Abstract
Objective
Using a prospective observational design, we assessed adverse events (AEs) after COVID-19 vaccination in Japanese patients.
Methods
Two doses of the mRNA-1273 (SPIKEVAX®) or BNT162b2 (COMIRNATY®) vaccine were administered to participants aged 12 to 18 years, and AEs after each dose were recorded for 14 days. Data on the duration and nature (local vs. systemic) of AEs were collected using a questionnaire. Sex-based differences in AE frequency were also analyzed.
Results
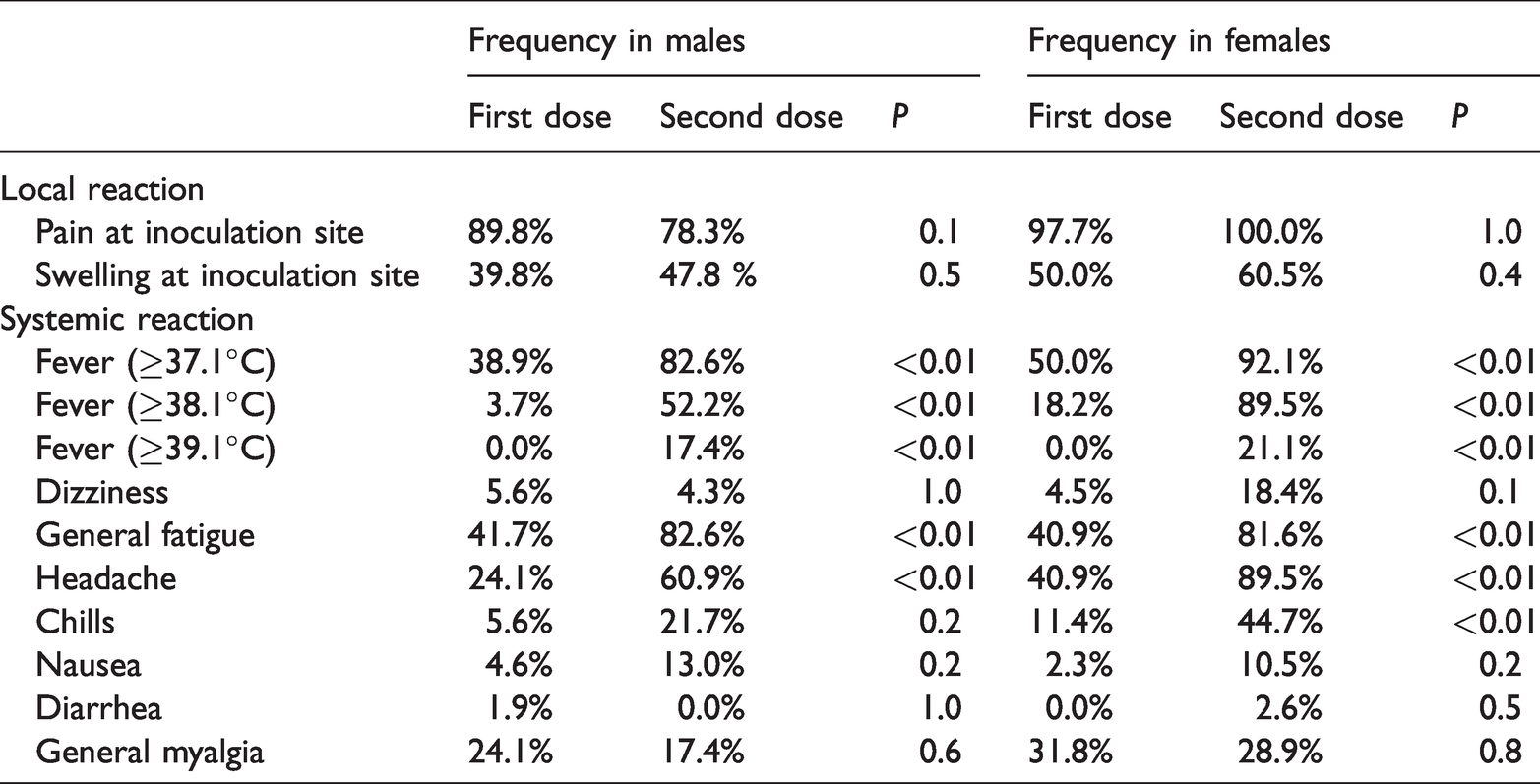
After the first and second doses, 152 and 135 vaccinees were enrolled, respectively. After the first dose, fever (>37.1°C) occurred in 38.9% of males and 50.0% of females, whereas local pain occurred in 89.8% and 97.7% of males and females, respectively (only SPIKEVAX® was used as the first dose). After the second dose, fever (>37.1°C) occurred in 77.8% and 82.6% of males vaccinated with COMIRNATY® and SPIKEVAX®, respectively, and 82.6% of females (all received SPIKEVAX®). The local pain rates in these groups were 80.6%, 76.3%, and 100%, respectively. After the second dose, local pain, fever (>38.1°C) and headache were significantly more common in female participants, and the median symptom duration was 3 days.
