Abstract
Background:
Neisseria meningitidis is the main cause of bacterial meningitis in Brazil, where the main serogroups isolated are B and C; however, the serogroup W has recently emerged. LPS and type IV pili are important virulence factors that increase meningococci pathogenicity.
Methods:
The characterization of Lipopolysaccharide (LPS) and type IV pili in 19 meningococci strains of serogroup B, 21 of serogroup C, 45 of serogroup W and 28 of serogroup Y, isolated in Brazil between 2011 and 2017, was conducted using the Enzyme-linked Immunosorbent Assay (Dot- ELISA) technique and monoclonal antibodies.
Results:
We would like to emphasize the importance of characterizing relevant antigens, such as pili and LPS, the use of monoclonal antibodies to support it, and how such studies improve vaccine development and monitoring. Most of the strains studied presented L3,7,9 LPS and type IV pili; both antigens are associated with the capacity to cause invasive disease.
Conclusion:
Due to the impact of meningococcal disease, it is important to maintain and improve vaccine studies. Epitopes characterization provides data about the virulence of circulating strains. The use of monoclonal antibodies and serological techniques are relevant and support vaccine development.
Introduction
Neisseria meningitidis or meningococci (MC) is a Gram-negative, aerobic, immobile, and non-sporulating bacteria that can colonize the oropharynx of healthy individuals or provoke invasive meningococcal disease (IMD), causing meningitis and/or meningococcemia. Its virulence has been attributed to several factors, as the polysaccharide capsule, outer membrane proteins, such as pili, the lipopolysaccharide (LPS), mechanisms of horizontal gene transfer and antigenic variation.1–3
Serological typing of N. meningitidis strains by Dot-ELISA and a panel of monoclonal antibodies is well established. 4 The characterization of meningococci antigens by monoclonal antibodies uses the capsule, Porin B, Porin A and LPS to characterize the strains, respectively, in serogroup, serotype, subtype and immunotype.5,6
IMD is considered a public health problem, especially in developing countries. It is estimated that every year, there are 1,2 million new cases and 135 thousand deaths. 7 The distribution of serogroups and the impact of diseases caused by MC in the world are heterogeneous; however, the impact of IMD is greater in developing countries. Adequate surveillance has great importance in estimating the impact of IMD, outlining strategies of prevention, which is the better strategy for control, and evaluating the available diagnostic and monitoring systems.8,9 Although there are molecular tools to classify the pathogen, 10 serological techniques present lower costs, are easy to perform and reproducible, being suitable especially in low-income countries.4–6
We performed this study to verify the expression of L3,7,9 immunotype and type IV pili (T4P) among circulating strains of N. meningitidis in Brazil in recent years, because, even though they are important for IMD, being related to high pathogenic levels, they are not routinely tested.11,12 This was an important step for the study of the pathogenic potential of most prevalent Brazilian strains, serogroups B and C, 13 as well as emerging serogroups W and Y, which have recently increased in the Americas and, because of that, got our focus in this study.7,8
Materials and methods
Whole cell suspensions
Whole cell suspensions and its phenotypic characterization were performed at the Bacteriology Center of Adolfo Lutz Institute (São Paulo, Brazil). We used 113 Brazilian strains (19 of serogroup B, 21 of serogroup C, 45 of serogroup W and 28 of serogroup Y), isolated from cases of IMD, between 2011 and 2017. During this period, 12,677 cases of IMD were reported and 6354 of them were caused by the serogroups studied (the difference being caused by ‘ignored serogroups’), according to the Brazilian National Notifiable Disease Surveillance System (SINAN). 14
Strains were isolated in chocolate blood agar (5% sheep blood), followed by cultivation in Müeller–Hinton agar containing horse serum, incubated at 37°C ± 2°C, in 5% CO2 atmosphere, during 24 hours. Whole cell suspensions were prepared in PBS/azide solution, with an OD ~2.0, at 620 nm. Following that, the suspensions were inactivated in a water bath 56°C for 30 minutes and maintained under refrigerator temperature. Serogrouping was performed by slide agglutination with polyclonal sera and serosubtyping was performed by Dot-blotting with monoclonal antibodies, serological techniques routinely performed at the Bacteriology Center. 15 Table 1 shows the phenotypic characterization of the strains used.
