Abstract
Multiple sclerosis is a disabling and progressive neurological disease that has significant negative effects on health-related quality of life. This exploratory efficacy study examined the effects of Healing Light Guided Imagery (HLGI), a novel variant of guided imagery, compared with a wait-list control in patients with relapsing-remitting multiple sclerosis. Changes in the Beck Depression Inventory, Fatigue Severity Scale, and Multiple Sclerosis Quality of Life instrument (physical and mental components) were compared between groups. Patients who completed HLGI (N = 9) showed significant reductions in depressed mood (P < .05) and fatigue (P < .01) and showed significant gains in physical (P = .01) and mental (P < .01) quality of life compared with journaling (N = 8). Our results suggest that HLGI can improve self-reported physical and mental well-being in patients with relapsing-remitting multiple sclerosis. Further research is needed to study the effectiveness of this therapy, as well as its mind-body mechanisms of action.
Multiple sclerosis (MS) is a disabling and progressive neurological disease that affects approximately 400 000 people in the United States. 1,2 MS has a significant negative effect on health-related quality of life, including in domains of emotional, functional, physical, social, and spiritual well-being. 3,4 In a survey of more than 11 000 individuals with MS conducted by the MS Foundation, 57% of patients reported having tried at least one complementary and alternative medicine modality (now commonly called integrative medicine when incorporating complementary and alternative medicine practices into mainstream medical practice). 5 Other studies suggest higher rates (67%) of use of such practices by MS patients. 6,7 MS patients often seek out mind-body therapies, and studies indicate benefits including improved quality of life and physical functioning and moderation of common MS symptoms such as fatigue. 6,8 –12 Recent reviews focusing on potential benefits of mindfulness meditation, for example, suggest an association between meditation practice and lower risk of depression and improved quality of life in people with MS. 9,13 –15 Given known associations between measures of MS disease activity and stressful life events, and the moderating effect of stress management programs on these measures of disease activity, 16,17 it is been postulated that integrative therapies mediate benefits through the reduction of autonomic arousal and stress. 18,19
Among integrative modalities, Guided Imagery (GI) is considered a “relaxation modality” by the National Center for Complementary and Integrative Health, because it can profoundly activate the body’s natural relaxation response as characterized by slower breathing and lower blood pressure as well as increased feelings of well-being. 20 A critical review of the literature by Trakhtenberg 21 suggested that GI can influence the immune system. 21 There is increasing interest in the literature on the efficacy of GI as part of a broader biobehavioral model for disability and depression, which includes its use in the management of inflammatory diseases. 22 –24 Given the potential immune-mediating effects of GI via changes in the hypothalamic-pituitary-adrenal axis, 24 –26 there is a rational foundation for the use of GI in the treatment of immune mediated disorders such as MS to therapeutically modulate immune responses. 21,27 Important indirect effects of GI also include adherence to other positive lifestyle activities and medication recommendations. 28,29
A recent trial in clinical groups examining the effectiveness of GI as compared with usual care on fibromyalgia symptoms demonstrated that patients assigned to GI showed significant decreases in fatigue, pain, and depression. 30 Studies on GI effects in breast cancer patients suggest improvements in depression and immune function. 23,31
Despite the promise of the literature suggesting such benefits of GI, few studies have explored the potential benefits of GI in MS. GI and affirmations are believed to stimulate a light self-hypnotic state that has the potential to provide a healing modality to accompany current MS treatment. In a prior quasi-experimental trial comparing the effects of self-hypnosis training with relaxation on pain in 22 MS patients, Jensen et al 32 reported reductions in pain and pain interference associated with self-hypnosis.
The current study examined a novel GI modality, Healing Light Guided Imagery (HLGI). HLGI was developed by an MS patient specifically for MS patients and their symptoms. Unlike hypnosis scripts developed by generalized hypnotherapy practitioners who seek to treat a broad range of conditions using several combinations of scripts available from published sources, HLGI was developed specifically to treat the concerns related to an MS diagnosis. In addition to the relaxation component, the therapeutic use of GI includes using active visualization or directed imagery for changing behavior and the use of receptive imagery to help relieve symptoms and improve mood.
The purpose of this exploratory efficacy study therefore was to examine the potential of HLGI to enhance self-reported quality of life. We hypothesized that HLGI, as compared to an active wait-list control, would improve symptoms of depression and fatigue and improve self-reported quality of life.
