Abstract
The objective of this manuscript was to review and evaluate the efficacy and safety data of Dengvaxia for the treatment of severe secondary dengue infection. Dengvaxia is the brand name for chimeric yellow fever-dengue-tetravalent dengue vaccine (CYD-TDV). A literature search through PubMed was conducted using the keywords ‘dengue vaccine’, ‘Dengvaxia’, ‘efficacy’ or ‘safety’. Trials were selected if they appropriately assessed vaccine efficacy or were related to the vaccine approval process for CYD-TDV. Findings from this review underline the evolution of vaccine efficacy against seroprevalence, serotypes, and various ages. There are currently no preventive measures or antiviral treatments for dengue; CYD-TDV is the first vaccine to receive US Food and Drug Administration approval. Protective responses seen with the complete administration of CYD-TDV can become a standardized tool as part of a world vaccination program.
Keywords
Introduction
The dengue virus (DENV) is a type of mosquito-borne flavivirus transmitted from the bite of an infected female belonging to the Aedes genus. A select number of species serve as vectors for dengue transmission, however, the Aedes aegypti species is most responsible for viral circulation. Four serotypes (DENV-1, DENV-2, DENV-3, and DENV-4) have been identified. 1
Alongside vector-control methodologies, a recently approved vaccine for dengue immunizes against all four serotypes. Dengvaxia is the brand name for chimeric yellow fever-dengue-tetravalent dengue vaccine (CYD-TDV), manufactured by Sanofi Pasteur (Paris, France), and is the first and only of its kind that has been licensed for use in 19 countries, but is only available in 10. 2 Several phase II and phase III studies have led to particular vaccination limitations whereby CYD-TDV is only administered to people with a previous dengue infection and living in endemic areas. Newly evaluated data from secondary analyses reveal the relationship between vaccine-induced protection and baseline serostatus.
Pharmacology
Mechanism of action
CYD-TDV is a live vaccine that, through the use of recombinant DNA technology, contains the structural proteins Thailand PUO-359/TVP-1140 (type 1), Thailand PUO-218 (type 2), Thailand PaH881/88 (type 3), and Indonesia 1228 (TVP-980) (type 4) that are embedded in a yellow fever RNA polymerase backbone. 3 The premembrane and envelope proteins that are normally found within the 17D strain of yellow fever polypeptide backbone are replaced with four uniquely structured dengue wildtype serotype genes, commonly expressed as DENV-1, DENV-2, DENV-3, and DENV-4. While the exact mode of action is unknown, an immune-mediated response occurs as a result of the neutralizing antibody titers that are formed against each viral strain component in the vaccine. As with any live, attenuated vaccine, the risk of experiencing an aggravated immune response is inevitable and often related to the antibody-dependent enhancement seen with the introduction of these viral antigens. Serotype-specific antibodies formed and possibly maintained during the first exposure encourage viral binding and pathogenic destruction via the recruitment of white blood cells. Neutralizing dengue in this newly formed genetic composition will require the overcoming of the dangerous aspects of cytotoxicity when passive immunity agents such as CYD-TDV are used to target all four viral serotypes. A precautionary approach to ensure the safety of CYD-TDV’s formulation is to test for dengue serostatus prior to administration.
Host response
Based on the US Food and Drug Administration (FDA)-approved indication that CYD-TDV is reserved for individuals with an established prior infection, mechanisms involved in vaccine functionality are secondary and often two-fold. Immediate activation of an immune response is seen with an increased expression in innate receptors such as macrophages and dendritic cells. Synergistically, memory CD8 T cells formed during subsequent initial exposure and are serotype specific begin reproducing a faster, more robust response towards that same antigen found in the vaccine. Tetravalency is the unique property of CYD-TDV but inevitably it makes it a fragile and potentially life-threatening treatment opportunity for those for whom it is indicated.
