Abstract
Dengue is a mosquito-borne flaviviral disease that is endemic to tropical and subtropical regions, affecting hundreds of millions of people worldwide. Although it was once considered a neglected disease, the incidence and mortality rates of dengue have surged over the past decade, in part due to the expanding distribution of the Aedes spp. vector facilitated by changing climatic factors. While most infections are asymptomatic or cause mild flu-like symptoms, some cases can develop into severe forms, leading to serious complications. The burden of the disease is gradually shifting from primarily affecting children, whose immune systems are immature, to increasingly impacting the older population, who typically experience waning immune responsiveness and comorbidities. With no specific treatment available, the development of a prophylactic vaccine is crucial for long-term control and prevention. School-age children are the primary target group for immunization programs of the two recently licensed dengue vaccines. However, there is limited information on the efficacy of either vaccine among the elderly or of two further immunogenic preparations currently undergoing clinical trials. This review gives an update on dengue vaccine implementation and provides recommendations for the vaccination of persons aged 60 years and above.
Plain language summary
Dengue is a debilitating and often life-threatening disease caused by a virus that is transmitted between people by the bite of an infectious Aedes mosquito. With the global rise in dengue cases, especially in regions previously considered non-endemic, the elderly are increasingly vulnerable due to such factors as weakened immune systems and comorbidities. Recommendations for implementing a vaccine in the elderly population focus on addressing the unique challenges posed by this demographic group, particularly as they are at higher risk for severe dengue infections.
In order to roll out the vaccine effectively, it is crucial to conduct thorough seroprevalence studies to identify individuals who have previously been exposed to the virus. This information can help tailor vaccination strategies, ensuring that those most at risk receive the vaccine while minimizing the potential for adverse reactions in dengue-naïve individuals.
Moreover, education and awareness campaigns are essential to improve vaccine acceptance among the elderly and their caregivers. People may be worried about the safety and efficacy of vaccines, especially following previous controversies. Providing transparent information about the advantages of vaccination, as well as details on how it can help prevent serious complications from dengue, can reduce anxiety and promote acceptance of the vaccine.
In summary, a comprehensive approach that includes targeted vaccination strategies using either of the two recently regulatory authority-approved vaccines, public education programs, in combination with concurrent vector control measures, is vital for protecting the elderly population from dengue. By addressing these factors, we can improve health outcomes and reduce the burden of dengue in this vulnerable group.
Introduction
Dengue virus (DENV) has long posed a public health threat, particularly in tropical regions such as Southeast Asia, South Asia, and South America. 1 Over the past 5 years, outbreaks of infection have become more frequent, and the geographical distribution of DENV has gradually expanded to countries that hitherto were rarely affected. 2 In 2023, the World Health Organization (WHO) reported a record high global incidence of dengue, exceeding 5 million, with many more suspected cases going unreported. 3 During this period, outbreaks occurred in European countries that had not experienced dengue in over a decade, including Italy, France, and Spain. 4 In the first 4 months of 2024 alone, the WHO reported 7.6 million dengue cases, already surpassing 2023 numbers, with over 3000 deaths. 3 This expansion is due to various anthropogenic factors, including globalization, urbanization, and population growth, with the accelerating rate of climate change considered as the most significant cause. 5 For DENV, the most dangerous complications of clinical infection include dengue hemorrhagic fever (DHF) and dengue shock syndrome, which can lead to multiple organ failure, severe bleeding, and ultimately death. 6 Global mortality from dengue has increased fivefold since 2000, with the annual death toll in 2023 surpassing 5000 people. 2 These alarming statistics for both morbidity and mortality highlight the critical need for vaccination among the most affected populations.
The dengue vaccines that are now or soon to become commercially available are discussed later. The stated recommendations for each of these vaccines indicate that its primary target group is young people, to whom it should be principally limited. 7 However, this does not serve the older population, a vulnerable group that suffers the most in terms of dengue-associated hospitalization rates. 8 While the dengue-related death rate among the geriatric population varies between countries, the number of deaths in older adults is significantly higher than in younger age groups and general populations. Studies in East Asia and the Americas indicate that the death rate in older adults can be up to nine times higher than in other age groups. 9 Therefore, it is crucial to focus on this demographic to reduce the burden caused by DENV infection. However, data on this vulnerable target group are still scarce, which is critically required to inform vaccination recommendations for persons aged 60 years and above.