Methods
Patients and Procedures
Participants were a convenience sample recruited through study flyers distributed to local and UC San Diego MS clinics and organizations. The ad sought volunteers for a study on “complementary and alternative techniques for improving quality of life in patients with Relapsing-Remitting Multiple Sclerosis (RRMS).” Potential participants underwent a phone screening where the study was further described, and age, sex, diagnosis, and duration of disease were collected. Inclusion criteria included if participants were between 18 and 70 years old, had a current medical diagnosis of RRMS, agreed they were able to read and write in English, and were available for 1 hour weekly at home or at the University of California San Diego School of Medicine.
Ethical Approval
The UC San Diego Institutional Review Board approved the study and all study participants provided written informed consent prior to participation.
Assessments
Screening assessments included the Beck Depression Inventory–II (BDI-II) and the Fatigue Severity Scale (FSS), which were administered over the phone. BDI-II is a measure of mood based on criteria found in the Diagnostic and Statistical Manual of Mental Disorders, Fourth Edition for depressive disorders 33 and is a strong predictor of cognitive performance in MS patients. 34 FSS is a measure of fatigue that differentiates MS patients from controls and is sensitive to changes in fatigue over time. As measured by the FSS, fatigue is largely independent of self-reported symptoms of depression. 35
Potential participants were excluded for severe depression (score of 31 or higher on the BDI) and/or very high levels of fatigue that might interfere with intervention adherence (greater than an average of 6 on the FSS).
Intervention assessments included the BDI-II, FSS, and the Multiple Sclerosis Quality of Life instrument (MS-QOL-54) which were completed at their intake session and again the end of the 10-week intervention. MS-QOL-54 is a multidimensional health-related quality of life measure that combines both generic and MS-specific items into a single instrument. This 54-item instrument generates 12 subscales along with 2 summary scores, and 2 additional single-item measures; we studied the physical health and mental health composite scores. The MSQOL-54 shows good test-retest reliability and good intraclass correlation coefficients (.66-.96.). 36
For this population, the Cronbach α reliability coefficients for the BDI-II and FSS and the physical and mental health MS-QOL-54 instruments were .367, .841, .857, and .736, respectively.
All assessment questionnaires were scored by an experimenter with intervention masked.
Interventions
Patients who met inclusion criteria were invited to enroll in the study and were put in contact with the study coordinator. Study enrollment occurred in person at the University of California San Diego School of Medicine. A study coordinator who was not involved in phone screening conducted the informed consent process and assigned participants to the study arms. Twenty-three individuals were enrolled into the study (Figure 1). Participants were assigned alternately (1:1) to the 2 study arms; 10 hours of HLGI or 10 hours of positive journaling (control condition). If enrolled patients dropped out of the study, we enrolled new patients in order to achieve our intended sample size.
