Abstract
Objective
To investigate CUE domain containing 2 (CUEDC2) expression in colorectal cancer with different invasion and migration abilities.
Methods
Fresh colon cancer tissues, obtained from patients with or without lymph node metastasis who were treated at the Department of General Surgery, Chinese People's Liberation Army General Hospital, and SW620 and HT29 colorectal cancer cell lines, were analysed for CUEDC2 expression.
Results
Real-time polymerase chain reaction showed significantly higher CUEDC2 mRNA levels in colon cancer tissue from patients with (n = 8) versus without (n = 8) lymph node metastasis, and in SW620 versus HT29 cells. Western blots revealed significantly higher CUEDC2 protein levels in colon cancer tissues from patients with versus without lymph node metastasis, and in SW620 versus HT29 cells. Colorectal cancer tissues from patients with lymph node metastasis showed more intense immunohistochemical staining and moderate staining of cell nuclei and cytoplasm versus less intense/weak staining in tissues from patients without lymph node metastasis.
Conclusions
CUEDC2 is highly expressed in colorectal cancer tissues and colorectal cancer cell lines with high invasion and migration ability. CUEDC2 may be involved in promoting invasion and metastasis in colorectal cancer.
Background
Colorectal cancer has become the third most common malignant tumour in men and is second only to breast cancer in women, with an annual death toll as high as 861 663 according to the latest World Health Organisation cancer statistics. 1 Approximately 50% of patients with colorectal cancer will have liver metastases, with 25% prevalence each of synchronous and metachronous liver metastases. 2 In patients with colorectal cancer, distant metastasis is the leading cause of death, and the liver is the most common distant metastatic site.3,4 There is no accurate method to predict the risk of metastasis in patients with colorectal cancer and 90% of patients diagnosed with metastases will not be able to undergo radical surgery at the time of initial diagnosis.5,6 The mechanism of invasion and metastasis of colorectal cancer remains unclear. 7 In order to improve the prognosis of patients diagnosed with colorectal cancer, it is imperative that the mechanism of colorectal cancer invasion and metastasis be determined.
CUE domain containing 2 (CUEDC2) is a protein with a molecular weight of about 32 kDa that is expressed in all human tissues and organs. 8 It has been found that CUEDC2 is highly expressed in ovarian and breast cancer, and in renal carcinoma, and is closely related to patients' adverse prognosis and disease recurrence. As a result, it is thought that CUEDC2 expression may promote the development of tumours.9–11 CUEDC2 has been shown to play an important role in the development and progression of neoplasms, but varying mechanisms of action have been reported between different neoplasms, and its role in colorectal cancer invasion and metastasis has not been reported.
Clarification of the invasion and metastasis mechanism of colorectal cancer has important clinical significance for improving the prognosis of patients with colorectal cancer. Thus, the aim of the present study was to investigate the expression of CUEDC2 in postoperative fresh colon cancer tissues and in colorectal cancer cell lines with different invasive abilities, in order to further explore the role of CUEDC2 in colorectal cancer invasion and metastasis, and find a certain theoretical basis.
Patients and methods
Study population and specimen collection
Tumour tissue specimens were collected from consecutive patients with primary colon cancer with or without lymph node metastasis, who underwent surgical resection at the Department of General Surgery, the Chinese People's Liberation Army General Hospital, between December 2015 and February 2016. Tumour tissues were diagnosed as colon carcinoma by preoperative imaging and postoperative pathology.
Patients were included if they met the following criteria: (1) 18–75 years of age; (2) had undergone surgery at the People's Liberation Army General Hospital and the operating surgeons were above the level of deputy chief physician; and (3) postoperative pathology confirmed the diagnosis of colon cancer according to the 7th edition of the American Joint Committee on Cancer Staging Manual. Exclusion criteria comprised: (1) acute or chronic infection; (2) multiple primary malignancies; (3) perioperative death; or (4) undergoing emergency surgery.
The study was approved by the ethics committee of the Chinese People's Liberation Army General Hospital, Beijing, China, and all patients provided written informed consent to participate in the study.