Virus characteristics, infection, and clinical manifestations
DENV is a single-stranded, positive-sense RNA virus from the Flaviviridae family, 10 primarily transmitted by Aedes mosquitoes. 11 There are four distinct serotypes (DENV-1, -2, -3, and -4), 12 each capable of causing dengue in individuals of any age. 13 The circulation of these serotypes varies across different global regions. 14 Often, more than one serotype co-circulates in the same location. The DENV envelope protein (E) is notable as a major target for vaccine development due to its role in viral replication and immune system evasion, 15 while non-structural protein 1 (NS1) is linked to severe dengue complications, 16 and detection of which is used in diagnostic tests. 17
The symptoms of DENV infection range from asymptomatic to severe disease. According to the WHO 2009 Dengue Classification, dengue can be classified into three groups: dengue (without complications); dengue with warning signs; and severe dengue. 18 Based on disease progression, DHF can be classified into three stages: febrile; critical; and recovery phases. 19 The initial febrile phase is marked by high fever, headache, nausea, rash, muscle/joint pain (indistinguishable from many other viral infections), and mild bleeding. 20 As the fever subsides, the critical phase begins with severe abdominal pain, restlessness, enlarged liver, vomiting, and decreased platelet count with increased hematocrit.21 –23 In severe cases, plasma leakage, respiratory distress, and organ impairment can occur.24,25 The final recovery phase involves gradual improvement in symptoms, increased appetite, and normalization of laboratory values. 26
This clinical presentation only reflects typical cases, and some patients may not experience all stages clearly. 27 Many infections are misdiagnosed due to the commonality of symptoms with other infectious diseases and the lack of availability of diagnostic tests, leading to delayed diagnosis, treatment, and management of complications. 28 This is especially concerning for older adults, whose symptoms are often atypical compared to clinical manifestations in younger people. 29
Difficulty of dengue diagnosis in the elderly
Elderly patients often present with atypical symptoms, such as a lower-grade fever or fewer classic signs. 30 This can be attributed to age-related declines in both adaptive and cell-mediated immunity, resulting in a lower response to infection due to reduced cytokine secretion. 31 Some individuals may only present with fever, without other typical symptoms, making diagnosis more challenging. The nonspecific nature of an isolated fever means that it can be misdiagnosed due to multiple infectious and non-infectious diseases. The most likely differential diagnoses include other mosquito-borne viral infections such as chikungunya and Zika. 32 In older adults with multiple comorbidities, differential diagnosis can be particularly difficult. When experiencing severe disease, elderly individuals are more likely to develop fatal conditions due to multiple underlying ailments and may be misdiagnosed with more commonly encountered health conditions. Dengue warning signs, such as abdominal pain and vomiting, may be confused with surgical conditions, 33 gastrointestinal bleeding may be misdiagnosed as cirrhosis with esophageal varices, and altered mental status due to dengue-associated encephalopathy may be mistaken for a stroke. 34
Elderly individuals are more likely to develop secondary DENV infections and experience longer hospital stays compared to younger patients. 35 This is primarily due to the age-related decline in their immune system over time, with impaired functions and lower responses from T cells and cytokines, plus the increased risk of complications associated with multiple comorbidities. Thus, the geriatric population faces a higher risk of severe dengue, which raises the mortality rate.
Changing epidemiology and why the older population is at risk
Dengue in elderly people is significantly understudied. While the incidence is not as high as in other populations, the mortality rate is remarkably high. 36 In a retrospective cohort study in Thailand from 2013 to 2018, patients aged 60 years and above were at a higher risk of developing DHF than adults under 60 years old (40.69% vs 30.71%). 37 A study conducted in Bangladesh comparing the case fatality rate (CFR) of dengue across different age groups found that the CFR generally increases with age. The highest CFR, over 6%, was observed in the “over 80” years age group, indicating that elderly individuals are at a significantly higher risk of fatal outcomes from dengue. 8
Historically, dengue was primarily known to affect children. 38 Most global infections occur in endemic areas, which require two factors: a large population or steady movement of people into the area to maintain a pool of individuals susceptible to infection, and the long-term presence of vector mosquitoes carrying the virus. In these regions, most elderly individuals are likely to have been infected at least once. 39 For instance, in Malaysia, DENV seroprevalence increases with age, from 22.7% in children under 5 years old to 94.9% in those over 60 years of age. 40 This indicates that children are more susceptible to dengue. However, recent reports show a shift in the incidence rate, with an increasing number of cases in the adult population, particularly among the elderly. 38 In Thailand, the mean age of DHF cases rose significantly from 8.1 years in 1981 to 24.3 years in 2017. 41 Similarly, in Singapore, the age-standardized seroprevalence of anti-dengue IgG antibodies in individuals aged 18–74 decreased from 63.1% in 2004 to 54.4% in 2010. 42 This indicates an increasing susceptibility to DENV infection in the adult population.
