Abstract
The flavonoid silymarin extracted from the seeds of Sylibum marianum is a mixture of 6 flavolignan isomers. The 3 more important isomers are silybin (or silibinin), silydianin, and silychristin. Silybin is functionally the most active of these compounds. This group of flavonoids has been extensively studied and they have been used as hepato-protective substances for the mushroom Amanita phalloides intoxication and mainly chronic liver diseases such as alcoholic cirrhosis and nonalcoholic fatty liver. Hepatitis C progression is not, or slightly, modified by silymarin. Recently, it has also been proposed for SARS COVID-19 infection therapy. The biochemical and molecular mechanisms of action of these substances in cancer are subjects of ongoing research. Paradoxically, many of its identified actions such as antioxidant, promoter of ribosomal synthesis, and mitochondrial membrane stabilization, may seem protumoral at first sight, however, silymarin compounds have clear anticancer effects. Some of them are: decreasing migration through multiple targeting, decreasing hypoxia inducible factor-1α expression, inducing apoptosis in some malignant cells, and inhibiting promitotic signaling among others. Interestingly, the antitumoral activity of silymarin compounds is limited to malignant cells while the nonmalignant cells seem not to be affected. Furthermore, there is a long history of silymarin use in human diseases without toxicity after prolonged administration. The ample distribution and easy accessibility to milk thistle—the source of silymarin compounds, its over the counter availability, the fact that it is a weed, some controversial issues regarding bioavailability, and being a nutraceutical rather than a drug, has somehow led medical professionals to view its anticancer effects with skepticism. This is a fundamental reason why it never achieved bedside status in cancer treatment. However, in spite of all the antitumoral effects, silymarin actually has dual effects and in some cases such as pancreatic cancer it can promote stemness. This review deals with recent investigations to elucidate the molecular actions of this flavonoid in cancer, and to consider the possibility of repurposing it. Particular attention is dedicated to silymarin's dual role in cancer and to some controversies of its real effectiveness.
Introduction
Research on plants and their possible curative properties is not new. It has been occurring since ancient times. In the last 200 years this search has become more scientifically oriented and led to discoveries such as curare, strychnine, atropine, salicylate, digitalis, and more recently taxanes, artemisinin, vitamins, and many others. These naturally originated molecules “have cellular targets similar to those of new drugs developed by pharmaceutical companies.” 1 Many of these natural products were so strikingly important for human health that they swiftly entered clinical practice. Sometimes, they were favorably modified by the pharmaceutical industry and then derivatives with enhanced benefits were born. While taxane compounds are one of the best examples of a success story in oncology, other compounds, not so blatantly effective as taxanes are on the waiting list. There is also a group of natural products that were, and are, used for known diseases other than cancer. In some cases, their antitumoral effects were slowly recognized and they were repurposed. Silymarin is one of this type of products, with some recognized antitumor effects however, repurposing has not yet occurred. Seeds of Silybum marianum, 2 popularly known as milk thistle, have been used since ancient times to treat diverse ailments, and more recently liver damage due to toxins, particularly Amanita phaloides poisoning (but including many others such as carbon tetrachloride, 3 metals, allylalcohol) and alcohol-induced damage, including hepatitis, cirrhosis, and jaundice.4–6 (From a technical point, what are commonly called seeds are actually fruits, but we shall call them seeds following other publication precedents). The last 15 years have witnessed a growing interest in silymarin and the plant it comes from: Silybum marianum (L.) Gaertn (also known as Carduus marianus and wild artichoke).
Although Silymarin is probably the most thoroughly studied nutraceutical, it is looked upon with skepticism by the medical profession for multiple reasons, such as:
ample distribution and easy accessibility to milk thistle; over the counter availability; the fact that it is a weed; some controversial issues regarding bioavailability, and pharmacological actions; its status as a nutraceutical rather than a drug according to FDA; its vulgarization through many nonscientific Internet pages dedicated to silymarin compounds; the enormous number of manufacturers, many of them scarcely known (Figure 1); the direct consequence of this “popularization” is that it is available over the counter at the herbalist shop or through the Internet, rather than with a prescription in the pharmacy;
7
the lack of striking effects on the disease; the fact that it is not usually considered in university-level pharmacology courses.

A glimpse of the multiple brands and presentations of silymarin compounds in the US and European markets with the resultant “vulgarization.” There are many “silymarins” developed in well accredited pharmaceutical laboratories, but there are also many produced by scarcely known sources. Most can be acquired through the Internet.
Silybin 60% to 70%
Silychristin 20%
Silydianin 10%
Isosilybin 5%
Taxifolin 1%
Small amounts of the flavonoids: quercetin, kaempferol, apigenin, naringin, eriodyctiol.
In 1959, Möschlin isolated silybin, 15 and then in 1968 silymarin chemistry was described in detail by Wagner et al,16, 17 and Pelter and Hansel. 18 Today, more than 50 years have elapsed since the initial hepatic antitoxic and protective function of the compound was discovered and now, its antitumor activity is under scrutiny (Figures 2 to 4). Silymarin, the active principal component of milk thistle, was originally thought to be one substance, until it was discovered that it is actually composed of a group of different flavolignans (Box 1).

Milk thistle and the chemical structure of silybin (C25 H22 O10), with its proprietary numbering. Of note, is the similarity between silybin and steroid hormones. The lower panel shows silibinin's structural formula where 3 different chemical groups can be identified: a taxifolin and a coniferyl alcohol united by an oxirane ring. According to Biedermann et al, 22 “the 20-OH group was established to be the most active radical scavenging moiety and also the most important group responsible for the lipoperoxidation inhibitory activity.” Positions 2-3 also play a role in antioxidant activity because these positions can be oxidized to produce 2-3 dehydrosilybin (see Figure 3). Silybin has 5 hydroxyl groups in positions 3, 5, 7, 20, and 23 which are the targets to produce silybin derivatives. Positions 7 and 20 are usual sites of glucuronidation of silybin during its conjugation in human metabolism.

2-3 dehydrosilybin has 25-fold more antioxidant activity than silybin. 23 Taxifolin and quercetin have more antioxidant activity than 2-3 dehydrosilybin. Small amounts of 2-3 dehydrosilybin are found in silymarin.