When assessing secondary dengue events, few researchers have been able to pinpoint the exact causation behind autoimmunity and predictors of suboptimal response. Rothman postulated an immunodominance effect that occurs when varying frequencies of T-cell responses from a primary DENV infection alter the response to secondary infections according to serotype. 4 Furthermore, an analysis of five phase II studies reported antibody responses following each dose of CYD-TDV in children in Latin America and Southeast Asia. 5 Seropositive-at-baseline subjects produced higher levels of DENV titers against all four viral strains compared with seronegative-at-baseline subjects 28 days after receiving the third dose. Dorigatti et al. also noted that previous exposure to the yellow fever virus may have been a contributing factor in children who produced higher DENV titers due to their ability to bypass what would normally be a heightened cellular requirement in response to the yellow fever component of CYD-TDV and instead, began to respond to specific envelope proteins. 6
A comparative study performed by Henein et al. assessed the effects of antibody depletion and the CYD-TDV-induced contributions of cross-reactive serotypes in both dengue-naïve and dengue-exposed individuals with the use of DENV antigen-coated polystyrene beads. 7 In dengue-naïve children, the neutralization of DENV-4 was determined by particular DENV-4 antibodies, whereas neutralization of DENV1–3 was determined by a mixture of both type-specific and cross-reactive antibodies, indicating that responses used to establish vaccine efficacy come with a certain degree of limitations. In dengue-exposed children, higher levels of neutralizing antibodies developed for all four serotypes with the majority being cross-reactive antibodies. 7
Together, these findings suggest that although mild cross-reactivity occurs in the presence of DENV neutralizing antibodies, responses to all serotypes are not equivalent and vaccine efficacy varies based on initial serostatus and the evolution of specific cross-reactive components.
Pharmacokinetics
No pharmacokinetic studies on CYD-TDV have been performed.
Indications, dosing, and administration
The vaccine presents as a suspension. Single dose vials of a sodium chloride solvent and antigen powder formulation are reconstituted before use. The complete vaccination schedule consists of three 0.5 ml subcutaneous injections, each separated by 6 months.
Data sources
We performed a literature search using MedLine via PubMed and Embase to identify clinical trials that were relevant to the drug approval, safety, and effectiveness of CYD-TDV. The search string consisted of ‘dengue vaccine’ [All fields] OR ‘Dengvaxia’[All fields] AND (Efficacy) AND (Safety). Filter restrictions were limited to humans, English, clinical trial, and a publication date within 10 years.
Clinical trials
Articles were selected if they met the search criteria listed above and demonstrated novel clinical data in support of CYD-TDV’s efficacy and long-term safety. The literature search yielded seven articles, of which one phase II and two phase III trials were included to evaluate the efficacy of the vaccine in children, and one observational study and one phase II trial were included to determine the safety of vaccine administration relative to immunogenetic factors such as serostatus (Table 1).
Overview of eligible Dengvaxia (CYD-TDV) clinical trials.
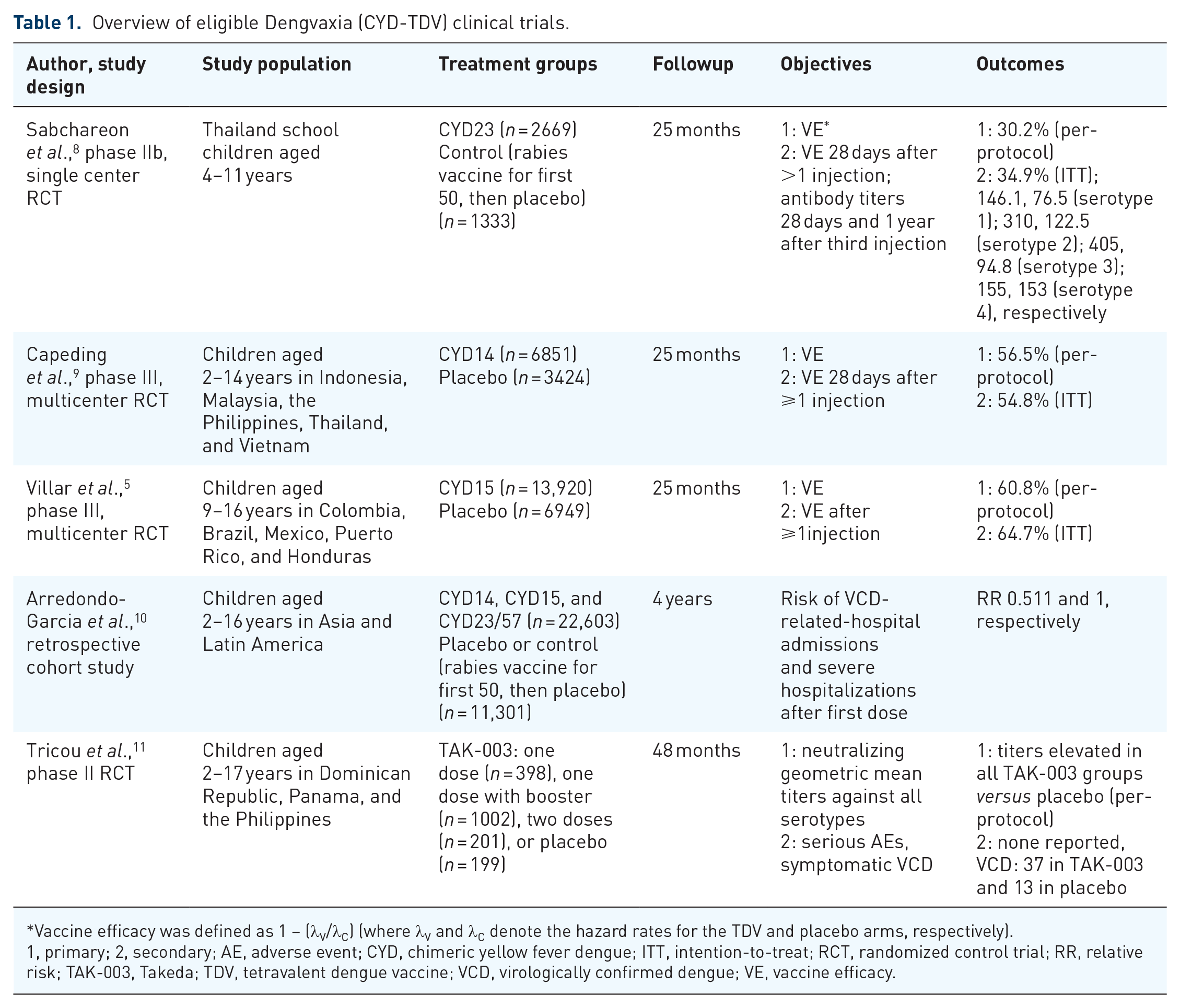
Vaccine efficacy was defined as 1 – (λV/λC) (where λV and λC denote the hazard rates for the TDV and placebo arms, respectively).
1, primary; 2, secondary; AE, adverse event; CYD, chimeric yellow fever dengue; ITT, intention-to-treat; RCT, randomized control trial; RR, relative risk; TAK-003, Takeda; TDV, tetravalent dengue vaccine; VCD, virologically confirmed dengue; VE, vaccine efficacy.
Results
