Abstract
Background:
Patients with plasma cell dyscrasia are at a higher risk of developing a severe Coronavirus-2019 (COVID-19) infection. Here we present a systematic review of clinical studies focusing on the immune response to the COVID-19 vaccination in patients with plasma cell dyscrasia.
Objectives:
This study aims to evaluate the immune response to COVID-19 vaccines in patients with plasma cell dyscrasia and to utilize the results to improve day-to-day practice.
Design:
Systematic Review
Methods:
Online databases (PubMed, CINAHL, Ovid, and Cochrane) were searched following the preferred reporting items for systematic review and meta-analysis (PRISMA) guidelines. Only articles published in the English language were included. Out of 59 studies, nine articles (seven prospective and two retrospective studies) were included in this systematic review. Abstracts, case reports, and case series were excluded.
Results:
In all nine studies (N = 1429), seroconversion post-vaccination was the primary endpoint. Patients with plasma cell disorders had a lower seroconversion rate compared to healthy vaccinated individuals and the overall percentage of seroconversion ranged between 23% and 95.5%. Among patients on active therapy, lower seroconversion rates were seen on an anti-CD38 agent, ranging from 6.5 up to 100%. In addition, a significantly lower percentage was recorded in older patients, especially in those aged equal to or greater than 65 years and those who have been treated with multiple therapies previously. Only one study reported a statistically significant better humoral response rate with the mRNA vaccine compared to ADZ1222/Ad26.Cov.S.
Conclusion:
Variable seropositive rates are seen in patients with plasma cell dyscrasia. Lower rates are reported in patients on active therapy, anti-CD38 therapy, and elderly patients. Hence, we propose patients with plasma cell dyscrasias should receive periodic boosters to maintain clinically significant levels of antibodies against COVID-19.
Registration:
PROSPERO ID: CRD42023404989
Plain language summary
Introduction
Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), initially detected in Wuhan, China (December 2019), rapidly spread worldwide, infecting numerous people. As of September 2022, SARS-CoV-2 has affected more than 609 million cases across the world, claiming more than 6.5 million lives. Coronavirus-2019 (COVID-19) infection predominantly causes pneumonia, leading to acute respiratory distress syndrome (ARDS) which is the most common cause of mortality. At the beginning of this pandemic due to this disease’s contagious nature and the absence of targeted antiviral medications, mortality and morbidity were substantial. The pathophysiology of ARDS is immune mediated secondary to the cytokine release syndrome resulting in endothelial injury. 1 One of the comorbidities associated with worse outcomes is an immunocompromised state. 2
Patients with hematological malignancies are at higher risk of severe infection. 2 Patients with leukemia, lymphoma, myeloproliferative disorder, and multiple myeloma (MM) usually have prolonged immunodeficiency. The disease process itself causes an inability to mount a substantial immune response. In addition, treatment modalities including chemotherapy and bone marrow transplant can also independently suppress the immune system. 3
The development of the COVID-19 vaccine has allowed the public to develop antibodies that protected them from severe illness. Vaccine response, however, is not similar in all recipients. Patients with hematological malignancies may respond differently to the vaccine compared to otherwise healthy individuals. In this systematic review, we aim to discuss the immune response to the COVID-19 vaccine in patients with plasma cell dyscrasia (PCD). This review aims to discuss in depth, the efficacy and safety of COVID-19 vaccines in patients with MM. We plan to discuss the positive and negative risk factors associated with vaccine response including the chemotherapies which can affect antibody production. In addition, many studies discuss the humoral response, however, few focus on T-cell-mediated response. We plan to bring forth both types of responses and attempt to explain heterogeneous data in a way that is easy to understand.