Nomenclature diagram of the different components of milk thistle seed and silymarin. 24 Isosilybin B seems to be the most powerful anticancer fraction. 25 Silymarin and Silibinin are different concepts, however some older publications use both terms interchangeably. Silibinin is the more active form of the silymarin extract. Only silymarin extracts are available in pharmacies while researchers usually use silibinin for their experiments. Standard silymarin extracts usually contain 33.5% silybin, 13% silychristin, 8.35% isosilybin, 3.5% silydianin 26 (see Figure 5).
Silybin is stable in acidic conditions but unstable under alkaline conditions. Alkaline media disrupt flavolignan's skeleton. This is important because the extracellular matrix of tumors has a low pH (approximate pH = 6.8), while intracellular tumor pH is alkaline (approximate pH = 7.5), but only slightly more alkaline than normal cell intracellular pH (approximately = 7.2). 19 Normal cells, on the other hand, have an alkaline extracellular milieu (approximate pH = 7.35). We presume, without evidence to sustain the presumption, that silybin can reach the malignant cell's acidic extracellular space without degradation. This singular feature, the acidic extracellular pH of tumors, 20 may explain why silybin effects differ in normal versus malignant cells. Silymarin may be able to better access the malignant cell compared with normal cells. This theory needs experimental confirmation.
In spite of the evidence favoring its benefits in chronic liver disease, “the overall efficacy of silymarin remains unclear” according to Tighe et al.
39
However, there are many, and some potentially beneficial, known biochemical effects of silymarin and silybin. For example, free radical scavenging and antioxidative properties of silybin are well known and have been thoroughly investigated.
40
It is considered 10-fold more antioxidant than vitamin E. In 1977, Machicao and Sonnenbichler
41
showed that silybin increased RNA synthesis in rat liver cells and mainly increased the production of ribosomal RNA and polymerase A. Shriever et al
42
found that silymarin inhibited fatty acid synthesis in rat liver: fatty-acid-synthase and ATP-citrate-lyase, 2 of the main lipogenic enzymes, were diminished by about 50%. Fiebrich and Koch43,44 described silymarin as a blocker of prostaglandin production in vitro through inhibition of both prostaglandin synthetase and lipoxygenase. This reduction of lipoxygenation was confirmed on liver ribosomes and mitochondria as well and probably explains silymarin's hepatoprotective actions.
45
A few years later Sonnenbichler et al 46 presented the first evidence that silymarin acted in a different way in noncancerous hepatic tissue and malignant cells: in the first case it stimulated DNA synthesis, in the second it did not. Silymarin is also a potent blocker of cyclic AMP breakdown (in vitro) by a phosphodiesterase preparation, 47 an inhibitor of histamine release from human basophil leukocytes, 48 dose-dependent downregulator of in vitro lymphocyte blastogenesis 49 and alters the mitochondrial electron transport chain through mitochondrial calcium release, 50 in addition to its antioxidant properties. 51 Immunostimulatory effects of silymarin were also described in experimental models, but not in the context of cancer treatment.52,53
Silymarin and Other Diseases (Table 1)
Silymarin has been investigated and proposed for the treatment of many different diseases, from Alzheimer dementia 54 to SARS 2 Covid-19, including diabetes, 55 diabetic complications,56–58 hyperlipidemia, and hypercholesterolemia,50–61 among others. However, in the last 15 years, the main focus has been cancer.
Silymarin Research Beyond Hepatoprotection and Cancer: A Summary.

