Abstract
Plain Language Summary
Generalized pustular psoriasis (GPP) is a skin condition that causes blisters filled with pus over large areas of the body. People living with GPP have painful flareups, which can be life-threatening if they are not treated. GPP seriously affects quality of life, even when the person is not currently having a flareup. Right now, there is only one medicine, spesolimab, that is approved to treat GPP. However, researchers and doctors still do not understand the best ways to treat GPP flareups over the long term or what advice to give people living with long-term GPP. This report gives an overview of current research on GPP and what still needs to be studied to help people with this condition. This report also explains the results of a recent study called EFFISAYIL® 2. The study showed that one year of spesolimab treatment reduced the number of flareups in people with GPP. Research still needs to be done on how to help people with GPP at all times, how to prevent flareups from coming back, and how to control the disease over a long period of time.
Introduction
Generalized pustular psoriasis (GPP) is a rare chronic inflammatory skin disease characterized by persistent symptoms and sudden flares of painful sterile pustules, which may be accompanied by systemic inflammation. 1 The clinical course of GPP is unpredictable as flares may occur due to a particular trigger or without any evident cause. 1 Patients with GPP often experience significant chronic disease burden even in the absence of flares, including ongoing symptoms such as scaling and erythema.2-5 GPP is also associated with persistent effects on quality of life (QoL) and a high prevalence of chronic comorbidities.6-9 GPP flares can be life-threatening if left untreated. 5 The impairment of the functional skin barrier can result in the percutaneous loss of fluids, nutrients, and electrolytes, culminating in multisystem organ failure. 10 Patients with GPP flares are also at greater risk for sepsis due to the compromised defensive barrier functions of the skin. 10 Therefore, continuous treatment to prevent flares is critical.
The approval of spesolimab, a first-in-class, humanized, selective antibody that targets interleukin-36 receptor signaling, represented a milestone for the management of GPP. 11 It is the first approved treatment for GPP evaluated in statistically powered, randomized, placebo-controlled clinical trials (EFFISAYIL™ 1 and EFFISAYIL® 2).12,13 To date, intravenous (IV) spesolimab has been approved by regulatory authorities in 48 countries, including the United States, Japan, China, and the EU, to treat GPP flares in adults.11,14-16
However, current GPP guidelines do not address chronic disease management, including how to maintain long-term control of GPP and prevent future flares.3,17,18 This results in uncertainty regarding how to approach management of GPP after flare treatment. Here, we present an overview of current literature describing the chronic disease burden of GPP, with a focus on current unmet needs, and a discussion of future directions for long-term GPP management. We also present a review of EFFISAYIL® 2, which was the first randomized, placebo-controlled clinical trial to systematically evaluate the long-term treatment of GPP between flares with spesolimab as a subcutaneous (SC) injection.
Review of Current Literature
GPP is a Chronic Persistent Disease
GPP presents as a chronic persistent disease with episodic recurrent flares (Figure 1).19,20 In a survey of 29 dermatologists experienced in GPP, 83% (24/29) agreed that patients experience continued disease burden post-flare, with symptoms such as skin scaling (76%), skin lesions (66%) and erythema (66%) commonly requiring up to 3 months to resolve.
5
This is consistent with a retrospective multicenter study where 74% (63/86) of patients had GPP symptoms at the time of their last visit, while another study found that 34% (32/95) of patients had persistent pustular lesions or erythematous thin plaques despite the use of off-label systemic therapies.2,7 Likewise, 40% (6/15) of patients in the placebo group of EFFISAYIL® 2 who were not experiencing a flare had ≥1 GPP Physician Global Assessment (GPPGA) total score of 2 (ranking of severity from 0-4) over the trial period.
