Abstract
The transmission of diseases between wildlife and livestock poses a major challenge to both conservation and livestock sectors in Southern Africa. Focusing on the cases of foot and mouth disease and trypanosomiasis in the Kavango–Zambezi Transfrontier Conservation Area, this article explores the complexity of coexistence between humans, livestock, wildlife, vectors and pathogens. Multispecies coexistence, we suggest, is best understood not only through the relations between species, but also as characterized by a collision of modes of ordering. Drawing on expert interviews and a discourse analysis of policy documents and reports, we identify three modes of ordering coexistence: a categorical and increasingly disfavoured mode of species eradication, a territorial mode focused on containment and separation, and an infrastructural mode premised on connectivity between populations, landscapes and ecosystems. Together, these different modes of ordering pose a challenge to scientific knowledge production; where uncertainties present themselves not so much in the form of ignorance or knowledge gaps, but rather in the form of ambiguity: of knowing diseases and species differently. In this view,
Introduction
Debates on human–wildlife conflict as a consequence of large-scale conservation have been much focused on human–carnivore or human–elephant conflict (Knox et al., 2021). It is often characterized by crop damage and livestock loss or by injuries and deaths suffered by humans and animals. A less prominent aspect of this tense coexistence has been the (potential) transmission of diseases between coexisting species. Yet, the transmission of diseases between wildlife and livestock poses a major challenge to both conservation and livestock sectors in Southern Africa and across the globe. A key challenge here is finding a balance between the conservationist quest for habitat connectivity, wildlife corridors and wildlife dispersal areas and the containment of infectious diseases linked to wildlife mobility.
This article aims to offer an understanding of animal health and conservation as modes of ordering multispecies coexistence in an area where these challenges are particularly salient: the Kavango–Zambezi Transfrontier Conservation Area (KAZA TFCA). The KAZA TFCA spans across Angola, Botswana, Namibia, Zambia, and Zimbabwe (known as its partner states), and hosts a great number of species and ecosystems. In this area, as elsewhere, diverse wildlife species are considered to be hosts to a range of diseases that can be transmitted to livestock and in some cases also to humans.
In earlier times the control of animal diseases often meant the eradication of wildlife; prominently highlighted for northern Botswana by Gumbo (2010) and Morton (1996). In their accounts, they showed how the killing of potentially diseased and infectious wildlife was done in order to provide a safe space for agricultural expansion. Since the 1960s disease control has been commonly practised through an intensification of vector eradication programmes via spraying, the containment of wild animals and the separation of domestic and wild spaces through the creation of separate land use zones and the erection of fences to divide such zones and inhibit livestock/wildlife mobility across them. These fences are today a pain in the eyes of conservationists, who see the ideal of connectivity between landscapes and animal populations being obstructed and who raise concerns about the detrimental effect of fences on wildlife populations and the ecosystem (Ferguson and Hanks, 2012; Gadd, 2012; Mbaiwa and Mbaiwa, 2006; Stoldt et al., 2020; Thomson et al., 2013). They advocate that new forms of coexistence must be developed. Challenging in this regard is that zoonotic and livestock diseases emanating from wildlife expand the range of relevant actors, knowledge, and economic sectors to consider, presenting an exceedingly complex situation where the aspired multispecies relations potentially multiply and diverge significantly.
In this contribution, we approach diseases as part of multispecies sociality, in which host species, vectors, microbes, humans, and the wider ecosystem form a complex network of exchanges and dependencies – and where the idea of individual, autonomous organisms and species is replaced by a relational ontology of co-constitution (Gilbert, 2017; Hinchliffe et al., 2016; McFall-Ngai, 2017; Tsing et al., 2017). Hence, our multispecies approach to animal health, following Keck, involves taking ‘pathogens crossing barriers between species as a starting point for an inquiry about transformations in our relationships with nonhuman animals’ (Keck, 2020: 2). Modern disease control appears largely based on containing and exterminating individual host, vector, or pathogenic species. This happens against a reality of such assemblages characterized by continuous movement, exchange, transformation and co-becoming. Here, different arrangements of the ‘wildlife–livestock interface’ (e.g. fenced, open, and buffered) structure interspecies contact and transmission pathways, as well as the potential for human–wildlife conflict (Kock et al., 2014), to create what Hinchliffe et al. (2016) call a ‘disease situation’.
Moreover, we consider diseases to be subject to discursive framing and particular regimes of knowledge production. The very construction of a disease, its social and economic relevance, and the role it comes to play in (inter)national politics, interlaces with its biology, thus generating uncertainties at different bio-social levels. In her historical and political analysis of foot and mouth disease (FMD) outbreaks in the UK, Woods concludes that ‘it is the thoughts and actions of humans that have “manufactured” FMD into the particular political, economic, social and psychological problem that it is today’ (Woods, 2004: XIV), stressing further that ‘FMD was not, and is not, simply an animal issue; the issues it raised always involved economic and political circumstances and calculations’ (Woods, 2004: 147). Accordingly, we approach diseases as co-constructed by socially produced narratives and material dynamics of pathogens, taking into consideration their political ramifications and imaginary frameworks as well as their physical characteristics.
The complexity of disease control, then, does not emerge only from the intimacies between and among species, because what a specific disease or species is, is not given (Law & Mol, 2011; Kirksey, 2015). Is a buffalo a maintenance host of a contagious disease? Or is it, first of all, a species to be conserved? And cattle, are they encroachers? Important commodities? Or members of social life? We might live well with some versions of these species, but not necessarily with others. How we may then ask, do not only these different species, but also their different ways of being known and enacted figure in our attempts to coexist with them?
In this article, we explore the complexity of living with diseases through the different sets of knowledge, values and meanings associated with the coexistence of humans, livestock, wildlife and pathogens. Particularly in this context, we agree with anthropologist Schroer that the study of coexistence needs to focus on ‘relations and the
Analysing the frictions between these fields, this article explores how the resultant complexity may best be approached by scientists who are working in this landscape and beyond, reflecting on the challenges of interdisciplinarity for our interlocutors. In the following, we first introduce our study site, the KAZA TFCA, before elaborating further on our multispecies approach to disease and complexity, and subsequently our methodology. Our analysis then proceeds in four parts, discussing various aspects of the complexities of multispecies coexistence: (1) the discursive framing of wildlife as a disease reservoir; (2) ideals of the connectivity of species versus the containment of pathogens; (3) specificities of vector-borne diseases with regard to the previous two topics; and (4) the role of science in producing this complexity. In the concluding section, we discuss the very possibility of coexistence with pathogens by reflecting on the different modes of ordering (infrastructural, territorial, and eradicating) through which scientific communities have been addressing the problem.
