Abstract
Objective:
Meningiomas represent the most common intracranial extraaxial neoplasia in adults, accounting for a third of all diagnosed primary tumors of the brain. Despite decades of research, relatively little data on demographics of meningiomas exist. The aim of our study was to undertake an analysis of demographics and outcome of patients who underwent meningioma surgery over an 8-year time period at our institution.
Methods:
We reviewed 187 consecutive patients with histologically confirmed meningioma. Demographic data, tumor location and side, surgical resection grade, and histopathological and radiological data were collected and assessed for all patients. Furthermore, recurrence, morbidity, and mortality were evaluated.
Results:
Of the 187 consecutive patients undergoing meningioma resection over a period of 8 years, 131 (70.1%) were women (p < 0.001). Meningiomas were classified as World Health Organization (WHO) grade I, grade II, and grade III in 66.8%, 31.0%, and 2.1%, of the cases, respectively (p < 0.001). MIB-1 proliferation index was <1 in 7.5%, 1–5 in 52.9%, 6–10 in 22.4%, >10 in 11.8% of the lesions (p < 0.001). In 82.4% of the cases, gross total resection was achieved. Recurrence occurred in 23 patients (12.3%), while overall morbidity and mortality rate was 41.2% and 7.7%, respectively.
Conclusion:
Based on our results, women are more than twice as likely to be affected, and the peak age is between 60 years and 70 years. Recurrence rate in our cohort was relatively low when compared to the data in the literature. The diagnosis of WHO grade II meningiomas, 31% in our cohort, is increasing since the 2007 WHO criteria have been published.
Introduction
Meningiomas are the most common primary brain tumor in adults with an age-adjusted incidence rate of 7.61 per 100,000 individuals per year. 1 –3 These are lesions arising from meningothelial cells of the arachnoidal layer of the brain, mostly located in the skull base, the parasellar region or the cerebral convexities. 4 Most meningiomas are slow growing, classified as Word Health Organization (WHO) grade I, while atypical (WHO grade II) and anaplastic (WHO grade III) meningiomas show more aggressive behavior. 5 Although most meningiomas are histologically benign, depending on their location, they can cause neurological symptoms affecting the quality of life and even leading to death. 6 Mutations of the neurofibromatosis gene (NF2), female sex, increasing age, and high-dose ionizing radiation exposure are well-established risk factors. 6 Furthermore, an association between hormones (both endogenous and exogenous) and tumor occurrence has been suggested. 4 Despite high clinical relevance and decades of research, little data exist on the epidemiology and demographics of intracranial meningiomas. 5 Among others, this might be due to the long latency of meningioma recurrences, the fact that it is a relatively rare disease and that the prevalence of subclinical disease is up to 2.8% of the population according to autopsy studies. 6 –8
The goal of our study is to present recent demographic and outcome data of histologically confirmed intracranial meningiomas in a single-center cohort.
Materials and methods
We retrospectively reviewed 187 consecutive patients with a histologically confirmed diagnosis of meningioma between June 2008 and March 2015 at our institution. All patients were operated on using classic microsurgical techniques. Mean clinical and radiological follow-up time was 705.6 ± 559.0 days (range 1–2290 days). Demographic data, anatomical tumor location and side, surgical resection grade (Simpson resection grade 9 ) assessed through surgical reports, histopathological and radiological data, morbidity and mortality, and recurrence rates were collected and assessed for all patients.
All statistical analyses were done using SPSS Statistics version 21.0 (2012; IBM Corp., Chicago, Illinois, USA). Correlations were done using Pearson’s test; Fisher’s exact test or χ 2 test was used for contingency analysis, while all other calculations were done using the Mann–Whitney U test. A p value of <0.05 was considered significant.
The study protocol was approved by the local ethics committee (EKNZ, Basel, Switzerland). For this retrospective study, patient consent was not sought because the administrative burden would be disproportionately large, and therefore, consent is not necessary according to local ethics guidelines.
Results
Demographics
Of the 187 consecutive patients with histologically confirmed meningioma, 131 (70.1%) were women and 56 (29.9%) male (p < 0.001), leading to a male:female (m:f) ratio of 1:2.3 (Figure 1). The mean age was 61.5 years (±13.3 years), with 20 patients (10.5%) being younger than 45 years, 57 patients (30.5%) between 45 years and 59 years, 78 patients (41.8%) between 60 years and 74 years, and 32 (17.1%) patients older than 75 years (p < 0.001) (Figure 2).

Sex distribution and female:male ratio by age. n = number, y = years.