Vaccine efficacy against virologically confirmed dengue (VCD) cases was examined by Sabchareon et al. 8 in a phase IIb, proof-of-concept, randomized controlled trial of 4002 children living in Thailand. A reactogenicity subset was analyzed to determine vaccine-related adverse events and an immunogenicity subset for correlating serotype-specific antibody titer production to the occurrence of dengue. There were no significant differences in baseline characteristics between the two treatment arms. Mean age was 8.2 years, and 48% (per-protocol analysis) and 44.5% (full analysis for immunogenicity) were male. Of the 134 VCD cases reported 4 weeks following the last dose of the vaccine, 77 cases occurred in total (45 cases during 2522 person-years at risk in the vaccine group, 32 cases during 1251 person-years at risk in the control group). Interestingly, children with serotype two accounted for the majority of VCD cases after at least one injection, >28 days after two injections, and >28 days after three injections in the vaccine group [52 of 76 (68%), 44 of 61 (72%), 31 of 45 (69%), respectively] and placebo group [27 of 58 (46%), 22 of 47 (47%), 17 of 32 (53%), respectively]. Vaccine efficacy was at its lowest against DENV-2 after the third injection [9.2% (95% confidence interval (CI) −75.0 to 51.3)], producing an overall diminished outcome in determining vaccine efficacy [30.2% (95% CI −13.4 to 56.6)] against all serotypes in which authors did not meet their primary objective with a threshold of 70%. Vaccine-induced protection against VCD was noted against DENV-1, DENV-3, and DENV-4 serotypes. There were no statistically significant differences in duration of fever, hospital admissions due to VCD, and episodes with any hemorrhagic signs (i.e. spontaneous bleeding and plasma leakage) between the vaccine and placebo group. In addition, vaccine-related serious adverse events were examined and authors found no worsening signs of dengue progression in more than 2600 vaccinated children. 8
Rather than explicitly reviewing vaccine efficacy, Capeding et al. examined levels of neutralizing antibodies with age-stratified immunogenicity endpoints. 9 Findings illustrated a proportional association between age and the number of children that express higher levels of neutralizing antibodies [2–5 years: 348 of 678 (51%); 6–11 years: 507 of 706 (72%); 12–14 years: 485 of 599 (81%)], implying that older children have the potential to exhibit a stronger defense against viral antigens introduced by the vaccine when compared with children of younger ages. Indeed, vaccine efficacy was higher in older cohorts than in younger cohorts. In addition, country-stratified data for the primary objective revealed that overall vaccine efficacy was consistent across all five countries; however ranges varied from 51% (Vietnam) to 79% (Malaysia). Although children living in Malaysia and Indonesia were given a 2-month extension during a 6-month enrollment period to be allocated in the reactogenicity and immunogenicity assessments as part of the secondary objectives, skepticism regarding validation of the primary objective for establishing true vaccine efficacy lingers. 9 While the incidence of hemorrhagic fever was noted to be an insignificant outcome in the Sabchareon et al. 8 study, Capeding et al. 9 reported 28 total VCD cases of hemorrhagic fever (8 cases in vaccine group, 20 in placebo group) as defined by the 1997 World Health Organization (WHO) criteria. Higher vaccine efficacy was established against hemorrhagic fever in the per-protocol analysis than in the intention-to-treat analysis [88.5% (95% CI 58.2–97.9), 80.0% (95% CI 52.7–92.4), respectively], meaning that any conclusions surrounding the possibility for mortality benefit seen with CYD-TDV support a full and complete three-dose vaccination schedule for children at risk of secondary dengue infection. Reductions in the number of hospital admissions and median duration of admission were seen in the vaccine group and proved to be clinically significant sound. Participants who underwent randomization to receive CYD14 had a 82% less risk of developing plasma leakage and a 71% less risk of developing thrombocytopenia compared with those who received a placebo. The most predominant vaccine-related adverse events included systemic reaction (57% in vaccine group, 55% in placebo group) and injection-site reactions (47% in vaccine group, 43% in placebo group). After more than 28 days following the third injection, serotype-specific vaccine efficacy was strongest in cases against serotypes 3 and 4 (78.4%, 75.3%, respectively). 