Serogrouping, serotyping and subtyping characterization of N. meningitidis strains used in this study.

Monoclonal antibodies (mAbs)
We characterized LPS using 4BE12-C10 mAb (L3,7,9 immunotype), 16 disposable at the Neisseria.org database, 17 and pili using 7BE211-E12 mAb (T4P), both obtained by the hybridoma technique, 18 produced in mice as ascitic fluids. They were kindly given to Dr De Gaspari during her post-doctoral fellowship at the Walter Reed Army Institute of Research, where they were routinely used. Back in Brazil, the mAbs were lyophilized and kept in aliquots at 4°C. For use, they were resuspended, titled and maintained at –20°C. 19
SDS-Polyacrylamide Gel Electrophoresis (SDS-PAGE)
To verify specificity against pili, we performed an immunoblotting assay. For that, whole cells of meningococci strain B:4:nt were characterized by electrophoresis in a 10% polyacrylamide gel, in the presence of sodium dodecyl sulfate (SDS), following the protocol described by Laemmli 20 and using a molecular weight marker ranging from 11 to 245 kDa (Color Protein Standard Broad Range, New England BioLabs). After the electrophoresis, the gel was stained with Coomassie Blue (PhastGel Blue R; Pharmacia Biotech). Immunotype mAbs were previously tested in our laboratory. 21
Immunoblotting
A new gel was prepared following the same steps described earlier. After electrophoresis, proteins were transferred to a 0.45 µm nitrocellulose membrane (BioRad Laboratories) at 100 V for 18 hours at 4°C. After that, the membrane was stained with Ponceau-S and discolored with PBS. The membrane was blocked with skimmed milk (La Sereníssima) 5% for 2 hours, at room temperature. mAb was diluted at 1:2000 in skimmed milk 2.5% and incubated overnight at 4°C. Then, the membrane was washed five times with PBS pH 7.2 and incubated with goat anti-mouse IgG (whole-molecule)-alkaline phosphatase (Sigma Aldrich) diluted at 1:5000 in skimmed milk 2.5%, for 2 hours, at room temperature. The membrane was washed again and the substrate 5-bromo-4-chloro-3-indolyl phosphate/nitro blue tetrazolium (BCIP/NBT-plus) (Mabtech) was incubated for 20 minutes, at room temperature and protected from light. The reaction was stopped by adding distilled water and was considered positive by the appearance of color in the membrane. 22
Dot-ELISA
Dot-ELISA was conducted pipetting 1 µL of whole cell suspension in nitrocellulose membrane 0.45 μm (BioRad Laboratories). After that, Ponceau-S staining was proceeded. Membranes were blocked with skimmed milk (La Sereníssima) 5% for 2 hours at room temperature. mAbs, diluted at 1:2000 in skimmed milk 2.5%, were incubated overnight at 4°C. Membranes were washed five times with PBS pH 7.2 and incubated for 2 hours, at room temperature, with goat anti-mouse IgG (whole-molecule)-alkaline phosphatase (Sigma Aldrich) diluted at 1:5000 in skimmed milk 2.5%. After washing, membranes were incubated with the substrate BCIP/NBT-plus (Mabtech) for 20 minutes, at room temperature and protected from light. We added distilled water to stop the reaction, which was considered positive by the appearance of color in the membrane. 4
This project was performed at the Immunology Center of Adolfo Lutz Institute and was approved by the Technical and Scientific Committee of this institution (CTC number 41D-2011).
Results
Accessing 7BE211-E12 specificity, Figure 1 shows meningococci strain B:4:nt electrophoretic profile in SDS-Polyacrylamide Gel Electrophoresis (SDS-PAGE) and immunoblotting reaction with the referred mAb, recognizing only one band in immunoblotting. LPS mAb specificity was previously tested in our laboratory. 21

Immunoblotting of mAb 7BE211-E12. On the left, molecular weight and electrophoretic profile of strain B:4:nt, on the right, mAb Immunoblotting. mAb, monoclonal antibody.
