Abstract
Background:
Immunization in the elderly population is critical due to the high frequency of health outcomes related to COVID-19.
Objectives:
This study aimed to compare the effectiveness levels of COVID-19 vaccine schedules in preventing SARS-CoV-2 infection in the older adult group who received at least one booster dose.
Design:
Retrospective cohort study.
Methods:
This study evaluated 8969 adults aged 65 and over in the Sultanbeyli district of İstanbul. COVID-19 vaccination and SARS-CoV-2 polymerase chain reaction testing data between January 14, 2021 and December 2, 2022 were obtained from the National Public Health Management System.
Results:
The median age of participants was 71 years. The vaccines were mostly administered as CoronaVac for the first and second doses (81.4% and 82.2%, respectively) and BNT162b2 for the third and fourth doses (61.8% and 73.1%, respectively). Turkovac was administered only in booster doses (third dose 0.6%, fourth dose 4.8%). The adjusted relative vaccine effectiveness (rVE) was found to be 61.8% (95% confidence interval (CI) 51.5–69.9) in two doses of inactivated vaccine and one dose of mRNA vaccine schedule compared to the homolog booster of CoronaVac primary vaccine schedule. In two booster doses receipts, the adjusted rVE was found to be 45.4% (95% CI 13.8–65.4) in three doses of inactivated and one dose mRNA vaccine schedule and 43.0% (95% CI 20.5–59.2) in two doses of inactivated and two doses of mRNA vaccines schedule compared to the two homolog boosters with CoronaVac primary vaccine schedule.
Conclusion:
In this study, the effectiveness of the mRNA vaccine as a booster dose was higher than that of the homologous boosters in participants receiving the CoronaVac primary series for those aged 65 and over.
Introduction
The World Health Organization (WHO) declared the COVID-19 pandemic in March 2020, and vaccine research against SARS-CoV-2 started in the next few months.1,2 As of early 2021, vaccination programs have begun in various countries. The vaccination program in Türkiye started with CoronaVac, an inactivated whole-virus SARS-CoV-2 vaccine developed by Sinovac Biotech, in January 2021.3,4 In the first weeks of the program, healthcare workers and people aged 65 and over were the target population. Türkiye started administering the BNT162b2 (mRNA vaccine) developed by Pfizer–BioNTech in April 2022.3,5 The Turkovac, an inactivated whole-virion COVID-19 vaccine, was developed in Türkiye and approved by the Turkish Ministry of Health for emergency use in December 2021. 6
Booster dose vaccination started on July 1, 2021, in Türkiye. According to the strategy of the Turkish Ministry of Health, the first two doses must be the same vaccine type; however, the types of booster doses were based on personal choice. 3 Vaccinations were administered in health institutions such as family health centers, district health centers, and hospitals. In addition, older adults were vaccinated in their homes if requested. Only inactivated vaccines were administered in home visits. Vaccination and SARS-CoV-2 PCR (polymerase chain reaction) testing were free and widely available in İstanbul/Türkiye. 3
Individuals aged 65 and over were the population most affected by the SARS-CoV-2, as the group with a high incidence of chronic diseases. 7 Although older adults had the most prolonged period of pandemic restrictions in Türkiye, hospitalizations and deaths due to COVID-19 were most common in this group.8,9
Vaccines developed with different technologies, including mRNA, inactivated, and viral vector vaccines, were administered worldwide during the pandemic. Türkiye is among the countries where BNT162b2 and CoronaVac vaccines were both applied at high rates. This study aimed to assess the effectiveness of vaccination schedules in older adults who received at least one booster dose of the COVID-19 vaccine.
Materials and methods
Study group and participants
The study group of this retrospective cohort study consisted of the population aged 65 and over residing in the Sultanbeyli district of İstanbul/Türkiye. Sultanbeyli has the lowest socio-economic development index rank among the districts of İstanbul. 10 The population of Sultanbeyli was 349,485 people in 2021 and 358,201 people in 2022. According to the Turkish Statistical Institute, the population aged 65 and over corresponds to approximately 3.8% of the district population. 11 After people whose SARS-CoV-2 PCR test data could not be reached were excluded, 8949 elderly adults were included in the study.