Silymarin and Cancer
The first observation of silymarin's possible benefits in cancer is the 1991 publication by Mehta and Moon. 118 They showed that silymarin could act as a preventive (antipromoter) of cancer in mouse mammary glands treated with DMBA (dimethylbenzanthracene) and TPA (tetradecanoylphorbol acetate). The treatment protocol they employed made it possible to differentiate whether the chemoprevention worked at the initiation stage of carcinogenesis (DMBA phase) or during promotion (TPA phase). A 1991 review on the advances in pharmacological studies of silymarin by Rui, 119 did not mention anticancer activities. But in 1994, Agarwal et al 120 performed a study on skin treated with TPA confirming the protective effect of this flavonoid against tumor promotion. Silymarin protected against induction of ornithine decarboxylase by TPA. Ornithine decarboxylase inhibition protects against tumor promotion. A protective effect of silymarin was also found in colon and small intestine adenocarcinoma cells induced by 1,2-dimethylhydrazine. 121 Silymarin and its components also inhibit beta-glucuronidase. 122
Valenzuela and Garrido
123
proposed 3 levels for silymarin's action in experimental animals:
(a) as an antioxidant, by scavenging prooxidant free radicals and by increasing the intracellular concentration of the tripeptide glutathione; (b) through a regulatory action of cell membrane permeability and increase in its stability against xenobiotic injury; (c) through nuclear expression, by increasing ribosomal RNA synthesis, by stimulating DNA polymerase I, and by exerting a steroid-like regulatory effect on DNA transcription.
Silymarin also inhibits rat liver cytosolic glutathione S-transferase,
124
although this function does not clearly hint towards anticancer activity. On the other hand, silymarin scavenges reactive oxygen species as noted above, and inhibits arachidonic acid metabolism in human cells,
125
has antiinflammatory effects similar to those of indomethacin,
126
protects skin against carcinogenic agents127,128 and ultraviolet radiation.129–131 These publications strongly suggest a cancer-preventive activity and silymarin is slowly emerging as an anticancer drug. For example, Scambia et al
132
tested the antiproliferative activity of silymarin on human ovarian and breast cancer cell lines and found a growth-inhibiting effect on both. Silymarin also showed synergism with the commonly used anticancer compounds doxorubicin and cisplatin.
In DU145, prostate carcinoma cells, silymarin showed inhibition of Erb1 (eukaryotic ribosome biogenesis protein 1) signaling and G1 arrest. 133 In MDA-MB 486 breast cancer cells, G1 arrest was found due to increased p21 and decreased CDKs activity. 134 In advanced human prostate carcinoma cells, silymarin decreased ligand binding to Erb1 135 and NF-kB expression was strongly inhibited by silymarin in hepatoma cells, 136 as well as in histiocytic lymphoma, HeLa and Jurkat cells. 137
According to Zi and Agarwal, low doses of silymarin inhibited ERK1 and ERK2 Map kinases in a skin cancer cell line (human epidermoid carcinoma A431) and at higher doses activated MAPK/JNK1. This means that at lower doses the effect was antiproliferative and at higher doses proapoptotic. 138
Treating prostate carcinoma cells with silymarin the levels of PSA were significantly decreased and cell growth was inhibited through decreased CDK activity and induction of Cip1/p21 and Kip1/p27. 139
Silymarin has also been shown to have a variety of other protective effects in various cell types, such as anti-COX2 and anti-IL-1α activity, 140 antiangiogenic effects through inhibition of VEGF secretion, upregulation of Insulin like Growth Factor Binding Protein 3 (IGFBP3), 141 and inhibition of androgen receptors. 142 In leukemia HL-60 cells, silymarin inhibited proliferation and induced differentiation into monocytes in a dose-dependent manner. 143 Another important effect of silymarin in cancer is the downregulation of the STAT3 pathway which was seen in many cell models. STAT3 is active in many types of cancer and is associated with poor prognosis and resistance to treatments.144–146 Telomerase activity is another important factor in promoting carcinogenesis and evading senescense, thus inducing cancer cell immortality; silymarin has the ability to decrease telomerase activity in prostate cancer cells. 147
Silymarin and Apoptosis
The apoptotic mechanism silymarin employs on cancer cells is generally p53 dependent, and follows the usual steps: increased proapoptotic proteins; decreased antiapoptotic proteins; mitochondrial cytochrome C release-caspase activation. 148 Caspase inhibitors terminate silymarin apoptotic activity. Malignant p53 negative cells show only minimal apoptosis when treated with silymarin. Therefore, one conclusion is that silymarin may be useful in tumors with conserved p53.
Silymarin and Cancer Cell Migration
Enhanced cell migration is an important part of cancer progression. The antimigratory effects of silymarin in cancer cells are the result of mechanisms that
149
:
inhibit histone deacetylase activity;
149
increase histone acetyltransferase activity;
149
reduce expression of the transcription factor ZEB1;
149
increase expression of E-cadherin;
149
increase expression of miR-203;
149
reduce activation of sodium hydrogen isoform 1 exchanger (NHE1);
150
target β catenin and reduce the levels of MMP2 and MMP9;
151
reduce activation of prostaglandin E2;
152
suppress vimentin expression;
153
inhibit Wnt signaling;
154
modulate β1 integrin signaling.
155
Silymarin and Angiogenesis
Angiogenesis is important in cancer growth because solid tumors need a blood supply to grow. Silymarin inhibits angiogenesis. There are various postulated mechanisms:
Decreased migration of endothelial cells.
156
Flt1 (VEGFR1) upregulation.
157
(VEGFR1 upregulation may act as a negative regulator of VEGFA that is upheld by this receptor with low protein kinase activity and therefore VGEFA is unable to bind to KDR [VEGFR2] with much higher kinase activity).
158
VEGF downregulation.
40
Silymarin and Epithelial–Mesenchymal Transition (EMT)
EMT is involved in tumor progression and metastatic expansion. In a transcriptome study of nonsmall cell lung cancer (NSCLC) cells, Kaipa et al 159 found that silibinin had no effect on EMT. However, the opposite was found in other malignant tissues160–162 where it showed inhibitory effects.
Silymarin and TIMP1
High expression of the tissue inactivator of metalloproteases I, or TIMP1, in cancer is a marker of poor prognosis163,164 54 because it is involved in tumor progression, metastasis, and shorter overall patient survival. TIMP1 also promotes accumulation of tumor-associated fibroblasts. 165 Therefore, it may be considered a target in cancer treatment. Silymarin has the capacity to decrease TIMP1 expression166–168 in mice.
Silymarin and LPAR1
LPAR1 and 3 (lisophosphatidic receptors 1 and 3) are related to cancer invasiveness.169–172 Silymarin has the ability to downregulate LPAR1. 173
Silymarin and TGF β2
Silibinin reduces the expression of TGF β2 in different tumors such as triple negative breast, 174 prostate, and colorectal cancers. 175 TGF β2 downregulation impedes the TGF β2/Smad pathway reducing cellular motility and MMP2 and MMP9 (metalloproteases) reducing invasion. In the liver, TGF β2 downregulation results in an antifibrotic effect, preventing hepatic fibrosis induced by inflammatory liver diseases.166,176
Silymarin and Hypoxia Inducible Factor-1α (HIF-1α)
When cells are exposed to hypoxia, HIF-1α accumulates in the nucleus activating transcription of many genes and this plays an important role in tumor progression. Silymarin was found to decrease HIF-1α expression in rainbow trout brain 177 and in rat lung under hypoxic conditions. 93 In prostate cancer cells silibinin inhibited HIF-1α translation. 178
Silymarin and CD44 and EGFR
CD44, the transmembrane receptor for hyaluronan, is increased in breast cancer and many other tumors, due to EGF (epidermal growth factor) stimulation. Silibinin decreased CD44 expression and the activation of EGFR (epidermal growth factor receptor) by EGF. 179 In prostate cancer, silibinin decreased/inhibited CD44 expression as well. 180 CD44 binding with hyaluronan triggers important protumoral signaling from its intracellular segment, inducing cancer cell survival, angiogenesis, migration, and invasion. The CD44 antigen (synonym HCAM) is a glycoprotein acting as an adhesion molecule 181 on the cell surface. Cell adhesion molecules play an important role in cell migration. In fact, CD44 has been shown to be strongly correlated with invasion182,183 and metastasis.184–186
Silymarin Modulation of TNFα (Tumor Necrosis Factor Alpha)
Tyagi et al 187 showed that silibinin pretreatment of lung cancer cells inhibited TNFα induced “phosphorylation of STAT3, STAT1, and Erk1/2, NF-κB-DNA binding, and expression of COX2, iNOS, matrix metalloproteinases (MMP)2, and MMP9, which was mediated through impairment of STAT3 and STAT1 nuclear localization.”
Silymarin Inhibition of the Wnt/β-Catenin Signaling
The Wnt/β-catenin pathway is critical in cell proliferation, migration, and differentiation. It is a powerful regulator of embryonic development and tumorigenesis. Lu et al 188 showed that silibinin inhibited the Wnt/β-catenin pathway in both prostate and breast cancer cells.
Silymarin Potentiation of TRAIL-Induced Apoptosis
