterol (B) Levels in Participants Before and After the Intervention of the Study. *Significant Reductions in Total Cholesterol and LDL Cholesterol (P < 0.05), #Significant Difference Between the THF Group and the Placebo Group (P < 0.05).
Effects of High-fat Foods Intakes on Total Cholesterol and low-Density Lipoprotein Cholesterol Levels Before and After the Intervention in Thai Herbal Formulation and Placebo Groups.

THF, Thai herbal formulation; TC, Total cholesterol; LDL-C, Low-density lipoprotein cholesterol.
Values are mean ± SD.
*Significant in P < 0.05.
P-value between groups.
Discussion
Traditional Thai herbal medicine acts as an alternative and complementary approach for controlling hyperlipidemia due to its low toxicity and therapeutic effects.5,6 Notably, multicomponent traditional Thai herbal medicine targets multiple pathways and biological processes to tackle complicated problems due to its diversity and complexity.5,8 In this study, based on the principles of traditional Thai medicine, ten herbal medicines including A. sativum, C. zedoaria, D. scandens, G. hanburyi, P. emblica, T. arjuna, T. bellirica, T. chebula, T. citrina, Z. officinale have been selected for the patients with borderline hyperlipidemia (Table 1). The present study aimed to investigate the biological and clinical efficacy of Thai herbal formulation for lowering blood lipids in patients with borderline hyperlipidemia.
Ten medicinal plants, including A. sativum, C. zedoaria, D. scandens, G. hanburyi, P. emblica, T. arjuna, T. bellirica, T. chebula, T. citrina, Z. officinale, have been used in traditional Thai medicine as healthy herbal remedies for centuries. The chemical constituents present in ten medicinal plants have been recognized to provide beneficial health effects, especially as anti-oxidant and anti-inflammatory compounds with the capacity to act as immunomodulators (Table 1). Moreover, bioactive compounds, mostly phenolics and flavonoids derived from medicinal plants (Tables 3 and 4), have antioxidant and anti-inflammatory properties that effectively protect the vascular endothelium, inhibit lipid oxidation, and reduce lipid levels. 8
Many studies have shown that A. sativum,11,12 C. zedoaria, 13 P. emblica,18,19 T. arjuna,20,21 T. bellirica,22,23 T. chebula, 24 Z. officinale 28 and could individually reduce blood total cholesterol, low-density lipoprotein, and triglycerides while elevating high-density lipoprotein levels in humans as well as in animals. This study demonstrated that a Thai herbal formulation could regulate serum levels of total cholesterol, low-density lipoprotein cholesterol, triglycerides, and high-density lipoprotein cholesterol to improve dyslipidemia, mainly due to the phenolic and flavonoid compounds derived from medicinal plants. As shown in Tables 3 and 4 and Figure 3, the main effective components of Thai herbal formulation for hyperlipidemia treatment were phenolic compounds such as ((+)-chebulic acid, 4-glucogallic acid, gallic acid, ellagic acid, piscidic acid, 6-gingerol) and other bioactive components. Prior researches have established that numerous phenolic compounds significantly impact human cardiovascular health by neutralizing and inhibiting the production of reactive oxygen species, lowering blood pressure, enhancing endothelial function through elevated plasma epicatechin levels, promoting the synthesis of endothelial vasodilators, increasing high-density lipoprotein cholesterol in prehypertensive individuals, decreasing low-density lipoprotein cholesterol and very low-density lipoprotein cholesterol, downregulating pro-inflammatory cytokines to mitigate inflammation, and resulting in reductions of low-density lipoprotein cholesterol levels in humans.47,48 Moreover, gallic acid and 6-gingerol, naturally occurring compounds, have lipid-lowering effects via many mechanisms, including the regulation of squalene monooxygenase and HMG-CoA reductase, which are essential enzymes in cholesterol manufacture.49,50 Gallic acid has shown the capacity to improve lipid profiles and reduce markers of metabolic syndrome in animal studies. 49 6-Gingerol has shown the capacity to reduce cholesterol levels and may inhibit pancreatic lipase activity, hence lowering cholesterol absorption. 50
Thai herbal formulation has been thoroughly examined as therapeutic targets for hyperlipidemia. The four mechanisms involving herbal medicines reduce lipid levels have been determined that blockage of cholesterol absorption in enterocytes, reduction of cholesterol production, enhancement of reverse cholesterol transport, and facilitation of cholesterol excretion in the liver. 7 According to the lipid-lowering mechanisms, Thai herbal formulation may reduce cholesterol absorption and synthesis, along with fatty acid production, resulting in decreasing cholesterol levels. Thai herbal formulation may inhibit human enzymes critical for cholesterol synthesis, such as squalene monooxygenase and HMG-CoA (3-hydroxy-3-methylglutaryl-coenzyme A) reductase. In addition, Thai herbal formulation may lower low-density lipoprotein cholesterol levels by inhibiting hepatic cholesterol 7α-hydroxylase and HMG-CoA reductase, reducing pentose-phosphate pathway activities, enhancing bile acid excretion, and modulating microsomal triglyceride transfer protein and cholesteryl ester transfer protein activities, while also preventing hepatic fatty acid synthesis, mainly through its main phenolic compounds and/or other constituents.7,8,47,48 In conclusion, the Thai herbal formulation effectively reduced detrimental lipid levels, particularly low-density lipoprotein cholesterol and triglycerides, while enhancing beneficial high-density lipoprotein cholesterol levels, therefore establishing its efficiency in controlling hyperlipidemia. Therefore, Thai herbal formulation may reduce the risk factors associated with cardiovascular disease and diminish cardiovascular morbidity. The findings indicated that the Thai herbal formulation could potentially act as a novel therapeutic medication for managing borderline hyperlipidemia. Further investigation through large-scale randomized controlled trials in hyperlipidemic patients is necessary to understand the efficacy and therapeutic effects of the Thai herbal formulation.
Conclusions
The findings indicated that the Thai herbal formulation including A. sativum, C. zedoaria, D. scandens, G. hanburyi, P. emblica, T. arjuna, T. bellirica, T. chebula, T. citrina, and Z. officinale exhibited potential as a herbal therapeutic approach for borderline hyperlipidemia management. The therapeutic benefits of the Thai herbal formulation could be attributed to phenolic compounds as the major compounds in this formulation. However, further investigation through large-scale randomized controlled trials in hyperlipidemic patients is essential to establish the efficacy and therapeutic advantages of the Thai herbal formulation.
Footnotes
Acknowledgements
Not applicable.
Ethics Approval and Consent to Participates
Ethical approval for a clinical study was obtained from the Human Research Ethics Committee of Faculty of Traditional Thai medicine, Prince of Songkla University (reference number: EC.63/TTM.01-001). The protocol was also registered in Thai Clinical Trials Registry (registration ID: TCTR20241111003).
Author Contributions
PB, SS, TS conceptualized, designed and executed of study, PB, NN, TS analyzed and wrote the first drafted of manuscript, PB, NN, TS participated in the tables production. TS supervised, wrote, reviewed and edited the manuscript. All authors have reviewed and approved the manuscript prior to submission.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Faculty of Traditional Thai Medicine, Prince of Songkla University, (grant number TTM6404242S).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
