Abstract
While vaccination against COVID-19 has significantly improved the morbidity and mortality of the disease, with the increase in the administration of COVID-19 vaccines, it is more likely to observe their rare side effects in the clinical settings. Herein, we report a case of an 82-year-old man with history of coronary artery disease, prostate cancer in remission, gastroesophageal reflux disease, and hypothyroidism, who presented with acute pancreatitis few hours after receiving the third dose of Pfizer-BioNTech BNT162b2 mRNA COVID-19 vaccine, without other identified etiology. His symptoms were mild and he was discharged in a stable condition after improvement in his condition with supportive care.
Introduction
Coronavirus disease 2019 (COVID-19) caused by severe acute respiratory syndrome coronavirus 2 (SARS-COV-2) is a global health concern. Vaccination against COVID-19 has significantly contributed to the decrease in the hospitalization and mortality due to this disease. 1 Adverse gastrointestinal effects following COVID-19 vaccination are generally mild and may include nausea, vomiting, abdominal pain/discomfort, and diarrhea.2–4 Nevertheless, with the increase in the rate of vaccination, less common side effects of vaccination may be encountered clinically. Herein, we report a case of acute pancreatitis following administration of the third dose of Pfizer-BioNTech BNT162b2 mRNA COVID-19 vaccine without other identified etiology.
Case presentation
An 82-year-old man presented with a sudden onset of dull epigastric abdominal pain with radiation to back. The pain was associated with nausea and two episodes of non-bloody non-bilious vomiting. He reported no history of trauma or prior cholelithiasis and reported receiving his booster (third) dose of BNT162b2 vaccine earlier on the day of admission. With prior doses, he had experienced mild headache and myalgia.
His medical history was significant for coronary artery disease with one drug-eluting stent placed about 30 months ago, prostate cancer treated with radiation therapy complicated by radiation proctocolitis followed by partial colectomy with end anastomosis about 20 years ago, hypothyroidism for about 36 years, and gastroesophageal reflux disease for about 50 years. He had been taking daily aspirin 81 