The Kavango–Zambezi Transfrontier Conservation Area
The KAZA TFCA covers a vast area of 520,000 km2 (for reference, its official website refers to it as being ‘larger than Germany and Austria combined and nearly twice as large as the United Kingdom’ (KAZA TFCA Secretariat, 2019)). The KAZA TFCA is not a park in itself though, but rather envelops 36 national parks and an additional host of game reserves, communal conservation initiatives and tourism areas in the partner states. Furthermore, it is home to around 2.7 million people living in rural settlements and towns. Particularly Botswana is a large beef producer, and together with Namibia dependent on the export of beef to the European Union (EU) thereby complying with the strict regulations of the EU and the World Organization for Animal Health (OIE) for red meat. Also, in the other partner states, livestock plays an important role in the economy and is essential to many rural people's livelihoods.
Of interest to this paper is the framing of the KAZA TFCA as a ‘coexistence landscape’ – a space that has not been territorialized for a single purpose such as either conservation or grazing, but that is explicitly recognized to encompass both and more. Discussions around coexistence landscapes – alternatively ‘working landscapes’ (Kremen and Merenlender, 2018) – are key in the development of twenty-first-century conservation. A key premise of the coexistence approach is that conservation does not happen only in secluded, protected areas but also in working lands that are humanly dominated, wherein conservation-orientated land use and sustainable land use interpenetrate each other (Marris, 2013). Crucial is also the (somewhat regretful) realization that ‘protected areas will remain islands of “pristine nature” in a sea of profound human transformations to the landscape through logging, agriculture, mining, damming, and urbanization’ and that such islands are not capable of supporting biodiversity in the long-run (Marvier et al. 2012). As a result, large-scale conservation is most promising when considered in terms of integrative, connective, and patchy landscapes in the eyes of coexistence landscape proponents (Kremen and Merenlender, 2018; Marvier et al., 2012). In the KAZA TFCA, as in other conservation areas worldwide, this means potentially divergent stakes, logics and aims are brought together in a single geographic, legislative, and ideological framework, namely that of (transfrontier) coexistence conservation. Furthermore, given that the post-2020 Global Biodiversity Framework establishes that 30% of the globe's terrestrial surface should be protected by 2030 (CBD, 2022), the situation we observe in the KAZA TFCA will be typical for many regions of the world and foreshadows intense discussions on the challenges and chances that coexistence landscapes present.
Complexities of coexistence and the challenges of uncertainty
In order to understand complexity, Mol and Law (2002) suggest, it is imperative to look in detail at the multiplicity of simplifications. Simplifications are at the basis of technology, the production of scientific knowledge, and modern governance, which can be recognized as distinct ‘modes of ordering’ that seek to reduce, tame, or repress the boundless complexity of the world. Simplifications, modes of ordering, ‘diagrams’, or alternatively ‘logics’ (we will use the terms interchangeably) do not simply impose an order on the world, however. They are
From an epistemological point of view, the complexity of multispecies coexistence is problematic as far as it fosters uncertainty. The unintended consequences of complex processes are hard to predict and difficult to manage and may include transmission of viruses, newly emerging infectious diseases, and transformations of other kinds. The challenge for scientific research (but also governance) is not complex as such, but how to embrace the uncertainty it inevitably entails (Scoones, 2019). In the case of the KAZA, the complexity of multispecies coexistence presents a particularly grave challenge for scientific knowledge production and environmental governance, because the ongoing transformation of the landscape is exceptionally rapid, comprehensive, and potentially harmful for livestock and humans. The primary issue here is not limited knowledge about pathogens and the mechanisms of contagion, however, nor even ignorance related to societal responses and behaviours of the affected populations. While addressing these ‘knowledge gaps’ certainly contributes to improving environmental governance, a narrow focus on disciplinary knowledge misses the point. Disciplinary engagements, however expansive or intricate they may be, will always understand and enact coexistence in terms specific to the discipline. As such, we argue, they still simplify the issue, even if this disciplinary knowledge may be very difficult to understand for outsiders (Thompson, 2002). Complexity, as we understand it, may be more accurately associated with the multiplicity of simplifications and their frictions, and as such, the challenge for knowledge production is not about knowing
Here, we focus on conservation and animal health, as distinct ways of understanding and categorizing, simplifying and managing coexistence at the interface of wildlife, livestock, pathogens, and humans. Our point, then, is not to produce an alternative mode of ordering, so as to simplify the bewildering complexity that is the KAZA TFCA as a landscape, international organization, geopolitical zone, and multispecies assemblage (although we certainly cannot escape doing so either). Neither do we claim to present a holistic, all-inclusive view of this complexity. Rather, we seek to follow and draw lines of connectivity between materials, subjects, species, and scales, exploring the friction that occurs between existing modes of ordering. How have animal and zoonotic diseases been ‘manufactured’ in KAZA? How does (potential) disease transmission between wildlife and livestock affect ideas of coexistence? And how is this coexistence perceived, managed, and contested by different stakeholders in and beyond the region? At the core of this particular manifestation of human–wildlife conflict, we suggest, are differential understandings and enactments of human–nonhuman relations. To put this differently, we approach the wildlife–livestock interface not merely as an interface of species and organisms, but also consider how this interface is continuously ordered and contested, as different modes of ordering interface themselves and create complexity along the way. Here, uncertainty is the product of the ambiguity of how scientific research knows diseases and species in the first place.
Methodology
This article draws primarily on 15 remote, in-depth interviews with local and international experts in conservation, animal health, and livestock industries, government officials of the KAZA TFCA member states and international institutions such as the South African Development Community, the OIE and the Food and Agriculture Organization of the United Nations (FAO). Interlocutors were recruited through their respective institutions. The interviews were held via video calls (due to the prevailing COVID-19 situation in 2020/21) and lasted between 45 min and 2 h. Recordings of these conversations were transcribed verbatim and analysed carefully looking for attempts to order reality and to deduct assumptions about causality. Oral consent was sought from each participant at the start of the conversation.