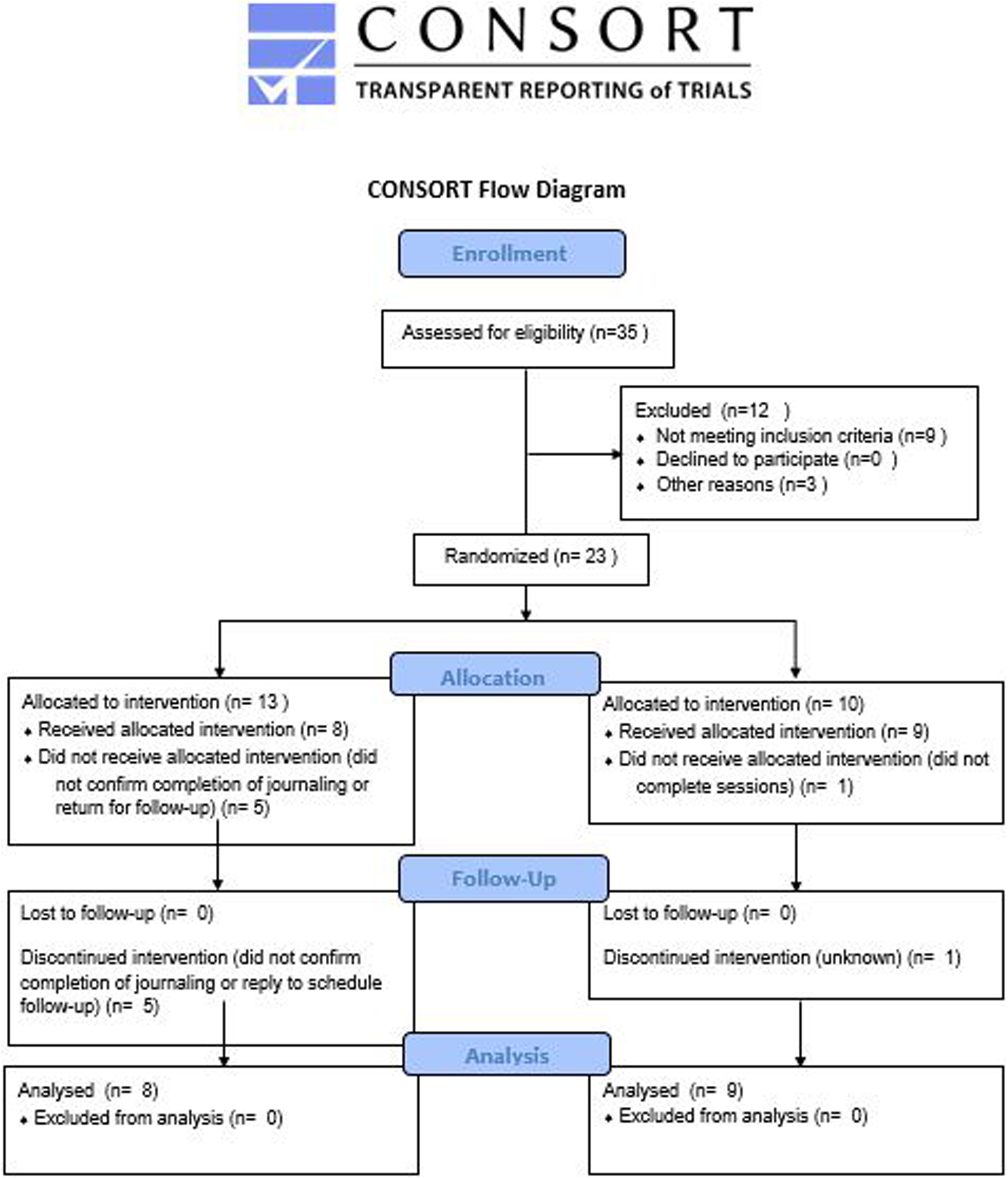
CONSORT (Consolidated Standards of Reporting Trials) diagram.
Patients in the HLGI condition participated in 10 HLGI sessions, which were conducted by a qualified practitioner. Eight of the sessions involved active HLGI; the first and final sessions were intake and completion sessions with little or no active treatment. HLGI followed a common sequence for each participant (see Appendixes A and B) but was highly individualized depending on patients’ responses.
Journaling was chosen as an active wait-list control activity. Patients in the journaling condition were instructed to write at home for 1 hour a week for 10 weeks. Participants were provided with a positive journaling prompt each week. Participants returned their journals to the study investigators at the end of the 10 weeks. Several participants completed the journaling verbally (audio recording) rather than by hand due to pain or limited hand mobility. As part of enrollment, patients were told that those assigned to journaling would receive HLGI following the study, if they so choose.
Data Analysis
This was an exploratory study to examine the effects of HLGI as compared to a wait-list control in patients with RRMS. The purpose was to examine potential effectiveness and, if the findings were significant and promising, to generate data for a subsequent effectiveness trial based on a power analysis using data from this study. Data were analyzed by t tests, 1-way analysis of variance (ANOVA), and 2-way (group × time) repeated-measures ANOVAs (SPSS Version 24). Per examination of box plots and skewness and kurtosis values, data were found to be normally distributed. We report effect sizes in the form of the partial η2 statistic and 95% confidence intervals (CIs).
Results
Nine participants completed HLGI and 8 completed positive journaling. Five participants dropped out (or fell out of contact) in the journaling condition and 1 in the HLGI condition (38% and 10%, respectively). Mean enrollment scores did not differ between participants who completed versus those who did not complete the study for the BDI II (completed M = 8.2, dropped M = 11.6, t(20) = 0.90, P = .38), FSS (completed M = 38.7, dropped M = 48.0, t(20) = 1.22, P = .24), MS-QOL-54 physical composite (completed M = 53.3, dropped M = 49.0, t(19) = 0.47, P = .64), or MS-QOL-54 mental composite (completed M = 69.8, dropped M = 64.3, t(19) = 0.61, P = .55).
Patients ranged in age from 33 to 69 years (mean = 50.8 years, SD = 11.5). Disease duration was 1 to 32 years (mean = 12.1 years, SD = 10.2). There were no significant group differences in age, gender distribution, disease duration or screening scores for depressed mood or fatigue (Table 1).
Relapsing-Remitting Multiple Sclerosis Patient Characteristics According to Group.a

aData are presented as mean ± standard deviation; no significant differences.