Colorectal cell lines
The following two human colorectal cancer cell lines were obtained from the Chinese People's Liberation Army General Hospital of the General Surgery Institute: SW620 colorectal adenocarcinoma cell line, originally derived from lymph node metastasis of colon tissue and HT29 colorectal adenocarcinoma cell line, originally derived from a primary colon tumour by explant culture. For the experiments detailed below, cell lines were cultured in Corning 25 cm2 culture flasks at 37°C/5% CO2 in HyClone Dulbecco's Modified Eagle's medium (Fisher Scientific, Waltham, MA, USA) supplemented with 10% HyClone fetal bovine serum (Fisher Scientific).
Reverse transcription and real-time polymerase chain reaction (PCR)
To investigate levels of CUEDC2 mRNA in different types of colorectal cancer cells, total RNA was extracted from fresh colorectal cancer tissue (200 mg per sample) and human colorectal cancer cells (1 × 106 cells/cm2) using RNAiso™ Plus (Takara Bio, Kusatsu, Shiga, Japan) according to the manufacturer’s instructions. Total RNA was synthesized to cDNA by reverse transcription using PrimeScript™ RT Master Mix (Takara) according to the manufacturer’s instructions. The cDNA was then used as a template for real-time PCR amplification of CUEDC2. Each 20 µl reaction mix contained the following: 10 µl SYBR Premix Ex Taq™ II (with dNTPs; Takara), 2 µl cDNA, 7.2 µl ddH2O and 0.2 µmol/l of the following primers: CUEDC2 forward GATGCCAGGAACAAAGAGAA and reverse CTCATCTTGGGTGTCTGC; or GAPDH forward GAAGGTGAAGGTCGGAGTC and reverse GAAGATGGTGATGGGATTTC as internal control. The PCR reactions were performed using an Applied Biosystems real-time PCR system (ThermoFisher Scientific, Waltham, MA, USA) and the following cycling programme: preliminary denaturation at 95°C for 30 s, followed by 40 cycles of denaturation at 95°C for 5 s, annealing at 60°C for 34 s, and elongation at 72°C for 10 s. The mRNA levels were analysed using the comparative Ct method (2-△△Ct). The experiment was repeated three times for each tissue sample and each cell line.
Western blot
Total protein was extracted from fresh colorectal cancer tissues and human colorectal cancer cell lines (1 × 106 cells/cm2) using radioimmunoprecipitation assay (RIPA) buffer for cell lysis, and protein was quantified by a standard bicinchoninic acid (BCA) method. Aliquots of total protein (20 µg each) were separated by sodium dodecyl sulphate (SDS)-polyacrylamide gel electrophoresis (PAGE), transferred to nitrocellulose membranes (CWBIO Company, Beijing China), and blocked for 1 h at room temperature using 5% skimmed milk powder (Mengniu Dairy Co., Ltd., China) in Tris-HCl buffered saline (TBS)-0.1% Tween-20 (Beijing Suolai Bao Technology Co., Ltd). Membranes were then incubated with rabbit anti-CUEDC2 (1:500 dilution) and anti-GAPDH (1:1000 dilution) primary polyclonal antibodies (both Abcam, Cambridge, UK) overnight at 4°C and washed three times 10 min each in TBS-Tween-20 buffer at room temperature. Membranes were then incubated with horse-radish peroxidase (HRP)-conjugated goat anti-rabbit secondary antibody (1:1000 dilution; CWBIO Company) for 45 min at room temperature, and washed three times 10 min each in TBS-Tween-20 buffer. The immunoreactive signal was visualised using an enhanced chemiluminescence kit according to the manufacturer’s instructions reagents. Results were evaluated using a G:BOX imaging system (Syngene, Cambridge, UK) with Kodak X-Omat film, followed by relative quantification by determining the mean optical density of each sample and calculating the CUEDC2:GAPDH ratio. The experiment was repeated three times.