Methods
Online databases including PubMed, CINAHL, Ovid, and Cochrane were searched from the inception of literature through 3 August 2022, following the PRISMA (preferred reporting items for systematic review and meta-analysis) guidelines. We included articles published in English. Randomized, quasi-randomized, and nonrandomized clinical trials, prospective, retrospective, and case–control studies were included. Systematic reviews, meta-analyses, conference abstracts, case reports, case series, review articles, and articles in other languages were excluded. MeSH terms for multiple myeloma included Multiple Myelomas, Myeloma Multiple, Myeloma Plasma-Cell, Myeloma Plasma Cell, Myelomas Plasma-Cell, Myelomatosis, Myelomatoses, Cell Myeloma Plasma, Cell Myelomas Plasma, Myelomas Plasma Cell, Kahler Disease, Myeloma-Multiple, and Myeloma-Multiples. MeSH terms for covid-19 vaccine included COVID 19 Vaccines, COVID-19 Virus Vaccines, COVID 19 Virus Vaccines, COVID-19 Virus Vaccine, COVID 19 Virus Vaccine, COVID19 Virus Vaccines, COVID19 Virus Vaccine, COVID19 Vaccines, COVID19 Vaccine, SARS-CoV-2 Vaccines, SARS CoV 2 Vaccines, SARS-CoV-2 Vaccine, SARS CoV 2 Vaccine, SARS2 Vaccines, SARS2 Vaccine, Coronavirus Disease 2019 Vaccines, Coronavirus Disease 2019 Vaccine, Coronavirus Disease 2019 Virus Vaccine, Coronavirus Disease 2019 Virus Vaccines, Coronavirus Disease-19 Vaccines, Coronavirus Disease 19 Vaccines, Coronavirus Disease-19 Vaccine, Coronavirus Disease 19 Vaccine, COVID 19 Vaccine, 2019-nCoV Vaccine, 2019 nCoV Vaccine, 2019 Novel Coronavirus Vaccines, 2019 Novel Coronavirus Vaccine, 2019-nCoV Vaccines, 2019 nCoV Vaccines, COVID-19 Vaccine, and SARS Coronavirus 2 Vaccines. MeSH terms for antibody formation included Formation, Antibody, Antibody Response, Antibody Responses, and Antibody Production. Fifty-nine articles were identified. Two authors participated in the primary and secondary screening leading to the inclusion of nine studies in the final systematic review. Data were extracted manually onto Microsoft Excel software. Two separate sheets were maintained for efficacy and safety. Once collected, data were analyzed by the two researchers. Discrepancies were resolved by discussion between the two researchers. Data were collected and the abstract was formulated by December 2022. The manuscript was then completed by January 2023.
The risk of bias was assessed by using the ROBIS 1.2 risk assessment tool. The risk of bias was low in domains 1, 2, and 3 which focused on study eligibility criteria, identification and selection of studies, and data collection and study appraisal, respectively. Domain 4 was not applicable to our study. The overall risk of bias was deemed to be low for this review. A limitation was the lack of sufficient sample of patients with seroconversion post-vaccination, due to the relative rarity of the disease as well as the exposure to COVID-19 vaccines in patients with PCD. To maximize sample size, meta-analysis was an option; however, the heterogeneity of published risk estimates would make the understanding and interpretation of the findings challenging. Prior research projects have shown mixed results regarding the importance of molecular-based monitoring among patients on active therapy, and seroconversion rates in patients taking anti-CD38 medications. In addition, there are a number of other measures of body composition that have received less attention. It is the objective of this article to shed light on the risk of variable seropositive rates observed in patients with PCD through a review of nine articles (seven prospective and two retrospective). It is important to note, however, that most of the individual studies conducted to date were small and did not possess enough power to detect associations. This led to the fact that the pooled analysis exhibited a lowered power with an increased heterogeneity, especially when the same covariates were adjusted and the unconditional regression analysis was applied across these studies.
These study methods have been registered at PROSPERO, registration ID: CRD42023404989.
Results
We included seven prospective and two retrospective studies in this systematic review. Screening process is further explained in the PRISMA table (Figure 1). Out of the 1492 patients, 1213 had MM. Patients received both mRNA (BNT162b2/mRNA1273) and vector-based (ADZ1222/Ad26.Cov.S) vaccines.

Preferred reporting items for systematic review and meta-analysis table.
