Abstract
Background
The Tibetan medicine Ershiwuwei Guijiu Pill (EWGP), a classic Tibetan medicine prescription for the treatment of postmenopausal osteoporosis (PMOP) in the Qinghai–Tibet region, has attracted extensive attention due to its curative effects on gynecological diseases. However, its chemical ingredients and molecular mechanism are still unclear.
Aim of the study
To analyze the chemical constituents and effective serum chemical metabolites of EWGP and to explore the molecular mechanism of EWGP in treating PMOP through network analysis and experimental validation.
Methods
The ethanol extract of EWGP and its drug-containing serum were detected by liquid chromatography-mass spectrometry (LC–MS), and the chemical constituents were analyzed and identified. SwissTarget prediction was used to predict the corresponding potential target genes of the identified chemical components. Thereafter, a visualization network of the components and corresponding targets was constructed with Cytoscape software. Moreover, a specific disease database for animals was used to search and filter for osteoporosis (OP) targets, and a drug-disease target protein–protein interaction (PPI) network was constructed. Cytoscape 3.7.0 was used for visualization and cluster analysis, and R Studio was used for GO and KEGG enrichment analysis. AutoDock Tools were applied for molecular docking of the serum metabolites and specific target proteins. The potential mechanism of EWGP in preventing and treating PMOP was predicted by network pharmacology analysis and was experimentally studied and verified in vivo and in vitro.
Results
A total of 199 chemical substances were identified in the ethanol extract, and 11 were found in the serum. A total of 419 predicted targets and 128 target genes related to osteoporosis were screened. There were 16 common targets identified between the predicted targets and OP genes. Following the enrichment analysis, 16 KEGG signaling pathways and 63 GO biological process items were identified. The results of molecular docking showed that the main active compounds may be Protopine, Hetisine, Piperine, Visaminol, Boldine, and Trigonelline, and the specific targets may be CYP17A1, ESR2, MAPK14, and the vitamin D receptor (VDR). The results of cell and animal experiments showed that EWGP may improve bone metabolism via estrogen and calcium signaling pathways regulated by estrogens and calcium ions.
Conclusions
EWGP contains multiple herbal drugs and treats PMOP through multiple targets and signaling pathways. We preliminarily tested the chemical compounds of EWGP, especially in the serum, to determine the chemical metabolites of EWGP and revealed the molecular mechanism of EWGP in preventing and treating PMOP; moreover, we used computer-virtual molecular docking and experiments for preliminary verification of the core targets of network pharmacology analysis.
This is a visual representation of the abstract.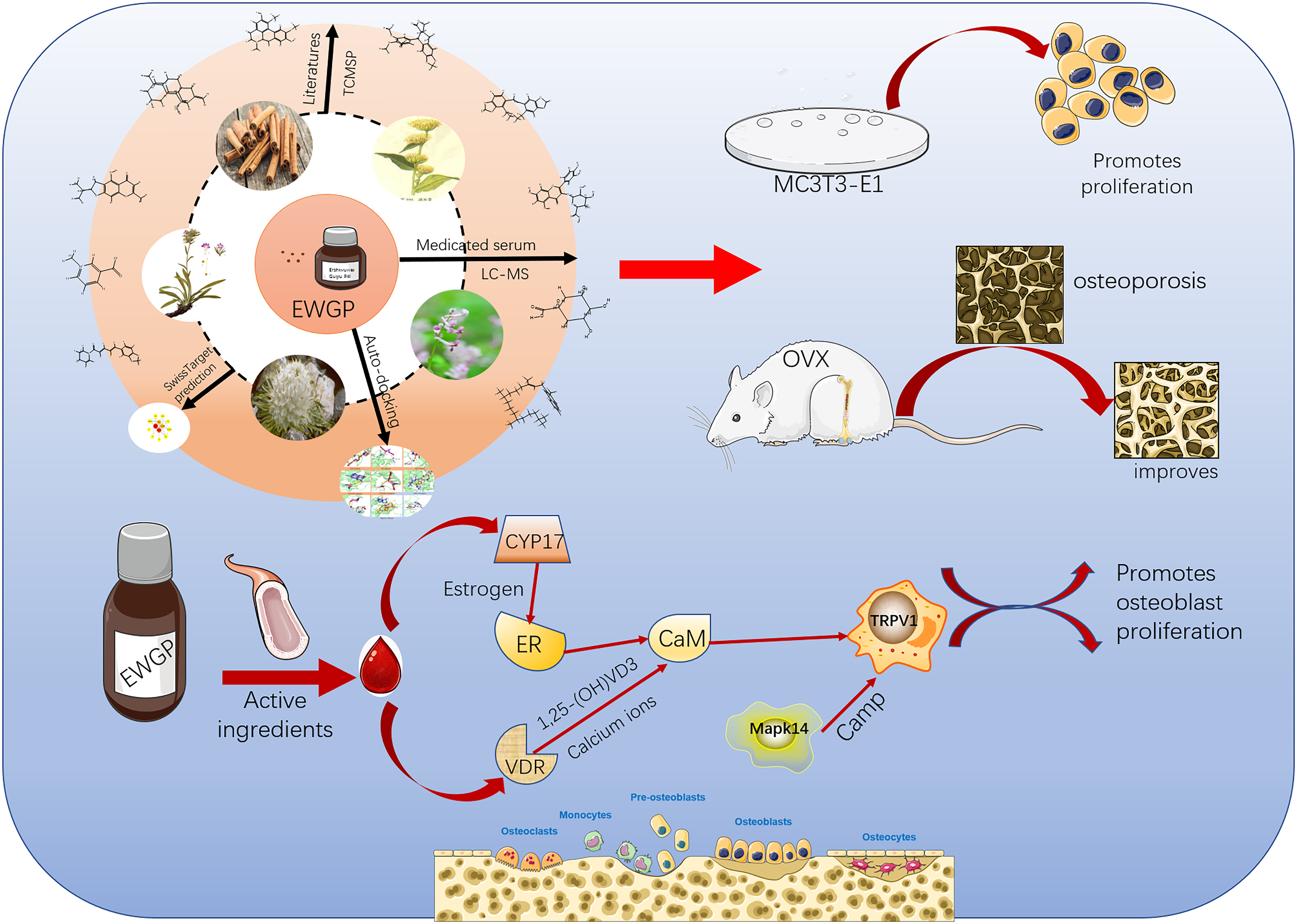
Keywords
Background
Osteoporosis (OP) is a systemic and progressive disease. An imbalance between bone resorption and bone formation is a major cause of decreased bone mineral density (BMD) and deterioration of the trabecular bone structure and is characterized by low bone strength and increased fracture risk. 1 The “cliff-like” decline in estrogen levels during the perimenopausal period leads to an upward trend in the incidence of OP in postmenopausal women. 2 According to the 2018 epidemiological survey report on postmenopausal osteoporosis (PMOP), the prevalence rate of OP is 16% in individuals aged 40 to 49 years, 37.5% in individuals aged 60 to 69 years, and 52.9% in individuals aged > 70 years. 3 In the clinical guidelines for PMOP, antiresorptive and anabolic drugs are the main recommended treatments. However, long-term use of these drugs induces gastrointestinal symptoms and increases the risk of cardiovascular disease (CVD) and obesity, which reduce the quality of life of patients.4–6
Traditional Chinese medicine (TCM) has unique advantages in treating chronic diseases such as osteoporosis. The Tibetan medicine Ershiwuwei Guijiu Pill (EWGP), sourced from the Qinghai Provincial Tibetan Hospital (Qingyao: Z20140539, specification: 0.35 g × 45 pills per bottle), is an ethnic prescription for the treatment of PMOP in the Qinghai–Tibet region. It was described in the treatises of Tibetan medicine and has been applied in clinical treatment for more than 300 years. The EWGP includes multiple special medicinal herbal drugs and materials from the Qinghai–Tibet region, such as Sinopodophyllum hexandrum, roots of Inula racemosa, pomegranate, Chebulae fructus, cinnamon, and Phyllanthus emblica. 7 Mollugin (the main chemical ingredient of Rubia tibetica Hook., 8 piperine (pepper), 9 cinnamic acid (cinnamon), 10 and pomegranate seeds 11 can improve the BMD in ovariectomized rats. The herbal drugs of EWGP are shown in Table S4. In the future, single-herb drugs and extracts of TCMs may be potential substances for the prevention and treatment of PMOP, but these substances have been less studied in terms of the exact mechanism of action of these natural medicines.12,13