9 Compared with the Sabchareon et al. trial, reports of vaccine efficacy against serotype 2 were greater in this trial (35% versus −0.3%, respectively), but still considered to be the lowest of all serotypes according to Capeding et al. 9 (35.0% in serotype 2 versus 50% in serotype 1, 78% in serotype 3, and 75% in serotype 4).8,9 Authors hypothesized that the differences found in vaccine efficacy for DENV-2 between these two studies were a result of greater serotype distribution data as well as the lower overall seroprevalence among five countries compared with just one.8,9 A 5-year active follow up to assess the safety profile is still ongoing.
Both serotype-dependent and dose-to-dose vaccine efficacy were studied by Villar et al. in a larger, multicenter, randomized controlled trial of 20,869 children. 5 Subjects were stratified based on country and age (9–11 years or 12–16 years). No observable differences were noted among baseline characteristics between both groups. More than 95% of all subjects received three injections. In the per-protocol analysis, 176 cases of VCD in the vaccine group and 221 in the control group were reported, for an overall vaccine efficacy of 60.8% (95% CI 52.0–68.0). Between-dose analyses of vaccine efficacy proved a slightly weakened effect between 28 days after receiving the second injection and receiving the third injection as opposed to 28 days after receiving the first injection and receiving the second injection (64.5%, 74.3%, respectively), per the supplementary index. As with previous trials, vaccine efficacy was lowest against serotype 2 [42.3% (95% CI 14.0–61.1)]. Importantly, authors revealed a near double vaccine efficacy among children that were seropositive compared with children that were seronegative at baseline (83.7%, 43.2%, respectively). Four in the vaccine group and one in the placebo group vaccine-related serious adverse events were documented. In seropositive-at-baseline children, the greatest positive change in geometric mean titers between baseline and 28 days after the second injection occurred against serotype 2, whereas the least positive change occurred against serotype 4. 5
Major differences between these efficacy trials include criteria for febrile episodes, which investigators of the first trial defined as two readings of a temperature of 37.5°C at least 4 h apart as opposed to 38.0°C on >2 consecutive days established thereafter, the underestimation of sample sizes needed to meet statistical power, differences in polymerase chain reaction (PCR) assays used leading to varying degrees of specificity and sensitivity, and the frequencies of antibody titer assessments.5,8,9
Participants from these three efficacy trials were later observed by Arredondo-Garcia et al. in a 4-year, retrospective study to potentiate the relationship between VCD-induced hospital admissions and severe hospitalizations in vaccinated children and unvaccinated children aged 2–16 years. 10 Immunogenicity subset group data were also pooled to establish any influence of baseline serostatus on safety outcomes. Objective criteria were defined based on years following the first CYD-TDV injection, individual age groups, and collective age groups less than as well as greater than or equal to 9 years. Investigators assessed the probability of hospitalizations and clinically severe cases via relative risk (RR), both individually and collectively. In children of all ages who received more than one dose of the dengue vaccine or placebo/control, the cumulative RR of both hospital admissions and severe hospitalizations was 51.1% (95% CI 0.42–0.62), regardless of serotype, indicating that children with the CYD-TDV were 50.1% less likely to suffer from a hospital experience. Despite this overall effect, differences in hospital admissions were noted among age groups followed in years 1–4. 10
In the CYD14 and CYD23/57 extension trial, 145 and 92 patients in the vaccine group and placebo group were hospitalized after one dose, respectively (2–5 years: 66 and 28; 6–8 years: 79 and 64). Data from the CYD15 trial were only available for ages 9–16 years, as Villar et al. targeted populations specific to vaccine age-related restrictions. 5 In children aged 9–16 years, 88 and 136 patients in the vaccine group and placebo group were hospitalized after 1 dose, respectively, with a RR of 0.324 (95% CI 0.24–0.43).