Data source and extraction
The study data were obtained from the National Public Health Management System (HSYS in Turkish). The HSYS is a registry system established by the Turkish Ministry of Health and includes the data of all SARS-CoV-2 PCR tests and vaccines applied in Türkiye. Data on sex, age, smoking, SARS-CoV-2 PCR testing dates, counts, and results, and SARS-CoV-2 vaccination dates, counts, and vaccine types were obtained from people residing in Sultanbeyli by the HSYS. The primary outcome of the study was SARS-CoV-2 PCR test positivity. Analyses focused on the period from January 14, 2021, when the COVID-19 vaccination was started to be administered in Türkiye, to December 2, 2022.
Statistical analysis
Statistical analysis of this study was performed using IBM SPSS Statistics for Windows, version 29 (IBM Corp., Armonk, NY, USA). The descriptive data were presented as counts (n), ratios (%), and median and range values. The period of administration of the booster doses was separated according to the number of COVID-19 cases in Türkiye, considering the wave periods. The effectiveness of the vaccine schedules against SARS-CoV-2 PCR positivity was calculated using Cox regression analysis (proportional hazard model) as 1 − hazard ratio formula. Vaccination schedules of the participants were evaluated in two regression models for those who received three doses and four doses. In both regression models, only those vaccinated with all doses of inactivated whole-virion SARS-CoV-2 vaccines were considered as reference, and the relative effectiveness of the other schedules was calculated. Age and sex variables were included in the regression analyses, along with SARS-CoV-2 PCR test results. Only two participants vaccinated with four doses were not included in the analysis due to the missing data on the fourth dose vaccine. Insufficient counts of vaccine schedules for analysis were also not included in the models. In the regression analysis, CoronaVac and Turkovac vaccines were evaluated in the same category as they were both inactivated whole-virion SARS-CoV-2 vaccines. Considering the optimal protective periods of the COVID-19 vaccines, SARS-CoV-2 PCR test results performed in the 90-day period from ⩾14 days after the vaccination were included in the regression analyses. 12 The statistical significance level was accepted as p < 0.05 in analyses.
Results
The median age of the participants was 71 years (65–109 years), and 42.4% (n = 3804) were aged 65–69 years. Of the participants, 58.3% (n = 5232) were women. Almost half had at least one SARS-CoV-2-positive test result during the study period (n = 4132, 46.1%). The percentages of older adults who received one dose, two doses, three doses, and four doses of SARS-CoV-2 vaccines were 2.8% (n = 291), 31.7% (n = 2842), 38.1% (n = 3419), and 21.6% (n = 1936), respectively. Only 5.4% (n = 481) were unvaccinated (Table 1). The first and the second doses were mostly administered as CoronaVac (81.4% and 82.2%, respectively). Over half of older adults who received the booster doses preferred the BNT162b2 vaccine (61.8% for the third and 73.1% for the fourth doses). The number of individuals vaccinated with Turkovac was 31 (0.6%) for the third dose and 92 (4.8%) for the fourth dose (Table 2).
Demographic and clinical characteristics of the study population (n = 8969).

Missing = 1804.
PCR, polymerase chain reaction.
Frequency of the vaccine types, n (%).