Network pharmacology connects active ingredients with disease targets through network topology, reflecting the potential molecular mechanism of prescriptions via network informatics. It is widely used for complex prescription screening of targets and mechanisms of action. 14 At the same time, network pharmacology can be combined with a variety of bioinformatics methods to quickly screen the core targets and mechanisms of herbs; for example, Xiaobo Zhang combined network pharmacology, metabolomics, and transcriptomics to explore the effective targets and drug metabolism characteristics of Huanglian-Hongqu herb pair in the prevention and treatment of non-alcoholic fatty liver disease. 15
Moreover, serum chemistry provides another way to screen active components of prescriptions. 16 Combined with serum chemistry and network pharmacology, the exploration of the potential mechanisms of complex prescriptions could be effectively improved. Molecular docking, a computer simulation technique, can simulate the interaction between small-molecule chemicals and proteins and calculate the binding energy and binding sites between two proteins. This approach can predict the affinity between small-molecule compounds and target genes well.
We used a combination of serum chemistry, network pharmacology, computer-virtual molecular docking, and other methods to analyze the potential targets and molecular mechanisms of EWGP in the prevention and treatment of PMOP. The chemical substances and main active metabolites of the EWGP were detected by liquid chromatography-mass spectrometry (LC–MS) and preliminarily verified by animal and cell experiments.
Materials and Methods
Tibetan Medicine Ershiwuwei Guijiu Pills (Qinghai Provincial Tibetan Hospital, Qinghai, China), estradiol valerate tablets (Bayer Pharmaceutical Healthcare Co., Ltd, China), chemical standards (HPLC ≥ 98%) and estradiol (HPLC ≥ 98%) were obtained from Shanghai Yeyuan Biotechnology Co., Ltd, Shanghai, China. The cell counting kit-8 (CCK-8) assay kit, 5-ethynyl-2′-deoxyuridine (EdU) assay kit, phosphate-buffered saline, and 4,6-diamino-2-phenyl indole (DAPI) were used (Beyotime Biotechnology Co., Ltd, China). We used a chemiluminescence (ECL) reagent from Absin Biotechnology Ltd. (Shanghai, China). The primary antibodies used were CYP17A1 (GB112095, Servicebio Co., Ltd, Wuhan, China); ESR2 and Mapk14 (41007-1-AP,66234-1-lg, Proteintech Group, Inc., Biotechnology Co., Ltd, Wuhan, China); and VDR, TRPV1 and CAMKIIα (WL03312, WL02734, WL03453, Wanleibio Co., Ltd., Liaoning, China).
Detection of Chemical Constituents in the EWGP and rat serum
The chemical composition of the Tibetan medicine EWGP and serum was examined using UPLC-Q-Exactive-Orbitrap-MS. The components of the medicinal herb drugs used in the prescription were retrieved from the Traditional Chinese Medicine Systems Pharmacology Database and Analysis Platform (TCMSP, https://tcmspw.com/tcmsp.php) database and collected, and the literature supplied other active components.
Preparation of test samples: 200 mg of EWGP was accurately weighed, added to 10 mL of 75% ethanol, ultrasonically treated at room temperature for 1 h, centrifuged at 3000 × g and 4 °C for 10 min, and the supernatant was collected. Nitrogen was blown to dry the ethanol, and ethanol was added to 2.5 mL. The liquid was filtered through a 0.22 µm filter membrane, and 10 µL was injected continuously for 3 injections.
Preparation of standard substances: The preparation of chemical standard substances is shown in Supplementary Table S1. After preparing the mother liquor of each chemical standard, 20 µL of each standard mother liquor was taken, and a total of 380 µL and 620 µL of 50% methanol were added to prepare the mixed standard mother liquor. The mixed standard mother liquor was diluted ten times with 50% methanol to obtain the working liquid, and 10 µL was injected.
Preparation of serum samples: The administration group was given EWGP suspension liquid by gavage, and the blank group was given normal saline. Blood samples were collected from the orbital vein at 30 min, 1 h, 2 h, 3 h, 4 h, and 5 h after intragastric administration. The samples were centrifuged at 4 °C and 3000 rpm for 10 min. Afterward, 800 μL of methanol was added to the mixture, and the mixture was oscillated for 1 min, sonicated in an ice bath for 10 min, and centrifuged at 10 000 × g for 10 min. Afterward, the supernatant was dried with nitrogen, redissolved in 100 μL of 50% methanol, and centrifuged at 10,000 × g at 4 °C for 10 min. Then, 90 μL of the supernatant was collected, and 10 μL was injected.