Conclusions
AEs were more frequent after the second dose and in females.
Introduction
COVID-19 is an acute respiratory illness caused by SARS-CoV-2 that has spread globally since the end of 2019.1–3 In Japan, the COVID-19 pandemic is ongoing, and it has featured six major waves of spread, resulting in 15.9 million infections and more than 35,000 deaths. 4 Two types of messenger RNA (mRNA) vaccines against SARS-CoV-2 have been approved in Japan, and vaccination was initiated in February 2021. Data on the types of adverse events (AEs) occurring in adults after administration of COVID-19 vaccines are gradually being accumulated.5,6
It should be noted that there is insufficient knowledge available on the characteristics of AEs in adolescent vaccinees. We thus collected data on AEs following COVID-19 vaccination in children aged 12 to 18 years. Several studies reported that AEs occur more frequently in females than in males and that they are more common in individuals who received the mRNA-1273 (SPIKEVAX®) vaccine than in those who received the BNT162b2 (COMIRNATY®) vaccine. We thus also examined differences in AEs based on sex and the type of vaccine received.7,8
Materials and methods
Vaccine type and schedule
Osaka Medical and Pharmaceutical University (OMPU) provided mass COVID-19 vaccination to students at Takatsuki Junior and Senior High Schools who were 12 to 18 years old. The family members of the students and staff members at the schools could also receive the vaccine based on individual preference. Vaccination was conducted by doctors and nurses affiliated with OMPU. Initially, two doses of elasomeran (0.1 mg [mRNA-1273] SPIKEVAX®; Moderna, Inc., Cambridge, MA, USA) were administered at intervals of 27 to 29 days. However, the Ministry of Health, Labour and Welfare (MHLW) allowed males in their teens and twenties to be vaccinated with tozinameran at a dose of 0.225 mg (BNT162b2, COMIRNATY®; Pfizer Inc., New York, NY, USA) as a second dose. This decision was based on the higher likelihood of developing AEs including myocarditis after receiving SPIKEVAX® than after receiving COMIRNATY®. 9 Therefore, some males received COMIRNATY® as the second dose. Because this MHLW policy was announced immediately before the second dose was made available, the vaccine interval was set at 28 to 33 days even if COMIRNATY® was chosen for the second dose.
Study participants
We distributed printed information about a questionnaire-based, prospective, observational study of AEs occurring during the first 14 days after COVID-19 vaccination to the parents or guardians of the participants. The vaccinees had to be accompanied by at least one of their parents or a guardian to the vaccination venue. Printed explanatory documents of our study and the questionnaire used to record AEs after vaccination were provided to the vaccinees or their guardians. These documents were distributed so that each vaccinee would receive one copy. Regarding study recruitment, vaccinees aged 12 to 18 years who received the COVID-19 vaccine at OMPU, who were observed for AEs over a 14-day period following vaccination, and who submitted a paper-based questionnaire or data registration through the Internet using Google Forms were considered to have participated in the study based on their submission. All participants meeting these criteria were eligible for study participation. Children aged 12 to 15 years who wanted to participate in the study were required to check a box indicating that their parents or guardians had agreed to their participation in the study. For participants 12 to 15 years old who received the COVID-19 vaccine, we obtained written informed consent from their parents or guardians prior to study enrollment. If data were submitted electronically via Google Forms, a checkbox stating that parental consent was obtained was required. People who had received or who were scheduled to receive the COVID-19 vaccine at other medical facilities were excluded. Submission of a completed questionnaire and data registration through Google Forms was accepted at any time from 14 days after vaccination until December 15, 2021.