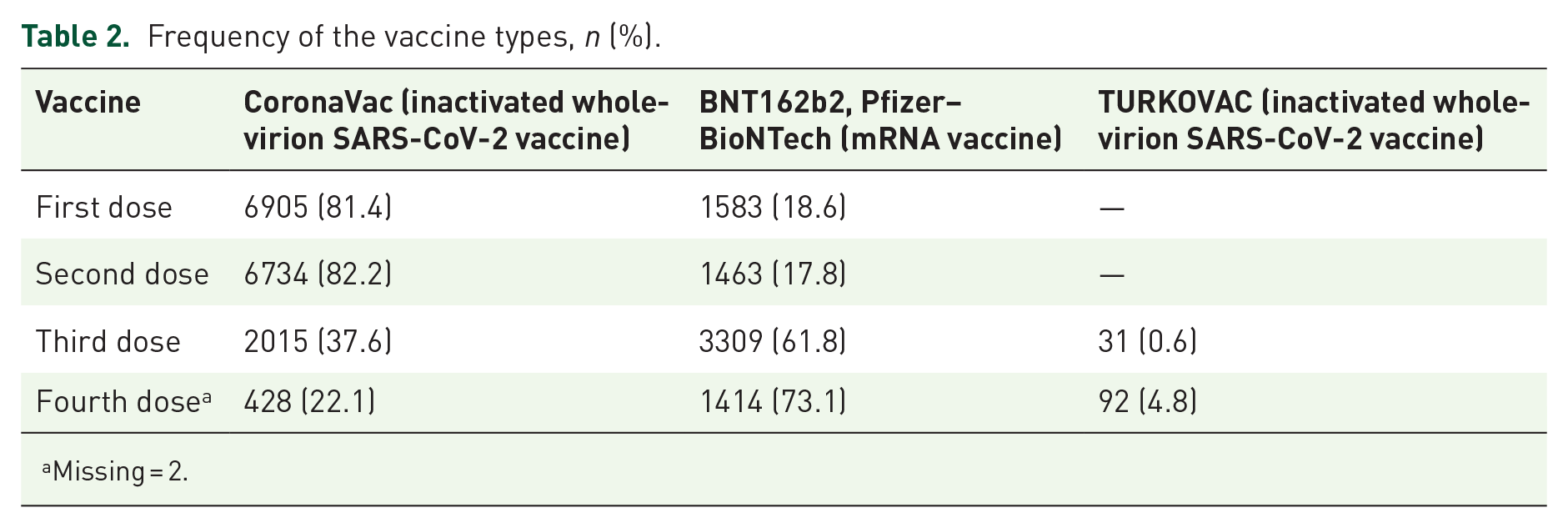
Missing = 2.
The percentages of the three-dose vaccine schedules for CoronaVac–CoronaVac–BNT162b2 was 60.6% (n = 3244), CoronaVac–CoronaVac–CoronaVac was 37.2% (n = 1992), and BNT162b2–BNT162b2–CoronaVac was 1.6% (n = 87; Table 3). In those who had four doses of vaccine, the vaccine schedule was CoronaVac–CoronaVac–BNT162b2–BNT162b2 for 59.5% (n = 1150), CoronaVac–CoronaVac–CoronaVac–CoronaVac for 18.7% (n = 362), and CoronaVac–CoronaVac–CoronaVac–BNT162b2 for 13.1% (n = 254) of the participants (Table 4).
Frequency of three-dose vaccination schedules (n = 5355).

Frequency of four-dose vaccination schedules (n = 1934).

The periods in which the vaccines were administered most frequently were July 1 to December 15, 2021, for the third dose (91.5%, n = 4899) and December 16, 2021 to February 15, 2022, for the fourth dose (86.0%, n = 1664; Figures 1 and 2). The most common three-dose vaccination schedules of the 65–74 and 75–84 age groups were two doses of inactivated and one dose of mRNA vaccine (62.8%, n = 2362 and 58.0%, n = 773, respectively). However, more than half of the participants in the ⩾85-year-old group had completed three-dose inactivated vaccination series (58.0%, n = 152; Table 5).