mg, atorvastatin 80 mg (for coronary artery disease), rectal mesalasine 1,000 mg (for radiation colitis), pantoprazole 40 mg (for gastroesophageal reflux disease), and levothyroxine 88 µg (for hypothyroidism); however, he did not report over-the-counter medications. He reported no smoking, recreational drug use, or consumption of alcoholic beverages.
On examination, his vital signs were stable. Physical examination was unremarkable other than abdominal examination which revealed a soft abdomen, mild epigastric tenderness, without rebound or gaurding. Laboratory investigations are summarized in Table 1. In brief, serum lipase was elevated to 2,257 U/L (reference range: 11–82), serum triglyceride was 57 mg/dL (reference range: <150), serum calcium was 9.3 mg/dL (reference range: 8.6–10.3), and serum ethanol level was less than 10 mg/dL.
Laboratory findings at presentation.
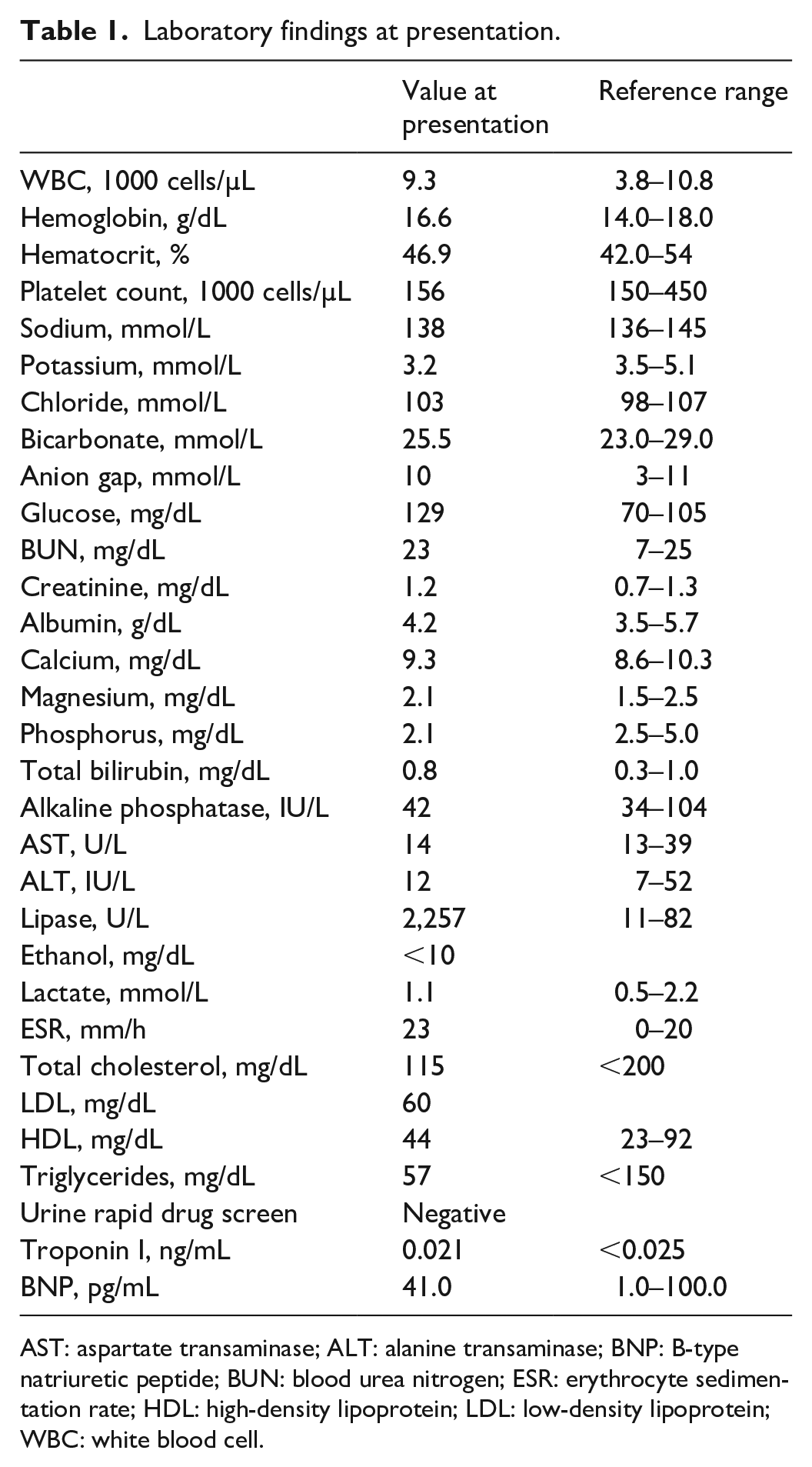
AST: aspartate transaminase; ALT: alanine transaminase; BNP: B-type natriuretic peptide; BUN: blood urea nitrogen; ESR: erythrocyte sedimentation rate; HDL: high-density lipoprotein; LDL: low-density lipoprotein; WBC: white blood cell.
Abdomen and pelvis computed tomography scan with contrast showed moderate peripancreatic fat stranding with tracking fluid into the mesenteric root, suggestive for acute interstitial pancreatitis, without evidence of necrosis (Figure 1). Of note, no calcified cholelithiasis, gallbladder wall thickening, or biliary dilatation was observed (Figure 1). Right upper quadrant ultrasound also showed normal gall bladder without evidence of cholelithiasis and a normal common duct measuring 5 mm.

Computed tomography findings.
He was treated with intravascular fluids, symptomatic therapy with acetaminophen, hydromorphone, and metoclopramide. He tolerated oral diet without further abdominal pain, and his serum lipase was decreased to 268 U/L at his discharge, 2 days following the admission.
Discussion
Vaccination remains as one of the most powerful tools in our battle with COVID-19 pandemic.1,5 Nevertheless, with more common use of the vaccine, it is more likely that rare side effects of vaccination are observed in the clinical setting. Thus, awareness of potential side effects and monitoring of the vaccinated individuals can improve patient care.
