Abstract
Introduction.
Moleac (MLC) 901 is a traditional Chinese medication approved by the Sino Food and Drug Administration in 2001 for treating stroke. This study aims to analyze the efficacy of MLC901 in animal stroke models after medial cerebral artery occlusion (MCAO).
Methods.
Literature selection was performed according to the guidelines of the Preferred Reporting Items for Systematic Review and Meta-Analysis Protocols (PRISMA) 2015. Inclusion criteria for the experimental studies were the use of animal models, publication in English between 1990 and 2020, information regarding the intervention technique used, and outcomes regarding the efficacy of MLC901 administration.
Results.
MLC901 administration resulted in significantly less infarction volume by a mean difference of 17.17 compared to the control group (p < .00001). The MLC901 group resulted in significant improvement in 5-bromo-20-deoxyuridine (BrdU)-positive cells expression by a mean difference of 662.79 (p < .00001) and neurological function, which was indicated by a mean difference in the Bederson Neurological Outcome Score of 1.40 (p < .00001).
Conclusions.
MLC901 administration in an animal stroke model resulted in a better reduction in infarction volume and improvement in BrdU expression and neurologic function. These data could help in further determining the efficacy of MLC901 for acute ischemic brain injury in humans.
Introduction
Strokes can be classified into 2 major categories: ischemic stroke and hemorrhagic stroke. Ischemic strokes account for about 80% of stroke cases, while hemorrhagic strokes account for 20%, although the proportions of types depend on the population. 1 In the United States, 795,000 people experience a stroke, and 610,000 of these are new strokes. 2 In Europe, the incidence of stroke ranges from 95 to 290 per /100,000 per each year based on recently published studies. 3 The incidence of transient ischemic attack (TIA) ranges from 28 to 59 per /100,000 each /year. 3
Stroke is the cause of 15.4% of all deaths in Indonesia. Based on age and sex, it has a mortality rate of 99/100,000 cases. 4 The prevalence itself has reached 0.0017% and 0.022% in rural and urban parts of Indonesia, respectively. 4 Although stroke remains a leading cause of morbidity and mortality worldwide, numerous failures in stroke trials have led to some pessimism regarding stroke care. In this vein, NeuroAiD (MLC601) has previously been reported to have some beneficial clinical effects in stroke patients. 5
Moleac (MLC) 901 is a traditional Chinese medication approved by the Sino Food and Drug Administration in 2001 and used widely as a treatment for post-stroke patients to improve neurological function in China, especially in the non-acute phase. 6,7 MLC901 is a simplified version of a previous form, MLC601, which contains 9 herbal components, namely Radix Astragali, Radix Salviae Miltorrhizae, Radix Paeoniae Rubra, Rhizoma Chuanxiong, Radix Angelicae Sinensis, Carthamus Tinctorius, Prunus Persica, Radix Polygalae, and Rhizoma Acori Tatarinowii. 5 The traditional Chinese medicine added to the MLC901 capsule has the effect of Qi restoration and blood circulation improvement after ischemic stroke based on traditional Chinese medication theory. 8 Several pharmacologic studies have shown that NeuroAiD II® has neuroprotective effects on neurons undergoing ischemic damage, leading to cognitive function and post-stroke recovery. The neurotropic effects of this medication are derived through stimulation of cell proliferation, neurite growth, and synaptogenesis and the prevention of hippocampal damage from global ischemic injury. 7,9 Previous studies have also reported its effectiveness in improving outcomes in stroke through the promotion of neuroprotection and brain plasticity as well as increased cerebral blood flow (CBF). 10 A correlation between MLC901 administration and decreased infarct volume, the stimulation of basal neurogenesis, 11 and neurological outcome (as shown as Bederson neurological outcome score) has also been reported. 12,13
Rodents have been known to serve as excellent models for studying human stroke. Tagaya et al in 1997 demonstrated the first reported increase in significant cellular damage of the striatum in rodent animals after performing middle cerebral artery occlusion (MCAO). 14 The damage from brain injury was assessed by Bederson et al in 1986 after performing the same procedure, which led to the development of a grading system through several tests on rats called the Bederson Neurological Outcome Score for assessing and classifying the degree of brain injury. 15 Infarct volume has long been associated with the outcome of ischemic stroke in animal models and humans. 16 It has been studied with the use of multiple therapeutic approaches to treating cerebral infarction, such as endovascular recanalization therapy. 17,18 MLC901 has been reported to have a beneficial effect on total infarct volume, with a study by Heurteaux et al confirming reduced infarct volume after MCAO after MLC901 administration. 5 Further, 5-bromo-20-deoxyuridine (BrdU) has been evaluated as an indicator for infarct volume in animal models through immunohistochemical staining along with the use of Ki67, a protein specifically expressed during the active phases of the cell cycle. BrdU is a nucleotide analog incorporated into DNA during the process of replication. 19
This meta-analysis aims to analyze the efficacy of MLC901 in reducing infarction volume, increasing neuroploriferation as shown as BrdU (5-bromo-20-deoxyuridine)-positive cells in the dentate gyrus subgranular zone (SGZ), and neurological function through the Bederson Neurological Outcome Score in animal models with acute ischemic stroke.
Methods
Information Source and Search Strategy
The literature selection was performed according to the guidelines of the Preferred Reporting Items for Systematic Review and Meta-Analysis Protocols (PRISMA) 2015. 20 A literature search was conducted through MEDLINE/PubMed, ScienceDirect, Cochrane Library and Google Scholar (Table 1).
Literature Search Date.