In addition to the interviews, we draw on informal online and offline conversations with international scholars with extensive research experience in Southern Africa, among whom were biologists and social scientists.
Furthermore, we conducted a discourse analysis of a range of policy documents and reports from various (non)governmental and international organizations associated with the question of disease transmission, animal health, and conservation in the KAZA TFCA. Combining interviews and informal talks with this analysis of documents has allowed us to place official standpoints and guidelines next to the experiences of specialists, not with the aim of testing either narrative, but to sort out the sometimes-uneasy alignment between policy and practice.
This article, further, is the result of an interdisciplinary collaboration between the authors. van Dam, who is an anthropologist and environmental humanities scholar, conducted the interview study and discourse analysis. van Engelen, who is a conservation social scientist, contributed to the article with further insights on multispecies studies, FMD and the KAZA TFCA based on the literature review. The scope and questions of the research were decided on in collaboration with Bollig who is an anthropologist, and Müller-Mahn, who is a geographer. Agha and Borgemeister, who are entomologists, and Jungeln, who is a virologist, brought in a natural sciences perspective, reviewing the findings in light of their expertise on the topic of (vector-borne) disease transmission in southern and eastern Africa. In an academic context where disciplines have drifted apart and withdrawn into their own particular jargon and modes of inquiry, interdisciplinary work has the potential to irritate everyone and satisfy no one, but also to bring together fragmented, specialized knowledge and produce new ones. As our analysis will demonstrate, this is not a matter of synthesis, but rather involves careful ‘negotiation of […] collaborative possibilities’ (Mol and Hardon, 2020: 1), based on the recognition of the partiality of disciplinary knowledge and positionalities of knowledge producers. Engaging with these challenges we sought to conceptualize complex bio-social challenges in a more adequate way, even if the resulting knowledge remains ever partial.
Finally, regarding our approach to the materials that informed our analysis, our study departs from traditional Foucault-inspired studies of knowledge and power in that – besides stressing the way in which science is used to control populations – we also consider how knowledge practices may constitute practices of care. Crucially, as our case demonstrates, science does not always work in the service of the state, and neither state nor science is unitary in itself – posing a challenge to the tight association between knowledge and (state) power (Nustad & Swanson, 2021). Scientific knowledge, as others have argued, may also simply be driven by curiosity or care (Cassidy, 2019; Tsing, 2015; Van Dooren, 2014). Thus, while we find it important to remain critical of the ways powerful actors categorize, map and understand multispecies coexistence, and how these frequently serve their interests, we also want to keep open the possibility that these practices provide insights into the lives of (nonhuman) others to protect, promote or care for them. As such, our approach is typified by a critical awareness of the knowledge/power nexus, but without it necessarily overshadowing the ways in which knowledge may also help us learn about the worlds of others.
Wildlife as a disease reservoir
It is well-documented that certain diseases are preponderant in biodiverse areas and particular wildlife populations (Keesing et al., 2010; Kock, 2014). Expanding wildlife populations may carry pathogens into settled areas, and – vice versa – humans encroaching upon wildlife areas may find themselves confronted with the risk of livestock and zoonotic diseases. Prominent examples of this are FMD being carried by cape buffalo populations; wildebeest carrying catarrhal fever; canine diseases, such as rabies, being exchanged between wild dogs, jackal, and domestic dogs; African swine fever being carried by wild pigs, elephants carrying anthrax and tuberculosis; and a range of diseases being spread by migrating bird species (see Bengis et al., 2002; Vosloo et al., 2005). Staying with the example of FMD, this is a viral disease affecting numerous species of cloven-hoofed animals including cattle and other livestock, while buffalos remain the only confirmed wildlife reservoir host (Paton et al., 2018). Buffalos are born with maternal antibodies against the disease but this quickly wanes leaving young animals (less than a year old) particularly susceptible to infections. This cohort of animals is involved in ‘childhood epidemics’ (Vosloo and Thomson, 2019), which play an important role in both the persistence of the disease within the reservoir host population as well as the disease transmission to other susceptible hosts. While cattle and other livestock suffer from fever, blisters and lesions when infected, the buffalos themselves remain largely unaffected by the disease (Kock et al., 2014). Similarly, tsetse-fly-transmitted trypanosomes are considered to have relatively little effect on other wildlife host species such as waterbuck and kudu, while cattle suffer from a loss of weight and strength and produce less milk.
Boxes 1 and 2 serve to give readers insight into some of the more pertinent aspects of FMD and trypanosomiasis.
Foot and Mouth Disease (FMD).