The chromatographic conditions used were as follows: For the ACQUITY UPLC® CSH C18 (2.1 × 100 mm, 1.8 μm) (Waters, Milford, MA, USA) chromatographic column, the column temperature was 40 °C, and the flow rate was 0.3 mL/min. The chromatographic mobile phase A was a 0.1% formic acid aqueous solution, and B was 0.1% formic acid acetonitrile; the chromatographic gradient elution procedure was as follows: 0–5 min, 5% B; 5–20 min, 5% 100% B; 20–25 min, 100% B; 25–25.1 min, 100% 5% B; and 25.1–30 min, 5% B.
Mass spectrometry conditions: The samples were detected by electrospray ionization (ESI) in positive ion (+) and negative ion (-) modes. The ionization conditions were as follows: spray voltage, 3.8 kV (+) and 3.2 kV (-); capillary temperature, 320 (±); sheath gas, 30 (±); aux gas, 5 (±); probe heater temperature, 350 °C (±); and S-Lens RF Level, 50.
The mass spectrometry acquisition time was 30 min. The parent ion scan range was 70–1050 m/z, the MS resolution was 70,000 @ m/z 200, the automatic gain control (AGC) target was 3e6, and the maximum IT was 100 ms. Secondary mass spectrometry analysis was performed according to the following methods. The MS2 scan was triggered after each full scan to collect 10 parent ions with the highest intensity: MS2 resolution, 17,500 @ m/z 200; AGC target, 1e5; MS2 maximum IT, 50 ms; MS2 activation type, HCD; and isolation window, 2 m/z. The normalized collision energies (Setpped) were 20, 30, and 40.
Qualitative analysis was conducted by importing the UPLC-Q Exactive experimental data into Xcalibur software. This analysis utilized the relative molecular mass (δ < 10 ppm), which was integrated with secondary fragmentation information, retention time, and corroborative literature references.
Network Pharmacology-Based Analysis
Prediction of Potential Drug Targets and Construction of a Component-Target Network
Based on the qualitative analysis results of the chemical constituents of EWGP, the 3D/2D molecular formulas of the compounds were searched for and downloaded from the PubChem database (https://pubchem.ncbi.nlm.nih.gov). The molecular formula was uploaded to the Swiss Target Prediction database (www.swisstargetprediction.ch) for target prediction, and the species was selected as Rattus norvegicus. The top 15 targets were collected according to the prediction results, and all the targets with a credibility value of 1. The website makes online predictions based on the functional groups of the chemical formula, sorting the results based on probability. A comprehensive observation reveals that the top 15 targets are high-probability targets, effectively screening the effective targets of EWGP for PMOP prevention and treatment. The targets and active components were imported into Cytoscape 3.7.0 software to construct the “drugs-active components-targets” network of EWGP.
Acquisition of OP Target Genes and Construction of a Protein–Protein Interaction (PPI) Network of Common Targets
The keyword “osteoporosis” was used to retrieve information on osteoporosis disease-related genes in rats from the Ensembl database (https://asia.ensembl.org), which includes professional animal disease research data. A Venn diagram was constructed to show the intersection targets of EWGP and OP. To fully demonstrate the interactions between the targets, the intersection targets were uploaded to the STRING database (https://string-db.org/). The species was selected as ‘Rattus norvegicus, and the credibility was set to 0.400. The PPI information was obtained and imported into Cytoscape 3.7.0 software. According to the degree value and the Cytohubbe plug-in, the core target PPI network was constructed.
GO Analysis and KEGG Pathway Enrichment Analysis
The gene symbol IDs of the targets were determined by Perl, after which Gene Ontology (GO) analysis and Kyoto Encyclopedia of Genes and Genomes (KEGG) pathway enrichment analysis were performed by using the “ClusterProfiler” package in R Studio software. We selected the top 15 GO terms and pathways to construct bar graphs and bubble graphs.
Computer-Virtual Molecular Docking
The chemical metabolites absorbed in the serum were regarded as the active metabolites of EWGP, and the top 10 target genes were calculated by the PPI network for molecular docking. The active metabolites served as the ligand, and the top 10 specific targets were used as the receptors. The 3D protein structure of the target gene was downloaded from the Protein Data Bank (PDB) database (www.rcsb.org), and PyMOL software was used to remove redundant conformations and water molecules from these proteins. In AutoDock 4, these selected receptor proteins and drug ligands were subjected to computer-virtual molecular docking, after which, according to the binding energy-ranked results, we recorded and analyzed the affinity values. The PDB IDs of these proteins are shown in Table S5.
Animal Experimental Verification
Animals
A total of 40 female specific pathogen-free (SPF) nonpregnant Sprague–Dawley (SD) rats, 4 months old, weighing 220 ± 20 g (Certificate Number: SCXK 2023-002), were obtained from the Experimental Animal Center of the Medical Department of Xi’an Jiaotong University. All rats had access to sufficient food and water and were maintained at a controlled ambient temperature of 22 ± 1 °C under a 12/12 h light/dark cycle. The experimental procedures adhered to the guidelines set by the Experimental Animal Ethics Committee of Qinghai University’s College of Medicine (QHDX2021000541).
Experimental Animal Modeling and Administration
After one week of acclimatization, 40 nonpregnant female SD rats were randomized into four groups: sham-operated (sham), ovariectomized (OVX), estradiol (E2), and EWGP. For the sham-operated group, only the adjacent adipose tissue of comparable size to the ovary was excised, while in the other groups, the ovaries were removed to establish the PMOP animal model.
Before the surgical procedure, the rats were subjected to a 12-h fasting period, but water was not withheld. A small animal-breathing anesthesia machine was used for anesthesia, and the anesthetic was isoflurane. Following deep anesthesia, the abdominal hair was removed, and the underlying skin was disinfected using iodophor. An incision approximately 2 to 3 cm in length was made along the midline of the rectus abdominis, revealing the V-shaped uterus, the distinctive mulberry-like ovaries, and the uterine horns. In all groups except for the sham-operated group, bilateral ovaries were excised to establish the PMOP animal model. For the sham-operated group, only adipose tissue of a size comparable to the ovaries was removed. To prevent postoperative infections, penicillin was administered intramuscularly at a dose of 200,000 units per rat for three consecutive days. Three days post-surgery, vaginal smears were obtained daily for five days from each rat. The absence of an estrus reaction indicated successful modeling. Oral dosing commenced one-week post-surgery. The E2 group received a daily dosage of 0.1 mg/kg based on equivalency to the clinical dose, while the EWGP group was given a dosage of 441 mg/kg. The remaining two groups were administered an equivalent volume of distilled water via gavage for 12 weeks. The group details are provided in Supplementary Table S2. After the last administration, the rats in each group were fasted for 12 h and anesthetized by a small animal respiratory anesthesia machine, after which blood was taken from the abdominal aorta. The femur was separated, the attached fascia was removed, and the samples were stored in a refrigerator at 80 °C.