Acute pancreatitis is commonly caused by obstructing common bile duct stones, alcohol consumption, hypertriglyceridemia, medications, and traumas. Nevertheless, infectious etiologies may also contribute to development of acute pancreatitis.6,7 Notably, SARS-COV-2 has been suggested as an etiology of acute pancreatitis in certain cases.8,9 Although a critical review of the presented cases in the literature suggests that attributing SARS-COV-2 as the etiology of acute pancreatitis is questionable in many of the reported cases, some evidence suggests that this virus may be implicated in development of acute pancreatitis in at least a fraction of the reported cases.9,10 Several mechanisms are postulated for the development of acute pancreatitis by SARS-COV-2. Direct viral invasion to the components of exocrine pancreas and its cytopathic effects on pancreatic cells may serve as the underlying mechanism of developing acute pancreatitis in individuals with COVID-19. For the entry of the virus to human cells, viral spike protein binds to cell surface angiotensin-converting enzyme 2 (ACE2) receptor. Subsequently, proteolytic activity of the type 2 transmembrane serine protease (TMPRSS2) facilitates viral entry to human cells.11–13 Pancreatic ductal cells express both ACE2 and TMPRSS2 molecules. 14 Although co-expression of these molecules by pancreatic ductal cells is rare, viral nucleocapsid protein has been detected in pancreatic ductal cells.14,15 Thus, direct viral invasion and subsequent cytotoxic effects of the virus may stem the development of acute pancreatitis. Nevertheless, bystander damage to pancreatic tissue due to local activation of the immune system following invasion of the virus to this tissue or severe systemic inflammation leading to non-specific bystander damage to the pancreas may also contribute to development of acute pancreatitis in individuals with COVID-19.
Although microbial agents constitute a minor fraction as the etiology of acute pancreatitis,6,7 reports on potential association of vaccination with acute pancreatitis are more rare.16,17 In terms of COVID-19 vaccination, to the best of our knowledge, currently four cases of acute pancreatitis have been reported in the literature, including the case stated in the BNT162b2 vaccine trial report. These cases occurred within few hours to days following administration of the first or second dose of BNT162b2 vaccine.18–21 Of note, in one case, acute pancreatitis occurred after both first and second doses of the vaccination. 21 Nevertheless, the association of COVID-19 vaccination and acute pancreatitis and its potential etiologic role is not clear in such cases. For instance, in one of the cases, 19 the diagnosis of acute pancreatitis is questionable based on Atlanta classification, 22 since despite the symptoms and elevation of urine amylase, serum amylase was not elevated more than three times of normal limits and also ultrasound and magnetic resonance imaging did not suggest pancreatic inflammation. The case of acute pancreatitis in the BNT162b2 vaccine trial is reported as obstructive pancreatitis, which suggests against an etiologic role of the vaccine for development of acute pancreatitis in this case. 18 One of the other cases had experienced a prior episode of presumably alcoholic pancreatitis prior to the episode associated with the vaccination and the patient has also consumed about 15 g of ethanol prior to onset of the symptoms. 20 Similarly, in the case of acute pancreatitis following both doses of vaccination, offending medication may also have contributed to development of the acute pancreatitis. 21 In our case, acute pancreatitis occurred after a short while after receiving the third dose of the vaccine, without evidence of consumption of alcoholic beverages, cholelithiasis, hypertriglyceridemia, hypercalcemia, or trauma. Although potential side effects of medications such as mesalasine and pantoprazole may have been the etiology of acute pancreatitis in our case, long-term use of these medications in our case rather than recent initiation of the agents may make them less likely to be the cause of acute pancreatitis.
Despite the higher prevalence of vaccination compared to COVID-19, fewer cases of acute pancreatitis have been reported following vaccination which may suggest that different mechanisms contribute to the development of acute pancreatitis after COVID-19 vaccination, compared to the development of acute pancreatitis during the disease. In cases of development of the acute pancreatitis after COVID-19 vaccination with viral mRNA, dissimilar to application of live-attenuated virus, direct cytopathic effects of the virus cannot explain pancreatic tissue damage and resultant acute pancreatitis. In such cases, considering that pancreatic tissue has been the only solid tissue involved, antigen mimicry or to less extent severe inflammation leading to non-specific activation of immune system may serve as the etiology of the acute pancreatitis. Nevertheless, our knowledge is currently limited on the cellular and molecular mechanisms through which COVID-19 vaccine may cause acute pancreatitis. Further studies may unravel whether other types of COVID-19 vaccines may also cause acute pancreatitis and whether potential pancreatic damage via these vaccines may occur via similar mechanisms.
Conclusion
Acute pancreatitis should be considered as a differential diagnosis in individuals with abdominal pain after receiving COVID-19 vaccine. Further studies are paramount to evaluate potential etiologic role of COVID-19 vaccine in development of acute pancreatitis.
Footnotes
Acknowledgements
Authors wish to express their gratitude to the patient for consenting for this publication.
Author contributions
R.C., A.J., R.J., K.N., K.V., and Z.B. concepted the report. R.C., A.J., and R.J. drafted the manuscript. K.N., K.V., and Z.B. critically revised the manuscript. All authors approved the final version of the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