Search strategy was using MeSH terms in MEDLINE/PubMed (Table 2). Articles were excluded if the title or abstract did not fulfill the inclusion criteria. Full-text reviews were carried out on several articles depending on the inclusion eligibility criteria.
MeSH Terms (MEDLINE/PubMed).

Eligibility Criteria
Criteria for inclusion of the experimental studies were the use of animal models, publication in English between 1990 and 2020, information regarding the animal model and technique of the intervention used, and outcomes regarding the efficacy of MLC901 administration. Studies with ischemic stroke animal models and the administration of NeuroAiD II (MLC901) were also included (Table 3).
Population-Intervention-Control-Outcome (PICO).

Review articles, editorials, commentaries, or publications without a peer-review process were excluded from this systematic review. Meta-analyses, systematic reviews, and randomized controlled trials were also excluded. Studies on cases other than acute ischemic brain injury in samples were also excluded.
Data Extraction
Extracted data included study characteristics (publication year; sample size), efficacy results (infarction volume; BrdU expression; Bederson Outcome Neurologic Score). Data extraction was conducted independently by 3 investigators. Any differences were resolved by senior author.
Risk of Bias Assessment
We used Systematic Review Center for Laboratory Animal Experimentation (SYRCLE)’s risk of bias tool for experimental studies on animal models to rate risk of bias of the included studies. 21 We syntesised data in narrative and summary of findings in tabular formats.
Statistical Analysis
This meta-analysis utilized Statistical Package for the Social Sciences (SPSS) version 24 and Revman version 5.4 from the Cochrane Review for statistical analysis. 20
Results
Study Selection and Bias
The search strategy identified a total of 105 studies after the duplicates were excluded, 4 of which were selected for further evaluation on the basis of the inclusion criteria and content, as shown in Figure 1. The risk of bias was assessed using Systematic Review Centre for Laboratory Animal Experimentation (SYRCLE)’s risk of bias tool for experimental studies on animal models. 21 The results of the assessment of the risk of bias are displayed in Table 4.

PRISMA flowchart.
Risk of Bias Assessment Tool Using SYRCLE.

In all the studies analyzed for full-text review, the baseline characteristics of the samples, including age and weight, were provided. However, the randomization process of the assignment to the treatment or control group was not clearly elucidated. All studies explained the blinding of the researchers when administering the intervention. However, all studies except for one (Widmann et al, 2018) mentioned the blinding of the researchers with respect to the outcome data extraction. Biases of attrition and data reporting were not clearly elucidated.
MLC901 administration resulted in significantly less infarction volume by a mean difference of 17.17 mm3 compared to the control group (p < .00001; Figure 2).

Forest plot of the effect of MLC 901 on infarction volume (in mm3).
In terms of BrdU expression, MLC901 administration resulted in a significantly higher number of expressions by a mean difference of 662.79 positive cells (p < .00001; Figure 3).