Individual AE records were not linked to personal information when submitted to us; therefore, our study was performed with the raw data, which was anonymized. This study was approved by the Ethics committee of OMPU (address: 2–7 Daigakumachi, Takatsuki, Osaka 569-8686, Japan; approval No.: 2021-089-1, date of approval: 18 October 2021), and the protocol complied with Japanese domestic law, the Declaration of Helsinki, and the equator-network STROBE guideline. 10
Collected information of AEs after vaccination
Information was collected on local reactions (pain and swelling) and systemic reactions (urticaria, dizziness, chest pain, general malaise, headache, chills, vomiting or nausea, diarrhea, generalized myalgia, disturbance of consciousness, and convulsions) 14 days after each vaccination. For fever, vaccinees were asked to report their maximum body temperature daily. If AEs other than those previously mentioned occurred, they were described freely. In addition, height, weight, underlying medical conditions, regularly used medications, AEs occurring during the first 15 minutes (or the first 30 minutes if deemed necessary by the physician) after vaccination, and clinically significant illnesses requiring hospital visits or hospitalization were also documented by the participants.
Health observation forms on which the health status could be written for 14 days after vaccination were distributed at the vaccination sites. Study participants who opted to submit paper forms directly to the authors had them submitted by their parents/guardians, who are employees of OMPU. Study participants who opted for data entry through the Internet were requested to enter the contents of their completed 14-day health observation forms simultaneously on the 14th day by themselves.
Procedures for collecting information on AEs
Because the study participants were recruited separately for the first and second vaccinations, the participants did not always provide data on AEs for both doses. The presence or absence of symptoms did not require confirmation by a parent or guardian. Therefore, even among participants younger than 15 years, AEs were left to the individual judgment of each participant. The questionnaire used in this study is presented in Figure 1. Internet data were documented in a pen-and-paper format or captured in an electronic format.

This chart was distributed to all vaccine recipients to record any adverse events that occurred over 14 days following vaccination. The chart could be submitted in paper form, or the information could be provided electronically via Google Forms.
Study design and statistical analysis
This study was designed as a prospective observational study.
The appropriate sample size necessary to ensure sufficient statistical power was not considered. The frequency of AEs other than fever occurring after the first vaccine dose were compared between male and female participants using Fisher’s exact probability test. After the second vaccination, we also used Fisher’s exact test to perform a pairwise comparison of participants stratified by sex and vaccine type (SPIKEVAX® vs. COMIRNATY®). For SPIKEVAX® recipients only, each AE was compared after the first and second dose using Fisher’s exact test. All tests were two-sided, and