21
GPP is a chronic disease. CVD, cardiovascular disease; GPP, generalized pustular psoriasis; PsO, psoriasis; QoL, quality of life; T2D, type 2 diabetes.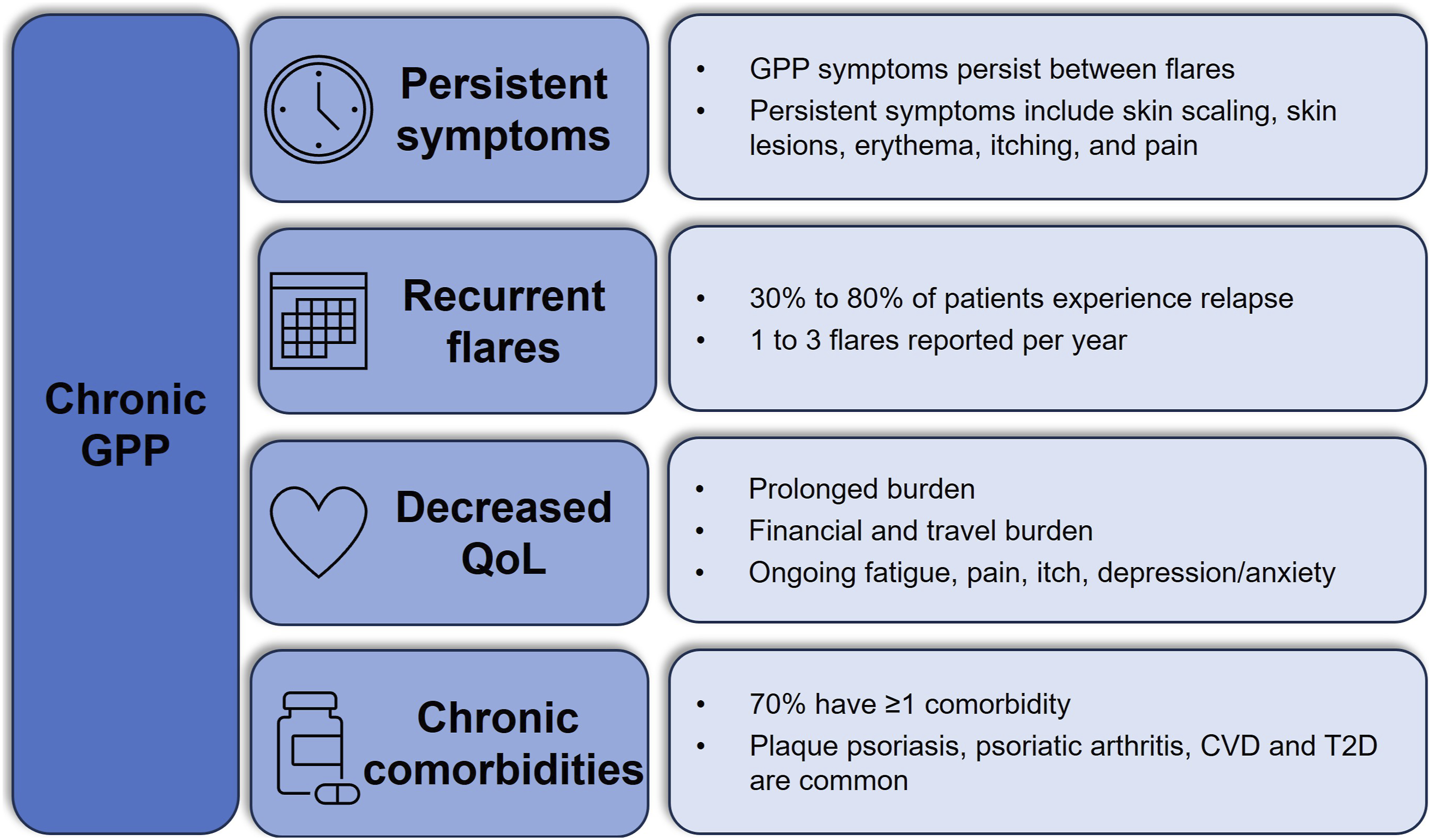
Patient-reported outcomes also reflect persistent disease in GPP. In an online survey of 66 patients with GPP in the United States, 24% (16/66) and 6% (4/66) reported symptoms of moderate and high severity, respectively, even when GPP flares were under control. 4 Approximately one-quarter of survey participants (27%, 18/66) identified as disabled. 4 Chronic symptoms of GPP, such as itching and pain, interfered with patient perception of treatment success. 22 In a cohort study that included 7 patients with GPP, patient-reported treatment satisfaction was significantly affected by ongoing disease severity. 22
Recurrent GPP Flares
Many patients with GPP experience periodic flares which can last over 3 months (Figure 1).2,9,23,24 While most patients experience an average of 1 flare per year, surveys have shown some patients experience 2-3 flares per year.4,5,25,26 Triggering factors for GPP flares are reported in 41%-85% of cases,1,7,27 though not all triggers are identifiable or avoidable, highlighting the need for consistent disease management to mitigate flare occurrence. Flare triggers can include medications (i.e., vaccines, non-steroidal anti-inflammatory drugs, antibiotics, and hydroxychloroquine), infections (bacterial and viral), pregnancy, menstruation, and corticosteroid withdrawal, with some evidence for flares triggered by seasonal changes, stress, and hypocalcemia.1,3,6,28-38 However, cases of GPP flares may also be idiopathic. 39 A recent review of real-world evidence (RWE) reported there was no identifiable cause for GPP flares in 15%-62% of patients. 6