Immunohistochemistry
Fresh tissue samples were fixed with 4% formalin followed by routine dehydration and paraffin embedding. Tissue blocks were cut into 4 µm thick sections using an automatic microtome and then underwent dewaxing in xylene and rehydration by alcohol gradient (anhydrous C2H5OH→95% C2H5OH→90% C2H5OH→80% C2H5OH→70% C2H5OH) for 5 min at each concentration. Sections were then washed with 0.01 mol/l phosphate buffered saline (PBS; pH 7.4), followed by antigen retrieval in 10 mM sodium citrate buffer (pH 6.0) and autoclave at 120°C for 2.5 min. Endogenous peroxidase activity was blocked by incubating the sections for 10 min at room temperature in 3% H2O2 solution. Sections were then blocked with goat serum for 30 min at room temperature, followed by incubation with rabbit anti-human CUEDC2 polyclonal primary antibody (1:200 dilution; Abcam) at 4°C overnight. Sections were then washed three times in TBS-Tween-20, incubated with goat anti-rabbit biotinylated secondary antibody (1:200 dilution; CWBIO Company) for 1 h at room temperature, then incubated with HRP-conjugated streptavidin at 37C for 30 min. One drop of DAB colour solution (Sigma, USA) was added per section, covered and incubated at room temperature 15 min. Sections were counterstained with haematoxylin (CWBIO Company) and dehydrated in gradient alcohol (5 min per concentration) followed by xylene fixation (xylene I and II, 10 min each). PBS was used as a negative control and breast cancer tissues known to express high levels of CUEDC2 protein, were used as a positive control.
A semi-quantitative scoring system, based on staining intensity and range, was used to evaluate the immunohistochemistry results. Staining intensity scoring criteria were as follows: no staining = 0; light yellow (low intensity staining) = 1 point; yellow or dark yellow (medium intensity staining) = 2 points; and brown or dark brown (high intensity staining) = 3 points. Staining range scoring criteria comprised the number of positive cells out of all cells counted in five high power fields (400 × magnification), to obtain the proportion of positive cells stained, where: 0% = 0; 1–24% = 1 point; 25–49% = 2 points; 50–74% = 3 points; and 75–100% = 4 points. Immunohistochemical results were scored by multiplying the staining intensity score by the staining range score, where: 0 points is negative (–), 1–3 points is weakly positive (+), 4–7 points is moderately positive (++), and ≥8 points is strongly positive (+++). In the present study, immunohistochemical scores were defined as follows: low expression, 1–7 points; high expression, ≥8 points; and not expressed, 0 points.
Statistical analyses
The Western blot immunoreactive signals were analysed using Quantity One software, version 4.6.6 (Bio-Rad Laboratories, Hercules, CA, USA). Statistical analyses were performed using IBM SPSS software, version 17.0 (SPSS Inc., Chicago, IL, USA) and graphs were generated using GraphPad Prism software, version 5 (GraphPad Inc., CA, USA). Quantitative variables are presented as the mean ± SD. Measurement data with a normal distribution and homogeneity of variance were analysed for statistical differences using Student’s t-test. A P-value < 0.05 was considered statistically significant.
Results
Tissue specimens were collected from 16 patients diagnosed with primary colon cancer (mean ± SD age, 61.4 ± 5.4 years; male:female ratio, 11:5). Eight cases were associated with lymph node metastasis and eight were not, with no statistically significant between-group differences in terms of age or sex.
CUEDC2 mRNA levels
Reverse transcription with real-time PCR showed that levels of CUEDC2 mRNA in tumour tissues from patients with colorectal carcinoma and lymph node metastasis was significantly higher than in tissue from patients without lymph node metastasis (P <0.05, n = 8; Figure 1a). Levels of CUEDC2 mRNA were also significantly higher in SW620 cells, originally derived from lymph node metastasis, compared with HT29 cells (P <0.05, n = 3; Figure 1b).

CUE domain containing 2 (CUEDC2) mRNA levels in colorectal cancer: (a) fresh colon cancer tissue from patients with lymph node metastasis (LN+) versus without lymph node metastasis (LN−) (each bar represents one sample, each assayed in triplicate); and (b) colorectal cancer cell lines HT29 versus SW620; Data presented as mean ± SD, #P <0.05 versus HT29 cell line (Student’s t-test).
CUEDC2 protein levels
Western blot analysis showed that CUEDC2 protein levels were significantly higher in patients with colorectal carcinoma and lymph node metastases compared with patients without lymph node metastasis (P <0.05, n = 8) (Figure 2a). CUEDC2 protein levels were also significantly higher in SW620 cells compared with HT29 (P < 0.05, n = 3) (Figure 2b).