In regions historically less affected by dengue, the escalating incidence among adults can be attributed to several factors. Climate change has expanded the range of Aedes spp. mosquitoes, the primary vectors of dengue transmission, by creating warmer temperatures and altering rainfall patterns. 43 Rapid urbanization has also increased exposure to these habitually peridomestic mosquitoes, especially for adults who have moved from rural areas with lower dengue prevalence. 44 By contrast, for endemic areas, one of the contributing factors is the success of residential vector control programs, which can lower the seroprevalence in the adult population. This shift in the acquisition of DENV infection from around the home environment to more external habitats of the Aedes vector explains why dengue occurs more frequently as individuals age. 29
Antibody-dependent enhancement — A challenge for vaccine development
Primary DENV infection is often self-resolving, and lifelong immunity against the infecting serotype may be achieved following exposure. In some individuals, however, homologous reinfection may still result in symptomatic dengue. 45 Following a person’s first DENV infection with any of the four serotypes, as cross-protection is short-lived, there is usually limited protection against heterotypic infection by any of the three other serotypes. 46
Severe dengue is most likely during a secondary infection with a different serotype to that which caused a person’s primary infection due to the phenomenon of antibody-dependent enhancement (ADE). 47 ADE occurs when antibodies from a previous infection recognize but do not neutralize DENV, allowing virus particles to enter cells and abrogate the immune response, leading to severe manifestations like DHF. Primary DENV infection triggers the innate immune response, followed by the adaptive immune response whereby T cells activate B cells to produce specific antibodies, starting with IgM and later IgG, which neutralizes the virus. Some B cells become memory cells, providing long-term immunity to the initial DENV serotype. 48 However, during a secondary infection with a different serotype, cross-reactive antibodies from the first infection can enhance the new heterotypic infection, leading to increased viral replication and severe illness. 49 This severity can be attributed to elevated levels of pro-inflammatory cytokines and vascular leakage. 50 Studies have shown that longer intervals between successive DENV infections can correlate with significantly higher disease severity, posing a relatively greater risk to older adults, particularly due to ADE during secondary or subsequent infections.51,52 ADE even occurs reciprocally between infections with DENV and the closely related and often co-circulating flavivirus ZIKV that causes Zika. 53
Previous studies have shown that age correlates with the risk of severe dengue, which increases as age advances. 54 While ADE primarily concerns children and young adults, older adults are more susceptible to severe dengue due to the prevalence of multiple chronic diseases. 55 For elderly individuals with comorbidities, these chronic conditions further elevate the risk of developing severe disease. Diabetes, in particular, increases the risk of severe dengue more than fourfold. 54 This heightened risk can be attributed to changes in the immune system and altered vascular permeability, 56 which can lead to secondary bacterial co-infections, 57 exacerbating the condition and increasing the risk of death. However, further studies are necessary to determine whether immune system dysfunction in diabetes directly causes ADE or not, with potential mechanisms including Fcγ-receptor interaction, direct endothelial damage, complement activation, and unregulated production of cytokines, among others. In addition to diabetes, multiple chronic diseases in the elderly, such as renal diseases, 58 cardiac disorders, 59 and asthma, 59 can serve as risk factors for severe dengue and organ failure.
As vaccine development is predicated on mimicking a protective immune response, the risk of inadvertently triggering ADE presents a significant challenge to effective immunization, especially for vaccines targeting dengue-naïve individuals. The antibodies produced by vaccines can cross-react during secondary infection, potentially causing ADE and exacerbating the disease. Currently, no vaccine, either licensed or prototype, can produce antibodies that neutralize all four DENV serotypes equally well. An unbalanced immune response to different serotypes can lead to partial immunity, increasing the risk of ADE. 60 The ideal dengue vaccine should be both safe and effective for all community members, be they naïve or previously exposed individuals. However, how each person responds is not straightforward to predict due to their exposure history, which often might not be fully known, complicating the development of a universally effective vaccine. 61
What vaccines are currently available?