Quality of Life
The chronic nature of GPP and prevalence of persistent symptoms detrimentally impacts patient QoL (Figure 1). A review of RWE on the humanistic burden of GPP reported a mean Dermatology Life Quality Index (DLQI) score from 7.8-12.4, indicating moderate to very large effects on QoL. 6 GPP can negatively impact a variety of physical, mental, and financial factors. 40 For instance, substantial costs may be associated with traveling long distances for specialist care, obtaining specialty medications, and undergoing long-term treatment. Patients may also face barriers to treatment due to misdiagnosis or prolonged insurance authorization procedures. Chronic symptoms such as fatigue, pain, and itching also contribute to poor QoL, and the debilitating anxiety or depression stemming from the unpredictability of GPP flares can affect patients’ mental and emotional well-being. When surveyed, patients reported that GPP impacted their daily lives even when flares were absent, with 36% reporting prolonged disease burden for months and 38% reporting the burden for years due to delays in diagnosis. 4 Furthermore, several respondents felt their physician did not understand the psychological and emotional or physical pain caused by their GPP. 4
Comorbidities
Prevalence of Most Common Comorbidities in Patients With GPP.
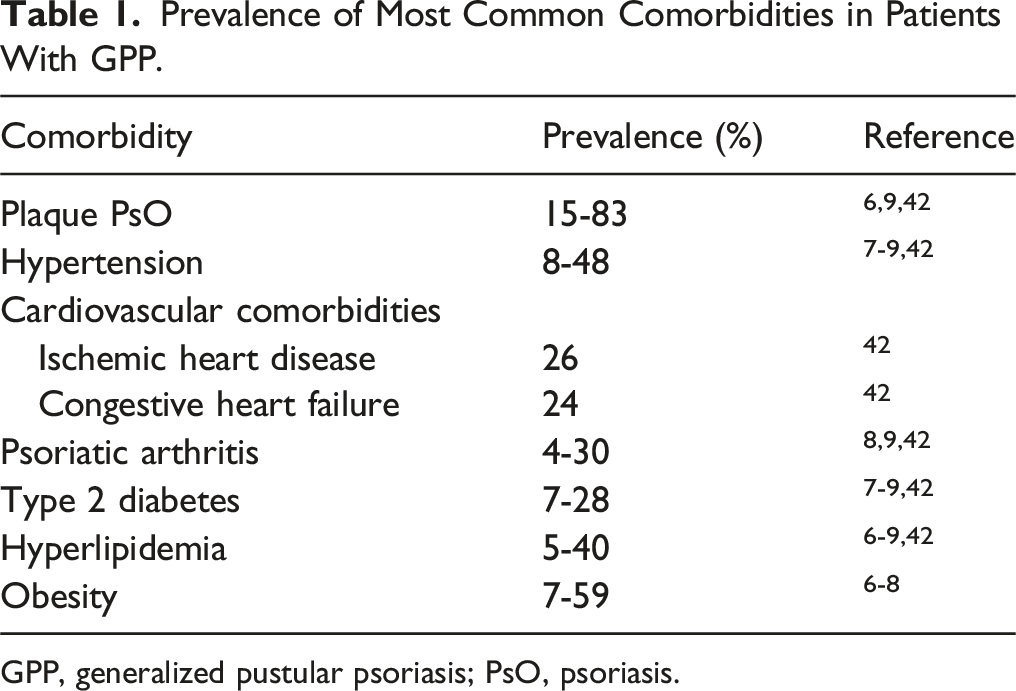
GPP, generalized pustular psoriasis; PsO, psoriasis.