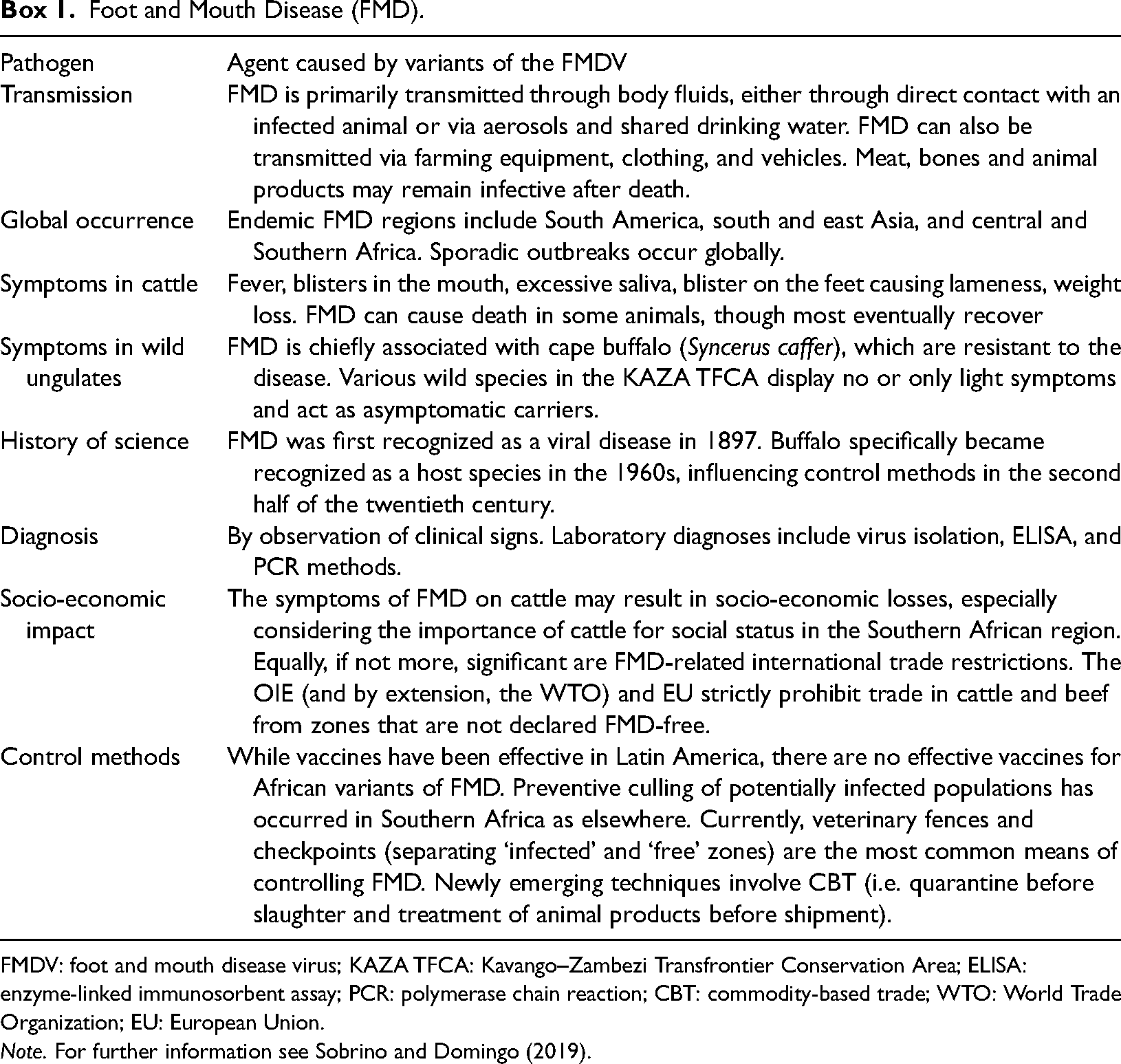
FMDV: foot and mouth disease virus; KAZA TFCA: Kavango–Zambezi Transfrontier Conservation Area; ELISA: enzyme-linked immunosorbent assay; PCR: polymerase chain reaction; CBT: commodity-based trade; WTO: World Trade Organization; EU: European Union.
Animal African Trypanosomiasis (AAT) (Nagana).

KAZA TFCA: Kavango–Zambezi Transfrontier Conservation Area; PCR: polymerase chain reaction.
Framing wildlife as a reservoir suggests that wildlife and conservation areas are a source of the problem of livestock diseases and zoonosis. This, however, is incomplete. Helliwell et al. (2021: 12–13) recognize the ‘reservoir’ as a distinct scientific ‘environmental imaginary’: as a space that forms a ‘latent environmental threat existing independent of human interventions’ and which ‘suggests that the environment may act as a space of “holding” or “collection”’ of diseases, as well as of antimicrobial resistance. Furthermore, as Keck (2019, 2020) has argued, framing an environment or species as a ‘reservoir’ for a particular disease, effectively frames it as pathogenic. The idea of wildlife reservoirs draws focus to the
Different approaches to animal health are practised in these artificially separated landscapes. Our interlocutors pointed to the inability to intervene with disease control measures in conservation areas, as diseases are considered part of a balanced ecosystem. For example, in the words of one veterinarian working for an international non-governmental organization (NGO): In an ecological system it would just sort itself out, wouldn't it? Anthrax has been a real problem with hippos. Well, why don't you go and vaccinate the hippos? But wildlife conservationists will say: No, it's got to do its thing and let it do its normal cycle of up and down.
Furthermore, insofar as wildlife is framed as a reservoir for diseases, and livestock disease is understood as an area of human–wildlife conflict, humans have facilitated the proliferation of this conflict with their efforts to expand conservation into settled areas and to promote the cohabitation of wildlife, livestock and humans in some landscapes. Further, the intensification and expansion of human activities, including settlement, herding, and deforestation in areas populated by wildlife, is likely to increase pressure on wildlife populations and increase the likelihood of human–wildlife contact (either directly or via their livestock), and thus also to increase the risk of disease transmission. This view was shared among most, if not all, of our interlocutors. One Zambian parasitologist said: The human population is expanding. The interaction between wildlife, vectors of diseases, and man is going to increase. People want land and they will move closer and closer to the national parks and game management areas. And this settlement has resulted in serious economic losses, because people move in with their cattle, and they lose cattle over time because of trypanosomiasis and other diseases. Because of this interaction, again, more outbreaks are expected. Therefore, we put ourselves in a future of more outbreaks of disease.
Connectivity and containment
One of the primary aims of the KAZA TFCA is, according to its treaty, realizing connectivity between conservation areas of different kinds (national parks, community conservation areas, wildlife management areas, community forests, etc.) within a vast territory. This objective reflects a reorientation in the conservation sector globally. It moves away from its traditional focus on wilderness devoid of its original human inhabitants and bounded territories of nature conservation (also known as ‘fortress conservation’), towards the creation and maintenance of patchy working landscapes of conservation in which core conservation areas are connected by wildlife corridors necessarily increasing the interface between humans and wildlife. Allowing species to migrate is considered to strengthen the resilience of biodiverse ecosystems and stabilize wildlife populations. This effort of promoting movement and connectivity in the KAZA TFCA includes attempts to ‘restore’ migration pathways of wild animals in so-called ‘wildlife dispersal areas’ (KAZA TFCA, 2016). This is also thought to alleviate human–wildlife conflict. As one conservationist working with elephant populations in Botswana commented: Elephants could be allowed to start to move again. Out of Ngamiland, where they've been literally bottled up, literally exploding because they have nowhere to go [due to veterinary fences]. To move across the Zambezi into Angola, alleviating human wildlife conflict and expanding their numbers. That could be a win–win–win for tourism and for mitigating conflict. Because when elephants are bottled up, they cause conflict. In conservation they will say that you are expanding the biodiversity, which is very, very good. But at the same time, you are expanding the biodiversity of pathogens. So, as a veterinarian you have a problem, because now your situation is complicated.