Tumor necrosis factor-related apoptosis-inducing ligand (TRAIL) is part of the TNF superfamily. It is known to selectively induce apoptosis in cancer cells without having significant toxicity toward normal cells. Kauntz et al189,190 found silibinin potentiated TRAIL-induced apoptosis in human colon adenocarcinoma cells. Furthermore, this potentiation was also found in TRAIL-resistant cells. Silibinin upregulated Death Receptors 4 and 5, thus increasing the number of receptors for TRAIL binding. Silibinin had the ability to induce not only the extrinsic apoptotic pathway, but also the intrinsic pathway. TRAIL sensitization by silymarin was also found in glioblastoma cells 191 and in hepatocarcinoma. 192
Silymarin and Phospholipase A2
Secreted phospholipase A2 participates in inflammation and carcinogenesis. Silibinin downregulates secreted phospholipase A2 in cancer cells. 193
Silymarin and Platelet-Derived Growth Factor (PDGF)
PDGF and its receptor are required for fibroblast proliferation and differentiation. It was found that silibinin had the ability to downregulate PDFG in fibroblasts, thus decreasing proliferation. 194
Silymarin Decreases the Levels of Interleukin 8 (IL-8)
Interleukin 8 has been identified as a protumoral cytokine195–198 and there is evidence showing that inhibition of IL-8 reduces tumorigenesis. 199 Flavonoids, in general, reduce levels of IL-8. Curcumin, 200 apigenin, 201 and silybin showed the ability to decrease IL-8 levels.150,202,203
Silymarin Inhibits the Signal Transducer and Activator of Transcription 3 ( STAT3) Pathway
STAT3 exists in the cytosol of cells and is a focal point of multiple oncogenic pathways. Silymarin inhibited STAT3 phosphorylation and decreased the expression of intranuclear sterol regulatory element binding protein 1 (SREBP1), decreasing lipid synthesis. The final consequences of these inhibitions were growth arrest and apoptosis. 204
Silibinin Acts as a Mitochondrial “Poison” in Malignant Cells
Si et al, 205 experimenting with 2 human breast cancer cell lines, MCF7 and MDA-MB-231, found that silibinin produced morphological and functional changes in mitochondria: decreased mitochondrial mass, condensed crests, reduced membrane potential and ATP content, and decreased mitochondrial biogenesis.
Silibinin and Metalloproteases
MMP2 and MMP9 play an important role in extracellular matrix remodeling and their levels correlate with progression of neuroblastoma tumors. 206 Yousefi et al 207 found that silibinin decreased MMP2, MMP9, and urokinase plasminogen activator receptor level (uPAR) in neuroblastoma cells. uPAR is also a marker of cell invasion.
Silymarin and COX2
COX-2 expression in cancer can stimulate angiogenesis and is associated with tumor growth, invasion, and metastasis. 208 Silymarin decreased the expression of COX2 in a model of chemically induced hepatocarcinoma in rats. 209
Silibinin and Programmed Death-Ligand 1 (PD-L1)
The programmed cell death protein and its ligand (PD-L1) complex play a key role in tumor progression being involved in growth regulation disturbance. This results in a defect in programmed cell death, apoptosis. 210 Silibinin inhibits PD-L1 by impeding STAT5 binding in NSCLC. 211 This hints at the possible usefulness of silymarin as a complement to immune checkpoint inhibitors. A similar effect was found in nasopharyngeal carcinoma. 212 In renal carcinoma cells, silibinin decreased PD-L1 in murine renal cancer cells in vitro and in vivo. 213
Silybin and Notch Signaling
The Notch signaling pathway is highly conserved, regulating development and is involved in angiogenesis and metastasis. 214 Silybin inhibited Notch signaling in hepatocellular carcinoma cells showing antitumoral effects. 215 However, dibenzazepine is much more powerful in this respect. 216 Notch was also downregulated by silibinin in breast cancer cells impeding notch-1/ERK/Akt signaling 217 and inducing apoptosis.
Silymarin and SIRT1
SIRT1 can deacetylate histones and other substrates and may act in a dual manner: as tumor suppressor or tumor promoter.218,219 Silymarin has the ability to increase hepatic SIRT1 expression. 220 Silymarin can also increase SIRT1 expression in other tissues, such as hippocampus, 221 articular chondrocytes, 222 and heart muscle. 223 Silymarin seems to act differently in tumors: in lung cancer cells SIRT downregulated SIRT1 and exerted multiple antitumor effects such as reduced adhesion and migration and increased apoptosis. When SIRT1 was independently downregulated with siRNA the silymarin's antitumoral effects were increased. 224
Silymarin and VEGF/VEGFR
The angiogenic cytokine vascular endothelial growth factor (VEGF) and its receptor (VEGFR) play critical roles in vasculogenesis and angiogenesis. Jiang et al 225 found that adding silymarin to prostate and breast cancer cells swiftly reduced the secretion of VEGF to the medium in a dose-dependent manner. Silymarin also prevented VEGF expression in myocardial cells exposed to doxorubicin toxicity 226 as well as other manifestations of cardiotoxicity. A similar decrease in VEGF and VEGFR levels was found with silymarin in preeclamptic placenta, however the effect was very modest. 227 There is evidence showing that silymarin reduces VEGF expression at the transcription level. 228
Silymarin and Myc
C-Myc is a multifunctional master regulator transcription factor; it is activated by oncogenic pathways, drives many functions for rapid cell division, and inhibits antiproliferative pathways. 229 There is direct and indirect evidence that silymarin interacts with c-Myc, 230 in some cases increasing its expression in liver cells as a response to hepatic chemical injuries 231 or decreasing it in malignancies. 232 Rajamanickam et al233,234 found that silymarin could prevent spontaneous tumorigenesis in an APCmin/+ mouse model (prone to develop intestinal tumors) by decreasing β-catenin, cyclin D1, c-Myc, phospho-glycogen synthase kinase-3β expression, phospho-Akt, and cyclooxygenase-2 in polyps. This report confirms silymarin's multitargeting effects in tumors and its different behavior in nonmalignant cells.
Silymarin and Carbonic Anhydrases
Carbonic anhydrases (CAs) play an important role in cancer progression, particularly those associated with the cell membrane (membrane CAs), namely isoforms CA9 (CAIX) and CA12 (CAXII). These CAs intervene in acidifying the extracellular substance and, working in tandem with sodium bicarbonate cotransporters, increase the intracellular pH. Downregulating or inhibiting membrane CAs has become a valid target for cancer therapy. Silymarin has the ability to inhibit CA isoforms CA I and CA II.235,236 However, we could not find any publications specifically addressing silymarin's role as a possible inhibitor of membrane CAs.
Silymarin and Mitochondria
This is a controversial relationship. On one side, silymarin showed ability to reduce oxygen consumption in mitochondria NAD-dependent substrates, while on the other hand stimulating respiration in mitochondria oxidizing succinate. 50 Silymarin increases mitochondrial release of Ca++ and lowers mitochondrial membrane potential in cancer cells 237 and increases the transmembrane potential in toxic aggressions. 238 Regarding mitochondria we may presume that silymarin has context-dependent effects.
Antimetastatic Potential
Many of the features discussed above hint towards silymarin's antimetastatic potential. In a TRAMP (Transgenic Adenocarcinoma of the Mouse Prostate) model of prostate carcinoma, when mice were fed with silibinin invasion and metastasis were reduced. 239 The antimetastatic effect was due to less invasion, less EMT, less collagen I-cancer cell adhesion, and less expression of CD44. 240 In a randomized clinical study with patients harboring solid tumors, silymarin was added to standard chemotherapy. Although silymarin failed to improve the results, there was a a slight—not significant—trend towards reduced metastasis. 241 We think that this study had some flaws which included a small sample size (15 patients with silymarin and 15 with placebo), tumors being present in different organs, and very low dosage (420 mg/day). In spite of these flaws, the trend towards a decrease in metastasis is still interesting and further study with a larger sample population was suggested by the authors.
Silymarin: Decreasing Side Effects and Toxicity of Chemotherapeutic Drugs
Silymarin coadministered with chemotherapeutic drugs has the ability to reduce toxicity in normal organs242,243:
It protects against liver and kidney toxicities induced by methotrexate in children and adults treated for leukemia.244,245 Silibinin decreased cisplatin's nephrotoxicity without affecting its antitumoral effectiveness.246,247 There is also evidence that it protects the heart from doxorubicin toxicity, however, it is less potent than quercetin in this effect.
248
Silymarin reduced docetaxel central and peripheral neurotoxicity.
249
Silymarin was able to decrease diarrhea produced by irinotecan treatment.
250
Silymarin reduced hepatotoxicity in patients with nonmetastatic breast cancer receiving doxorubicin /cyclophosphamide-paclitaxel.
251
Silymarin and Resistance to Treatment
Rho et al 252 found that adding silymarin to epidermal growth factor receptor tyrosine kinase inhibitors could overcome resistance produced by the T790M mutation in NSCLC xenografts. The mechanism of action seems to be impeding EGFR dimerization. It was found that bladder cancer cell lines resistant to cisplatin could be resensitized with silymarin. 253 A similar result was obtained with ovarian cancer cells resistant to paclitaxel. 254 The mechanism involved in resensitization to chemotherapeutic drugs is not fully known, however possible factors are: inhibition of NF-kB nuclear migration, 136 inhibition of survivin protein levels, downregulation of Pgp (MDR 1),255–258 and multidrug resistance-associated protein 1 (MRP 1).258,259 Silychristin (a component of silymarin) and silychristin derivatives have shown the particular ability to inhibit Pgp activity in a concentration-dependent manner 260 (see Tables 2 to 12).
Melanoma.