The results of Dot-ELISA show a high expression of the L3,7,9 immunotype and type IV pili. From 19 MenB strains, all were reactive to L3,7,9 immunotype and 16 were reactive to T4P. From 21 MenC strains, L3,7,9 mAb recognized 19 strains and T4P recognized 14 strains. Of 45 MenW strains, all presented L3,7,9 immunotype and 35 presented T4P. Of 28 MenY strains, L3,7,9 mAb recognized 26 strains and T4P recognized 23 strains. Figure 2 shows the results obtained.
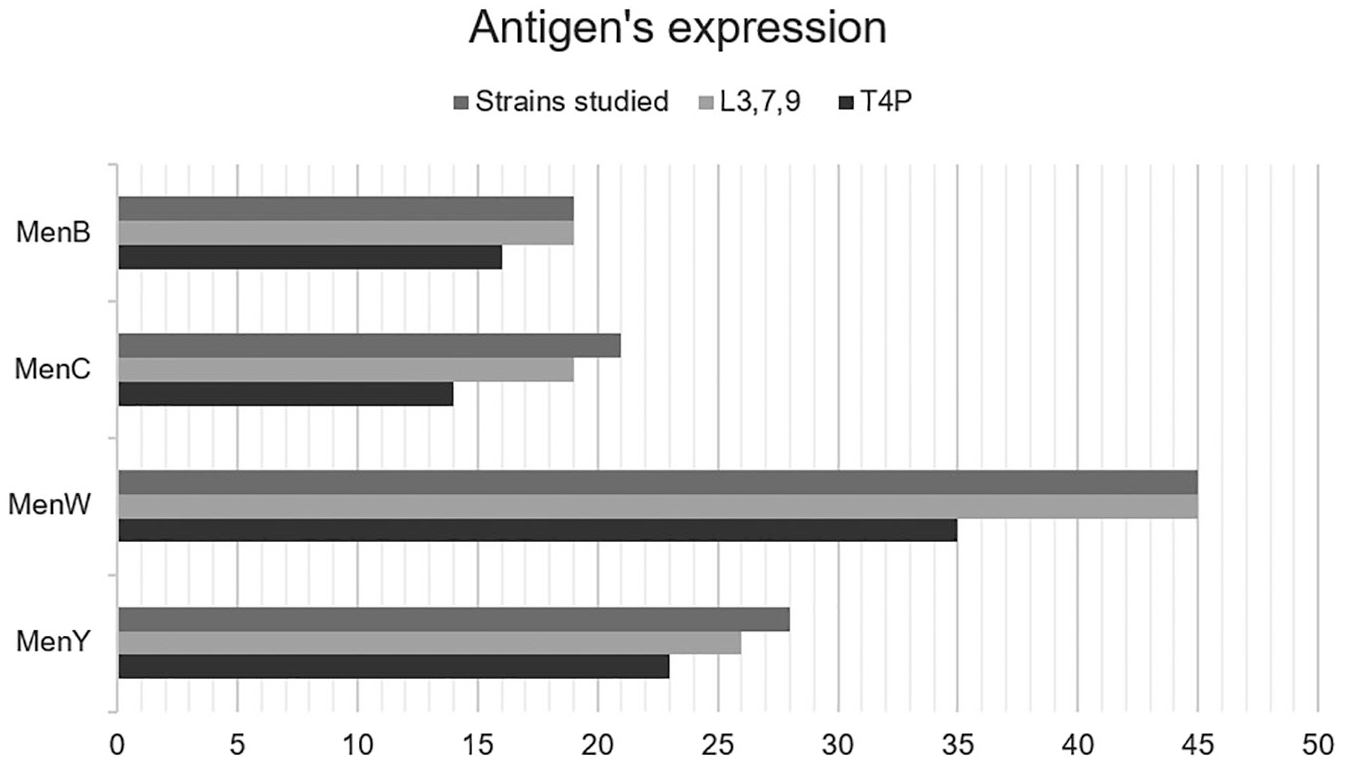
L3,7,9 and T4P expression in Brazilian strains of serogroups B, C, W and Y studied. T4P, type IV pili.
Discussion
As expected, mAbs were specific, recognizing only one band in immunoblotting. The pili epitope was approximately 17 kDa according to molecular weight. 23 LPS mAb was previously tested in our laboratory, also recognizing only one band, at 5.9 kDa. 24 The Dot-ELISA technique proved to be advantageous due to its technical simplicity, dispensing with the use of specific equipment, using a low volume of monoclonal antibodies and requiring little manipulation of samples, as proposed by Wedege et al. 4 Given that low-income areas are still linked to a higher burden of IMD, Dot-ELISA characteristics strength that even in the molecular biology era, serological techniques may still be applicable, especially in developing countries.