This concern with the spread of diseases also reflects in regional policy documents (e.g. SADC, 2008). The key case here is FMD. Although preventive vaccines are available, the diversity of strains of the FMD virus circulating in Southern Africa, a lack of data and underreporting pose a significant challenge to the selection and distribution of effective vaccines (Maree et al., 2014). Accumulating losses of up to US$21 billion a year in endemic countries, and an additional US$1.5 billion in “FMD-free” countries and zones, FMD is considered one the most economically important infectious animal diseases worldwide (Knight-Jones and Rushton, 2013; Maree et al., 2014). Accordingly, FMD is subject to strict OIE trade regulations. Some hosts are asymptomatic carriers of FMD, making zoo-sanitary control difficult, while at the same time increasing the risk of importation of infected animals into FMD-free zones. Since the 1950s and 1960s, many veterinary fences have been erected across Southern Africa to separate potentially infected wildlife species, buffalos in particular, from livestock, so as to conform to OIE trade standards and secure the possibility of international trade in beef (see Figures 1 and 2). Arguments for upholding these fences also extend to other diseases that are transmitted through direct contact between wild and domestic animals. These include, among others, wildebeest-associated catarrhal fever, anthrax, canine diseases such as rabies, and bovine tuberculosis (Thomson and Penrith, 2011).
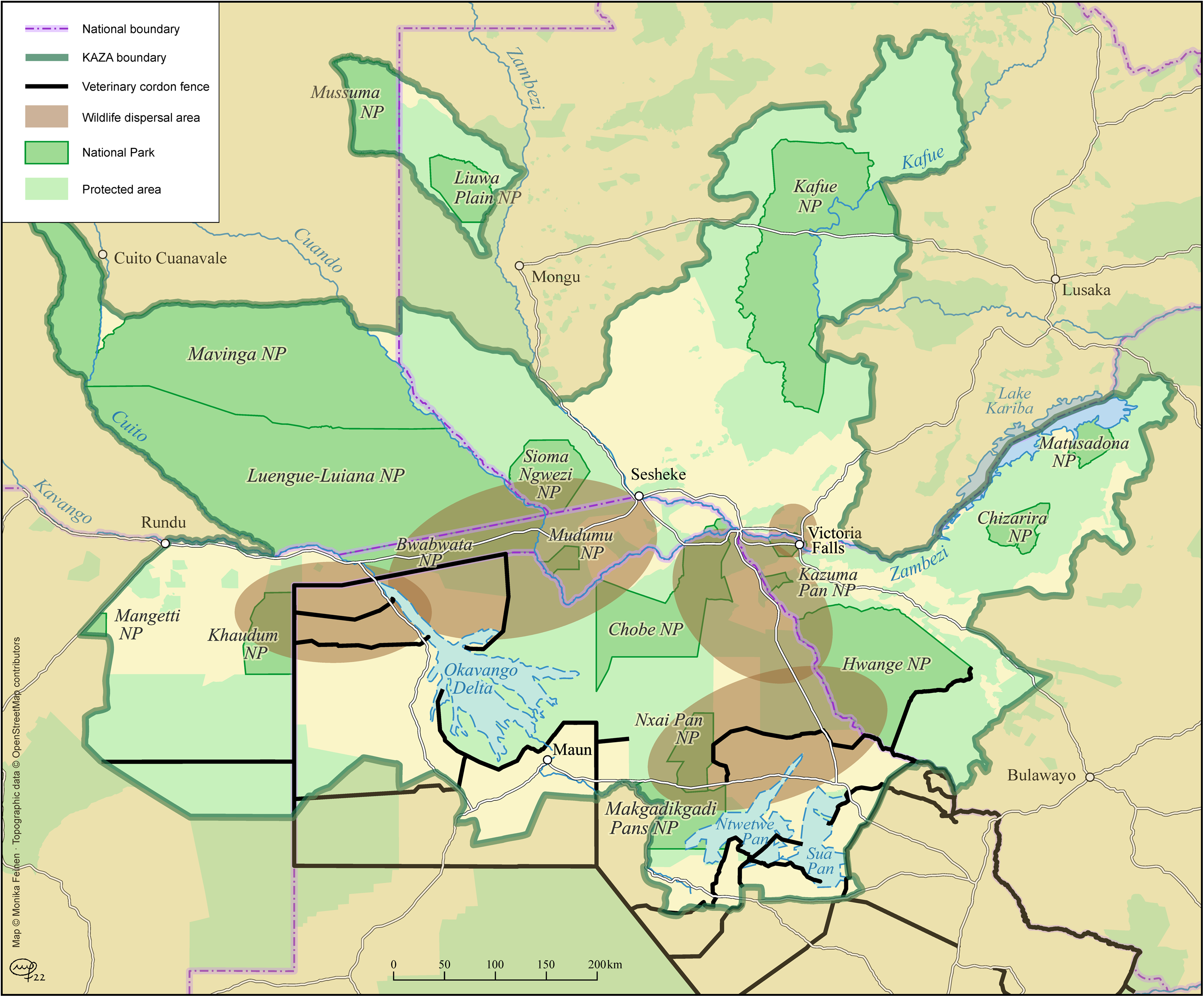
Veterinary fences in the Kavango-Zambezi Transfrontier Conservation Area (KAZA TFCA). Credit: Monika Feinen, 2022.

Veterinary cordon fence in Botswana. Credit: Wisse van Engelen, 2022.