Not only were seronegative pediatric populations younger than 9 years of age likely to experience an increased risk of VCD-related hospitalization, including clinically severe cases [RR 1.707 (95 CI 0.53–7.19)], but any child who received CYD-TDV without having a previous dengue infection had a 32.7% increased risk of hospitalization or clinically severe symptoms compared with those who received a placebo [RR 1.327 (95% CI 0.56–3.48), respectively]. In seropositive children of any age, the RR was just 0.292 (95% CI 0.14–0.58) for all participants in all three clinical trials.
These statistical evaluations demonstrate that the cumulative hospitalization risk for vaccinated children, regardless of age or serostatus, is reduced when compared with unvaccinated children. Safety analyses for postmarketed data are continuing to be monitored for a period of 6 years following the first administration of the dengue vaccine, which can provide additional follow-up data points in favor of CYD-TDV’s administration and serious concern in children not previously exposed to dengue.
Unlike the clinical trials mentioned thus far, Tricou et al. studied the tetravalent dengue vaccine, Takeda (TAK-003), which is scientifically designed with a DENV-2 backbone rather than the yellow fever virus, in 1794 patients living in dengue-endemic areas. 11 Healthy children aged 2–17 years were subject to a 1:2:5:1 randomization to receive either TAK-003 as one dose, one dose with a booster, or two doses, or a placebo, and were stratified by age. Among them, randomly selected participants were included in an immunogenicity subset group in order to assess both safety and vaccine efficacy. Baseline characteristics were similar across all age groups and were not noted to be significantly different. Average mean age for the safety analysis set was 7.2 years and the per-protocol, immunogenicity set was 8.1 years. The most commonly enrolled age group in the immunogenicity subset was 6–11 years with 76 of 171 (44%) receiving TAK-003 one primary dose, 76 of 174 (44%) receiving one primary dose plus a booster, 34 of 83 (41%) receiving TAK-003 two-dose series, and 34 or 81 (42%) receiving placebo. Viral-specific serotypes against all four strains of dengue (DENV 1–4) were not identified in seropositive children. Around 45% of all participants were seronegative. 11
Geometric mean titers in seropositive-at-baseline children who received two primary doses of TAK-003 were not significantly less than any other treatment group. Contrastingly, geometric mean titers against DENV-1, DENV-3, and DENV-4 in seronegative-at-baseline children who received either one primary dose plus a booster or two primary doses of TAK-003 were higher than in children who received a single vaccine dose at month 48 (DENV-1: 271 in one primary dose plus booster group, 175 in two-dose group, 103 in single-dose group; DENV-3: 74, 67, 37; DENV-4: 66, 58, 38). The primary endpoints for vaccine-induced antibody responses against DENV-2 were of smaller-scaled margins in all treatment arms, regardless of baseline serostatus. Around 2% of TAK-003-treated participants experienced VCD compared with 7% in placebo-treated participants [RR 0.35 (95% CI 0.19–0.65)]. Vaccine-related serious adverse events were not observed. 11
Based on the results examining the relevance that serostatus inflicts on vaccine response, Tricou et al. 11 proved that antibody titers remain elevated in all TAK-003-treated groups, regardless of dosing regimens, of both seronegative and seropositive children versus placebo recipients at 48 months. As opposed to the DENV-2 backbone vaccine Takeda, the typical vaccination schedule with CYD-TDV includes several, carefully spaced out administrations over the course of 12 months in children with a prior exposure to dengue. Therefore, the diminished safety concerns investigated in this phase II clinical trial propel vaccine use with Takeda in dengue-naïve and dengue-exposed children. Speculation surrounding vaccine efficacy and reduced VCD hospitalization burden may be attributed to the biological engineering of each vaccine’s backbone.