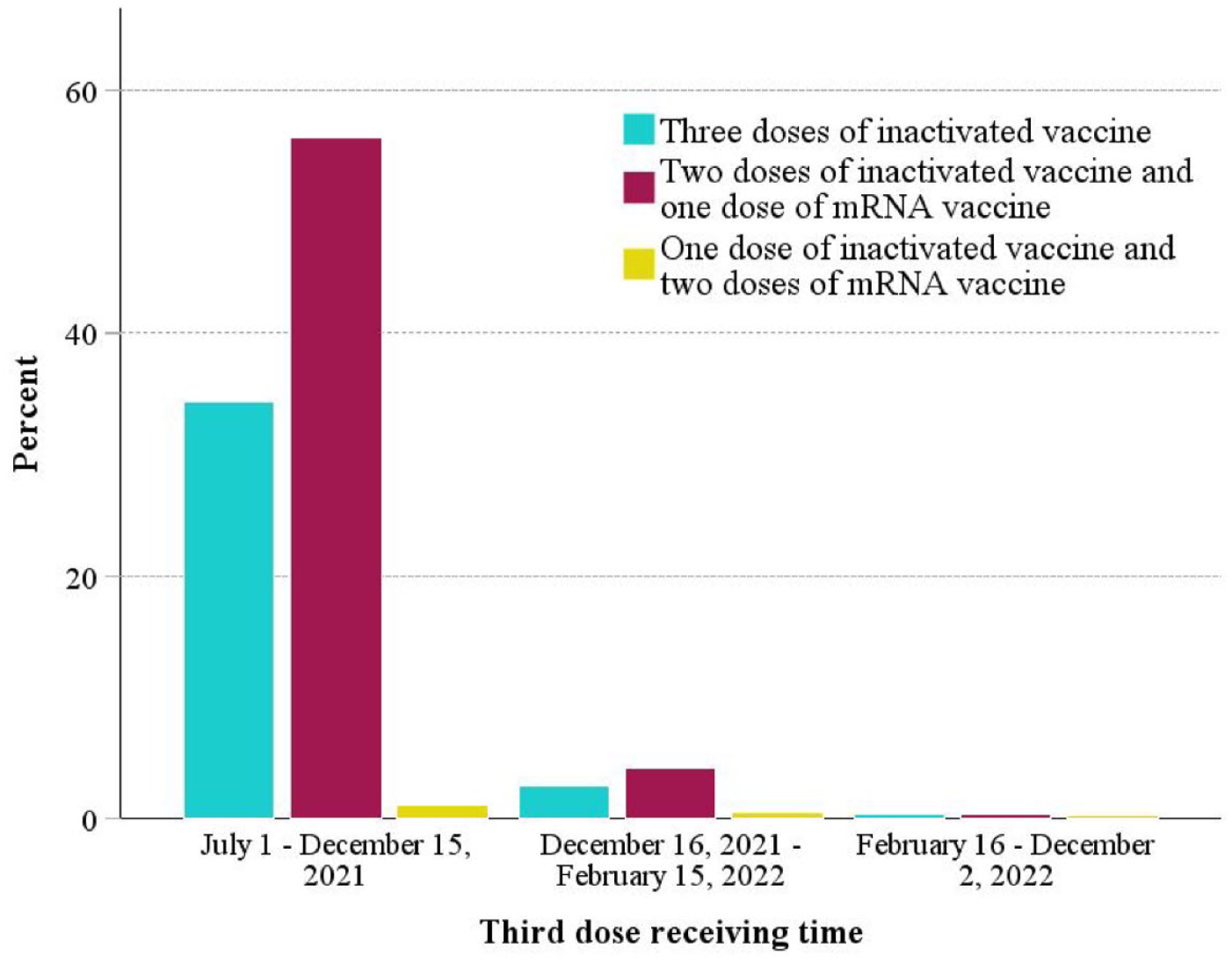
Analysis groups and the third dose of vaccines receiving times.

Analysis groups and the fourth dose of vaccines receiving times.
Booster doses applied in different age groups, n (%).

The incidence rates of positive SARS-CoV-2 PCR test were 8.6% (172/2005) in three doses of inactivated vaccine group, 3.5% (112/3244) in two doses of inactivated and one dose of mRNA vaccine group, and 1.9% (2/105) in two doses of mRNA vaccine and one dose of inactivated vaccine group. The adjusted relative vaccine effectiveness (rVE) was found to be 61.8% (95% CI 51.5–69.9) in two doses of inactivated vaccine and one dose of mRNA vaccine schedule and 79.8% (95% CI 18.4–95.0) in two doses of mRNA vaccine and one dose of inactivated vaccine schedule compared to three doses of inactivated vaccine schedule (Table 6). The incidence rates of those with a positive SARS-CoV-2 PCR test were 16.2% (54/333) with four doses of inactivated vaccine group, 9.3% (28/301) with three doses of inactivated and one dose of mRNA vaccine group, and 9.6% (98/1019) with two doses of inactivated and two doses of mRNA vaccine group. The adjusted rVE was found to be 45.4% (95% CI 13.8–65.4) in three doses of inactivated and one dose of mRNA vaccine schedule and 43.0% (95% CI 20.5–59.2) in two doses of inactivated and two doses of mRNA vaccines schedule compared to the four doses of inactivated vaccine group (Table 6).