Representative Western blots and summary data of CUE domain containing 2 (CUEDC2) protein levels in colorectal cancer: (a) fresh colon cancer tissue from patients with lymph node metastasis (LN+) versus without lymph node metastasis (LN−); and (b) colorectal cancer cell lines HT29 versus SW620; Data presented as mean ± SD, #P <0.05 compared with (a) LN- group and (b) HT29 cells (Student’s t-test).
Immunohistochemistry results showed moderate CUEDC2 staining in the nuclei and cytoplasm of the lymph node metastasis group (Figure 3a), and weak staining in the non-lymph node metastasis group (Figure 3b).
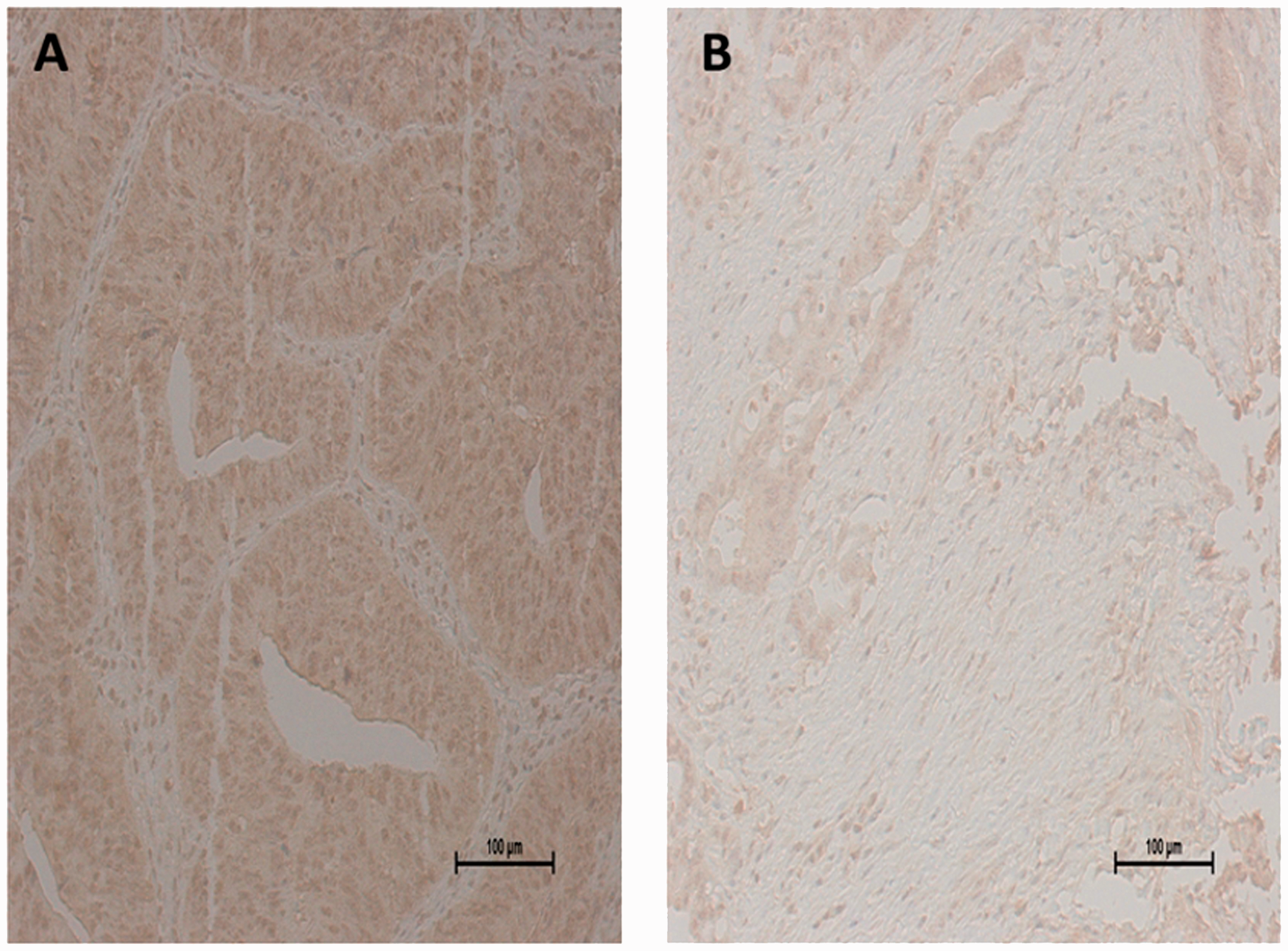
Representative photomicrographs showing immunohistochemical staining of CUE domain containing 2 (CUEDC2) protein in fresh colon cancer tissue from patients (a) with or (b) without lymph node metastasis; 200 × Magnification.