Clearly, erecting fences to restrict the movement of wild animals counteracts the ideal of free-roaming herds. The fences here function as an ‘essentially static disease intervention strategy that intentionally disrupts the contact structure between wild and domestic animals and pathogens’ (Ferguson and Hanks, 2010: 12). The fences put up to contain diseases, then, obstruct the aspired (transfrontier) connectivity. Nevertheless, connectivity and containment, as seemingly opposite modes of approaching wild species, cannot simply be attributed to either the livestock or conservation industries. Pastoralists, our interlocutors indicated, might also cross fences that have been put up to protect their cattle, in order to reach better grazing grounds. Consequently, some conservationists have argued to maintain these fences, even if they restrict the movement of wild animals, because they consider them to protect wildlife from the encroachment of humans and livestock. One veterinarian from Botswana phrased this concern as follows: In KAZA, there's what's called the southern buffalo fence, and it goes around the southern part of the Okavango Delta. You'll have lots of conservationists arguing that if we took down that fence, cattle would be inundating the delta. And there's some truth to that, I mean, the fence blocks wildlife from coming out of the delta and it keeps cattle from going in.
At the core of the tensions in multispecies coexistence, then, is not simply the fact of intimate exchanges between species or sectors, but conflicting modes of ordering: on the one hand,
Containing vector-borne diseases
Vector-borne diseases, such as tsetse fly-transmitted trypanosomiasis and tick-transmitted anaplasmosis and babesiosis, have been associated with natural parks and reserves. Although these vectors might, as some of our interviewees indicated, travel along with migrating herds of ungulates, the vectors themselves are not necessarily restricted by fences, which means the territorial containment of diseases is further complicated in the case of vector-borne diseases. We will zoom in on the control of trypanosomes for a moment. These Protozoan parasites can cause sleeping sickness in humans (Human African Trypanosomiasis) and
While these methods have proven effective in relatively isolated areas such as the island of Zanzibar, where tsetse flies were eradicated completely in 1997 (Vreysen et al., 2000), they remain largely ineffective in mainland Africa, even if removal of tsetse has been shown to result in a rapid, yet localized and often temporary increase in cattle in that region (Bollig and Vehrs, 2021). As the various tsetse experts we spoke with indicated, the different ‘tsetse belts’ of western, central, and eastern Africa, are interconnected and reach southward into the Zambezi river basin. Of the KAZA TFCA partner states, then, mostly Zimbabwe and Zambia are affected by tsetse. Historical documents suggest that in the late nineteenth and the first half of the twentieth century also northern Botswana and north-eastern Namibia had been massively affected. Due to this interconnection of tsetse populations across sub-Saharan Africa, eradicating the flies from particular areas in many cases only has temporary benefits, as the population is likely to soon replenish. ‘Many African countries were led to believe that they could eradicate tsetse flies’, one critical tsetse expert said, ‘and I think that put a lot of countries off track. After 10, 15 years, you can see it's not been eradicated’. Connectivity between populations, then, is an unavoidable concern, implying that total eradication is not feasible, and some form of coexistence is inevitable.
In addition to this, tsetses are strongly associated with wildlife species that are considered to act as hosts for the trypanosomes and from which the flies take their blood meals. ‘Definitely, the wild animals are reservoirs’, said a Nigerian ecologist and international tsetse control expert, reiterating what several of his colleagues in this field also emphasized, namely that they were unable to intervene with traps or pesticides inside natural parks, so as not to disturb sensitive ecosystems. Eradicating species from nature-protected areas would go against the principles of habitat and species conservation – an effort that is principally centred on the prevention of species extinction. Accordingly, within KAZA TFCA, as elsewhere, tsetse populations are primarily concentrated in various nature reserves and parks.
Rather than pursuing a logic of total eradication, then, the general strategy for tsetse control has shifted towards more focused control in specific areas outside the parks. ‘We use what we call disposable targets’, a Zambian tsetse ecologist explained, ‘They should be effective up to 12 months in the tsetse habitat. And within these 12 months they should reduce the population to a point where, without re-invasion, the population will be brought down to zero’. To prevent re-invasion of tsetse from the conservation areas, the idea is to create a buffer zone between the sanitized area and the conservation area. ‘We create a barrier of four to eight kilometres of a very high concentration of targets’, he explained.
Here, it becomes apparent that the way of thinking about the origin and spread of vector-borne diseases, such as trypanosomiasis, does not differ fundamentally from diseases transmitted through direct contact such as FMD. Much like ungulates carrying FMD, tsetses are thought of as connected, their ‘belts’ functioning as lines of connectivity of and between tsetse populations. Here too, the logics of disease control are strongly linked to place: to the inside and outside of conservation areas, and as such territorialized into ‘clean’, ‘infested’ and ‘reservoir’ zones. With the aim of containment, buffer zones around conservation areas (neither fully dedicated to conservation nor suitable for livestock raising), share much the same ontology as veterinary fences. However, the Zambian expert quoted above added a note of caution: But Zambia has some of the largest national parks, and there are very different types of land, so that to put targets around them is extremely costly and challenging. Sometimes we are not able to maintain the barrier as well as we should, for it to stop the tsetse coming from a national park into a cleared area.
Such a lack of capacity to maintain buffer zones, combined with the realization that it will most likely not be possible to eradicate tsetse completely, puts into question the logic of containment. ‘Essentially we are growing to live with tsetse’, he added.
Following this realization that coexistence and connectivity are inevitable, and with an increasing number of parties looking for new ways to organize this, the clash between the logics of connectivity and containment is currently probed by large-scale conservation projects such as KAZA that are oriented towards creating and restoring lines of connection between various conservation areas. KAZA's coexistence approach, as such, provokes a de-territorialization of disease control. Signs of this can be found for both FMD and tsetse control.
Commodity-based trade (CBT), whereby animal products are processed in ways that ensure freedom from disease in the final product, has been promoted as an alternative to area-based FMD control (see Barnes, 2013; Naziri et al. 2015; OIE, 2015; SADC, 2016). New approaches to tsetse control are emerging, such as the recent development of collars fitted with tsetse repellents, which have been found to significantly protect cattle in tsetse-prone areas (Saini, 2020; Saini et al., 2017).