Forest plot of the effect of MLC 901 on BrdU expression.
In terms of neurological function, MLC 901 resulted in a lower Bederson Neurological Outcome Score, indicating better neurologic function, by a mean difference of 1.40 (0-3) (p < .00001; Figure 4).
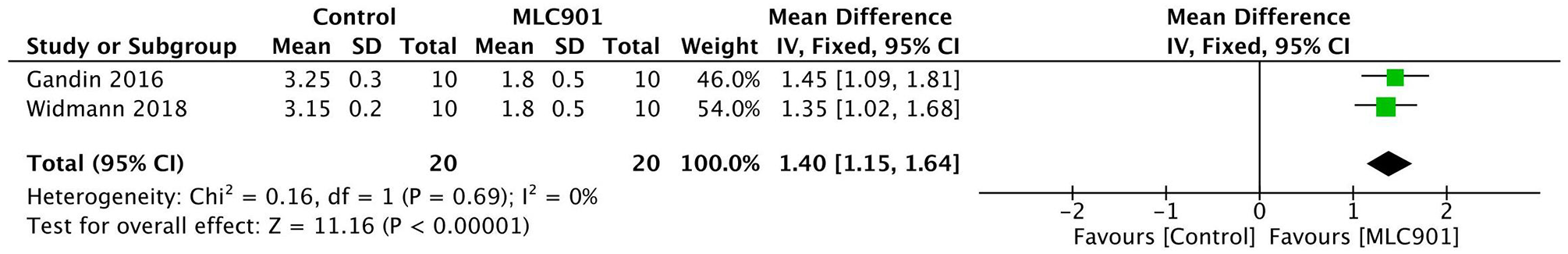
Forest plot of the effect of MLC 901 on Bederson Outcome Neurologic Score.
Discussion
In this systematic review, we evaluated the effect of MLC901 on infarction volume reduction, BrdU-positive cells expression, and Bederson Neurological Outcome Scores. MLC901 is known to be widely used as a treatment for post-stroke patients to improve neurological function, especially in the non-acute phase in China. 6,7
Studies have shown that MLC901, the active component of the drug NeuroAiD II®, improves recovery in the acute and recovery phases of stroke. It is also considered to be safe and well-tolerated, and it can reduce early vascular events since it has been shown not to modify hemostasis, hematology, and biochemistry properties in normal subjects and stroke patients. 22 Several studies on animal models of acutely ischemic brain injury have also shown promising results in regard to MLC901 administration. 9,11 –13 These studies included infarction volume reduction of the brain, BrdU-positive cells expression as an indicator of neuroploriferation capability, and Bederson Outcome Neurological Score as an indicator of neurological function.
Infarction Volume
Three out of the 4 studies were analyzed for their results on the effect of MLC 901 on infarction volume. 5,12,13
Gandin et al (2016) compared the infarction volume of the brains in rats after MCAO and treatment with MLC901 for 5 weeks before the MCAO induction and sacrifice of the mice for analysis. 12 The reported total infarct volume was lower in the group administered MLC901 compared to the vehicle-treated group (56.1 ± 0.3 vs. 39.8 ± 0.2; p < .01). Furthermore, the treatment group also showed significantly reduced brain edema compared to the vehicle group. This is consistent with the theory that MLC901 promotes neurogenesis and synaptogenesis with an increase of brain-derived neurotrophic factor (BDNF), thus preventing further brain damage and stimulating brain plasticity and subsequent neuro-repair after brain injury. 12 Although this study administered MLC901 5 weeks prior to MCAO, several other studies confirmed the efficacy of MLC901 administration after the incidence of acute brain ischemia itself. 7,11
Heurteaux et al (2010) reported significantly decreased infarction volume on acute ischemic stroke in mice treated with MLC901 (p < .01). In this study, the acute administration of MLC901 induced a survival rate of 90% compared to 69.5% in ischemic vehicle-treated mice. 5 Additionally, the study also reported a markedly increased survival rate in rats with a six-week pre-treatment protocol of MLC901 during the MCAO procedure. 5 This could support the hypothesis that MLC901 works not only in improving stroke-related health problems but also in preventing them.
Consistent with the previous studies, Widmann et al (2018) also reported a significantly reduced cerebral infarction volume in mice treated with MLC901. 13 Several other protective effects of MLC901 on acute ischemic brains in mice were also reported, including blood-brain-barrier (BBB) breakdown and cerebral edema. 13
All 3 studies yielded results consistent with the theory that MLC901 exerts neuroprotective effects moments after acute ischemic brain injury based on a particularly thorough assessment of brain infarction volume reduction. Moreover, the studies also reported its protective effect on acute ischemic brain injury during the event itself, as indicated through increased survival rates during MCAO procedures.
BrdU (5-Bromo-20-Deoxyuridine)
Detection of BrdU was performed using immunohistochemistry assessment. BrdU is a nucleotide analog incorporated in DNA during replication, and its presence could help indicate cells and, in this case, neuroproliferation of rat’s neuron cells of hippocampus. 19 Lorivel et al mentioned, BrdU assessment was performed by first injecting BrdU into the animal along with the experiment. After the brain of the animal is removed and prepared for the examination, an anti-BrdU antibody is administered, which will help BrdU detection later on. Two studies were analyzed for their reports on the effect of MLC901 on BrdU expression. 5,19
Heurteaux et al (2010) reported a clear increase in the number of BrdU-labeled cells in the subgranular zone (SGZ) on mice treated with MLC901. This indicated enhanced basal neurogenesis since BrdU itself is a DNA synthesis marker. 5
Lorivel et al (2015) reported a significant increase in the number of hippocampal BrdU-positive cells in samples treated with MLC901 compared with controls. 19 The presence of profound BrdU-positive cells also indicated the promoted differentiation of newborn cells into neurons, which is an important aspect of neuroproliferation in the recovery of neurological disease. 13 Moreover, mice treated with MLC901 also showed an increased amount of BrdU in extended periods of the course of the disease. In fact, there was as much as a 2.1-fold increase in the number of mature neurons as compared to the vehicle-treated/control group of mice. 13