The first reported evaluation of a dengue candidate vaccine was in 1930. 62 Since then, many different formulations have been developed, several tested in animal models, and some entering clinical trials. 63 In the last decade, two vaccines have been approved for public implementation, Dengvaxia® (CYD-TDV) and Qdenga® (TAK-003), both of which are now licensed in many jurisdictions.64,65 A third formulation, TV003/TV005, is under evaluation in phase III clinical trials. 66 All these vaccines are recombinant, live-attenuated, and tetravalent, aiming to provide immunity against all four DENV serotypes and thereby reduce the risk of ADE. 67 Compared to other vaccine types, live-attenuated formulations can elicit a stronger immune response because exposure via immunization closely mimics the natural acquisition of infection. This helps generate both humoral and cellular immunity, eliciting robust and long-lasting protection and reducing the need for booster doses. 68 Given the complexity of immunity to dengue, however, it is only to be expected that any new vaccine raises apprehension about viral toxicity, an increased risk of severe disease, and a higher chance of hospitalization for seronegative individuals. 69 In addition, there are safety concerns for specific populations, including pregnant women, individuals with weakened immune systems, and the elderly. 64 Yet, for all three of these vaccines, few major side effects have been documented in the targeted population. 70 For example, in a report on the clinical safety of the TAK-003 vaccine only 2.72 serious adverse events per 100 person-years were observed in TAK-003 recipients, compared to 3.28 in the placebo group. The most common side effects include headache (30%–50%) and mild discomfort at the injection site (43%), along with fever, fatigue, and muscle pain. 71 These symptoms typically resolve within 1–3 days. Although each vaccine is recommended for different age groups, none is indicated for elderly individuals (age ⩾60 years) due to insufficient clinical trial data, challenges addressing safety concerns, and/or low efficacy in this age group. 72
Dengvaxia (CYD-TDV), developed by Sanofi Pasteur, was the first licensed dengue vaccine, approved for public use in December 2015. This is the only vaccine approved by the FDA for use in the USA. 65 It uses the yellow fever virus YF17D (also a flavivirus) as a backbone, onto which grafted sequences encode pre-membrane (prM) and envelope (E) proteins from each of the four DENV serotypes. The vaccine employs a sequential attenuation strategy, whereby each serotype is individually weakened before being combined. Vaccination is by administration of three doses, 6 months apart. Dengvaxia shows promising efficacy against DENV-3 (71.8%) and DENV-4 (76.9%), but lower efficacy against DENV-1 (50.0%) and DENV-2 (43.0%). 7 Its modest efficacy can be explained by its composition lacking non-structural or capsid proteins, which might not induce a sufficient T-cell response. 73
While Dengvaxia is recommended for individuals between 9 and 45 years of age who have previously been infected with DENV, its efficacy varies significantly based on a person’s serostatus. The overall efficacy for seronegative individuals is only 35.5%–43.2%, compared to 74.3%–83.7% in seropositive individuals. 74 Furthermore, Dengvaxia can increase the risk of hospitalization for dengue-naïve individuals when they first experience a natural infection after vaccination. 75
A phase III trial focusing on the pediatric population (under 18 years old) showed that in seronegative individuals, while they produced neutralizing antibodies to all four serotypes of DENV after receiving one dose of the vaccine, the only serotype-specific neutralizing antibodies were against DENV-4. The other serotypes were only neutralized by cross-reactive antibodies. 76 This explains the immunodominance of antibodies against DENV-4, which can pose a threat to dengue-naïve individuals, potentially leading to ADE, especially when infected with DENV-2. Besides a person’s serotype status and which DENV serotypes they are exposed to, the efficacy of Dengvaxia is also age-dependent. Vaccine efficacy in children of 9 years and younger is significantly lower compared to in older children, with efficacy rates of about 45% versus 65%. 77 Younger children are more likely to require hospitalization for severe dengue. This may be related to their reduced risk of exposure to DENV before vaccination. In contrast to this detailed data on juveniles, however, there is minimal information from trials involving the elderly population.