Avivi et al. enrolled 171 patients and 64 controls in a prospective case–control study. Twelve patients had smoldering multiple myeloma (SMM) and 159 had MM. All patients received two doses of the mRNA vaccine (BNT162b2). A cut-off value of ⩾0.80 U/mL of anti-SARS-CoV 2 specific immunoassay was considered positive. Patients with SMM had a 100% seroconversion rate. A lower seroconversion of 69% was reported in patients on anti-CD38 therapy. Factors associated with statistically significant lower seroconversion rates included low absolute lymphocyte count ⩽1.5 K/µL [odds ratio (OR) 0.4 (0.17–0.94), p = 0.036], hypogammaglobinemia ⩽10 g/L [OR 0.15 (0.03–0.67), p = 0.013], ⩾3 lines of therapy [OR 0.16 (0.04–0.58)], and partial response to therapy [OR 0.43 (0.19–0.98), p = 0.04]. Limitations of the study include variability of treatment regimens and focus on humoral response only. 4
Ghandili et al. enrolled 82 patients in a single-center prospective study. A total of 63 patients received the mRNA vaccine (BNT162b2 or mRNA1273) and 19 received a vector-based vaccine (AZD1222). Seroconversion after one dose of vaccine was determined based on anti-SARS-CoV-2S antibody titer level with a cut-off value of ⩾33.8 BAU/mL. The longer duration of anti-CD38 therapy was related to lower antibody production (p-value = 0.006). A higher CD-19 lymphocyte count (⩾30/µL) was associated with a higher seroconversion rate of 35% compared to 12.5% in patients with a low CD-19 lymphocyte count (p-value = 0.027). No statistically significant difference between seroconversion rates was reported between mRNA and vector-based vaccination. Limitations include a smaller cohort size, nonstandardized measurement of COVID-19 antibody level after the first dose and focus on humoral response only. 5
Greenberg et al. enrolled 44 MM patients in a prospective study. All patients received two doses of mRNA vaccine (BNT162b2 or mRNA1273). The anti-SARS-CoV antibody titer was checked at a median of 29 days after the last vaccine. The cut-off of seroconversion was set at ⩾0.79 U/mL. Patients on active therapy and those not on active therapies had a comparable seroconversion, 94% versus 93%, respectively. Three patients had undetectable antibody levels. One of these patients was not on therapy, while the other two received ixazomib (proteasome inhibitor) and teclistamab (a B-cell maturation antigen × CD3-bispecific antibody), respectively. Limitations of this study included a small sample size and social media recruitment strategy. 6
Nooka et al. prospectively studied the antibody response in 238 patients with MM. Antibodies against CoV-2 receptor-binding domain (RBD-Ab) were assayed, as were neutralizing antibodies (nAbs), which are more predictive of the protection against symptomatic COVID-19 infection. Both antibody levels peaked around 1–2 weeks after the second dose of the vaccine and declined over the next 3 months. Antibody levels were higher in patients with prior COVID-19 exposure. While there was a strong correlation between anti-RBDs and nAbs, there were 33.6% of patients who had positive RBD-Ab, but negative nAbs. Antibody levels were found to have a negative association with hypogammaglobinemia (IgG ⩽ 700 mg/dL) [OR 1.80 (0.93–3.45), p = 0.08] and two or more prior therapies with anti-CD38 agent [OR 0.36 (0.16–0.80), p = 0.012]. Black patients were noted to have higher nAb response compared to their white counterparts. mRNA1273 vaccine recipients had higher nAb response (67%) as opposed to BNT162b2 (48%), though the patients had similar rates of seroconversion for anti-RBD. Patients on MM therapies were also noted to have a lower level of antibodies. There were also reduced RBD levels in patients on B-cell maturation antigen targeting therapies, such as belantamab mafodotin and venetoclax. However, patients on maintenance therapy had higher levels of both antibodies. The limitation of the study was the lack of healthy controls for comparison. 7