Observation of the Microstructure of the Femur by Scanning Electron Microscopy (SEM)
Glutaraldehyde (2.5%) was added to the 4% PFA of the fixed femur to continue to soak the femur for 24 h, after which the femur was treated with a 15% sodium hypochlorite solution for 6 h and then washed with distilled water repeatedly after ultrasonic cleaning for 10 min. The tissues were soaked in 50%, 70%, 90%, or 100% ethanol for 10 min each time they were subjected to gradient dehydration. After drying, the samples were sprayed with gold, which was a process of conductive coating, and coronal sections of the femurs were taken by SEM for bone microstructure observation.
Observation of Calcium Deposition in Undecalcified Femurs by Von Kossa (VK) Staining
The bone tissue was embedded in methyl methacrylate, and the embedded tissue was sliced with a robust tissue slicing machine with a tungsten steel blade. The slices were immersed in ethylene glycol ethyl ether acetate I for 6 h at 37 °C, ethylene glycol ethyl ether acetate II overnight at 37 °C, ethylene glycol ethyl ether acetate III at room temperature for 10–15 min, ethylene glycol ethyl ether acetate IV at room temperature for 10–15 min, 100% I ethanol for 10 min, 100% II ethanol for 10 min, 95% ethanol for 10 min, 90% ethanol for 10 min, 80% ethanol for 10 min, tap water washing, and slices with ultrapure water leaching 5 times. Staining was performed according to the instructions of Von Kossa’s staining kit.
In Vitro Experimental Verification
Preparation of the Ethanol Extract of the EWGP
Two hundred milligrams of EWGP were accurately weighed, 10 mL of 75% ethanol were added, and the mixture was ultrasonically treated at room temperature for 1 h. After centrifugation at 3000 × g at 4 °C for 10 min, the supernatant was collected. Nitrogen was blown to dry the ethanol, and a volume of anhydrous ethanol was added to prepare a stock solution of 1 mg/ml, which was filtered through a 0.22 µm filter membrane.
Cell Culture and Treatment
MC3T3-E1 subclone 14 cells (No. STCC20026G) were purchased from Wuhan Sevier Biotechnology Co., Ltd The cells were cultured in a medium supplemented with 10% FBS (α-MEM + 10% FBS + 1% P/S) at 37 °C in a 5% CO2 incubator. When the cell density reached 80%, the cells were passaged and cultured. Cells in the logarithmic growth phase were taken for the experiment.
Cell Counting Kit-8 (CCK-8) Assay
The cells were inoculated into 96-well plates at a density of 3 × 103 cells/well. After treatment with different concentrations of EWGP (100 µg/mL, 80 µg/mL, 60 µg/mL, 40 µg/mL, 20 µg/mL, 10 µg/mL, 5 µg/mL, 0 µg/mL) for 6 h, 24 h, or 48 h, 10 µL of CCK-8 solution was added to each well. After incubation at 37 °C for 1–2 h, the absorbance at 450 nm was measured.
After treatment with EWGP (at a suitable concentration and 0 µg/mL) and estradiol (0.1 nmol/mL) for 48 h, 10 µL of CCK-8 solution was added to each well.
5-Ethynyl-2′-Deoxyuridine (EdU) Assay
The cells were inoculated into 24-well plates at a density of 6 × 104 cells/well. After treatment with EWGP (80 and 0 µg/mL) and estradiol (0.1 nmol/mL) for 48 h, the cells were treated with 500 μL of 5-ethynyl-2′-deoxyuridine (EdU) for 2 h. After washing with phosphate-buffered saline (PBS) three times and 1 mL of 0.3% Triton X-100 in turn, the cells were incubated in 100 µL of click reaction solution in the dark at room temperature for 30 min, followed by 100 μL of DAPI for 10 min. The cells were washed with PBS after each step and then observed via microscopy.
Western Blotting
Total protein was extracted from the femur and cells according to the instructions of the total protein extraction kit. A BCA assay kit (Solarbio, Beijing, China) was used to determine the protein concentration of each sample. Protein electrophoresis was performed using an 8%, 12% SDS–PAGE gel. The electrophoresis conditions were as follows: 80–120 V gradient electrophoresis, transfer to a nitrocellulose membrane, and transfer to a membrane at 200 mA for 60–90 min. After the membrane was transferred, the membrane was blocked with serum-free rapid blocking solution for 5, 10 min, incubated with primary antibody for 12 h at 4 °C, eluted with TBST, and incubated with secondary antibody for 2 h. After the antibody incubation, strip development was performed under the development system.
Statistical Analysis
All the statistical data were analyzed using GraphPad Prism software V8.0. Differences between the two groups were analyzed by a t-test, and comparisons between groups were evaluated by one-way analysis of variance (ANOVA). The experimental data are expressed as the mean ± SD. p < 0.05 indicated statistical significance.
Results
Identification of Chemical Components of EWGP
As a classic Tibetan medicine, most of the medicinal materials in the prescription of EWGP are unique to the Qinghai-Tibet Plateau, which leads to few reports on related botanic herb medicines in English literature. Therefore, in the TCMSP database, we could only retrieve Piperis Fructus, Chebulae Fructus, cinnamon, Phyllanthi Fructus, Lycii Fructus, Myristicae Semen, Linderae Radix, Dalbergiae Odoriferae Lignum and sort out the chemical components of each medicine collected in the database, for a total of 189 chemical components. The other herbal medicines were retrieved by CNKI for data collection, with a total of 332 chemical components. Combined with ion retention time, molecular weight, protonation type, lysis fragment size, peak, mass spectrometry, etc., for comprehensive analysis and matching of the corresponding chemical components. LC–MS/MS detection identified 199 chemical components of the EWGP (Table S3, Figure 1). The chemical substances were divided into 9 main categories: 26 flavonoids, 7 isoflavones, 7 cinnamic acids and their derivatives, 22 fatty acyls, 18 isoprene glycol lipids, 15 organic oxygen compounds, 15 carboxylic acids and their derivatives, 14 benzene and substituted derivatives, and 75 other categories. When 75% ethanol is used as a solvent, it can be used to extract water-soluble and alcohol-soluble substances. The extraction of EWGP from ethanol can lead to the acquisition of more chemicals, thus comprehensively elaborating the material basis of EWGP. At the same time, considering that some Tibetan individuals have the habit of using Baijiu (white wine) to take traditional Chinese Tibetan medicines, so 75% ethanol is more suitable for extraction and impurities.