Prostate.

Lung NSCLC: Nonsmall Cell Lung Cancer//SCLC: Small Cell Lung Cancer.

Bladder.

Pancreas.

Breast.

Colon.

Liver.

Ovary.

Hematologic.

Miscellaneous.

Silymarin's Cancer Chemopreventive Actions
Table 5 summarizes the findings by Vinh et al that show that silymarin was able to significantly decrease the incidence of bladder neoplasms in male rats receiving the carcinogenic substance N-butyl-.N-(4-hydroxybutyl) nitrosamine. Interestingly, these results were achieved by oral administration of silymarin and were found in those animals that received silymarin not only at the initiation of carcinogenesis, but also in those of the postcarcinogenic period (for more examples on chemoprevention, see Tables 2 to 12).
Silymarin and Hormonal Receptors
Silymarin is a selective estrogen β receptor (ER-β) agonist. 261 However, it also has some estrogenic effects through ER-α. 262 Silymarin has strong binding affinity to ER-β and a mild affinity for ER-α. 263 Silymarin's estrogenic actions should be seriously considered as a problem in female hormone-dependent tumors. Furthermore, silymarin's estrogenic effects are confirmed by the observation that it produces benefits in menopausal women with hot flashes. 116 Contrario sensu, it may be advantageous in benign prostate hyperplasia264,265 and prostate cancer. However, in an experiment carried out in albino rats, silymarin increased testosterone and LH. 266 It also increased spermatogenesis in rats. 267 In spite of these 2 findings, the evidence for silymarin benefits in prostate cancer is abundant (see Table 3). Our conclusion is that the possible benefits found in prostate cancer are independent of silymarin's hormonal effects.
Silymarin Inhibits Clathrin-Dependent Trafficking
Endocytosis is an important mechanism of cell intercommunication which acquires major relevance in cancer. This process is initiated by the invagination of the plasma membrane. The protein clathrin provides A coat to this invagination (Figure 5). The clathrin coated vesicle has the ability to select for the adequate cellular receptor. 268 There is also endocytosis without clathrin coating. Silymarin has the ability to inhibit clathrin-dependent trafficking at least in the case of certain viruses such as Hepatitis C virus, reovirus, influenza virus,269,270 and Hepatitis B virus. 271 The mechanism behind this inhibition is through interference with the clathrin endocytic pathway. Actually, silymarin interferes with all the clathrin-dependent endocytic processes. Taxifolin, a close relative of silybin, was also found to inhibit receptor-mediated endocytosis of β-hexosaminidase in normal fibroblast culture. There were similar findings with other flavonoids. 272