Ramasamy et al. prospectively observed both humoral and T-cell responses to the COVID-19 vaccine in patients with MM and SMM. Data from 214 patients (204 MM, 10 SMM) were reported. Anti-spike antibody (anti-S Ab) levels were reported after two doses of vaccine. ADZ 1222, BNT162, and unknown vaccines were administered to 94, 66, and 54 patients, respectively. Anti-S Ab level of >50 IU/mL was considered positive. Overall seropositivity was reported as 92.7% for MM and 90% for SMM patients. Out of the 49 patients previously on therapy, 44 attained seropositivity (89.7%). This did not include those who received anti-CD38 therapy. It was concluded that those on chemotherapy had lower levels of anti-S Ab compared to those not on chemotherapy (p = 0.025). Patients with complete remission/very good partial remission had a higher response than those with partial response or relapsing disease (11.9% versus 3.9%, p = 0.051). Men were found to have lower anti-S Ab levels after the second dose compared to women, adjusted to age (11.3% versus 2.3%, p = 0.015). Antibody levels were negatively associated with increasing age. A comparable seropositivity rate was reported (89.4% versus 93.6%) in patients receiving the BNT162b2 versus ADZ1222 vaccine. However, a higher anti-S Ab titer was seen in patients receiving the BNT162b2 vaccine (p = 0.018). T-cell response was measured by interferon-gamma release assay (IGRA) which was measured after the second dose of vaccine. A positive IGRA response was significantly associated with higher anti-S Ab levels (p = 0.002). Predictors of a negative IGRA response were increased age, exposure to anti-CD38, and Pfizer vaccine. Limitations of this study include missing clinical data and a short follow-up. 8
Shah et al. retrospectively studied 89 patients with PCD, 41 had MM. A total of 78 patients received the mRNA vaccine (BNT162b2 or mRNA1273) while 11 received the vector-based vaccine (Ad26.Cov.S). Anti-spike SARS-CoV antibodies were measured with a cut-off of ⩾0.4 U/mL for seroconversion. Statistically significant lower rates of seroconversion were seen in patients with advanced age with a 1-year difference having an adjusted p-value of 0.0044. Higher seroconversion rates were seen in patients receiving BNT162b2 and mRNA1273 vaccines compared to Ad26.Cov.S with an adjusted p-value of 0.0465 and 0.0194, respectively. No statistically significant difference was found between mRNA1273 and BNT162b2 vaccines (p = 0.7903). Four patients did not seroconvert; two of the four patients had HIV with low T-cell counts, while the other two patients seroconverted after receiving intravenous immunoglobulins. Limitations of the study include a small study population and a focus on humoral response only. 9
Terpos et al. enrolled 276 patients with PCD including 213 patients with MM. The control group had 226 patients. SARS-CoV antibody titer (Nab) was measured using ELISA after two doses of the vaccine, a level of ⩾30% was deemed positive while a level ⩾50% was clinically relevant for viral inhibition. Levels were measured on day 1, day 22 (d22), and day 50 (d50) in both groups. On d22, the median Nab inhibition titer in PCD patients was lower than the controls, 27% compared to 39.7%, respectively (p < 0.001). On d50, the median Nab inhibition titer was 62% and 90% in cases and controls, respectively (p = 0.001). Patients with MM had a lower antibody titer compared to monoclonal gammopathy of undetermined significance (MGUS) patients on both d22 (p = 0.009) and d50 (p = 0.003). The percentage of patients versus controls developing ⩾50% Nab titers was 19.9 and 32.3, respectively (p = 0.002). No statistically significant difference was found between patients receiving mRNA vaccine versus vector-based vaccine. On d50, patients who did not receive any treatment had a higher Nab level compared to those on belantamab mafodotin (monoclonal antibody-drug conjugate comprises an antibody targeting B-cell maturation agent) based combinations or anti-CD38 therapy [(mean ± SD: 66% ± 25.4%) versus (mean ± SD: 28.2% ± 20.1%, p = 0.002) and (mean ± SD: 45.4% ± 29.4%, p = 0.013), respectively]. These patients also had lower Nab levels when compared to patients on lenalidomide (immunomodulatory agent) (p = 0.003 and p = 0.015, respectively). Limitations include a small study population, a focus on humoral response only, and a short follow-up. 10