Separate 2-way (group × time) repeated-measures ANOVAs showed that HLGI led to significant improvements in each of the 4 study outcomes (Figure 2). HLGI led to a 75% decrease (95% CI, 4.18-11.7) in depressed mood scores (vs a 15% decrease with journaling (95% CI, 1.68-5.44), F(1, 8.3) = 5.9, P = .04, partial η2 = 0.34), and a 24% decrease in fatigue scores (95% CI, 27.6-43.1) (vs a 6% increase with journaling (95% CI, 27.6-43.1), F(1, 15.3) = 11.1, P = .004, partial η2 = 0.35). Regarding quality of life, HLGI led to a 38% increase in physical quality of life scores (95% CI, 43.3-64.3) (vs a 3% increase with journaling (95% CI, 53.5-74.6), F(1, 14.2) = 8.7, P = .01, partial η2 = 0.38), and a 30% increase in mental quality of life (95% CI, 60.4-80.0) (vs 0% change with journaling (95% CI, 71.1-87.5), F(1, 14.4) = 9.8, P = .007, partial η2 = 0.41).

Change in scores of multiple sclerosis (MS) patients from before to after Healing Light Guided Imagery (HLGI) or positive journaling for outcome measures fatigue (Fatigue Severity Scale), depressed mood (Beck Depression Inventory II), and MS physical and mental quality of life (MS-QOL-54 domain scores). HLGI was associated with reductions in depression (P < .05) and fatigue (P < .01), and increases in physical (P = .01) and mental (P < .01) quality of life. Error bars display standard error of the mean.
Discussion
Guided imagery is a mind-body modality that has demonstrated benefits for stress, depression, and possibly inflammatory diseases. 20,21 The current exploratory study tested a variant of GI called “Healing Light Guided Imagery” (HLGI) and MS patients showed improvements in depression, fatigue, and physical and mental quality of life following practice. All 4 planned outcome measures suggested clinically relevant effects with strong effect sizes that require confirmation in larger controlled studies. These effects were obtained after 10 hours of once-a-week treatment, suggesting that HLGI may be a fruitful relaxation technique to explore for individuals with RRMS. While relaxation, in and by itself, is a benefit to individuals with MS, we find additional benefits of changes in depressed mood and fatigue as well as improvements in the client’s physical and mental quality of life.
The effects of HLGI are consistent with the broader literature on the efficacy of GI for illnesses associated with pain, depression and chronic disability. 30 Although in this efficacy study we did not evaluate potential mechanisms of action of HLGI, studies on other GI modalities suggest the practices support relaxation and beneficial changes in hypothalamic-pituitary-adrenal axis activation, 24 –26 which might be supporting the effects we’ve observed. While it is common for studies of mind-body and relaxation techniques to apply a generalized script or procedure to each individual, HLGI was administered in an individualized manner. While this allowed us to learn less about potential mechanisms involved in HLGI, we believe it is a demonstration of the strength of mind-body interventions in their natural state, that is, as typically individually practiced in the complementary and alternative medicine community.
There are several limitations of this efficacy study, including the modest sample size and a higher drop-out rate in the active wait-list group, which could have led to bias in the results. While patients in the journaling group had been offered HLGI following completion of the study, this did not lead to a high degree of adherence we were anticipating.
A prior phase II clinical trial of cognitive behavioral stress management training on measures of MS disease activity showed that the stress management programs not only lessened reported symptoms but also reduced disease activity compared to a control group, an effect which disappeared after the completion of therapy suggesting a need for ongoing therapy to maintain these benefits. 37 It will be of interest in future studies of GI modalities to determine their durability as well as effects on measures of MS disease–specific activity.
In sum, the findings from this exploratory study suggest that HLGI has potential to benefit the well-being of patients with RRMS. There is at present strong demand by MS patients for mind-body modalities and many patients in this study expressed their gratitude that there was research being conducted examining integrative approaches to improve their quality of life. Needed are future studies that include more standard active control groups and that incorporate relevant biomarkers and disease activity outcomes to further advance this work.
Footnotes
Appendix A
Appendix B
Acknowledgments
We are grateful to the patients who contributed their time to this study.
Author Contributions
LKC and PJ designed the study. PJ designed and administered the HLGI intervention. PJM managed and oversaw the conduct of the study. LKC analyzed the data. LKC and PJM wrote the manuscript and RK helped with further writing and editing. All authors read and approved the final manuscript.
Declaration of Conflicting Interests
The authors declared that there are no conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was supported by the University of California San Diego Department of Family Medicine and Public Health Center of Excellence for Research and Training in Integrative Health.
Ethical Approval.
This project was approved by the University of California San Diego Human Research Protection Program (IRB #121412).