Results
Study participants
In total, 469 vaccinees (352 males and 117 females) received the first dose and 456 vaccinees (342 males and 114 females) received the second dose at OMPU during the study period. Thirteen participants declined the second dose. Of the 342 males who received the second vaccine dose, 252 received COMIRNATY®, and 90 received SPIKEVAX®. In total, 152 participants (108 males and 44 females) who received the first dose and 133 participants (95 males and 38 females) who received the second dose were included in the study. The participation rates were 32.4% and 29.2% for the first and second doses, respectively (Figure 2).
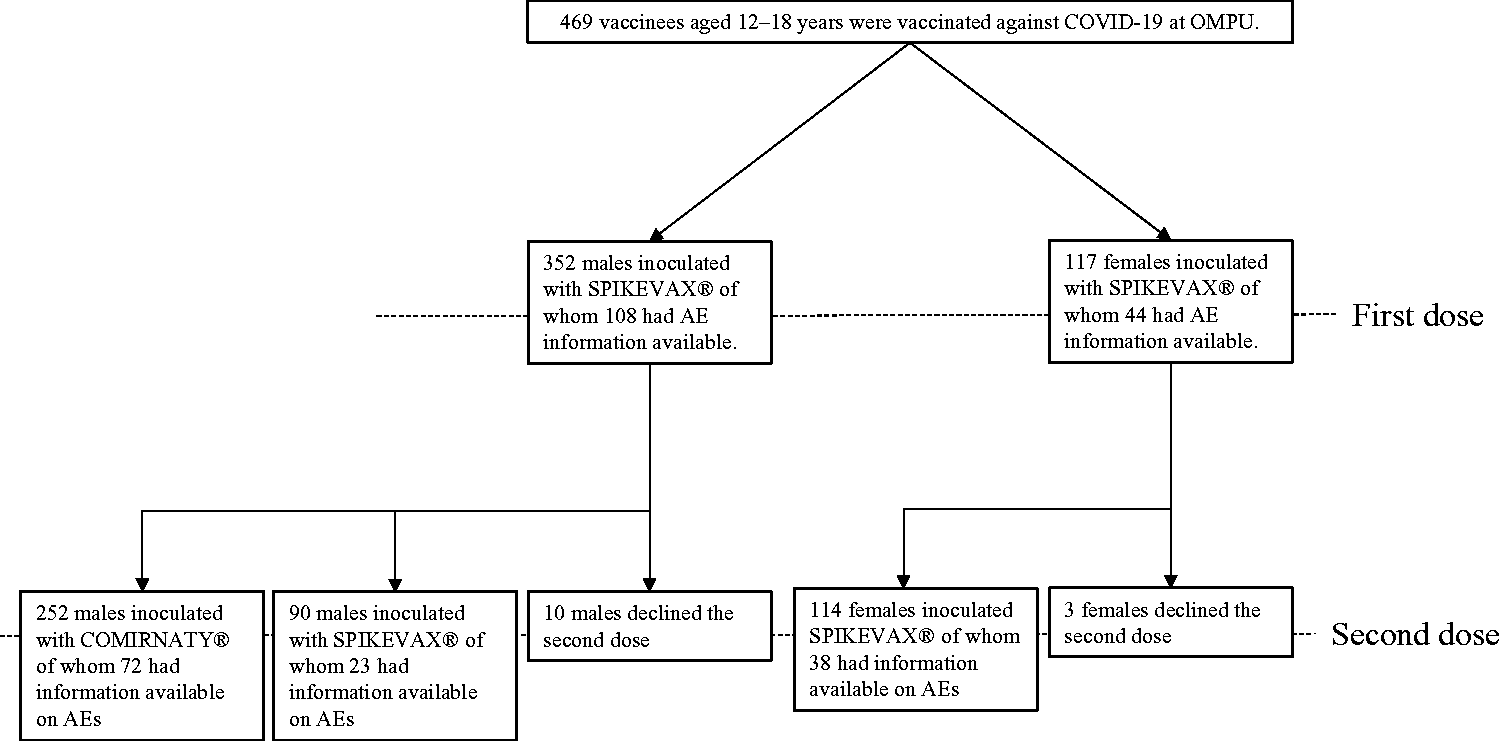
Participants aged 12 to 18 years who received the COVID-19 vaccine at Osaka Medical and Pharmaceutical University Hospital were categorized by the type of vaccine received, sex, and the number of study participants from whom adverse event data could be collected.
Among participants who received the first dose, 13 were aged 12 years (eight males and five females), 36 were aged 13 years (23 males and 13 females), 27 were aged 14 years (20 males and seven females), 30 were aged 15 years (20 males and 10 females), 22 were aged 16 years (17 males and five females), 18 were aged 17 years (14 males and four females), and six were aged 18 years (six males and no females). Among participants who received the second dose, 16 were aged 12 years (10 males and six females), 32 were aged 13 years (22 males and 10 females), 24 were aged 14 years (16 males and eight females), 21 were aged 15 years (14 males and seven females), 16 were aged 16 years (13 males and three females), 22 were aged 17 years (18 males and four females), and two were aged 18 years (two males and no females). For the first dose, 102 and 50 participants submitted data in paper form and through the Internet, respectively, compared with 90 and 43 participants, respectively, after the second dose.
Local reaction after vaccination
Pain at the inoculation site was reported by 89.8% of male participants and 97.7% of female participants after the first dose and by 78.3% of male participants and 80.6% of female participants after the second dose. Swelling at the inoculation site was observed in 39.8% of male participants and 50.0% of female participants after the first dose and in 22.2% of male participants and 47.8% of female participants after the second dose. The median symptom duration was 2 to 3 days (Tables 1–2). The frequency of AEs was compared between the first and second doses only among SPIKEVAX® recipients; however, there were no significant differences between males and females regarding the rates of pain and swelling at the inoculation site (Tpable 3).
Duration and frequency of adverse events after the first dose of the SPIKEVAX® vaccine.

IQR, interquartile range; Blank, no applicable research participants.
*
Duration and frequency of adverse events after administration of COMIRNATY® or SPIKEVAX® as a second dose.