Terao et al. conducted a three-center, prospective, case–control study involving 269 PCD patients (206 MM and 25 SMM) and 94 controls. Most patients including 95.2% of cases and 100% of controls received the BNT162b2 vaccine, while only 4.8% of PCD patients received the mRNA1273 vaccine. Values of anti-spike antibody (S-IgG) ⩾0.8 U/mL and neutralizing antibody (N-IgG) ⩾1.0 cut-off index were considered positive. S-IgG ⩾ 200 U/mL was considered clinically protective. The median S-IgG (U/mL) was reported as 694, 561, 268, and 116 in controls, MGUS, SMM, and MM, respectively. Overall seropositivity was reported as 88.7%, 96%, and 100% in MM, MGUS, and SMM patients, respectively. Clinically protective levels were reported as 72%, 76%, and 38.3% in SMM, MGUS, and MM patients, respectively. Patients who received anti-CD38 therapy within 30 days when compared with patients who received anti-CD38 therapy within 31–180 days of the first vaccine had a comparable seropositivity percentage (82.5% versus 81.3%); however, a lower clinically protective rate (23.8% versus 37.5%) was reported suggesting that a washout period improved antibody production. Older age ⩾65 years [OR 0.36, 95% CI 0.13–0.99, p = 0.048], insufficient treatment response [OR 0.24, 95% CI 0.08–0.74, p = 0.013], anti-CD38 therapy [OR 0.32, 95% CI 0.11–0.90, p = 0.031], immunomodulatory drugs [OR 0.26, 95% CI 0.12–0.54, p < 0.001], lymphopenia (<1000/µL) [OR 0.37, 95% CI 0.14–0.99, p = 0.048], and a lower polyclonal IgG (<550 mg/dL) [OR 0.29, 95% CI 0.14–0.58, p < 0.001] were associated negatively with a humoral response. Limitations of the study include a small study sample, short follow-up time, and focus on humoral response only. 11
Wu et al. retrospectively compared 45 PCD patients [22 MGUS, 3 SMM, 2 light chain amyloidosis (LCA), 18 MM] to 78 healthy vaccinated controls. The remnant serum samples from the PCD patients from routine blood tests, collected between February and July 2021 were used to measure IgG antibodies against COVID-19. A cut-off of 50 relative fluorescence units (RFUs) was considered as positive. Patients who had COVID-19 infection previously were excluded from the study. The antibody levels were noted to be lower in the cases, compared to controls. One MM patient (BNT162b2) and two patients with MGUS (mRNA1273 and BNT162b2) did not seroconvert. There were insufficient number of patients to detect seroconversion differences between MGUS, SMM, LCA, and MM patients, as well as between different types of vaccines. Limitations of the study were observational nature, small patient population, lack of data on breakthrough infections, and focus on humoral response only. All MGUS and SMM patients were not on any current therapy and comprised most of the patient population. No comparison was made between MM patients on CD38 therapy versus conventional therapies. 12
Only three studies commented on the safety profile of the vaccine. None of the three studies reported grade 3 or 4 adverse events. No deaths were reported in any of the studies. Avivi et al. reported a higher percentage of myalgia, headache, dizziness, rash, and chills in MM patients compared to controls. Greenberg et al. reported a higher percentage of fatigue, headache, and myalgia with the second dose compared to the first dose of the vaccine (39% versus 64%, 32% versus 50%, and 32% versus 41%, respectively). Similar results were reported by Terpos et al., a higher percentage of mild systemic reactions was noted with the second dose of the BNT162b2 vaccine (13% versus 21%). AZD1222 group had 32.8% local mild reactions, and no systemic reaction was reported.
A comprehensive review of the included studies is provided in the form of efficacy and safety tables (Tables 1 and 2).
Efficacy of COVID-19 vaccines in patients with plasma cell dyscrasia.

Seropositive with antibody titer >30%.
Seropositive with antibody titer >50%.
CC, case control; CP, clinically protective; CR, complete remission; d22, day 22; d50, day 50; IGRA, interferon-gamma release assay; LCA, light chain amyloidosis; MM, multiple myeloma; n, number; n/N, number of participants evaluated/number of participants enrolled; NA, nonapplicable; nAbs, neutralizing antibodies; ND, no difference; OR, odds ratio; P/R, progressive/relapsing; PCD, plasma cell dyscrasia; PR, partial remission; PS, prospective study; RBD-Abs, anti-receptor-binding domain antibodies; RS, retrospective study; SMM, smoldering multiple myeloma; y, years.
Safety of COVID-19 vaccine in plasma cell dyscrasia.