Ion Current Diagrams of the Ethanol Extract, Standard Substances, and Blood Metabolites of the EWGP. A. Positive ion Flow Diagram of Standard Substances; B. Positive ion Flow Diagram of EWGP; C. Positive ion Flow Diagram of Blood Metabolites; D. Negative ion Flow Diagram of Standard Substances; E. Negative ion Flow Diagram of EWGP; F. Negative ion Flow Diagram of Blood Metabolites.
Identification of Chemical Metabolites Absorbed into serum
By comparing the results of drug-containing serum mass spectrometry with those of the EWGP and blank serum control groups, 11 metabolites absorbed into the serum were identified (
Identification of Metabolites Absorbed in serum of EWGP.

Network Pharmacology Analysis of EWGP
EWGP Herbs Drug-Ingredient-Target Network Diagram
According to the credibility value in the predicted target results, the top 15 targets and all the targets with a credibility value of 1 were selected. After removing the repeated targets, a total of 419 predicted targets were obtained. According to the corresponding relationships of herbs drugs-chemical ingredients-targets, a network diagram of 622 nodes and 386262 edges was constructed (Figure 2A), where red dots represent medicinal herbs drugs, yellow squares represent medicinal chemical components, and blue diamonds represent disease targets.

The Results of Network Pharmacology and Molecular Docking. A. Diagram of the Herbs Drugs-chemical Ingredients-Targets Network, in Which Red Represents Medicinal Herbs Drugs in Prescription, Yellow Represents Chemical Substances, and Blue Represents Predicted Targets; B. PPI Network of Common Targets; C. GO Functional Enrichment Analysis Results of Predicted Targets; D. KEGG Pathway Enrichment Analysis Results of Predicted Targets.
To highlight the correlation between the ingredients of the drug and the predicted targets, the area of the point is related to the degree value when constructing the network diagram; namely, the larger the area of the point is, the greater the degree value. According to the degree values, piperanine, benzenepropanamide, pellitorine, 1-piperidine carboxaldehyde, tetrahydropalmatine, nerolidol, dibutyl phthalate, niacin, atropine, and podofilox were the top 10 metabolites and may be the core metabolites of EWGP in the prevention and treatment of PMOP. Podofilox is not only a metabolite with a large degree of value in network analysis but also one of the metabolites absorbed into the blood. According to the analysis of the combined prescriptions, Sinopodophyllum hexandrum is the main drug and induces the main therapeutic effect. Ca2, Alox5, Ptgs2, Akr1b1, Ar, Adora1, Parp1, Gsk3b, Adora2a, and Sigmar1 were the top 10 targets according to their degree of correlation, which may be the core targets.
The PPI Network of EWGP and OP Common Targets
In the Ensembl database, 128 disease-related genes linked to osteoporosis in rats were obtained. A total of 16 common genes were obtained, namely, Pnp, Mmp9, Ar, Cyp17a1, Ptger4, Htr2c, Ctsk, Cyp19a1, Ace, Vdr, Esr2, Esr1, Mgll, Mapk14, Cyp24a1, and Adcy5, which may play key roles in the treatment and prevention of PMOP by EWGP. The above 16 gene targets were uploaded to the STRING database for PPI prediction. The interaction information was imported into Cytoscape 3.7.0 software for visual network construction (Figure 2B). The calculation results of the ctyoHubba plug-in showed that the top 10 hub genes according to the Matthews correlation coefficient (MCC) algorithm were as follows: Esr1, Esr2, Cyp19a1, Ar, Mmp9, Cyp17a1, Mapk14, Ace, and Ctsk; the top 10 hub genes according to the multinetwork clustering (MNC) algorithm were Cyp17a1, Esr2, Mapk14, Vdr, Ar, Cyp19a1, Ace, Esr1, Mmp9, and Ctsk; and the top 10 hub genes according to degree ranking were Cyp17a1, Esr2, Mapk14, Vdr, Ar, Cyp19a1, Ace, Esr1, and Mmp9. According to the comprehensive ranking of the three algorithms, the top 10 genes were Cyp17a1, Esr2, Mapk14, Vdr, Ar, Cyp19a1, Ace, Esr1, Mmp9, and Ctsk.
GO Analysis and KEGG Pathway Enrichment Analysis
KEGG pathway enrichment analysis of 419 predicted targets revealed 83 signaling pathways, such as neuroactive ligand-receptor interaction, calcium signaling pathway, cAMP signaling pathway, serotonergic synapse, and glutamatergic synapse pathways (the top 15 enrichment results are shown in Figure 2-C). GO functional enrichment analysis revealed 214 biological functions, such as neurotransmitter receptor activity, postsynaptic neurotransmitter receptor activity, G protein-coupled amine receptor activity, amide binding, and serotonin receptor activity. The top 15 enrichment results are shown in Figure 2D.
To further screen the specific signaling pathways of EWGP in the prevention and treatment of PMOP, the KEGG pathway and GO functional enrichment analyses were performed on 16 common targets. Sixteen specific signaling pathways and 63 biological functions were obtained (the top 15 enrichment results are shown in Figure 3A-B). The two enrichment results were intersected to obtain 13 common signaling pathways. According to the KEGG pathway annotation and adjusted p-values, the main signaling pathways involved estrogen synthesis, estrogen receptor expression, and calcium ion regulation.

A. GO Functional Enrichment Analysis Results of Common Targets; B. KEGG Pathway Enrichment Analysis Results of Common Targets; C. Heatmap for Binding Energies of Molecular Docking; D. Binding Sites of Molecular Docking.