Systemic AEs
In total, 38.9% of males and 50.0% of females developed a fever of ≥37.0°C after the first dose. Of these participants, 3.7% of males and 18.2% of females had a fever of ≥38°C. After the second dose, the participants were divided into three groups, i.e., males who received COMIRNATY®, males who received SPIKEVAX® group, and females who received SPIKEVAX®. For these groups, the rates of fever of ≥37°C were 77.8%, 82.6%, and 92.1%, respectively, whereas those of fever ≥38.6°C were 2.8%, 17.4%, and 21.1%, respectively. The duration of fever was up to 4 days, and the median duration for each group was 1 to 2 days. Among the systemic AEs for which information was collected, none of the respondents reported urticaria, dyspnea, convulsions, or disorientation. General malaise was the most common after both the first dose (40%) and second doses (66.7%–82.5%). Systemic AEs other than fever generally resolved within 2 days after both the first and second doses. The frequency of AEs was significantly higher in females than in males after the first vaccination. After the second dose, the frequencies of headaches and chills differed significantly among the three subgroups. The rates of headache and chills after the second dose were significantly higher among males who received SPIKEVAX® than among males who received COMIRNATY®, and the rates were significantly higher among females than among males. The detailed results are summarized in Tables 1–2. Data on the maximum body temperature recorded during the study are summarized in Tables 4–5.
Comparison of the frequency of adverse events after the first and second doses of SPIKEVAX®.
Maximum body temperature after the first dose.

Maximum body temperature after the second dose.

The frequency of each AE was compared between the first and second sessions only in the SPIKEVAX®-vaccinated population. Fever was significantly more frequent after the second dose regardless of the severity and sex. Among other systemic AEs, fatigue and headache were more frequent after the second dose regardless of sex, and chills were significantly more frequent after the second dose only in females (Table 3).
Other AEs
A free field was provided to allow participants to report unexpected AEs. Axillary lymphadenopathy was reported in six vaccinees (four males and two females) after the first dose and six vaccinees (five males after COMIRNATY® vaccination and one female after SPIKEVAX® vaccination) after the second dose. Two vaccinees complained of itchy skin after the first dose (one male and one female), and two female vaccinees reported these AEs after receiving the second SPIKEVAX® dose. Cough was reported by three male vaccinees after receiving the second COMIRNATY® dose. In addition, abdominal pain was reported by three vaccinees (two males and one female) after the first dose and by one male vaccinee after the second dose of COMIRNATY®. A 14-year-old male who developed abdominal pain 3 days after the first dose was diagnosed with appendicitis, and he underwent surgery.
AEs requiring medical care
As previously mentioned, a 14-year-old male was hospitalized for appendicitis 3 days after the first dose. However, no other patient was hospitalized for post-vaccination illness within 14 days after vaccination. Excluding the aforementioned cases, no participants required outpatient visits for AEs after the first dose. Four male vaccinees visited the hospital after the second COMIRNATY® dose. Two of these participants had a persistent cough, one participant experienced vomiting, and the other participant exhibited dizziness.
AEs occurring at the vaccination venue
After the first dose, a 13-year-old male developed numbness in the vaccinated arm, which later improved (recovery time unknown). After the second dose, a 14-year-old male who received SPIKEVAX® developed numbness in a finger on the side of vaccination that disappeared the next morning. No other AEs were reported at the vaccination venue.
Discussion
In Japan, pain at the vaccination site and fever of 38.1°C or higher have attracted public attention as post-vaccination AEs. Pain around the inoculation site was observed in 89.8% of males and 97.7% of females after the first dose and in 78.3% of males and 100% of females after the second dose. Fever of 38.1°C or higher, which significantly affects daily life, was observed in 3.7% of males and 18.2% of females after the first dose and in 38.9% of males and 89.5% of females after the second dose. Although only SPIKEVAX® vaccinees were studied, fever was more common after the second dose than after the first dose regardless of the severity of the event or sex of the participant. It is noteworthy that fever, a high-profile AE, was more common after the second dose. A comparison between male and female participants illustrated that fever of >38.1°C and headache were significantly more common in females after the first dose. After the second dose, fever of >38.1°C and headache were more common in females than in males who received SPIKEVAX® as the second dose. A comparison of COMIRNATY®- and SPIKEVAX®-treated males revealed a significant difference only in the rate of swelling around the vaccination site. The median duration of fever was 3 days, which was considered acceptable. This finding is in agreement with those of previous studies conducted in Japanese adults >18 years old. 3
The rates of fever exceeding 37°C did not differ between the first and second doses, and this was independent of both the type of vaccine received and the sex of the participant. This result is consistent with those of previous reports.5–8 However, the frequency of fever of >38°C was significantly higher in females than in males after both the first and second doses. In general, fever of >38°C to 39°C is classified as grade 1 in the Common Terminology Criteria for AEs (CTCAE) and is considered a “mild” AE. Therefore, fever at a level that would affect daily functioning was more common in females than in males. This result is consistent with those of previous reports.8,12
The duration of local AEs, such as pain and swelling at the inoculation site, was mostly shorter than 4 days, which is generally acceptable. The duration of symptoms was not particularly long compared with those reported in previous studies. 7 The frequency of AEs was not significantly different between males and females after the first dose, but the rate was significantly higher after the second dose among females who received SPIKEVAX®. Local reactions and fever occurred more frequently in females. This result is consistent with those of previous studies examining AEs in Japanese participants older than 18 years.13,14 We did not evaluate the severity of AEs other than fever; therefore, we could not evaluate the CTCAE grade for the severity of local reactions. This point should be addressed in future studies.