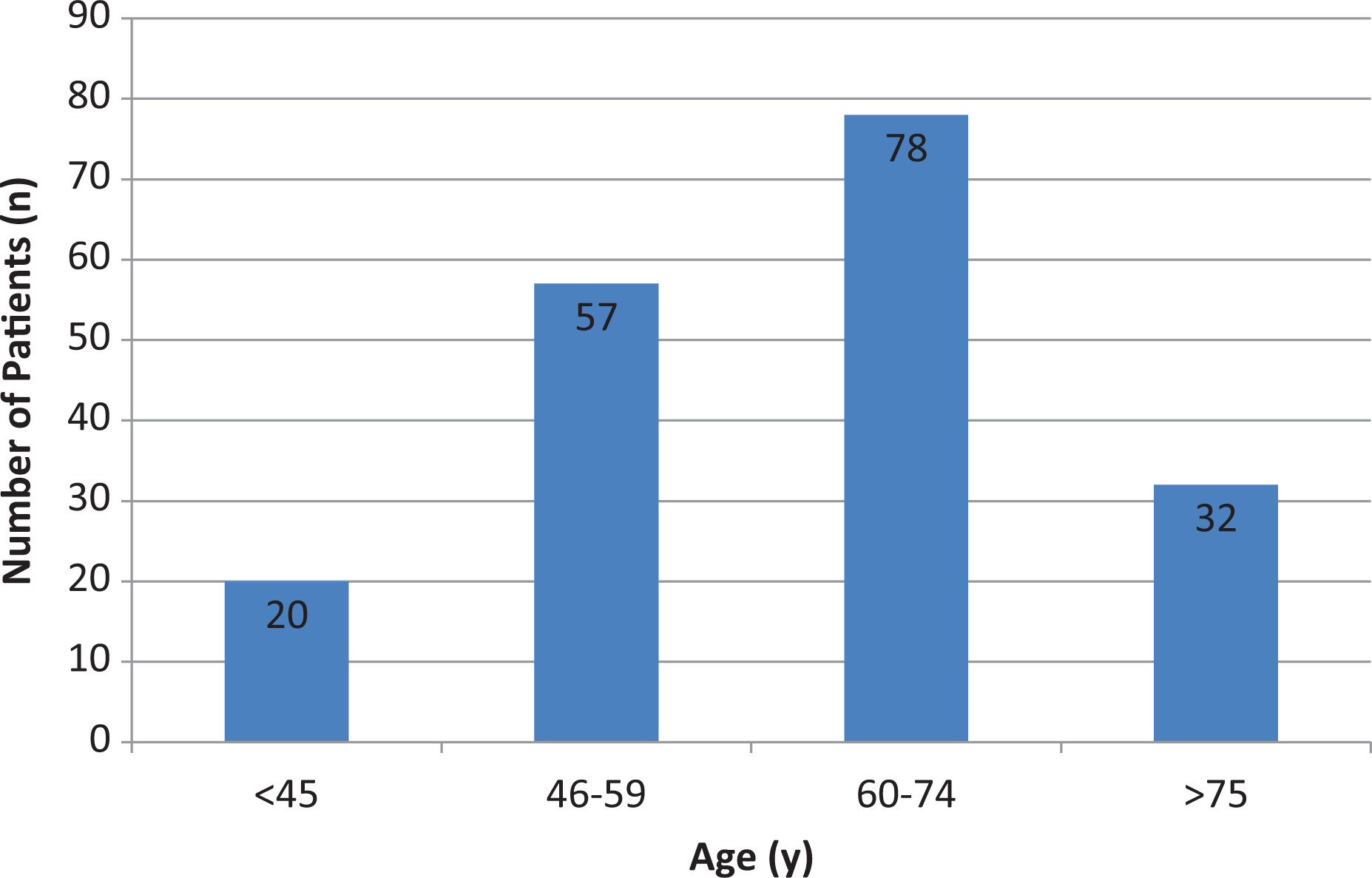
Age distribution. Amount of patients between the age of 46 years and 59 years differed significantly from those <45 years (p < 0.001) and >75 years (p = 0.008); amount of patients between 60 years and 74 years differed significantly from those <45 years (p = 0.01) and >75 years (p < 0.001). n = number, y = years
Location, histopathological findings, and resection grade
In 90 patients (48.1%), the lesion was located on the left side, in 77 patients (41.2%) on the right side, and in 19 patients (10.2%) at the midline (Table 1). The most frequent tumor location was at the convexity of the skull (37.4%), followed by the skull base (31.0%) and the falx (13.4%) (Table 1). WHO grade I, grade II, and grade III were seen in 125 patients (66.8%), 58 patients (31.0%), and 4 patients (2.1%), respectively (Figure 3). Of the WHO grade I tumors, the meningothelial subtype was the most common (58.3%), followed by the transitional (13.4%) and fibroblastic (10.2%) subtypes (Table 2). Molecular Immunology Borstel (MIB) 1 labeling index was <1 in 14 patients (7.5%), 1–5 in 99 patients (52.9%), 6–10 in 39 patients (20.9%), and >10 in 22 patients (11.8%) (Table 2). Simpson resection grades I, II, III, and IV were achieved in 95 patients (50.8%), 59 patients (31.6%), 6 patients (3.2%), and 24 patients (12.8%), respectively (Figure 4). Gross total resection (GTR; Simpson grade I and grade II) rates were significantly higher in patients presenting with non-skull base meningiomas (p < 0.001, Table 3). Higher WHO grade was significantly correlated with higher MIB 1 labeling index (0.68, p < 0.001), while no significant correlation was found between Simpson resection grade and WHO grade or MIB 1 labeling index. Three patients (1.6%) received the following adjuvant treatments: radiation, 1 case (multiple meningiomas at the convexity of the skull, WHO grade II, Simpson resection grade 1); combined treatment with radiation and Sandostatin, 1 case (falx meningioma, WHO grade II, Simpson resection grade 1); and CyberKnife, 1 case (convexity meningioma, WHO grade III, Simpson resection grade 2).