Molecular Docking
Ten blood-absorbed chemical metabolites were docked with the top 10 gene targets obtained by comprehensive calculation. The binding energy of the docking was determined, and the results were used to construct a heatmap (Figure 3C-D). It is generally believed that the binding energy is less than 4.25 kcal/mol, suggesting that the ligand and the receptor have a certain binding activity. The lower the binding energy is, the greater the affinity between the receptor and the ligand. There are 28 groups with binding energies lower than − 4.25 kcal/mol and only one group with binding energies greater than 0 kcal/mol. The heatmap showed that the binding energies between protopine, hepinine, piperine, visamminol, boldine, and trigonelline and the target were low. Therefore, it can be inferred that Protopine, Hetisine, Piperine, Visamminol, Boldine, and Trigonelline are the core active chemical metabolites of EWGP. The binding energies of ESR2, Mapk14, CYP17A1, and VDR are relatively low. Combined with the network analysis, one can speculate that ESR2, Mapk14, CYP17A1, and VDR are the hub genes of EWGP for the prevention and treatment of PMOP.
Effects of EWGP on Femoral Morphology in OVX Rats
Through scanning electron microscopy and Von Kossa staining of undecalcified sections, the efficacy of EWGP in improving the femoral microstructure and promoting femoral calcium salt deposition in ovariectomized rats was confirmed.
As shown in, Figure 4A, the thickness, shape, spacing, and arrangement of the whole trabecular bone in the femur can be observed under low magnification. Compared with that in the Sham group, the thickness of the trabecular bone in the OVX group gradually became thinner from the proximal to the distal end. The proximal trabecular bone was flaky and densely distributed. The distal end was unevenly rod-shaped, and the reticular arrangement was disrupted. Compared with those in the OVX group, the bone trabeculae in the E2 group and the EWGP group were arranged in an orderly manner, the overall shape and thickness from the proximal to the distal end were uniform, and the gaps between the bone trabeculae were scattered. At high magnification, bone collagen fibers, bone resorption holes, and fractures in the bone trabeculae on the surface of the bone trabeculae were observed. Compared with those in the Sham group, the trend of bone collagen fibers in the OVX group was inconsistent with the long axis of the bone trabecula, with more bone resorption holes and multiple transverse fractures of the bone trabecula. Compared with those in the OVX group, the trend of bone collagen fibers in the E2 group and the EWGP group was consistent with the long axis of the bone trabecula. As shown in, Figure 4B, the deposition of calcium and the formation of mineralization, it could be observed in the femoral epiphyseal line and trabecular bone. ImageJ software was used to measure the area indicated by the black line in the image. Compared with that in the Sham group, the calcium deposition area in the OVX group was significantly lower (p < 0.001). Compared with that in the OVX group, the calcium deposition area in the E2 group and EWGP group increased significantly (p < 0.01) (Figure 4C).
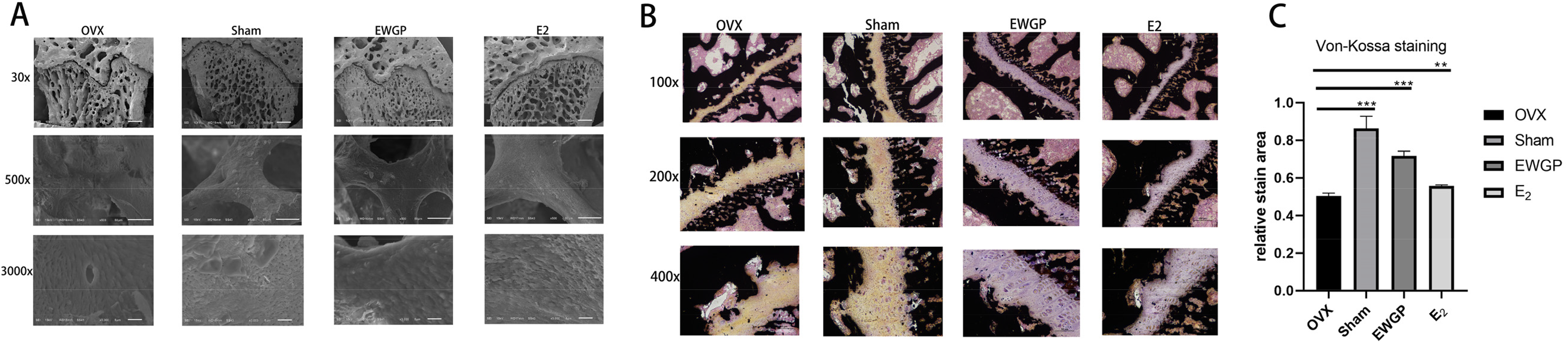
Femoral Morphology and Calcium Deposition were Observed in the Rats in Each Group. A. Electron Scanning Microscopy Images of the Femurs from Each Group. B. The Results of von Kossa Staining of the Femurs from Each Group. C. Statistical Analysis of the Relative Staining Areas.
Effect of EWGP on the Proliferation of MC3T3-E1 subcolony14 Cells
After treatment with different concentrations of EWGP (100 µg/mL, 80 µg/mL, 60 µg/mL, 40 µg/mL, 20 µg/mL, 10 µg/mL, 5 µg/mL, 0 µg/mL) for 6 h, 24 h, or 48 h, a CCK-8 kit was used to determine cell activity and proliferation at different concentrations (Figure 5A). After treatment with the same concentration of ethanol extract, the proliferation rate of MC3T3-E1 Subclone 14 cells increased gradually with increasing treatment time, indicating that the ethanol extract of EWGP could promote the proliferation of MC3T3-E1 Subclone 14 cells (Figure 5B). Three concentrations (70, 80, 90 µg/mL) promoted proliferation (p < 0.05, p < 0.01, p < 0.05); however, at 80 µg/mL, the concentration significantly promoted the proliferation of the MC3T3-E1 Subclone 14 cells. MC3T3-E1 Subclone 14 cells were treated with drug-free medium (con), 80 µg/mL of the ethanol extract of EWGP, or estradiol (0.1 nmol/mL, E2) for 48 h. Compared with those in the CON group (Figure 5C), MC3T3-E1 Subclone 14 cells treated with E2 or EWGP exhibited significant proliferation (p < 0.05).

Effects of EWGP on the Proliferation of MC3T3-E1 Subclone 14 Cells. A-B. the Optimal Concentration of the Ethanol Extract of EWGP for MC3T3-E1 Subclone 14 Screening; C. the Effect of the Ethanol Extract of EWGP on the Proliferation of MC3T3-E1 Subclone 14 Cells; D. the Statistical Results of the Percentage of EdU-Positive Cells; E. the Detection of the Effect of EdU on the Proliferation of the MC3T3-E1 Subclone 14 Cells.