PCR test results and adjusted relative vaccine effectiveness against SARS-CoV-2 infection for participants.

CI, confidence interval; PCR, polymerase chain reaction; VE, vaccine effectiveness (calculated as 100% × (1 − hazard ratio) for 3 months from day 14 after vaccination).
Adjusted for age and sex.
Between December 16, 2021 and February 15, 2022.
Discussion
Our study compared the effectiveness of vaccine schedules against SARS-CoV-2 in the population aged 65 and over and determined that the 90-day rVE of including at least one dose of mRNA vaccine in the vaccination schedule was higher than all inactive vaccine schedules. This finding was consistent with previous studies comparing homologous and heterologous additional dose effectiveness.13–16 In a study in Malaysia, the risk of developing SARS-CoV-2 infection was found to be lower in the group aged 60 years and above, when the group receiving two doses of BNT162b2 vaccine was taken as a reference, in those who received two doses of inactivated vaccine and one dose of BNT162b2 vaccine than those who received three doses of inactivated vaccine (adjusted odds ratio (aOR), 0.17 (95% CI 0.17–0.18), and aOR, 0.19 (95% CI 0.19–0.20), respectively). 15 In a study conducted in China in the age group 60 years and above, the primary CoronaVac series with BNT162b2 booster appeared to have higher vaccine effectiveness against infection than the homologous booster (60–79 years old: 30.9% (95% CI 28.5–33.2), 80 years and older: 61.4% (95% CI 55.8–66.3)), but no significant difference was detected in COVID-19-related hospitalization, severe complications, and mortality. 16 A meta-analysis that included 23 studies showed that the heterologous booster dose was more effective than the homologous inactive booster in preventing SARS-CoV-2 infection, symptomatic COVID-19, and COVID-19-related hospitalization. 13
This was the first booster dose vaccine effectiveness study focusing on the 65 and older age group in Türkiye. However, our research findings aligned with the prior studies conducted in different populations in Türkiye. A study on healthcare workers revealed that the rVE was lower in three doses of Sinovac receipts in comparison to those who received two doses of Sinovac followed by a single dose of BioNTech, with reference to the unvaccinated group (62% and 89% respectively). 17 Similarly, another study involving 25 hospitals across 16 cities found a decrease in hospitalizations among those who had two doses of Sinovac and a BioNTech booster, in contrast to those who had three doses of Sinovac. 18 The findings of our study that the effectiveness of the additional dose of BNT162b2 was higher than the homologous additional dose administered with CoronaVac were also aligned with research on antibodies in Türkiye. 12
This study found that the heterologous vaccine schedule showed higher effectiveness than the completely inactivated vaccine schedule in both three- and four-dose vaccine schedules. However, the relative increase in effectiveness was higher in the third dose schedules. In our research group, the majority of the third dose vaccines were administered in the July–December 2021 period, when the Delta variant dominated, and the majority of the fourth dose vaccines were administered in the January–February 2022 period, when the Omicron variant dominated in Türkiye. Besides, January–February 2022 was one of the peak periods for COVID-19 cases in Türkiye. During this period, the participants who often stayed at home received inactivated vaccines more than people with active lifestyles. That may have reduced the difference in the relative effectiveness of the mRNA vaccine. Similarly, previous studies found the effectiveness level of the mRNA vaccine to the Omicron variant to be lower than in the Delta variant period.19–21 A study conducted in England showed that Omicron had a higher rate of second attack rates inside and outside the household and clustering in households than Delta due to its success in evading the immune response and that it reduced the effect of the vaccine on the transmission rate compared to Delta. 21
Most of our study group (91.8%) had received at least two doses of SARS-CoV-2 vaccine. However, the proportion of that receiver booster dose dropped to 59.7%. In China, by November 2022, the rate of those aged 60 and over who completed the two-dose series was 88.6%, and those who received at least one booster dose was 72.4%. 22 In the United States, 89.7% and 84.4% of adults aged 65–74 and 75 and older, respectively, have received two doses. 23 In the United Kingdom, as of July 2022, the two-dose vaccination rate was 93.6%, and the booster dose rate was 90.4%. 24 Approximately one-third of those in our study group who completed the two-dose series did not receive a booster dose. In Türkiye, the COVID-19 vaccine was widely available and administered free of charge. However, considering the sociocultural characteristics of the study region, the low frequency of active lifestyles in older adults may have reduced seeking for booster doses. Hence, about a third of participants in a previous study conducted in our research area stated that the booster doses were unnecessary or ineffective. 25 Also, the significant drop in booster dose uptake, even with high primary vaccination coverage, could be attributed to most requirements mandating primary vaccination by the Turkish Ministry of Health do not extend to booster doses.