Location of meningiomas.

n: number.
a Sphenoid ridge, planum sphenoidale, tuberculum sellae, optic nerve, olfactory groove, petroclical/petrosal bone.

Distribution of World Health Organization (WHO) grade.
Histology of meningiomas.

n: number.

Distribution of Simpson resection grade.
Simpson resection grade and tumor localization.

n: number.
Outcome
Overall recurrence rate was 12.3% (n = 23). Recurrence rates did not differ when comparing supratentorial lesions with skull base or infratentorial lesions (Table 4). Recurrence rate was significantly influenced by WHO grade (p < 0.001), MIB 1 label index (p < 0.001), and Simpson resection grade (p < 0.001). Surgical complications occurred in 34 patients (18.2%), while systemic and infectious complications were present in 17 patients (9.1%) (Table 5). Bleeding events were seen in 11 patients (5.9%), and in 2 cases, revision surgery was required. (Table 5). Thromboembolic or cardiac complications occurred in 15 patients (8%). (Table 5). Postoperatively, 37 patients (19.8%) developed new neurological deficits, while in 8 of them a complete recovery was seen. Patients with Simpson resection grade III and grade IV had significantly higher rates of new neurological deficits compared to Simpson resection grade I (47.7%, n = 14) and grade II (14.9%, n = 23), respectively (p = 0.006). The occurrence of new postoperative neurological deficits in patients with skull base meningioma was not higher in patients with GTR (42.9%, n = 6) as opposed to those with STR (57.1%, n = 8; p = 0.75). Moreover, there was no significant difference between occurrence of new neurological deficits in patients with skull base meningioma compared to other tumor locations (p = 0.57). Overall mortality rate at follow-up was 7.7% (n = 4). Two patients died from systemic or cardiovascular complications, while one patient developed a brain abscess and one patient had a severe epileptic seizure with consecutive asystole.
Recurrence and tumor localization.

n: number.
a Sphenoid ridge, planum sphenoidale, tuberculum sellae, optic nerve, olfactory groove, and petroclical/petrosal bone.
Morbidity.