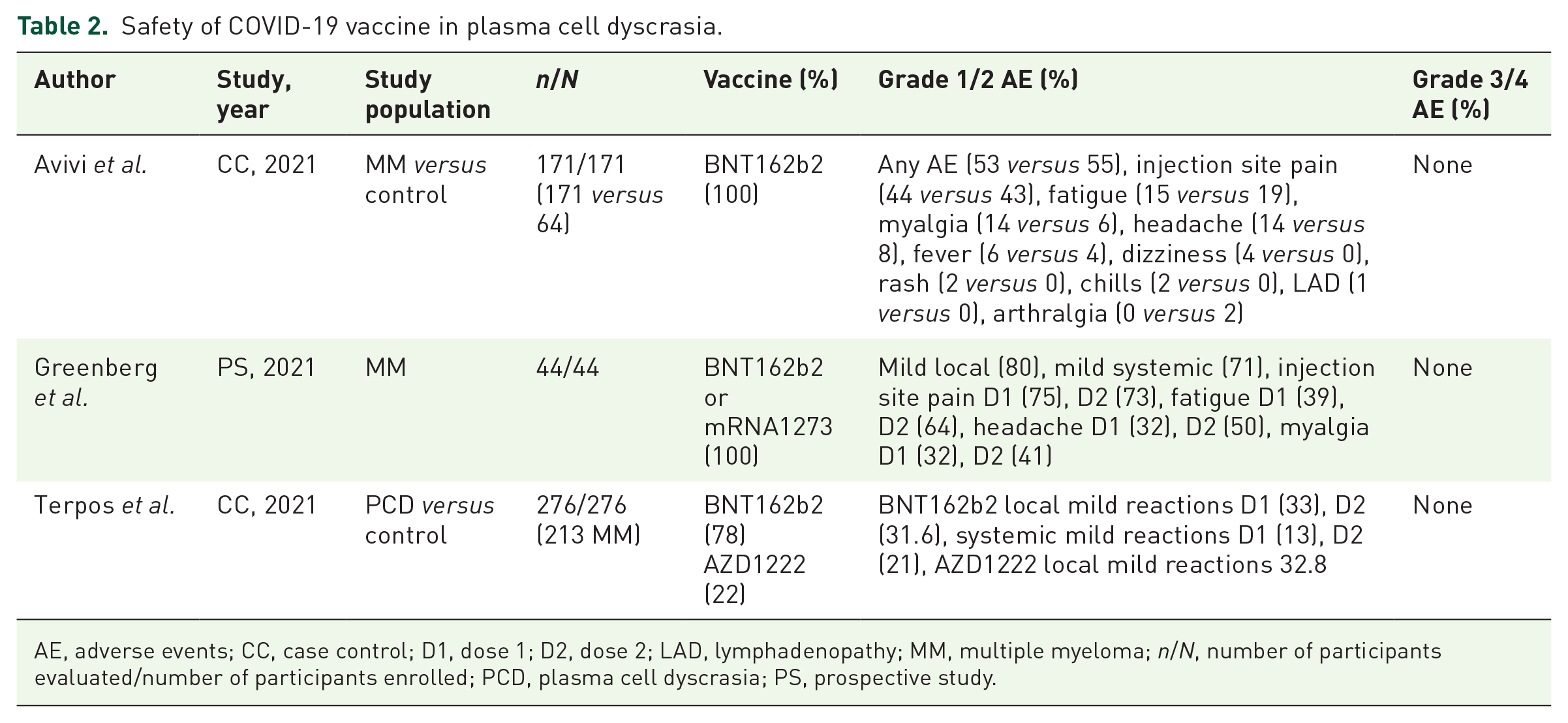
AE, adverse events; CC, case control; D1, dose 1; D2, dose 2; LAD, lymphadenopathy; MM, multiple myeloma; n/N, number of participants evaluated/number of participants enrolled; PCD, plasma cell dyscrasia; PS, prospective study.
Discussion
Patients with PCD are unable to mount a normal response to vaccines due to defective antibody production as seen previously with pneumococcal and influenza vaccines.13 –17 Compared to patients with oncological malignancies, seroconversion was reported to be inferior in patients with hematological malignancies.18 –20 All the four case–control studies included in this review reported a higher seroconversion rate in controls compared to cases. Antibody titer levels are often associated with the durability of the vaccine response; however, it is still unclear what level of antibody titer provides adequate protection from COVID-19.14,15,21 Real-world evidence shows a high risk of breakthrough COVID-19 infections in patients with hematological malignancies, especially in those with MM. Song et al. reports that the risk of COVID-19 infection decreases after the second dose of the COVID-19 vaccine in all cancer patients, especially in MM (OR 0.35; 95% CI, 0.15–0.72). 22 Antibody titers of ⩾50% were deemed clinically protective by Terpos et al. On day 50, only 57.3% of patients had a clinically protective antibody titer compared to 81% of the controls. 10 Similarly, Terao et al. considered an anti-spike antibody (S-IgG) titer of ⩾200 U/mL as clinically protective; in a previous study by Shields et al. (n = 246) an S-IgG titer of >195.2 U/mL provided 6-month protection against reinfection with COVID-19. The percentage of controls, MGUS, SMM, and MM patients who achieved a clinical protective S-IgG level was reported to be 92.6, 76, 72, and 38.3%, respectively.11,23 Hence, once again suggesting that as the plasma cell disorder progresses, the degree of impairment to respond to vaccine worsens.