Compared with that of the CON group (Figure 5E), the viability of the MC3T3-E1 Subclone 14 cells treated with E2 and EWGP for 48 h increased significantly. EdU fluorescence (green) was used to detect cell proliferation, and DAPI fluorescence (blue) was used to detect the number of cells. The ratio of proliferating cells to total cells was the percentage of EdU-positive cells. Compared with that in the CON group (Figure 5D), the percentage of EdU-positive MC3T3-E1 Subclone 14 cells was significantly greater after treatment with EWGP and E2 (p < 0.05, p < 0.01). Combined with the CCK-8 results, these findings suggested that EWGP and E2 treatment promoted cell proliferation.
Effects of EWGP on Estrogen Receptor Pathways and Calcium ion-Related Signaling Pathways
According to the enrichment analysis, the specific signaling pathway of the EWGP involved estrogen receptors and calcium ions. Therefore, the expression levels of four hub gene targets (CYP17A1, ESR2, MAPK14, and VDR) and calcium-regulated proteins (TRPV1 and CAMKII) in animal and MC3T3-E1 Subclone 14 cells treated with EWGP were assessed via Western blot analysis.
Compared with those in the Sham group (Figure 6), the relative protein expression levels of CAMKII and CYP17A1 in the OVX group were significantly greater (p < 0.0001), and the relative protein expression levels of ESR2, Mapk14, VDR, and TRPV1 were significantly lower (p < 0.05, p < 0.001, p < 0.001, p < 0.05). Compared with the OVX group, the relative expression of CAMKII protein in the EWGP group and the E2 group was significantly decreased (p < 0.001), and the relative expression of ESR2, Mapk14, VDR and TRPV1 protein in the EWGP group and the E2 group was significantly increased (p < 0.01, p < 0.001, p < 0.001, p < 0.001; p < 0.05, p < 0.001, p < 0.05, p < 0.001: the relative expression of CYP17A1 protein in the EWGP group increased significantly (p < 0.01), and the relative expression of CYP17A1 protein in the E2 group decreased significantly (p < 0.001).

Effects of EWGP on the Estrogen Receptor Pathway and Calcium-Related Signaling Pathway in OVX Rats.
Compared with those in the control group (Figure 7), the relative protein expression levels of CAMKII, ESR2, Mapk14 and TRPV1 in the EWGP group (p < 0.01, p < 0.05, p < 0.001, p < 0.001) and the E2 group were increased (p < 0.05, p < 0.05, p < 0.001, p < 0.05); the relative expression levels of the CYP17A1 protein in the EWGP group and the E2 group were decreased; the relative expression of the VDR protein in the EWGP group was increased (p < 0.05); and the relative expression of the VDR protein in the E2 group was decreased (p < 0.05).

Effects of EWGP on the Estrogen Receptor Pathway and the Calcium ion-Related Signaling Pathway in MC3T3-E1 Subclone 14 Cells.
Discussion
The European Society of Endocrinology (ESE) recommends that the choice of PMOP dosage regimen should be based on the risk of fracture. 17 Postmenopausal women with a high risk of fracture should choose long-term use of bisphosphonates. For women with a tendency toward osteoporosis and a high risk of fracture, they should take teriparatide for 2 years. Patients with osteoporosis and a high risk of fracture should be treated with romosozumab for 1 year. Bisphosphonates destroy the function of osteoclasts through farnesyl pyrophosphate synthase (FPPS) and hinder the isopreneylation of small GTPase proteins. Teriparatide, a recombinant human parathyroid hormone (PTH), serves as an intermittent exogenous PTH pulse to stimulate bone remodeling and regulate the reabsorption of calcium and phosphate in the kidney. Romosozumab stimulates osteoblast formation by inhibiting sclerostin. 18
According to a cross-sectional investigation 19 conducted by Linhong Wang and colleagues in mainland China from 2017 to 2018, the incidence of osteoporosis in men older than 40 years was 5.0%, whereas that in women was 20.6%. The incidence of vertebral fractures was 10.5% in men and 9.7% in women. Moreover, in an overall epidemiological study, the diagnosis rate of osteoporosis based on BMD was only 0.3%, and the incidence of fractures caused by anti-osteoporosis drugs was 1.4%. In daily life, most patients with osteoporosis are diagnosed with fractures caused by accidental minor injuries. Therefore, preventive treatment cannot be truly achieved in clinical practice, and most patients are treated after diagnosis.
TCM prescriptions have few side effects and lower prices and are suitable for long-term use in the treatment of OP. Moreover, TCM can not only improve bone microstructure and bone mass and promote bone growth but also relieve other symptoms, such as back pain. 20 Several components in TCM have demonstrated efficacy. Using dexamethasone, Akshoo and coworkers 21 constructed an osteoporosis model and compared the therapeutic efficacy of trigonelline and bisphosphonates. Trigonelline was found to increase BMD, estrogen, calcium, and phosphorus levels while reducing PTH, alkaline phosphatase (ALP), and acid phosphatase (ACP) levels. It has been verified that Trigonelline can prevent and treat OP by increasing BMD and restoring bone physiology. Quinic acid derivatives (3,5-dicaffeoyl-epi-quinic acid) can promote the osteogenic differentiation of bone marrow mesenchymal stem cells through the WNT/BMP signaling pathway and reduce lipid differentiation by activating AMPK. 22 In addition, Boldine can inhibit osteoclast differentiation induced by RANKL by repairing the AKT signaling pathway and regulating Th17 cells/Tregs to prevent bone resorption.23,24 Further, Piperine can reduce osteoclast formation through the p38/c-Fos/NFATc1 axis and can also induce osteogenic differentiation by activating AMPK-dependent RUNX2.9,25 Therefore, we conclude that EWGP may prevent and treat PMOP through multiple targets and multiple pathways, as it raises protopontin, hepinine, piperine, vamminol, boldine, and trigonelline levels in serum.
Through the first-pass effect of the liver and the phase I and phase II metabolic processes, only 11 prototype metabolites were identified in the blood. After network pharmacology and molecular docking analyses, it can be speculated that protopine, hepinine, piperine, vamminol, boldine, and trigonelline may play key roles in the prevention and treatment of PMOP. ESR2, Mapk14, CYP17A1, and VDR may be specific predicted EWGP targets, and the results of KEGG enrichment analysis showed that estrogen- and calcium-related signaling pathways, such as ovarian steroidogenesis, the estrogen signaling pathway, and inflammatory mediator regulation of transient receptor potential (TRP) channels, may be the main signaling pathways.