Immunohistochemical staining intensity and range-based scoring revealed that the mean CUEDC2 immunohistochemical score for colorectal carcinoma tissue in patients with lymph node metastasis was significantly higher than in patients without lymph node metastasis (P <0.05, n = 8) (Figure 4).

Immunohistochemical analysis of CUE domain containing 2 (CUEDC2) protein levels in fresh colon cancer tissue from patients with lymph node metastasis (LN+) versus without lymph node metastasis (LN−); Data presented as mean ± SD staining scores, #P <0.05 versus LN- group (Student’s t-test).
Discussion
The incidence of colorectal cancer is high, 1 and 50% of patients die from complications due to distant metastasis.3,4 Understanding the invasion and metastasis mechanisms in colorectal cancer is of great significance to facilitate early detection in patients with colorectal cancer, and for targeted diagnosis and treatment measures. 3 Although the mechanisms governing invasion and metastasis in colorectal cancer are known to be complex, they currently remain unclear.
The CUEDC2 protein has a molecular weight of about 32 kDa and an amino acid number of 287, making it acidic. 8 The amino acid sequence of CUEDC2 contains a coupling of ubiquitin conjugation to ER degradation (CUE) domain in the amino acid sequence from position 144 to position 187. 8 CUEDC2 is ubiquitously expressed in all tissues and organs, with high expression levels in the brain, heart and testis. 8 CUEDC2 plays an important role in the regulation of cell physiological activities and in tumour development. 8
Expression of CUEDC2 has been shown to induce anaphase-promoting complex (APC/C) activation, resulting in abnormal mitotic chromosomal separation, which may promote tumour progression. 8 The activation of signal transducer and activator of transcription (STAT)3 can prompt cell-induced tumour angiogenesis, inhibit the body's anti-tumour immunity, and promote tumour progression. CUEDC2 has been shown to enhance the stability of the janus kinase (JAK)-STAT pathway inhibitor, suppressors of cytokine signalling (SOCS)3, by promoting the binding of extension protein C to SOCS3. The interaction of CUEDC2 with SOCS3 inhibits the cytokine-mediated JAK1-STAT3 pathway.12–14 Thus, CUEDC2 plays a role both in promoting and inhibiting tumorigenesis and progression through the regulation of physiological activities. The specific role of CUEDC2 in tumours has attracted wide attention from scientists.
Zhang et al., 15 found that CUEDC2, through CUE domain and progesterone receptor (PR) binding in the 180–226 amino acid region, inhibits the PR activation-mediated mitogen-activated protein kinase (MAPK) pathway, thus reducing the effect of progesterone in breast cancer cell growth. CUEDC2 was also found to promote progesterone-induced PR degradation through the ubiquitin-proteasome pathway. 15 Another study found that CUEDC2-positive expression was associated with a shorter disease-free survival time, and that CUEDC2 may be a promising biomarker for evaluating the progression of serous ovarian carcinoma and to predict the likelihood of ovarian serous carcinoma relapse. 11 Conversely, decreased expression of CUEDC2 has been suggested to contribute to tumour growth in lung adenocarcinoma, leading to a poor clinical outcome. 16
To date, the role of CUEDC2 in the invasion and metastasis of colorectal cancer has not been reported. The present study showed that CUEDC2 expression, in terms of mRNA and protein levels, was significantly higher in colon cancer tissues from patients with lymph node metastasis than in patients without lymph node metastasis, suggesting that CUEDC2 might play an important role in the lymph node metastasis of colon cancer. The SW620 cell line was originally obtained from lymph node metastasis of colorectal cancer, while the HT29 cell line was obtained from the explant culture of a primary colorectal cancer tumour, and the invasion and migration capacity decreased gradually. Levels of CUEDC2 mRNA and protein in colorectal cancer cells with high invasion and migration abilities (SW620 cells) were significantly increased versus HT29 cells, suggesting that CUEDC2 expression intensified with the increased invasion and migration ability, and thus may promote invasion and migration in colorectal cancer. Immunohistochemical analysis of tumour tissues demonstrated that CUEDC2 expression was higher in patients with lymph node metastasis than in patients without lymph node metastasis. CUEDC2 was found to be strongly expressed in the nuclei and cytoplasm of cells from patients with lymph node metastasis and only weakly expressed in the cytoplasm of cells from patients without lymph node metastasis, suggesting that CUEDC2 may play a role in promoting the lymphatic metastasis of colon cancer through activities in the nucleus and cytoplasm.