Mansi et al. studied both humoral and T-cell-mediated responses in cancer patients (28 solid tumors and 11 hematological malignancies): 74% of patients produced COVID-19-specific antibodies; however, only 46% had a T-cell-mediated response suggesting that cancer patients might have defective T-cell-mediated protection against COVID-19 as well. 24 Only one of the included studies investigated the role of T-cell-mediated response of post-COVID-19 vaccine. Ramasamy et al. monitored IGRA in 167 patients as an indicator of T-cell-mediated response; >8 interferon-gamma releasing cells/106 peripheral blood mononuclear cells was considered positive: 61% of MM patients had a positive IGRA response. In addition, patients were more likely to have a positive IGRA response with ADZ1222 than with BNT162b2 (70.6% versus 44.2%). Positive IGRA results were strongly associated with a positive S-IgG response (p = 0.021) and only 6.5% of the patients were found to have both negative IGRA and S-IgG responses. 8 In the study by Shah et al., two out of four nonresponders had HIV and low T-cell counts indicating that T-cell-mediated immune response probably contributes to a stronger humoral response. 9 In a recent article, patients with chronic lymphocytic leukemia (CLL) were evaluated for both humoral and T-cell response post-vaccination with the BNT162b2 vaccine. A lower rate of T-cell response was noted in CLL patients compared to controls (29% versus 59%). Current therapy with ibrutinib and previous therapy with CD20 monoclonal antibody had worse responses (14% each). 25 The T-cell response needs to be evaluated in larger studies, especially in patients with plasma cell disorders.
Aging is associated with a decline in bone marrow lymphopoiesis as well as the B-cell diversity leading to a decrease in the efficacy of vaccine response.26,27 All studies included reported a lower seropositive percentage in elderly patients. This can also be explained by the fact that elderly patients with PCD might have been previously treated with multiple agents and have an exhausted immune system.8,28 –30 Better response in young patients is true for both the control and PCD groups.8,30
In many of the included studies, lymphopenia and hypogammaglobinemia were independently associated with lower seroconversion rates. Avivi et al. and Terao et al. reported that lymphopenia (⩽1500/µL, p = 0.036) and hypogammaglobinemia (<1000/µL, p = 0.048), respectively, was associated with statistically significant lower levels of antibody titers.4,11 Similarly, Ghandili et al. 5 reported that a higher CD-19 lymphocyte count (⩾30/µL) was associated with a higher seroconversion rate of 35% compared to 12.5% in patients with a low CD-19 lymphocyte count (p-value = 0.027). Hypogammaglobinemia suggests poor B-cell reserve to begin with and with fewer functional B-cells. These patients end up having a poor humoral response to the vaccine. In the three studies included (Avivi et al., 4 Nooka et al., 7 and Terao et al. 11 ) hypogammaglobinemia was independently associated with worse seroconversion rates.
Patients on therapy with a good disease control had a higher vaccine response rate than those with a poorly controlled disease, refractory disease, or the ones treated with multiple agents previously demonstrating that these patients have fewer defective B-cells and hence a better humoral response. A low response rate was seen in patients on anti-CD38 therapy which can be attributed to its B-cell-depleting effect causing hypogammaglobinemia. Daratumumab is usually used in advanced refractory cases, which can be an alternate explanation. Patients on anti-CD38 therapy have been previously reported to show a response to the pneumococcal vaccine and seen to do so for the COVID-19 vaccine but this response is lower compared to that of other treatments. 31
Impressive seroconversion rates were reported in the subgroup of patients on lenalidomide, as reported previously. 32 Seroconversion percentage was 94%, 100%, and 100% in studies by Greenberg, Avivi, and Shah, respectively. Shah et al. reported a statistically significant decreased response rate with the Ad26.Cov.S vaccine compared to the mRNA vaccine, which leads us to believe that maybe a single-dose vaccine might not be enough to produce detectible antibodies in PCD patients.