For long-term efficacy, Dengvaxia has been shown to reduce the risk of hospitalization and severe dengue for up to 6 years after the final dose. 78 In May 2024, Sanofi-Pasteur made the surprise announcement that its production of Dengvaxia is to be discontinued due to low global demand. 79 Prima facie, in view of the escalating worldwide incidence of dengue stopping manufacture appears counterintuitive, so it remains to be seen if this is a temporary measure. Without being privy to any commercial rationale for this decision, it is noted that the announcement coincided with the news of a second vaccine, Qdenga, receiving prequalification from the WHO. 80
Qdenga (TAK-003), developed by Takeda, recently completed phase III trials and was first licensed for public use in Indonesia in August 2022. The vaccine employs a single attenuation strategy involving all four serotypes. It uses DENV-2 PDK-53 as its backbone, substituting the DENV-2 backbone with the DENV-1, -3, and -4 coding sequences of prM and E genes. 64
Qdenga is recommended for individuals aged between 4 and 60 years, regardless of serostatus, which distinguishes it from Dengvaxia. It is administered in two doses, 3 months apart. Unlike CYD-TYD, TAK-003 shows very high efficacy against DENV-2 (97.7%) and lower efficacy against other serotypes, with DENV-3 being the lowest at 63%. 7
The cumulative efficacy 4.5 years after vaccination remains high in protecting against virologically confirmed dengue and hospitalization due to severe dengue. However, this efficacy varies by serotype to which an individual is exposed and whether they have had an infection, either homologous or heterologous, prior to vaccination. The weakest long-term protection is against DENV-4, and dengue-naïve individuals have lower protection (50%) compared to their seropositive counterparts (63%). 81
While the trials for the Qdenga vaccine included a larger age group than those for Dengvaxia, and it is relatively safer as it does not increase the risk of hospitalization and severe disease for seronegative individuals, there is still limited data on individuals over 60 years old.
TV003/TV005, which was developed by the US National Institute of Allergy and Infectious Diseases, is the latest dengue vaccine to undergo phase III trials. TV003 consists of rDENV-1Δ30, rDENV-2/-4Δ30, rDENV-3Δ30/31, and rDENV-4Δ30. TV005 is similar to TV003 but with a 10 times increased dosage of the DENV-2 component. These vaccines employ the same single attenuation strategy as TAK-003 but utilize a different genetic modification approach. They are derived from the Western Pacific strain of DENV-1. Similarly, Butantan-DV, licensed by the Butantan Institute in Brazil, is modeled after the TV003 vaccine. It is composed of attenuated DENV-1, -3, and -4 (rDENV-1Δ30, rDENV-3Δ30/31, and rDENV-4Δ30). Serotype 2 uses a chimeric virus that combines the genes encoding prM and E proteins from DENV-2 with an attenuated DENV-4 backbone (rDENV-2/-4Δ30[ME]). 66
The Butantan-DV version is recommended for people between the ages of 2 and 60 years. Like TAK-003, it is safe for individuals who have not previously contracted dengue. The advantage of the Butantan-DV vaccine is that it is administered in just a single dose. While its efficacy against different serotypes is still under investigation, preliminary indications are promising. In a trial conducted in Bangladesh, TV005 demonstrated the highest efficacy against DENV-2 (99%) and the lowest against DENV-1 (83%). 82 In a trial performed in Brazil, Butantan-DV showed an overall vaccine efficacy of 79.6%, 83 with specific efficacy rates of 89.5% and 69.6% against DENV-1 and DENV-2, respectively. 82 The vaccine showed higher efficacy in adults, achieving 90% efficacy in the 18–59 year group, which was more than in younger groups. This protection can last up to 3 years for all age groups from 2 years to 60 years, regardless of serostatus. The vaccine has proven to be relatively safe, with no deaths and few solicited systemic adverse events related to vaccination reported during phase III trials. 84 However, no data was available for the older population.
Why are no dengue vaccines available for the elderly?
At present, there is little data on the efficacy of available dengue vaccines in persons aged 60 years and above. In endemic areas, which are typically in low- and lower-middle-income countries, the elderly comprise a relatively modest proportion of the total population. Hence, despite the high mortality rate associated with dengue in this age group, the absolute numbers of clinical cases and deaths are still less than those reported for other age groups. 8 This has led to the older population being largely overlooked in vaccine trials and policy implementations.
There are also safety concerns when conducting trials on the elderly population. This group often has multiple underlying chronic conditions, necessitating extra caution. 85 Immunity in older individuals is diminished, characterized by decreased T-cell function and antibody production, along with a reduced overall immune response to pathogens. Furthermore, symptoms of DHF in older adults are often misdiagnosed, leading to an underestimation of the actual number of deaths caused by dengue.