Similarities and differences among silymarin's components.
Although there is no experimental evidence in this sense, we may presume that silymarin decreases endocytic trafficking in cancer cells too. Additionally, clathrin has protumoral effects beyond endocytosis: it switches TGF-β into a procancer role. 273
Figure 6 shows a simplified overview of the clathrin-dependent endocytosis.
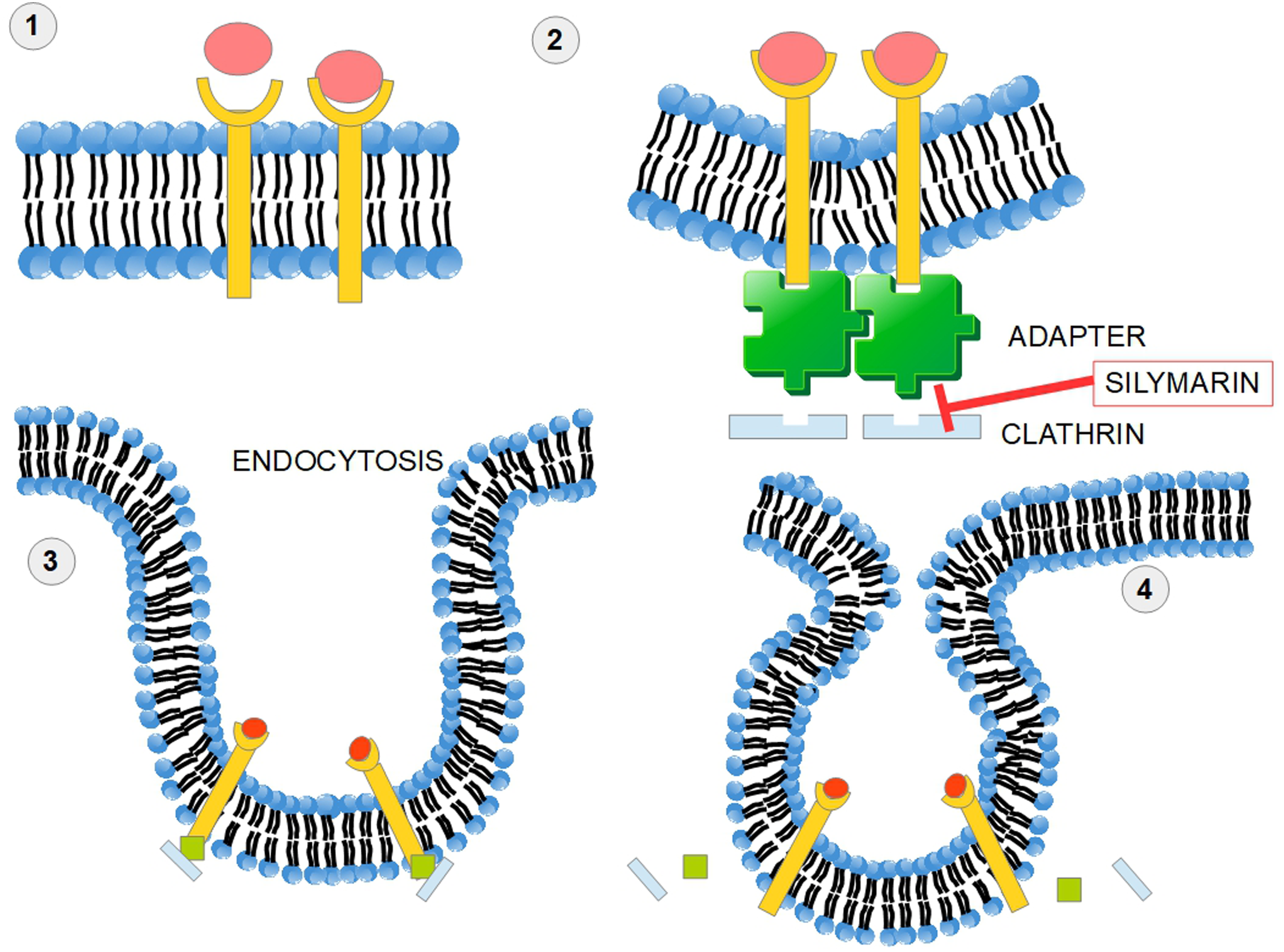
Silymarin impedes clathrin-dependent endocytosis. In receptor-mediated endocytosis, or clathrin-mediated endocytosis, cells incorporate hormones, proteins, and sometimes viruses in a selective manner that depends on a ligand-receptor interaction. (1) The ligand receptor interaction takes place on the cell surface. (2) An adapter protein and clathrin “coat” the internal surface of the receptors and a process of membrane invagination starts. (3) Advanced invagination process. (4) The adaptor protein and clathrin are released. Silymarin impedes this mechanism of endocytosis. Clathrin-dependent endocytosis of surface receptors participates in cellular signaling pathways. Clathrin seems to be a valid target in cancer therapy. 274
Silymarin and Renal Carcinoma
When targeting renal carcinoma cells with silymarin, migration and invasion were significantly decreased by inhibition of the EGFR/MMP-9 pathway: silymarin blocked phosphorylation of EGFR and ERK1/ERK2 and reduced expression of MMP-9. 275 This was confirmed by Liang et al 276 and by Chang et al in vivo 277 and in our unpublished observations with 2 patients with grade IV clear cell renal carcinoma, who experienced no new metastasis after they started on silymarin.
Silymarin and Pancreatic Cancer
Silymarin has not been extensively tested in pancreatic ductal adenocarcinoma (PDAC) (see Table 6). However, it does have an important antifibrotic effect. One of the major problems in PDAC is the intense stromal reaction with abundant production of stromal collagen fibers. 278 These impede delivery of the chemotherapeutic drug to the tumor mass and create interstitial hypertension through the strongly hydrophilic hyluronan. Therefore, silymarin's antifibrotic effects may provide an interesting complement to standard treatment. 279
Desmoplastic tumors are the consequence of the intense activity of cancer-associated fibroblasts (CAFs) producing collagen fibers. PDAC and the liver have specialized CAFs known as stellate cells considered the producers of the desmoplastic reaction. Silymarin has been found to inhibit/decrease the desmoplastic reaction through 2 mechanisms:
it inhibits TGF β2 that induces the desmoplastic phenotype of naïve fibroblasts;
280
it increases E-cadherin expression281,282 decreasing the invasive nature of the desmoplastic reaction.
Silymarin decreased fibrosis not only in 2 models of induced liver fibrosis37,283 but also in lung fibrosis induced by cigarette smoke.
284
In this last case, this occurred by downregulation of the TGF-β1/Smad 2/3 pathway signaling.
Although we could not find any publication showing that silymarin could reduce the desmoplastic reaction in pancreatic cancer, we may assume that it has the potential to do so, because the mechanisms behind this are similar to those found in liver and lung cancers. Long et al suggested this possibility, however they did not incorporate any evidence in their review. 279
Tables 2 to 12: Evidence of silymarin's anticancer effects. In vivo experiments are marked with an X in column 1.
In spite of the long list of publications mentioned in Tables 2 to 12, 5 cautionary notes should be added:
In a mouse model of induced mammary carcinogenesis, the administration of silymarin, slightly increased mammary tumor incidence.
369
This may be due to silymarin's estrogenic effects,115,261,370 however, the issue remains controversial because silymarin increases ERβ and decreases ERα expression.
264
In a model of mouse hepatic carcinogenesis (with diethylnitrosamine), silymarin showed no effects at all.
371
In a mouse model of alcohol-dependent hepatocarcinoma, silibinin increased tumor progression if chronic alcohol intake continued.
372
Many of the in vitro experiments described in Table 2 to 12 were performed at very high concentrations that are difficult or impossible to achieve in vivo. On the other hand, in vivo experiments (marked with an X in Tables 2 to 12) were mainly conducted with oral administration of silymarin or silibinin, so those results should have a more significant impact on future clinical research. Most of the published literature on silymarin and cancer does not mention the p53 status of the cells and this information is of capital importance (silymarin shows apoptotic effects on p53 positive cells but not on mutated p53).
Pharmacokinetics
Flavolignans (silymarin is a mixture of flavolignans) generally have poor bioavailability. This is the consequence of:
their strongly hydrophobic nature that does permit dilution to more than 50 μg/mL in water. Some organic solvents have a much better performance for this purpose. For example, ethanol shows a solubility of 225 mg/mL;
373
the fact that they are quickly metabolized; the fact they are poorly absorbed in the intestine.
The pharmacokinetic considerations we shall make refer to the standardized form of silymarin with known amounts of silybin.
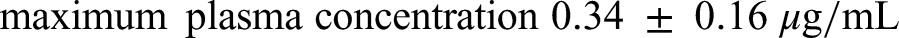
We think that there is no evidence that it can be. Oral administration of silymarin in humans achieves nanogram, but not microgram levels. Furthermore, we should not extrapolate Morazzoni's findings in rats to humans as their pharmacokinetics may differ.
Therefore, the evidence based on these high concentration experiments should be viewed with caution. On the other hand, experiments with xenograft models are more reliable (Tables 2 to 12, xenograft results are marked with an X).
Liver: 8.8 μg per gram of tissue Lung: 4.3 μg per gram of tissue Stomach: 123 μg per gram of tissue Pancreas: 5.8 μg per gram of tissue Prostate: 2.5 μg per gram of tissue after 1 h.
After an oral intake, silipide (the lipophilic SPC), achieved a maximum concentration of silybin in bile within 4 h and then declined with a mean time of approximately 10 h.
383
Silibinin complexed with the amino-sugar meglumine is water soluble and can reach a tissue concentration high enough to show clear antigrowth effects in NSCLC xenografts.
307
The distribution in different tissues also varies widely according to the type of tissue considered. It is higher in the liver and dimishes in lungs, pancreas, and prostate.
382
A relatively high concentration is achievable in colorectal muscosa (20-141 nmol/g of tissue).
384
The tissue levels obtainable compare unfavorably with those used in cell studies. To achieve apoptosis in cell studies, a concentration of more than 20 μM was necessary, 385 and this concentration does not seem easy to achieve by oral intake of standard preparations. It was also necessary to use a concentration of 100 μg/mL to induce apoptosis in Ramos cells (B lymphocytes). 386 Kamrani et al 387 used concentrations between 50 and 100 μg/mL to induce apoptosis in colon cancer cells. Therefore, while only a nanomolar concentration can be attained in tissues, micromolar concentrations were needed to induce apoptosis in these studies (the molecular weight of silybin is 482, 100 μg/ml = 207 μM).
In spite of this difficulty, Sing and Agarwal 298 found an important decrease in tumor volume in xenografted mice with human prostate carcinoma cells when the mice were orally fed with silymarin.
There are also different requirements for effects on cell migration versus proliferation. For endothelial cells, it was necessary to use a concentration of 48.1 μg/mL of silymarin to achieve a 20% reduction in proliferation and 16.1 μg/mL to achieve the same reduction in proliferation of LoVo colon cancer cells 156 to achieve a reduction of migration of 50%, it is necessary a concentration of 1.15 μg/mL on endothelial cells (with silibinin instead of silymarin 0.66 μg/mL were enough to achieve the same). 156 Our conclusion is that, from a bioavailability standpoint, it is much easier to achieve migration inhibition, than proliferative reduction.
In Europe one of the most used brands of silymarin is Legalon® L (silybin 3,23-O-bis-hemisuccinate) that comes in capsules of 150 mg. It also comes in vials containing 350 mg of silibinin for intravenous use. In the United States, silymarin is considered a nutritional supplement. 388 The intake of 5 of these capsules in 6 human volunteers, showed no adverse events. The concentration in plasma correlated with the dose and only 10% of it was unconjugated silymarin. A half life of 6 h was estimated. 389
In experimental conditions, many researchers dilute silybin in DMSO, a polar solvent in which silymarin is highly soluble. Unfortunately, this is not possible at the bedside.
Pharmaceutical Methods to Increase Bioavailability
Silymarin's low solubility, rapid metabolism, and quick excretion, led researchers and pharmaceutical industry to develop methods that could solve these very important drawbacks. Therefore, many compounds have been formulated mainly using nanotechnology. These compounds include nanosuspensions, solid dispersions, complexes with cyclodextrins and phospholipids, microemulsions, nanoemulsions, liposomes, polymer nanocarriers, solid-lipid nanoparticles and nanostructured lipid carriers, and polymer-based nanocarriers.
390
We shall discuss only a few of them.
The problem with silybin combinations is that although they increase water solubility at the same time, they may reduce other effects such as antioxidant properties.
23
There are many other mechanisms based on nano-particles that increase absorption, prolong half life, and improve water solubility of silym arin, that escape the scope of this article. For a review of the issue, see Di Costanzo et al
401
and Piazzini et al
402
(Figure 7).