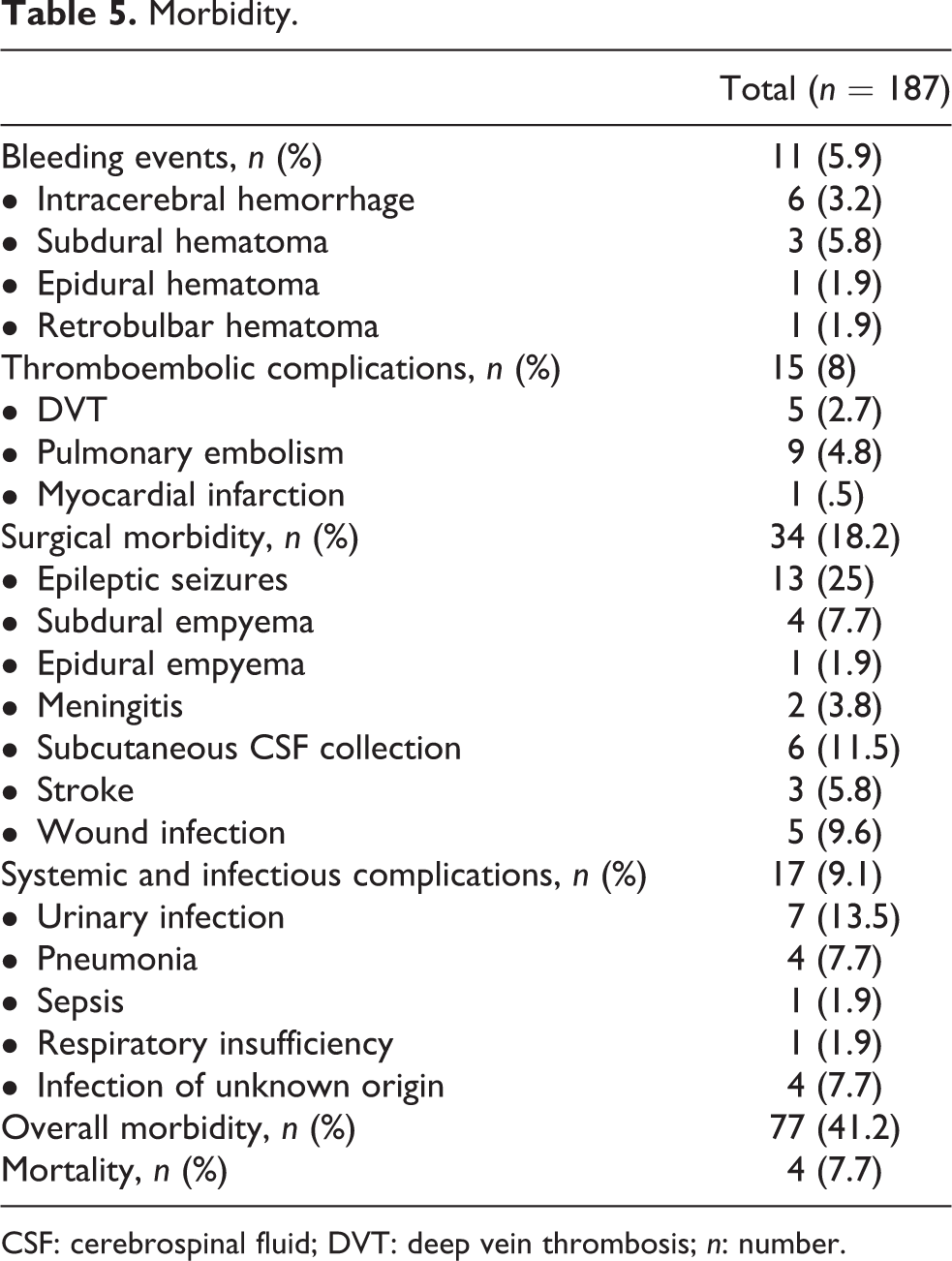
CSF: cerebrospinal fluid; DVT: deep vein thrombosis; n: number.
Discussion
Intracranial meningiomas represent approximately 20% of all intracranial neoplasms. 4,10 Despite of the relative high prevalence of meningiomas, only few studies describe their epidemiology, analyze the demographics, and investigate the risk factors for developing such lesions. 5,6,11,12 In our single-center cohort study with 187 consecutive patients undergoing intracranial meningioma surgery, the occurrence of meningiomas was higher with increasing age. Our findings are consistent with the data in the literature which shows increased risk of meningioma in the elderly. 6,13 However, while in the literature the peak incidence is described as the seventh and eighth decade of life, 14 in our cohort, meningiomas most often (41.8%) occurred between the age of 60 years and 74 years.
It is widely believed that the incidence among females is approximately twofold than in men, 2 analogous to our data. Women in our cohort were more likely to be diagnosed with a meningioma than men (m:f = 1:2.3). Interestingly, the m:f ratio was even higher in patients younger than 45 years (1:3) and those between 60 years and 74 years (1:2.7). Trying to find an explanation for the predominant meningioma incidence in females, a multitude of studies focused on its possible association with hormones, showing mostly controversial results. 15 –19 On the other hand, strong evidence exists, indicating that benign meningiomas express progesterone receptors (60–90%), 20,21 which has been also shown to be an indicator for recurrence. 20 While female sex hormones might be the reason for the highest meningioma incidence in younger women (<45 years), among our patients, the second highest peak incidence, namely between 60 years and 74 years cannot be sufficiently explained by hormonal influence.