In a study into the role of CUEDC2 in an animal model of inflammatory bowel disease, Wang et al. 17 revealed that CUEDC2 played a vital role in preventing excessive proliferation and inhibiting the nuclear factor (NF)-κB and STAT3 signalling pathways, suggesting that CUEDC2 may prevent colonic inflammation associated with cancer development. The research also revealed that CUEDC2 knockout mice had increased body weight loss, more severe colonic mucosal injury, worse diarrhoea and significantly elevated pro-inflammatory cytokines (interleukin [IL]-6, tumour necrosis factor-α, cyclooxygenase-2, and macrophage inflammatory protein-2) than control mice. 17 Moreover, the NF-κB and STAT3 signalling pathways were increasingly activated and the percentage of proliferating cell biomarkers (Ki67, CyclinD1, BrdU) in the inflammatory tissues was significantly raised in CUEDC2 knockout mice. 17 CUEDC2 has also been shown to inhibit tumour proliferation through inactivation of the phosphatidylinositol-3-kinase/protein kinase B(Akt) pathway, induction of p21 and down-regulation of cyclin D1. 16 Decreased expression of CUEDC2 was shown to contribute to tumour growth in patients with lung adenocarcinoma who had a poor clinical outcome and increased CUEDC2 expression was associated with decreased cell proliferation in vitro and inhibited tumour growth in nude mice in vivo. 16 Moreover, knockdown of endogenous CUEDC2 promoted tumour growth. 16 Another study reported that CUEDC2 inhibited invasion and metastasis of breast cancer by inhibiting phosphorylation of IκB kinase to block NF-κB pathway activation through regulation of the IL-4/miR-324-5p/CUEDC2 axis. 18
Conversely, a separate study reported that CUEDC2 played an important role in promoting tumour progression through aerobic glycolysis, or the Warburg effect. 19 CUEDC2 was shown to upregulate the two key glycolytic proteins (glucose transporter [GLUT]3 and lactate dehydrogenase (LDH]A) by interacting with the glucocorticoid receptor or 14-3-3ζ, respectively. The researchers further showed that increased aerobic glycolysis was essential for the role of CUEDC2 in driving cancer progression. 19 Moreover, they demonstrated a correlation between the abnormal expression of CUEDC2, and GLUT3 and LDHA in clinical human hepatocellular carcinoma samples, further illustrating a relationship between CUEDC2 and the Warburg effect during cancer progression. 19
In the present study, CUEDC2 was highly expressed in the nuclei and cytoplasm of colon cancer tissues from patients with lymph node metastasis, and colorectal adenocarcinoma cell lines with high invasive migration properties also expressed high levels of CUEDC2. CUEDC2 may play an important role in the invasion and metastasis of colorectal cancer. The results of the present study may be limited by the fact that this was a single centre study with a small sample size, and there may have been selection bias. Multicentre tissue sample evaluation with a larger sample size may yield more reliable results. The specific role and mechanism of CUEDC2 in colorectal cancer invasion and metastasis remains to be elucidated. Clear understanding of the invasion and metastasis mechanisms underlying colorectal cancer has important clinical significance for improving the prognosis of patients with colorectal cancer.
In conclusion, CUEDC2 is highly expressed in colorectal cancer tissues and colorectal cancer cell lines with high invasion and migration ability, and CUEDC2 may play a role in promoting invasion and metastasis in colorectal cancer via nuclear and cytoplasmic activity.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Footnotes
Funding
This work was supported by the National Natural Science Foundation of China (No. 61471397).