In terms of systemic reactions, headaches were more frequent in males than in females after the first dose, whereas headaches and chills were significantly more frequent in females than in males who received SPIKEVAX®. However, the prevalence of headache itself is higher in females than in males. 15 It was thus not possible to determine whether these differences were associated with vaccination. It is important to note that this study only recorded AEs without assessing their severity.
It is worth noting that axillary lymphadenopathy has been reported after the first and second doses in 61% of female vaccinees using ultrasound chest imaging. 16 However, the frequency of palpable auxiliary lymphadenopathy was reported as 0.4% 17 , and it was not common in our sample. However, six participants reported this AE in the open-ended questionnaire after the first and second doses. It is easy to understand that symptoms not included in the questionnaire may have been underestimated; therefore, it is possible that axillary lymphadenopathy occurred more frequently than reported. Reports of lymphadenopathy after the second dose might be characteristic of participants aged 12 to 18 years. It is difficult to compare SPIKEVAX® and COMIRNATY® in female vaccinees because COMIRNATY® was only used for the second dose in males. However, among males who received the second dose, swelling at the vaccination site and fever of 38°C or higher were significantly more common in the SPIKEVAX® group than in the COMIRNATY® group. This result is consistent with those of previous studies.18,19
This study had several limitations. First, this was a single-center study with a small sample size. Therefore, it was not possible to evaluate AEs such as myocarditis and pericarditis that occurred at low frequencies in this study. A major limitation of this study was the inability to stratify AEs by age because of the small sample size. Second, the participation rate in the post-vaccination health status survey was low. Participants who responded may have had some symptoms that prompted their participation, and others may not have responded because of AEs. Because only 30% of the vaccinees participated in the study, such self-selection bias might exist. Third, the MHLW stated that AEs such as myocarditis and pericarditis associated with SPIKEVAX® tend to occur more frequently in male recipients in their second and third decades of life. SPIKEVAX® is thus unlikely to be administered to many males in this age group in the future in Japan. It must be mentioned that few males in this generation will be vaccinated with SPIKEVAX® in the future. It is unclear whether our data can be directly applied to young males who will be vaccinated primarily with COMIRNATY®. Finally, it is worth noting that our study did not contain a sufficiently large sample size regarding AEs; therefore, there may be bias concerning analyses using sex and age. It should also be noted that a high percentage of the study participants were male.
In conclusion, fever was more frequent and severe after the second COVID-19 vaccine dose, with fever of >38.5°C being more frequent in females than in males. Headache was also more frequent after the second dose and significantly more frequent in females than in males. The median duration of all AEs was 3 days, which is generally considered acceptable. Based on the information gathered in this study, mRNA-based COVID-19 vaccines appear to be well-tolerated in Japanese participants aged 12 to 18 years.
Footnotes
Acknowledgements
Author contributions
TO designed the study, recruited the study participants, collected the information, performed the statistical analysis, and edited the manuscript. TN and AU supervised the study. TY, YM, KM, and FK collected information.
Data availability statement
The data cannot be shared publicly because of the privacy imposed by the Osaka Medical and Pharmaceutical University Ethics Committee. Data are available from the Infection Control Center of Osaka Medical and Pharmaceutical University for researchers who meet the criteria for access to confidential data. The representative email address for the Infection Control Center at Osaka Medical and Pharmaceutical University is
Declaration of conflicting interests
The authors declare that there is no conflict of interest.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by a personal research grant from the infection Control Center, Osaka Medical and Pharmaceutical University Hospital. The sponsors had no involvement in deciding the study design; collection, analysis, and interpretation of data; writing of the report; and the decision to submit the paper for publication.