LPS is considered the main virulence factor of Gram-negative bacteria. It induces an inflammatory response, contributes to adhesion, invasion and inhibits phagocytosis and complement activation, playing roles in infection establishment and progression.2,24 The arrangement of the LPS on the cell surface also influences the exposure of outer membrane proteins, which has implications in host–pathogen dynamics. 25 Studies showed that some immunotypes, such as L1 and L8, are more often related to carrier strains; however, meningococci can switch its expression during the disease. The L3,7,9 immunotype has already been described between virulent strains11,26 and had been associated with a high pro-inflammatory response. 27 Detoxified LPS had already been studied as a vaccine compound, given its adjuvanticity qualities, activating Toll-like receptor 4 (TLR-4), being relatively conserved among strains, inducing bactericidal antibodies and probably being able to confer protection against a range of Gram-negative bacteria.28,29
Pili is responsible for the first interaction between the bacteria and host cells, mediates motility, DNA exchange by transformation and allows the meningococci to cross the blood–brain barrier, establishing the infection. T4P is found in several Gram-negative pathogens, such as Pseudomonas aeruginosa and Vibrio cholerae.30–32 Studies showed that pili expression is required for colonization and its removal decreased the level of adherence between the bacterium and the host’s cells.33,34 A recent study proposed the use of T4P as a target for therapeutics, which reduced the colonization, spread and lethality of the bacterium. 35
According to previous studies, antigens of pathogenic species of Neisseria usually show great variability among strains. 36 However, keeping some antigens is important to maintain the virulence, as pointed out by Urwin et al. 37 Pili and LPS play important roles in colonization and spread in the host tissue, which are essential steps of invasive disease, 38 and several studies, described before, proved its importance to meningococci. Given our results, Brazilian strains express these antigens, suggesting high pathogenic capacity. The strains were isolated from IMD cases, which agrees with that assumption. The emergence of different serotypes and epidemiological changes, as we have seen with the emergency of serogroups W and Y, are usually connected to hypervirulent strains, 39 so it must be important that key factors to pathogenicity are maintained among them and, therefore, are characterized by surveillance programs. Weidlich et al. 40 and Lemos et al. 41 studied the hypervirulent strain W:2a:P1.5,2 in Brazilian cases of IMD. That strain was also seen in Argentina 42 and in Chile, where, in 2010, it became the most prevalent strain and led to an A, C, W and Y quadrivalent vaccine campaign. 43
All these data reaffirm how important it is to proceed with antigenic characterization, in order to recognize hypervirulent strains and take control initiatives. Besides the recognition of important antigens between strains, that led us to understand the pathogen better, phenotypic characterization may be applied to study vaccine targets, given that it would be interesting to induce an immune response against antigens that are key factors for virulence. Besides that, characterization allows us to check if current vaccine strains present such antigens, contributing to surveillance over immunization.37,44
Conclusion
Due to the impact of IMD, it is important constantly to maintain and improve vaccine studies. The characterization of epitopes provides data about meningococci virulence, the prevalence of circulating strains and contributes to immunization research and monitoring. Simple techniques, such as Dot-ELISA, and tools such as mAbs, are important and collaborate with strain selection for vaccine improvement and IMD control.
Footnotes
Acknowledgements
The authors would like to thank Dr Ana Paula Silva Lemos, from the Bacteriology Center of Adolfo Lutz Institute, for providing the whole cell suspensions and information about its serotyping, and Walter Reed Army Institute of Research, for kindly providing the mAbs used in this study.
Conflict of interest
The authors declare that there is no conflict of interest.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by São Paulo Research Foundation (FAPESP) (grants 12/15568–0, 13/11147-2, 14/11172-0, 14/07182–0, 18/04202-0) and the National Council for Scientific and Technological Development (CNPq) (132743/2014-6). São Paulo State Secretary of Health and Coordination of Improvement of Post-Graduate Level Personnel (CAPES) provided scholarships.