A model of PMOP was constructed via ovariectomy. In the team’s previous study, we compared the effect of 200 mg/kg, 400 mg/kg, and 800 mg/kg doses of EWGP in preventing and treating osteoporosis, of which 400 mg/kg doses were the daily oral dose calculated from the instructions for use of the EWGP and 800 was twice the daily oral dose (means high dose), and the experimental results showed that both 400 and 800 doses were effective in improving the bone microstructure in ovariectomized rats. 26 Therefore, in this article, we focus on the daily oral dose of 441 mg/kg/day, to observe the experimental pharmacodynamics of EWGP, and analyze the target effect of the network. After 12 weeks of drug gavage, SEM showed that EWGP improved the microstructure of the femur, and VK staining showed that the pill could increase the deposition of calcium in the femur, which proved that EWGP had the effect of preventing and treating PMOP. In the cell culture experiments, CCK-8 and EdU assays revealed that the ethanol extract of EWGP significantly promoted MC3T3-E1 Subclone 14 proliferation, which proved that EWGP could enhance the proliferation of osteoblasts. Western blot analysis revealed that EWGP promoted the expression of CYP17A1 in tissues, while estradiol inhibited CYP17A1, as CYP17 promoted the conversion of 17-hydrogen-progesterone to androstenedione during estradiol synthesis (Figure 8A, C). Direct estradiol supplementation can reduce the expression of the CYP17 protein through negative feedback regulation and maintain the stability of estrogen levels in the body. However, there is a lack of estrogen synthesis in cells. Therefore, when the cells were treated with ethanol extract of EWGP and estradiol, the biological function of CYP17 was inhibited, resulting in decreased expression. Cytochrome P450 (CYP) is involved in the biosynthesis of cholesterol, vitamin D, and estrogen, so CYP plays an important role in bone development. Among them, CYP17A1 is the key substance for catalyzing the production of dehydroepiandrosterone and androstenedione; therefore, in the body’s tissues, the loss of function of the CYP17A1 gene will lead to the deficiency of sex hormones and cause osteoporosis.25,26 According to the analysis of the mechanism of hormone synthesis, EWGP may promote estrogen biosynthesis by increasing the expression of CYP17A1 in the liver and gonads in ovariectomized rats. Xia et al 27 found that low concentrations of Rehmannia extract could significantly increase the protein concentration of CYP17A1 when they explored the prevention and treatment of glucocorticoid-induced osteoporosis by Rehmannia extract, so the concentration of EWGP promoting the proliferation of MC3T3-E1 cells may not be effective in promoting the protein expression of CYP17A1. Given the expression of CYP17 in tissues and cells, the mechanism of action of EWGP may resemble that of phytoestrogens. At low concentrations, it promotes the production of estrogen in the body, and at high concentrations, it antagonizes the estrogen synthesis pathway. 28 During the calcium ion transport process (Figure 8B), CAM is first activated, and TRPV1 also regulates calcium ions, resulting in the inhibition of CAMKII expression in response to drugs. Therefore, we speculate that EWGP may play a major role in the prevention and treatment of PMOP through estrogen and calcium-related signaling pathways (Figure 8D).

The Possible Mechanism of EWGP in the Prevention and Treatment of PMOP. A. Ovarian Steroidogenesis B. Inflammatory Mediator Regulation of TRP Channels C. Estrogen Signaling Pathway D. Possible Mechanism of EWGP.
In this study, bioinformatics methods such as network pharmacology and virtual molecular docking were used to analyze and screen the targets of EWGP for the prevention and treatment of PMOP. Although the target screening process was effectively shortened, there were certain limitations, such as the fact that research on the chemical compositions of Tibetan medicine unique to the Qinghai-Tibet Plateau was rarely reported, resulting in the inability to retrieve the chemical compositions of related medicinal herbs and materials in public databases such as TCMSP. Faced with this situation, the team used high-performance liquid chromatography-mass spectroscopy (HPLC-MS) for detection and analysis to compensate for the lack of chemical sources in network pharmacology. The principle of predicting drug targets on the online website is based on the data and reports collected on the website, using computational virtual prediction of the target and ranking the probability of the predicted target, where the probability of 1 (100%) is the target reported in the existing literature; therefore, when selecting the target, we try to select the target with the highest probability for subsequent analysis. Based on the results of bioinformatics analysis, we obtained several targets for the prevention and treatment of PMOP by EWGP, and combined with the results of network pharmacology analysis, metabolic component prediction, KEGG and GO enrichment analysis, and the existing literature reports, we selected the targets with the best possibility and operability for in vivo and in vitro experimental verification.
Conclusion
Our results demonstrate that EWGP improved estrogen and calcium levels in OVX rats through estrogen/TRP/calcium signaling and other related signaling pathways. Additionally, the chemical effects of EWGP were preliminarily verified, laying the foundation for the use of the effective active metabolites of the pill for treating PMOP.
Supplemental Material
sj-doc-1-chp-10.1177_2515690X251372707 - Supplemental material for Mechanisms of the Ershiwuwei Guijiu Pill in Treating Postmenopausal Osteoporosis Based on Network Analysis and Experimental Validation
Supplemental material, sj-doc-1-chp-10.1177_2515690X251372707 for Mechanisms of the Ershiwuwei Guijiu Pill in Treating Postmenopausal Osteoporosis Based on Network Analysis and Experimental Validation by Fanglin Duan, Li-Xue Zhang, Muhammad Naveed, Peifeng Wu, Yao Yu, Muhammad Zia Ahmad, Dongfang Dai, Jannat Bibi, Fenghui Li and Changxing Li in Journal of Evidence-Based Integrative Medicine
Footnotes
Acknowledgments
We are highly thankful to Dr. Jennifer Hill (UT) for critically reviewing the text of this paper. Moreover, all the authors thank and acknowledge their respective Universities and Institutes.
Ethical considerations
The animal study was reviewed and approved by the Experimental Animal Ethics Committee of Qinghai University’s College of Medicine [QHDX2021000541], China.
Author contributions
Fanglin Duan: Li-Xue Zhang: Conceptualization & writing-original draft. Yao Yu: Formal analysis. Dongfang Dai: Peifeng Wu: Jannat Bibi: Writing, review & editing. Md Naveed: Md Zia Ahmad: Fenghui Li: Writing, review & editing. Changxing Li: Conceptualization and supervision.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Qinghai Fundamental Scientific and Technological Research Plan (grant number 2023-ZJ-783), the Fundamental Research Funds for the Central Universities (31920230192), and the Gansu Province Youth Doctoral Fund for Higher Education Institutions (2022QB-026), China.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Availability of Data and Materials
The datasets used and analyzed during the current study are available from the corresponding author upon reasonable request.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