Moreover, it is axiomatic that older persons have a higher likelihood of prior exposure to DENV, resulting in an elevated seropositivity rate in this population. 41 Using live-attenuated vaccines in such individuals with compromised immune systems may trigger a similar immune response to natural exposure to DENV, potentially leading to ADE and severe disease outcomes.
Notwithstanding these concerns, it is important to acknowledge the shifting epidemiology of dengue and its impact on the elderly population. 86 Current data indicate that the latest available vaccines, such as TAK-003 and TV003/TV005, are relatively safe with few adverse reactions. 69 They also show positive efficacy against multiple serotypes and can provide long-term protection. In addition, several new vaccines are being developed, including inactivated, subunit, and nucleic acid-based vaccines, 63 which may offer safer alternatives for older persons.
Heterologous prime-boost vaccination: Is it realistic?
The concept of “mix-and-match” vaccines, or heterologous prime-boost vaccination, is well established in the field of vaccinology but gained particular attention during the coronavirus disease 2019 (COVID-19) pandemic. 87 This strategy involves using different vaccines for the initial and booster doses to enhance the immune response, making it stronger and more durable. 88 The flexibility of this approach also safeguards against a supply shortage of one vaccine hampering the vaccination schedule followed within a local community, an important consideration in rural and remote areas.
Applying this strategy to dengue vaccination programs may help to address the imbalance in efficacy against different serotypes that is seen with current vaccines when used on their own. 88 For example, Dengvaxia shows higher efficacy against DENV-3 and DENV-4, while Qdenga is more effective against DENV-2. A comprehensive assessment of Butantan-DV against all four serotypes awaits further trial data.
A heterologous dengue vaccine booster might elicit stronger cellular and humoral responses, especially if the first dose does not induce a robust response. This could be particularly beneficial for the elderly, who are at high risk of severe disease due to ADE. Combining multiple doses of different vaccines may balance and enhance efficacy across all four serotypes, potentially lowering the risk of severe disease. 88 However, it is also possible that mixing and matching vaccines could increase the risk of adverse events, especially since all current dengue vaccines are live-attenuated preparations. In addition, there is a concern that the vaccines might neutralize each other, the effect of which could be mitigated by spacing out the doses.
Although each is tetravalent, some qualities of Dengvaxia and Qdenga mimic monovalent vaccines. Dengvaxia primarily induces antibodies to DENV-4, while Qdenga invokes antibodies against DENV-2. Combining these vaccines could elicit a broader range of antibodies and thereby reduce the chance of neutralization. 89 Ongoing trials are exploring various combinations of live-attenuated, DNA, and inactivated vaccines. However, preliminary results have been mixed, with some combinations not eliciting a sufficiently strong or long-lasting immune response or causing serious side effects.
For the elderly population, who have a weakened immune response and typically suffer from multiple comorbidities, these combinations become even less viable as a vaccination strategy for this group. Regardless of any theoretical rationale, in-depth laboratory and clinical research on heterologous prime-boost vaccines is an essential requirement to establish safety and efficacy before being considered a realistic alternative to overcome the limitations of current dengue vaccines.
Vaccine perceptions in the elderly population
The global acceptance rate of the dengue vaccine is high at 88.3%, 90 surpassing the acceptance or coverage rates of other prominent vaccines such as those against influenza, severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), and human papillomavirus.91 –93 Nevertheless, dengue vaccination remains a challenge in many regions. A recent study on knowledge, attitude, and practices toward dengue vaccine acceptance in Latin American and Asia-Pacific nations revealed that while most people in endemic areas recognize dengue as a dangerous infectious disease, many remain hesitant to receive the vaccine. 94 In Argentina, Brazil, Colombia, and Mexico, people are generally more willing to vaccinate compared to those in Indonesia, Malaysia, and Singapore. 94
Unfortunately, the Dengvaxia controversy in the Philippines in 2016, where many children who had not previously been infected with dengue faced severe disease risks post-vaccination, led to this vaccine being banned in the country. 95 The lasting impact of this incident on Filipino public trust in vaccines, in general, has led to widespread vaccine hesitancy, contributing to a 2019 measles outbreak and indicative of a likely low dengue vaccination rate for future implementations. 96
Most research on dengue vaccine acceptance to date has excluded the elderly as they are not the recommended population for currently available licensed vaccines. Nevertheless, examining their perceptions through the lens of the recent COVID-19 outbreak reveals interesting insights. 97 In Peru and the UK, older adults demonstrated higher acceptance rates for SARS-CoV-2 vaccines compared to younger people. 92 Furthermore, the collective success of SARS-CoV-2 vaccines in combating the COVID-19 pandemic may have increased dengue vaccine acceptance. For instance, a report from Puerto Rico showed a 10% increase in dengue vaccine intention post-COVID-19 pandemic, with the highest acceptance rates for the 50+ age group (88.3% for themselves and 93.3% for their children). 98
Vietnam, located in an endemic region that has experienced several dengue outbreaks in recent years, began implementing the Qdenga vaccine on September 20, 2024. This is now available to individuals aged 4 years and above, including the elderly. According to a report from the Vietnam Vaccine Joint Stock Company (VNVC), the country’s leading provider of high-quality vaccination services, nearly 25,000 people were vaccinated during the first week of availability. 99 Despite its high cost relative to the Vietnamese average income, plus limited publicity about the availability and benefits of vaccination, the early uptake demonstrates a promising acceptance of this vaccine, including among persons aged 60 years and above.