Methods to increase silymarin's bioavailability.
Dosage and side Effects
A phase I study of silymarin in prostate cancer patients showed that 13 g daily per os divided into 3 doses was well tolerated. The most frequent adverse event was asymptomatic liver toxicity. 403 Side effects, although rare, were mainly related to the gastrointestinal tract, such as diarrhea, bloating, and nausea. 404 Abenavoli et al 405 found that daily doses beyond 1500 mg had laxative effects and increased bile flow. The usual dose of 400 or 800 mg a day is probably insufficient to achieve anticancer effects. It may be necessary to administer 800 mg 4 times a day because the half-life is short. However, the dose of silymarin for cancer treatment remains controversial. In one study, a high dose of silybinin was administered to patients prior to prostatectomy (13 g daily). They achieved high plasma concentrations, but nevertheless, low levels of silibinin were found in prostate tissue. 406 In an attempt to circumvent some of these problems one group used a silymarin-phosphatidylcholine compound administered orally as a daily dose of 2.8 g for 4 weeks prior to surgery. They achieved high levels in human breast cancer tissue. 407 This high bioavailability in this breast cancer study is an encouraging signal for a phase II clinical trial. It should also be noted that silymarin constituents have different anticancer abilities, 22 therefore a formulation of the strongest combination would represent a fundamental step in order to incorporate this flavonoid into standard treatments.
The Main Problems with Silymarin
Clinical Trials
The United States Clinical Trials web-page lists the following trials for silymarin in cancer:
NCT03130634: The Efficacy of Silymarin as Adjuvant Therapy on Colorectal Cancer patients Undergoing FOLFIRI Treatment. State: recruiting since 2017. Kaohsiung Medical University Chung-Ho Memorial Hospital. Taiwan. Study Design: This is an open-label, randomized, comparative, double arm, single center study to assess efficacy of Silymarin (150 mg 3 times a day) as adjuvant therapy on metastatic colorectal cancer patients undergoing FOLFIRI chemotherapy in Taiwan. NCT00487721: The Effect of High-dose Silybin-phytosome in Men With Prostate Cancer State: completed 2014 University of Colorado. Denver Subjects will take Silibin-Phytosome for 2 to 10 weeks. The dose of Silibin-Phytosome is 13 g daily, in 3 divided doses. Outcome: To determine if measurable silibinin tissue levels are detectable in the prostate glands of men treated with Silybin-Phytosome administered according to the protocol. Results: low concentration of silymarin in prostatic tissue.
117
NCT01829178: Evaluation of Effects of Silymarin on Cisplatin Induced Nephrotoxicity in Upper Gastrointestinal Adenocarcinoma. State: completed 2015 University of Tehran This study looked for possible protective effects of silymarin on kidney injury in patients receiving cisplatin. No results posted. NCT00055718: Silymarin (Milk Thistle Extract) in Treating Patients with Acute Lymphoblastic Leukemia Who Are Receiving Chemotherapy. State: completed 2013 Miami Children's Hospital, Winthrop University Hospital, Mount Sinai Medical School. This study looked for hepatoprotective effects of silymarin in patients receiving chemotherapy. No results posted. NCT02146118: A Phase II Study to Assess Efficacy of Combined Treatment with Erlotinib (Tarceva) and Silybin-phytosome (Siliphos) in Patients With EGFR Mutant Lung Adenocarcinoma. State: unknown Gosin University. Busan, Korea. No results. NCT01402648: Estrogen Receptor Beta Agonists (Eviendep) and Polyp Recurrence State: completed 2011 Ospedale Policlinico Consorziale—Gastroenterology Unit. Bari, Italy No results.
The conclusion we reach regarding clinical trials is:
There were only 2 clinical trials (4 and 5) to determine therapeutic possibilities of silymarin against cancer. Neither have published results. The dose used in the clinical trials showed differences of up to 1000% which clearly means that there is no standard dose.
Schröder et al 414 conducted a randomized double-blind, cross-over placebo-controlled trial (with 2 periods of 10 weeks with a wash out period in the middle) with 49 patients that showed rising PSA levels after radical prostatectomy (34) or radiotherapy (15). They received a supplement containing soy, different isoflavones, silymarin, vitamins, minerals, and antioxidants. While receiving the treatment the doubling time for PSA was 1150 days compared with 445 days with the placebo. The fact that the supplement contained many other components besides the silymarin makes it impossible to draw conclusions about this compound. But it is evident that the supplement modified the biochemical evolution of the disease, delaying PSA progression.
Four Clinical Cases
Four clinical case reports are available, which though they cannot in themselves constitute a proof for the efficacy of silymarin, are nonetheless interesting and suggest a need for further studies. Hsu et al 415 describe the case of a 66-year-old Taiwanese patient with a regression of an 11 cm diameter hepatocellular carcinoma. The patient was receiving 450 mg of silymarin daily, and no other medication. Even if we cannot consider this regression as a consequence of silymarin treatment, the fact that spontaneous regression of hepatomas is quite infrequent, makes us think of some intervention of silymarin in this unusual event. Moroni and Zanlorenzi 416 published another case of complete regression of an advanced unresectable hepatocellular carcinoma treated with sorafenib and silymarin. Additionally, Bosch-Barrera et al 417 presented 2 cases of brain metastases from lung cancer in which the treatment with silymarin decreased edema and the size of metastases, without improvement of the primary tumor.
Discussion
The concept of a tumor as a consequence of the mutation of one gene, and with one driver signaling or metabolic pathway, is flawed in most cases with the exception of cases such as chronic myeloid leukemia. Usually many genes and pathways are involved. The approach of attacking only one of the many hallmarks of cancer is also flawed. 418 Recent evidence suggests that multiple genes are usually involved along with many signaling pathways, all interconnected, and interdependent and generating an extraordinary ability of tumor cells to survive and resist internal and external threats. 419 This is one of the reasons why treatments made up of many different drugs are implemented in most treatment protocols.
Silymarin and its derivatives, through its multipronged attacks, allow one drug to reach many targets at the same time. Of course, we cannot expect silymarin to “cure” cancer all by itself, and it cannot replace any conventional chemotherapeutic treatment, but it is rather a privileged companion to therapeutic schemes in which it may develop useful complementary activity. This activity entails 3 concepts:
cancer prevention; synergy with some treatment protocols; decrease of collateral damage induced by chemotherapeutic drugs.
Silymarin's clinically achievable concentration in serum and at the tumor site, with the possible exception of the liver, seems insufficient for inducing apoptosis. However, xenograft model experiments showed that even with this low bioavailability drawback, silymarin could stop tumor growth.288,351,356
The first studies on silymarin activity in cancer were performed in hepatic cells showing some characteristics that cannot be really considered antitumoral such as increased ribosomal synthesis and RNA polymerase I activation. This did not happen in hepatoma cells or in other malignant cells (Figure 8).
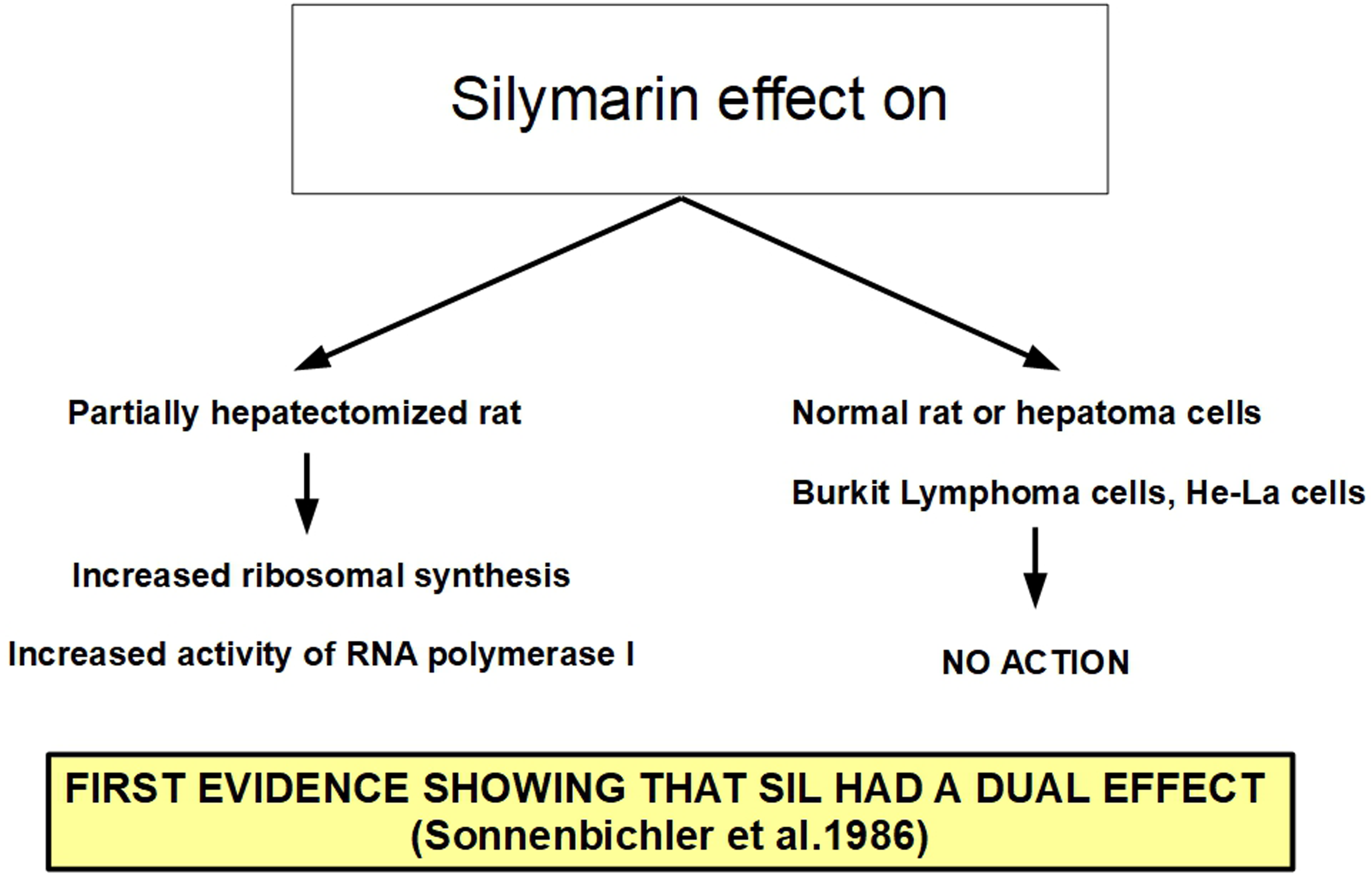
Different effects of silymarin according to context.
Antiproliferative activity was found against almost all types of tumors, whether solid or nonsolid (Tables 2 to 12). These findings were confirmed not only at cellular level but also
Silymarin has many other antitumor effects that can complement mainstream treatment protocols, such as:
reduction of cell motility and invasion through TGF-β2 inhibition; inhibition of HIF-1α translation; decreased TIMP1 expression, thus decreasing metalloproteases activation; inhibition of the EGFR-MMP9 pathway; decreasing the accumulation of MDSCs in the tumor; inhibition of ERK and AKT signaling; protection against off-target toxicity of chemotherapeutic drugs; synergistic or added effects with some chemotherapeutics; reduction of extracellular fibronectin production.
To this short list we must add that there is evidence sustaining clear benefits in clinical cases such as hepatocarcinoma and clear cell renal carcinoma.
However, silymarin also has some effects that work against classical chemotherapy. For example, its ability as an antioxidant reduces ROS production. Many of the drugs currently used against cancer are precisely based on the creation of an oxidative stress with increased ROS that induces apoptosis of malignant cells.
Therefore, we must ask: why is silymarin a useful complement to chemotherapy?
Evidence indicates that there may be 2 possible answers:
silymarin has context-dependent effects: its behavior is different in normal and malignant cells as can be seen in BOX 2. Its anticancer effects overwhelm those that seem favorable for cancer cell survival.