Certainly, diseases are here to stay, and so are vectors and host species. The main question on the table, then, is:
Producing and reducing complexity
The importance of scientific data was one topic that many research participants—veterinarians, conservationists, and government officials alike—brought up during the interviews, even though it had not been included in the topic lists that we used to guide the conversations. It became apparent that our interlocutors understood scientific knowledge as the prime way of handling the complexities and uncertainties of coexistence at the intersections of wildlife, livestock, pathogens, and humans. One veterinarian working for the OIE, for example, said: The bottom line is, we have to find a way, but guided by science. Instead of fighting, let's go and do science and come up with better ways of handling this.
The OIE itself is an organization that explicitly roots its legitimacy in its use of science, and that prides itself in using procedures for developing and updating trade standards that are ‘very flexible and allows for continuous improvement to standards as the supporting scientific information justifies it’ (OIE, 2006: 2). The advance of scientific knowledge, then, for this veterinarian, as for many of his colleagues, was fundamental to his line of work. He explained: I am a believer in growing science. In the last ten, twenty years there has been more new scientific research, and some of it has actually been able to influence the OIE standards. I think as more research is done, also in the meat value chain itself, maybe some of the processing methodologies can become better able to inactivate the [FMD] virus. So that things feel very safe. I think with science, as the years go by, we’ll see more improvement in the management of diseases, the control of diseases. So hopefully, researchers keep on working, and there will be more information coming.
Further, as several participants noted, there is a potential for advancing science to shed new light on old assumptions about disease transmission between wildlife and livestock. ‘In the past, if you found a particular disease in wildlife and then found it in livestock, we were quick to come up with this association’, a local government official, who was also a biologist by training. He elaborated that his constituents, like many of his colleagues in the local and national governments of the KAZA member states, commonly associated animal diseases such as FMD and trypanosomiasis with wildlife, but hesitated to comment on this relation himself. ‘I can't really talk about associating either environmental changes or land use changes with animal health’, he said, ‘because we've not done a specific study that actually addresses that connection. So, if I would say something it would be speculation rather than science to explain that there's an association between one and the other’.
Echoing this sentiment, an Australian veterinarian working for an international NGO, referred to her experience working with FMD control in Botswana, wondering aloud: We're trying so hard to keep the buffalo away from the cattle, but sometimes these buffaloes seem to get in contact with the cattle and they don't get FMD. But then these other ones seem to get in contact with one buffalo and they do get infected.
More fundamentally, however, she was concerned with the difficulty of acquiring this knowledge in the region – a concern shared by many research participants. They struggled with a perceived lack of data on the ecology and epidemiology of the KAZA TFCA and the difficulties in making science-based decisions in a region where comprehensive datasets are lacking. ‘This area is notoriously understudied’, commented a sociologist, while a veterinarian stressed that: We are working in an information-poor environment. In terms of getting a robust epidemiological picture, from a spatial epidemiological perspective, there's nowhere you can go to look at what outbreaks have happened in the last five years across this landscape.
Gaining more detailed information and narrowing down the scale at which relations are studied, down to the individual herd, animal, or even pathogen at a molecular level, as well as scaling up to cover the vast transboundary area as a whole, would potentially offer tools to revisit the trade restrictions and disease control measures in a more detailed manner. Indeed, advancing science may offer ‘new modes of noticing [as] new technologies are changing our ideas about the relationship between the micro and macro worlds by changing
However, ignorance, or a lack of knowledge, is only one form of uncertainty. As Scoones and Stirling (2020) argue, other forms of uncertainty also exist, namely in the form of ambiguity (knowing differently), and unpredictable future changes (not knowing what we don’t know). It is this ambiguity that was referred to when several of our interlocutors expressed their concern with the challenges of interdisciplinary knowledge exchange and of working according to One Health principles, even as they explicitly referred to themselves as advocates of One Health. Some pointed at the lack of social scientists involved in One Health programs, or the need to also involve (extractive) industries in the region in the conversation. The challenge here, it appears, is not so much rooted in the intricacies of multispecies coexistence, and a lack of knowledge of it, but rather in the difficulty of inter- and transdisciplinary communication and collaboration, and the uncertainty that emerges because of different understandings and approaches to the problem.
Discussion and conclusion
Coexistence landscapes are constituted not only by celebrated entanglements with ‘nature’, but also by sites of violent multispecies conflict and reconciliation (Kikon and Barbora, 2021), campaigns against disease-spreading arthropods (Reis-Castro, 2021) and the monitoring of ever-changing parasites, bacteria and viruses (Keck, 2020). The current scholarly and popular debate advocating better multispecies relations must, therefore, also find ways to incorporate a range of species and organisms that are considered pathogenic and antagonistic to human and nonhuman well-being – or, what Ginn et al. (2014) have termed ‘flourishing with awkward creatures’.
In this article, we have approached the wildlife–livestock interface not only as a ‘contact zone’, a site of interactions and relations between species (Haraway, 2008), but also a site where different understandings and enactments of multispecies coexistence meet, mingle and clash. These are expressed in scientific knowledge production, landscape development and international policy. Beyond identifying the wildlife–livestock interface as a site of human–wildlife conflict, then, we have focused on how humans, and more specifically, experts, scientists and policymakers, have made sense of and managed this coexistence: as a site of discursive and material human-to-human conflict. Multispecies coexistence, in this view, is characterized by a collision of multiple orderings, and infused with the moral imperative to preserve wildlife, ecosystems and landscapes, as well as the imperative to encourage development, secure livelihoods and protect human and animal health.
Here, we identified two modes of ordering of coexistence that both answer to these imperatives, albeit in different ways. The first, an ‘infrastructural’ mode of ordering, is focused on connectivity. It is characterized by a focus on movement and pathways of exchange, recognizing the unavoidable exchanges between species and seeking to channel these. It stresses the necessity of interconnection between ecosystems and materializes in the establishment of wildlife corridors and dispersal areas as infrastructures along which species are encouraged to move. In this, this logic is deeply aspirational: it aspires to a future where more connectivity and movement are made possible, wherein veterinary fences have become obsolete, and where ecosystems and communities thrive in coexistence. Yet, as others have argued, more connectivity in conservation is often accompanied by enforced attempts to control the mobility and activities of human communities (see, e.g. Ramutsindela, 2004, 2007). Furthermore, the logic of connectivity faces a socio-political reality in which the potential of disease transmission between wildlife and livestock, as well as the transmission of zoonotic diseases and other forms of human–wildlife conflict, has until now restricted the proper realization of this aspired future.