Studies have found that vaccine hesitancy among older adults may be related to cost, needle phobia, concerns about side effects, and fear of death, especially in rural areas. 100 For dengue specifically, factors such as previous severe infections and a higher likelihood of mosquito bites might influence decision-making. 101 Several strategies can be implemented to address these concerns, thereby overcoming barriers to dengue vaccination and protecting this vulnerable population. These include improving financial accessibility, building trust in healthcare professionals, assuring the public of the vaccine’s safety and efficacy, and educating people on the importance of vaccination in combating dengue.
Recommendations for dengue vaccine implementation in the elderly
While the three leading dengue vaccines have shown promise in certain populations (Table 1), there is a paucity of data regarding their safety and efficacy in the elderly. In recent years, several clinical trials have been conducted by the National Taiwan University Hospital to develop a dengue vaccine specifically for the older population. For instance, a phase II clinical trial of the TV005 vaccine in individuals aged 50–70 years in Taiwan was completed in 2022, with results pending publication [ClinicalTrials.gov identifier: NCT04133987]. Moreover, Takeda will launch a phase III clinical trial of the TAK-003 vaccine in 2025, targeting older adults aged 60–79 and adults aged 45–60 years [ClinicalTrials.gov identifier: NCT06579755]. The outcomes of these trials will provide crucial evidence for the implementation of dengue vaccines in this vulnerable group in the near future. As clinical trials in this age group continue, robust surveillance is essential to monitor adverse events, especially in people who have multiple underlying diseases or are in a frail condition.
Comparison of different vaccination strategies and their recommended use for elderly individuals.
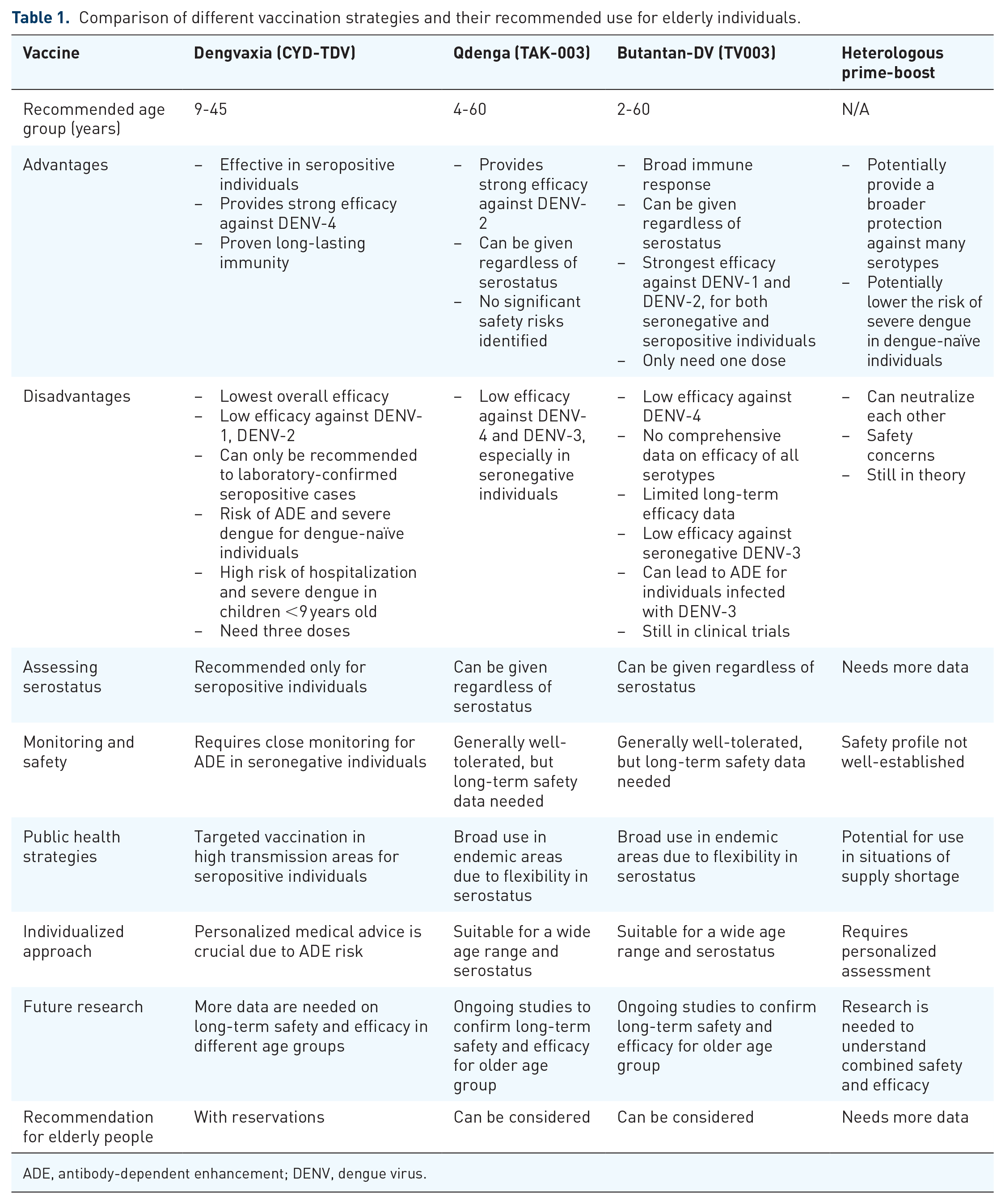
ADE, antibody-dependent enhancement; DENV, dengue virus.