Relevance to patient care and clinical practice
The development of a sustained, long-lasting immunity as well as adequate protection against the majority of dengue viral serotypes is emphasized through the antibody-dependent enhancement reaction, which is typically more pronounced in children who are dengue-positive who have received all three doses. Although the FDA recommendation to complete CYD-TDV as a three-dose series is paired with scientific data, several concerns arise that beg the question: can two doses be used instead of three?
A comparative investigation of the viral titers produced by incomplete and complete immunization in both flavivirus-naïve and flavivirus-experienced children may push healthcare providers in that direction, so long as the transmission rates are comparable if not more insufficient than the full vaccine schedule. In addition, the process of viral reversion against all four serotypes must be considered and monitored, especially DENV-2, which was proven to be the most prevalent and yet the least impacted by CYD-TDV in phase II and phase III clinical trials. Perhaps the cellular events that drive children to reach 90% or 100% immunogenicity against all four serotypes are comparable in a two-dose series.
As is evident by the number of children with a high baseline seropositive status in these trials, special emphasis is and continues to be placed on the astonishing and ever-growing burden of dengue. Although less prominent outbreaks have occurred in the USA, in territories such as Puerto Rico and the Virgin Islands, healthcare providers have the opportunity to provide further education regarding safe practices for those traveling to these countries as well as other dengue-prevalent countries in Central America and Southeast Asia. In hindsight, this also opens doors to making CYD-TDV more accessible.
Medically controlled strategies for populations at high-risk of dengue infection are a challenge due to limited priorities and financial support. Enhanced methods for ensuring the data used to evaluate and support the challenges seen with CYD-TDV, such as adequate vaccine efficacy with a satisfactory compromising safety profile, could provide benefit–risk assessments for both seropositive and seronegative children independent of vaccine indication. Examples of such methods are outlined in a review article by Wichmann et al. and entail systematic documentation of vaccination schedules, before and after evaluations of country-specific populations, post-vaccine virological surveillance of circulating genotypes, etc. 12 Authors stated the inevitable limitations of resources for designing such studies in order to accommodate these approaches. The capacity to detect dengue and reinforce methodological attempts for continuous monitoring, while encouraged, comes with an incredible forefront of planning. 12
Population surveillance carried out by the WHO led to the 2017 caution advisory from the manufacturer explaining the serious risk and worsened outcomes involved when CYD-TDV is administered to seronegative children. 13 With that being said, methods to establish baseline serostatus, such as rapid serological testing, would be extremely desirable, not only in terms of patient safety, healthcare resources, and eliminating indirect and direct medical costs, but by creating public confidence in CYD-TDV and ultimately reducing disease burden.
Conclusion
The available data derived from phase II and phase III clinical trials support the use of CYD-TDV in children with a previous dengue infection. Collectively, the evidence also suggests an enhanced immune effect in children of older ages, children suffering from dengue-induced hospital admissions and hemorrhagic fever, and children who complete the three-dose series as measured by the per-protocol analysis. There was little speculation regarding a cause and effect association between age and serotype-related vaccine protection in clinical trials designed to examine the effects of CYD-TDV on serostatus. The protective responses seen with the complete administration of CYD-TDV can become a standardized tool not only for children living in dengue-endemic areas but for those that travel to those areas as well. However, more definitive information about the effects on high-risk populations will be needed to justify the overall robustness of CYD-TDV in these individuals.