The second mode of ordering termed ‘territorial’, reflects the focus of its proponents on the maintenance of zones and the containment of distinct categories of species within these territories. Typically, it involves the separation of wild versus domestic species and activities. This logic materializes in fences, veterinary control points, trade restrictions, and administrative zones. It also resonates in governance structures, with wildlife management typically assigned to ministries of tourism or natural resources, and livestock being managed under ministries of agriculture and economy. The territorial mode of ordering, overall, appears conservative, aiming to retain what is, being protective of populations and markets considered ‘healthy’ or ‘clean’. This approach to biosecurity allows for substantial economic development in designated areas, as the beef industry in Botswana testifies. Yet, this may occur at the expense of ‘contaminated’ populations and zones, limiting opportunities for development in these areas.
The modes of ordering of containment and connectivity are accompanied by a third mode: one of categorical eradication. This logic has historically been prominent in various forms and at different times (i.e. the culling of trypanosomiasis-related wildlife populations in the 1930s, campaigns to eliminate tsetse flies in the 1960s to 1980s, and the introduction of sterile flies in the 2000s). However, eradication is premised on removing a singular agent from the landscape of multispecies relations, based on an understanding of these relations that is limited to the role of that one species in disease transmission, rather than a comprehensive approach to the ecological, and social network of relations that make up the landscape. Furthermore, in the current era where massive species extinction is a major concern, and where efforts at eradicating targeted species have failed consistently, eradication is increasingly disfavoured. This reflects an increasingly popular move towards
As our analysis suggests, framing wildlife and conservation areas as the source of livestock and zoonotic diseases does not do justice to the complexities of the coexistence between humans, livestock, and wildlife. In this, we agree with others that ‘it is urgent that we resist […] simplistic propaganda campaigns demonizing particular species and peoples’ (Skotnes-Brown, 2021: 496). Human expansion, through settlement and herding, into areas populated by wildlife, accompanied by deforestation, is likely to increase pressure on wildlife populations and increase the likelihood of wildlife–livestock contact (KAZA TFCA, 2016), and thus also to increase the risk of interspecies disease transmission. Hence, there is a pressing need for ‘finding an active, yet constructive role for humans in nature’ (Hourdequin, 2015: 19). This
In pursuing this moral imperative, it is generally accepted that animal health, public health and wildlife and ecosystem conservation would all benefit from better integration of programmes and aims – a position also shared by many of our interlocutors. With the emergence of One Health as a potentially holistic approach and an increasing interest in cross-disciplinary research, the future, on the one hand, characterized by potentiality (of contamination), and on the other hand by aspiration (of connectivity), offers opportunities for alignment of modes of ordering that merit further exploration, by policymakers, scientists, and practitioners (Osofsky et al., 2008). Animal health and conservation as we know it today initially developed as conjoined fields of knowledge and intervention (Keck, 2020; Skotnes Brown, 2020, forthcoming; Woods, 2004), but over the last century, they appear to have become increasingly antagonistic, each field withdrawing into its own academic subdisciplines, organizations, and ministries, forming, as one of our interlocutors put it, ‘silos within silos’. Reconciliation might be sought in better alignment of the modes of ordering that underpin contemporary interventions and policies, without presuming the moral superiority of one or the other.
However, while our interlocutors advocated the production of more detailed and up-to-date scientific data, this is, while crucial for informed decision-making, unlikely to alleviate tensions between the different modes of ordering at play. As Hourdequin has stated, ‘the idea that sound science and rigorous economic analysis are sufficient to resolve environmental controversies is too simple’ (2015: 4). And as Woods has also argued in the context of FMD in the UK, the suggestion that more scientific research would help improve control of the disease, may be too sanguine, especially if a ‘scientific view’ excludes the wider issues that have always shaped debates and, indeed, the scientific research itself. Additional investigations will not necessarily provide definitive answers to the problems of FMD prevention and control. […] The biology is but one component of an economic and social problem. (Woods, 2004: 150)
This discussion also reflects the typologies of uncertainty as outlined by Scoones and Stirling (2020) mentioned above. On one level, appeals to the production of scientific knowledge address uncertainties associated with knowledge gaps. These tackle the question of
This must have implications for policies of preparedness and risk mitigation. If uncertainty, at its most fundamental levels, cannot be controlled, and complexity cannot be tamed, it is better to learn how to live with it. In practical terms, since conservation is increasingly moving towards an infrastructural mode of ordering – the KAZA TFCA being a prime example of a conservation landscape reconceived not as a territory but as a network of zones and corridors – it might be worthwhile to reconsider also animal health, as materialized in veterinarian practices and state efforts to prevent (pan)zoonotic diseases, in infrastructural terms. What is the potential for networked, rather than territorial disease control? Can veterinarian practices begin to move along with the movement of wild and domestic species across landscapes, national borders, and continents, rather than seeking to confine species to disease-controlled spaces? And what is the potential for
Highlights
Considers disease transmission as a form of human–wildlife conflict.
Problematizes the disparity between a conservationist quest for connectivity and a veterinary quest for the containment of animal diseases.
Reviews the complexity of multispecies coexistence and the challenge it poses to the production of scientific knowledge on disease transmission.
Suggests the recognition of the partiality of knowledge as a requirement for learning how to live with pathogens, as well as the need for an alignment of modes of ordering.
Footnotes
Acknowledgements
We are grateful to all the interviewed experts who shared their knowledge and time in the midst of the COVID-19 pandemic. We would like to thank the three anonymous reviewers, whose in-depth comments and suggestions have greatly helped shaping our argument. Thanks also to all colleagues in CRC228: ‘Future Rural Africa’ for their support in the realization of this article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Agha, Bollig, Borgemeister, Junglen, Müller-Mahn and Van Dam received funding from the Deutsche Forschungsgemeinschaft (grant number CRC228: ‘Future Rural Africa'). Bollig and Van Engelen received funding from the European Research Council (grant number 101020976: ‘Rewilding the Anthropocene’). Views and opinions expressed are however those of the authors only and do not necessarily reflect those of the European Union or the European Research Council Executive Agency or the German Research Council.