Vaccine implementation plans for the elderly should be tailored to individual circumstances (Table 1), particularly in endemic areas where a significant proportion of the population is DENV seropositive and/or in which the prevalence of each serotype varies. Given that none of the current vaccines provides balanced efficacy across all four serotypes, careful assessment of individual risk factors and local DENV transmission epidemiology is essential. Preliminary indications suggest that a heterologous prime-boost vaccine strategy might lead to stronger immune responses and reduce the risk of ADE. However, it is important to await the release of a comprehensive report to confirm its safety and efficacy in the elderly.
In the meantime, traditional vector control measures remain essential for reducing the dengue burden in the elderly population. 102 Early diagnosis is crucial for timely management and prevention of severe complications. Raising awareness about dengue, increasing laboratory testing capacity, and closely monitoring warning signs can help limit misdiagnosis and ensure prompt treatment to prevent the disease from progressing to severe stages.
Conclusion
Recommendations for dengue vaccination in the elderly population are currently limited as they are not the primary target group for immunization. However, updated guidelines should emphasize the importance of protecting this vulnerable group, as they are at a higher risk for severe DENV infections. Given the increasing incidence of dengue globally, particularly in regions where the disease was previously uncommon, it is crucial to implement vaccination strategies tailored to people aged 60 years and above.
One key recommendation is to conduct serological screening to identify individuals who have previously been exposed to DENV. This is important because current vaccines are effective primarily in persons with prior DENV exposure. Vaccinating dengue-naïve individuals can increase the risk of severe dengue upon subsequent infections due to ADE. Therefore, careful screening can help to ensure that only those who are likely to benefit from the vaccine are vaccinated, thereby minimizing potential risks.
In addition, public education campaigns are essential to increase awareness and acceptance of the dengue vaccine among the elderly and their caregivers. Many individuals may have concerns about vaccine safety and efficacy, particularly in light of past controversies surrounding dengue vaccines. Clear communication about the benefits of vaccination, including its role in preventing severe outcomes from dengue, can help alleviate fears and encourage uptake.