The dual effects of silymarin can be clearly seen in an experiment by Su et al. 424 Here nasopharyngeal carcinoma cells were exposed to low concentrations of silymarin and this showed protumoral effects: “Silymarin increased the expressions of superoxide dismutase 1, catalase, and glutathione peroxidase. Consequently, the cell apoptosis was reduced markedly. An increase of Bcl-2 expression and a decrease of activated caspase-3….” Additionally, there is the problem of increased stemness induced by silymarin in pancreatic adenocarcinoma cells.
In spite of these isolated discouraging report,
424
the overwhelming sum of experimental evidence shows that silymarin has clear anticancer effect, and at the same time protects normal cells from the collateral damage of chemotherapy. There is reasonable doubt that the concentration that can be reached
As already mentioned, silymarin is not a stand-alone compound against cancer and it should be used alongside other medications. On this point, it is important to note that silymarin is an inhibitor of the Cytochrome P450 system, particularly silychristin. Therefore, caution should be exercised in this respect. 426
Silybin could be used as a scaffold or structure that can be modified improving its antitumoral effects. For example, Manivannan et al 427 have synthesized silybin analogues with increased anticancer capacity. One of these compounds named “15k,” was very potent and selective for ovarian cancer cells, where it bound to tubulin with high affinity. Subsequent experiments found that 15k induced growth arrest and apoptosis of ovarian cancer cells at a much lower concentration than silymarin. Furthermore, it showed no toxicity in animals. 428
Finally, it is important to note that many of silymarin's multipronged antitumoral actions are equally, or sometimes even better conveyed by other flavonoids such as genistein and epigallocatechin gallate.429,430
What Remains to be Done?
In the first place, some of silymarin's protumoral effects demand further research with the objective of ascertaining if they need to be counteracted. Then, the precise silymarin concentrations required for the different antitumoral effects need to be established. And finally, the tumor concentration achievable with the different pharmaceutical preparations has to be determined. Once these 3 pieces of information are combined silymarin will be ready for serious clinical trials as a complement to classical chemotherapeutic schedules.
Conclusions
Silymarin compounds have considerable antitumoral effects. Well-planned clinical trials should be necessary to finally asses its bedside indications. Its dual, antitumoral and protumoral, effects merit further research.
Silymarin should be used at very high doses because low concentrations may induce protumoral effects. There is no toxicity even with very high doses. Silymarin's low absorption and bioavailability make it preferable to use modified pharmaceutical forms, such as nanoparticles or conjugated with compounds that increase its water solubility. These combinations already exist even if they have not been marketed as yet. The abundant existing evidence shows that silymarin has a definite place in cancer treatment. It has the ability to interfere with the expression of proteins related to cell cycle regulation, apoptosis, angiogenesis, and multidrug resistance. These characteristics define an anticancer drug. On the other hand, its strong antioxidant activity makes it a useful drug in cancer prevention.
Silymarin's lack of toxicity, even at very high doses, and the lack of effects on normal cells are important reasons for its further development.
Is there any other drug, that with no toxicity at all that can:
Inhibit EGFR signaling Upregulate CDK inhibitors such as p21 and p27 Downregulate CDKs Induce growth arrest by interfering MAPkinases cascade Induce apoptosis Inhibit TGF-alpha Reduce the expression of VEGF and VEGFR Inhibit the AKT axis Decrease tumoral fibrosis and have many other antitumoral effects?
Probably no other drug can achieve all these results without adverse events or high toxicity. We cannot expect that such a nontoxic pharmaceutical work as a stand-alone drug against cancer. But it can be an important factor in a multidrug anticancer schedule. Having mentioned this, the reports of increased stemness problem remain an unsolved issue which needs further investigation.
The inability to patent the compound is no doubt a drawback for the pharmaceutical industry and will restrict investment in these types of compounds. In its bioavailable formulations, silymarin deserves to be tested on clinical grounds, not as a stand-alone pharmaceutical, but as part of a treatment schedule.
Finally, the low concentrations that can be achieved with silymarin extracts at the bedside (in the order of ng/mL) hints to a serious bias in much of the past and present research at the cellular level where the average concentration range used was between 50 and 100 μg/mL. As a precondition for repurposing silymarin, newer pharmaceutical formulations should be screened in order to establish whether they can reach the necessary therapeutic concentrations.
Footnotes
Acknowledgments
Ms. Julia Hanna Weiss cooperated in the revision and correction of the article.
Author Contributions
Both authors equally contributed in the writing of the article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
Not applicable, because this article does not contain any studies with human or animal subjects
Trial Registration
Not applicable, because this article does not contain any clinical trials.
Plagiarism
All the figures, tables, and boxes are original and were developed by the authors.