Nonuniform antibody titer measurements and cut-off values make it difficult to compare the included studies head-to-head. Avivi et al. and Greenberg et al. considered a cut-off of ⩾0.80 U/mL while Shah et al. and Ghandili et al. considered ⩾0.4 U/mL and ⩾33.8 BAU/mL, respectively. Terpos et al. measured neutralizing antibodies and used 30% as a cut-off for seropositivity while suggesting that a level of 50% would be associated with a clinical viral inhibition. 33 Ramasamy et al. used a cut-off of >500 IU/mL for anti S-Ab while Terao et al. used S-IgG ⩾ 0.8 U/mL and N-IgG ⩾ 1.0 U/mL cut-off index. Wu used the cut-off >50 RFUs and Nooka et al. used anti-RBD-Ab and Nab levels.
How well the vaccine response protects against COVID-19 variants is questionable. In a single-center retrospective study by Bronstein et al., 131 patients with lymphoproliferative disease were evaluated during the Omicron surge in Israel (2020). Thirty-six patients had MM. Only 14.5% of patients had severe/critical disease. Vaccination status was known for 124 patients, 53.7% had received three while 46.3% of patients had received four doses of vaccine. Of the 35 patients admitted to the hospital, 9 had MM. In the multivariate analysis, statistically significant variables included active hematological treatment and diagnosis of CLL. Of the seven patients (5.3%) who died within 30 days of diagnosis, one had MM. The overall mortality of 5% was reported to be much lower than the mortality in the early COVID-19 surge (34%). This was attributed to a better understanding of the disease and the development of vaccination and antiviral treatments. 34 In a similar study by Ennsle et al., humoral and T-cell response to wild-type (WT), Omicron, and Delta variants in MM patients receiving the third dose of BNT162b2 was explored. They concluded that patients receiving three doses of the vaccine mounted a higher serological response than those who received two doses only. However, a poor neutralizing capacity was reported for the Omicron variant. The T-cell response for Omicron and Delta variants was lower than that of the WT. However, these findings were also reported in the healthy controls. 35 Hence, evidence suggests that a milder disease is reported after vaccination in patients with lymphoproliferative disease, even with variants of concern. The response nonetheless is diminished and there is room for improvement by developing variant-specific vaccinations.
The role of COVID-19 booster vaccines in patients with hematological malignancies is crucial; studies have shown that receiving a booster after the completion of the initial vaccination series leads to seroconversion in more than 50% of the nonresponders. Greenberger et al. conducted a small (n = 49) study on patients with B-cell malignancy and reported that 55% of the patients who failed to develop anti-spike antibodies after the two doses of vaccination seroconverted after booster vaccination. 36 Similar results were reported by Ollila et al. (n = 181), 56% of the nonresponders seroconverted after a booster dose. The use of antivirals along with periodic boosters can help us protect MM patients against severe infections. 37
Variable response rates are observed in the included studies that can be due to nonstandardized seropositivity cut-off or the variability in patient characteristics. It has been previously seen that the booster influenza vaccine in PCD patients had better seropositivity rates compared to a single-dose vaccine.15,38,39 Considering our findings, it might be beneficial to administer serial booster doses to PCD patients especially those who are elderly, on anti-CD38 antibody treatment, and who have previously received multiple therapies and still have refractory disease.
For better understanding of B-cell and T-cell response to COVID-19 vaccine, there is a need for larger clinical trials. Although some studies try to answer what antibody titers against COVID-19 are clinically protective, most of these studies either exclude patients with PCD or have very few participants with PCD.40,41 The durability of response to vaccines is uncertain and the short duration of follow-up of the existing studies limits our understanding of the relation between breakthrough infections and mean time to the last vaccine. Also, with the approval of mRNA and vector-based vaccines for PCD patients, there is not enough data to suggest if one vaccine is better than the other. Looking at the literature, there is a negative association between anti-CD38 therapy and antibody titers. However, such relations with other commonly used therapeutic agents need to be explored. SARS-CoV-2 is expected to stay an endemic viral pathogen, with new variants continuously emerging. Poor control of infection because of immune compromise leads to the emergence of new variants which further complicates management.
This study was able to explore risk factors associated with poor response to vaccine and based on our findings we strongly recommend that PCD patients should be receiving booster doses. Our study has several limitations. Studies included are observational and the results are limited in terms of uniformity of patient characteristics, disease response, therapeutics used, types of vaccine, and antibody titer cut-off considered for seropositivity. We had limited data on T-cell response as only one study reported T-cell-mediated response. We were able to identify poor response in PCD patients. However, we were not able to conclude how many boosters are needed to achieve the desired protection from COVID-19 in MM patients.