The distribution of WHO grading in our cohort (WHO grades I, II, and III in 66.8%, 31.0%, and 2.1%, respectively) was consistent with the literature, where the majority of these tumors are considered histologically benign. 14 However, with adaptation of the 2007 WHO criteria, the rate of WHO grade II meningiomas has increased. 22 The rate of WHO grade II meningiomas in our study was 31%, which is comparable to recently published data. 22 When analyzing the meningioma grade II group, we noticed that according to the old WHO criteria 32 cases (55%) would have been graded as meningioma grade I, which would have lowered the rate of WHO grade II meningiomas to 13% (n = 26). In addition, when looking at these 32 cases, 12 suffered from recurrences (37.5%). Taken altogether, the recurrence rate of WHO grade I meningiomas according to the old WHO criteria would have been 12.1% instead of 5.6% when applying the new WHO criteria, leading to a nonsignificant difference in the recurrence rate between WHO grade I (12.1%) and grade II (8.3%) meningiomas. More so, WHO grade II meningiomas would show a tendency toward lower recurrence rates than WHO grade I meningiomas. These findings underline the validity of the new WHO criteria, which take into account brain infiltration and proliferation rate, among others. In our study, higher MIB 1 labeling index was associated with higher WHO grades. In the literature, too, a correlation between MIB 1 labeling index and grade of malignancy has been reported, since atypical and anaplastic meningiomas express a higher proportion of proliferative cells. 23,24 Even after GTR, a recurrence rate ranging from 10% to 32% within 10 years has been reported. 24,25 It seems that high MIB 1 labeling index shows strong association with recurrence rates and has to be taken into account, although not yet implemented as a formal WHO criteria for grade II or III meningiomas. According to the Simpson grading system, the extent of surgical removal represents one of the most significant indicators for meningioma recurrence. 9 In our cohort, 84% GTR was achieved with a relatively low overall meningioma recurrence rate of 12.3% (n = 23).
Although meningiomas are considered histologically benign lesions, they might strongly influence morbidity and mortality. 6 In our cohort, too, overall morbidity was high, reaching up to 41.2%. This emphasizes the fact that surgeons have to carefully take into consideration factors such as size, tumor location, and the preoperative condition of each patient when planning surgical resection. Especially when it comes to deep vein thrombosis (DVT) and pulmonary embolism (PE), an incidence ranging up to 32% has been reported in the literature. 26 However, in our cohort, these incidences were relatively low: 4.8% and 2.7% for PE and DVT, respectively. This might be due to the fact that all of our patients were mobilized early and received compression stockings and perioperative low-molecular-weight heparin. As it comes to neurological morbidity, studies have reported that approximately one-third of the patients with benign meningiomas had stable or worse neurological symptoms postoperatively, two-third suffered long-term neurological impairment, and a quarter were clinically disabled. 27 We observed new neurological deficits in 19.8% of the patients, while in 4.3% no recovery was seen. Interestingly, patients with STR had significantly more new neurological deficits compared to those with GTR. One possible explanation for this might be the tumor location. In eloquent regions associated with important structures at risk, surgeons operate less radically, while surgery-related neurological deficits are more likely to occur than in non-eloquent areas.
Mortality occurred in 7.7% of the patients during a mean follow-up period of 705.6 ± 559.0 days. These findings are slightly higher than those described in the literature (4% within 30 days 28 and 2% within 3 months 27 ).
Limitations
This retrospective study is subject to all the limitations of data collection inherent in such work. We included only histologically confirmed meningiomas which obviously lead to a detection bias. However, this problem is almost unavoidable, since many asymptomatic meningiomas are only detected through autopsy. Other meningiomas detected by coincidence and which are followed up by serial imaging without showing significant growth were also not included in this study, since the relevance of these meningiomas for clinical demographic and outcome studies is questionable. It is known that meningiomas can recur even after a period of 10 years. 29 Due to our relatively short follow-up period in some patients, the rate of recurrences might be higher than described.
Conclusion
In our cohort, the peak of occurrence of meningiomas is earlier than described in the literature, namely between 60 years and 74 years, and women were more often affected than men (ratio 2.3:1). Although meningiomas are mostly benign tumors, morbidity and mortality rates remain high. The rate of WHO grade II meningiomas is increasing since the 2007 WHO criteria were published, while in our cohort, 31% were WHO grade II meningiomas. Recurrence rate in our cohort was relatively low (12.3%) when compared to the data in the literature.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