Bederson Neurological Outcome Score
Aside from histological measures, functional assessment in animal models has proven useful for acquiring information about the neuronal cells biology and rehabilitation needed for experimental stroke. Rodents serve as an excellent animal model to study and research human stroke, aside from many other diseases of neurology. 15 Functional assessments are useful in identifying the disease progression and degree of damage over a certain period of time while also providing the data necessary to determine the appropriate therapeutic treatment for stroke. 23
Strokes that affect unilateral cerebral vascularization result in a contralateral neurological deficit in both humans and rodents and a compensatory reliance on the healthier side of the body. 24 This manifestation could be of use in performing tests for neurological impairment through the asymmetries detected. 25 In 1986, Bederson et al developed a neurologic grading system that can be used to rapidly evaluate the effects of cerebral ischemia on rats. 15 Bederson and his team developed this system by occluding various locations of the MCAs of rats, which also produced various degrees of cerebral infarction, and evaluating the neurologic status through several tests, which also resulted in various degrees of neurological impairment after MCAO.
The tests assess mainly the motoric function of the rats. For example, the rats are held gently by the tails, suspended one meter above the floor, and observed for forelimb flexion; normal rats tend to extend both forelimbs, while rats with the worst infarction consistently flex their wrists and elbows while also adducting and internally rotating the shoulder contralateral to the injury. The other tests include assessments of gripping a soft paper with a claw and determining the direction in which the rats moved freely, with impaired rats tending to circle toward the weaker side. The grading system ranges from 0 to 3, with 0 being normal (no observable deficit), 1 being moderate (forelimb flexion), and both 2 (decreased resistance of lateral push and forelimb flexion without circling) and 3 (same behavior as grade 2, with circling) being severe.
Of all the studies analyzed in this review, 2 were relevant for assessment regarding the effect of MLC901 on Bederson Neurologic Outcome Scores in animal models with acute ischemic brain injury.
Gandin et al (2016) reported significantly superior neurological function when evaluating the Bederson Outcome Neurological Scores. Moreover, MLC901-treated mice also showed a nearly similar behavior compared to the sham group on day 3 after the MCAO procedure, indicating a better performance compared to the vehicle-treated ischemic mice. 12
Consistent with the previous study, Widmann et al (2018) also reported neurological outcome improvement in MLC901-treated mice 30 hours after the MCAO procedure was performed, suggesting better functional recovery.
Study Limitations
This review was limited to experimental studies only due to the nature of MLC901 being relatively new drugs hence had not been used widely in clinical practice. Experimental model did not allowed us to study on heterogeneous effect of the medication due to homogenous sample of the animal model. Thus made this review not yet readily applicable to human. Several clinical trials are ongoing but still in early phase. This review marked the early phase of MLC901 efficacy as a reference for observing the ongoing studies ahead.
Conclusions
MLC901 administration on animal models of acutely ischemic brain injury has been shown to improve recovery when assessed through the infarction volume, BrdU expression of the brain, and neurological function testing using the Bederson Neurological Outcome Score. MLC901 could be of use in treating patients who undergo recovery from acute brain ischemic injury, with further research specifically including human samples.
Footnotes
Authors’ Note
Conceptualization, I G. M. Aswin R. Ranuh and Asra Al Fauzi; methodology, Gadis Meinar Sari, Budi Utomo. and Asra Al Fauzi; software, I G. M. Aswin R. Ranuh and Asra Al Fauzi; validation, Gadis Meinar Sari, Budi Utomo, Nur Setiawan Suroto and Asra Al Fauzi; formal analysis, Gadis Meinar Sari, Budi Utomo, Nur Setiawan Suroto and Asra Al Fauzi; investigation, I G. M. Aswin R. Ranuh, Gadis Meinar Sari and Budi Utomo; resources, I G. M. Aswin R. Ranuh and Asra Al Fauzi; data curation, Gadis Meinar Sari, Budi Utomo, and Nur Setiawan Suroto; writing—original draft preparation, I G. M. Aswin R. Ranuh and Asra Al Fauzi; writing—review and editing, I G. M. Aswin R. Ranuh, Gadis Meinar Sari, Budi Utomo, Nur Setiawan Suroto and Asra Al Fauzi; visualization, I G. M. Aswin R. Ranuh and Asra Al Fauzi; supervision, Gadis Meinar Sari, Budi Utomo, Nur Setiawan Suroto and Asra Al Fauzi; project administration, I G. M. Aswin R. Ranuh and Asra Al Fauzi; funding acquisition, Asra Al Fauzi. No ethical approval was required for this manuscript because this study did not involve human subjects or laboratory animals.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by MOLEAC Pte. Ltd., Singapore, the manufacturer of the product MLC901.