Since the CoronaVac was available in Türkiye when the vaccination program started, the first two doses of vaccines in the study group were mostly CoronaVac. The arrival of the BNT162b2 vaccine in Türkiye was in April 2022, and it had mostly been preferred as a booster dose. However, the frequency of preferring inactive vaccines as booster doses was higher in the group aged 85 and over compared to other age groups in our study. Providing only the CoronaVac during in-home visits might have raised the rate of inactive booster doses among individuals with a less active lifestyle. The most significant factors influencing vaccine choice were effectiveness and potential adverse reactions, as indicated in research. 26 The higher tendency toward the inactivated vaccine in the group aged 85 and over may have been impacted by adverse reaction perceptions of them. Besides, the occurrence of comorbidities increases with advancing age. Our study did not assess comorbidities; however, a previous study in the Turkish elderly population aged 65 and above demonstrated that the presence of comorbidities did not influence the selection between mRNA and CoronaVac vaccines. 27
Turkovac, developed in Türkiye, was in a small proportion of the booster doses in our study group. Although Turkovac development stages were followed with interest in the Turkish community, this low rate emerged because it started to be distributed to vaccination centers when the number of cases and vaccination rates decreased. 3
Limitations
The present study has some limitations: First, since COVID-19-related hospitalization and death data could not be obtained from the national HSYS system, the effectiveness level of the vaccines in these parameters could not be evaluated. Therefore, considering the divergent results of previous studies that evaluated SARS-CoV-2 PCR test positivity together with hospitalization and mortality, it is important to understand that it would be inappropriate to make inferences about COVID-19 hospitalization and mortality rates from our results. Second, chronic disease data were unavailable, so this variable could not be included in the regression analyses. However, in light of the lack of evidence linking comorbidities to the COVID-19 vaccine type preference and with age and sex variables included in our regression analyses, the effect of comorbidities on the outcomes was regarded as marginal level. Third, due to the unavailability of variant-specific SARS-nCoV-2 PCR positivity data in the national HSYS system, effectiveness levels of booster doses were interpreted based on dominant variants over time in Türkiye. Fourth, since most of the older age group had already completed the two doses of the vaccination until the BNT162b2 vaccine arrived in Türkiye, the number of individuals whose vaccination schedule was homolog booster with primary mRNA series was insufficient for analysis. Larger cohort studies should be conducted across Türkiye, with a higher number of those who received the homologous mRNA booster dose/doses vaccination schedules.
Conclusion
Our findings showed that in the population aged 65 and over, having at least one dose of BNT162b2 vaccine in the vaccination schedule had higher effectiveness in preventing SARS-CoV-2 PCR positivity compared to homolog boosters with primary CoronaVac series. Although the frequency of the two-dose series of vaccine receipt was high in our study group, one-third of those did not receive a booster dose. Future studies will evaluate SARS-CoV-2 vaccine acceptance and effectiveness levels in different districts of İstanbul to reveal the impact of sociocultural differences on this topic.