Furthermore, patients experiencing flares often require more frequent and prolonged hospitalization. In a Japanese retrospective cohort study, patients with GPP (N = 110) were more likely to require inpatient hospitalization compared to patients with plaque PsO (N = 20,254) or the general population (N = 436), with frequencies of 25% vs 6% vs 5%, respectively. 43 A French population-based study showed a median duration of stay of 8 days in the hospital and 12 days in the intensive care unit among 4195 patients with GPP. 42
Unmet Need For Chronic GPP Management
Prior to the availability of spesolimab, there was an unmet need for safe and effective treatment strategies for long-term control of GPP.44,45 Non-biologic systemic agents (i.e., retinoids, methotrexate, and cyclosporine) were often used to treat GPP flares, while several biologics gained approval for GPP in Japan based on small studies without validated measures for endpoints, and were used off-label in other countries.46,47 Patients were often switched from agent to agent, indicating a lack of evidence-based guidelines to manage GPP long-term. 48 Evidence from 2 US databases (Optum Clinformatics Data Mart and IBM MarketScan Commercial) from 2015-2020 showed that most patients did not receive systemic therapy within the first month of diagnosis, and treatments changed frequently over time, with low adherence over 2 years.49,50
Moreover, many non-GPP-specific treatments are associated with substantial toxicities that deter long-term off-label use. 51 Cyclosporine, while viable as a short-term therapy option, is associated with nephrotoxicity and hypertension with prolonged use. 52 Prolonged therapy with methotrexate can lead to liver and hematological toxicity, 52 and retinoids are associated with liver toxicity, alopecia, diffuse skeletal hyperostosis, and osteoporosis. 53 Both methotrexate and retinoids are contraindicated in pregnancy and should be used with caution in individuals of child-bearing potential.53,54 Methotrexate can also impact fertility in both sexes. 54 Withdrawal of systemic corticosteroids may trigger GPP flare, and the prolonged use of topical corticosteroids (particularly those of high potency) is associated with adrenal insufficiency, Cushing’s Syndrome, and osteoporosis.55,56 Prior to EFFISAYIL® 2, aside from some case reports and open-label studies, data were limited regarding the long-term management of chronic GPP.57-67
Discussion of EFFISAYIL® 2
EFFISAYIL® 2 was the first randomized, placebo-controlled clinical trial in which GPP flare prevention was systematically evaluated.
13
The aim of the study was to assess the efficacy and safety of SC spesolimab for GPP flare prevention over 48 weeks. Patients with a history of GPP were randomized 1:1:1:1 to 3 groups receiving a SC dose of spesolimab (300 mg every 4 weeks [q4w] after a 600 mg loading dose; 300 mg every 12 weeks [q12w] after a 600 mg loading dose; and 150 mg every 12 weeks [q12w] after a 300 mg loading dose), and 1 group receiving placebo q4w (Figure 2(a)). Endpoints included time to first GPP flare by week 48, and the occurrence of ≥1 flare by week 48. EFFISAYIL® 2 study design (A) and likelihood of flare up to week 48 when treated with spesolimab versus placebo (B).
14
. CI, confidence interval; GPP, generalized pustular psoriasis; HR, hazard ratio; q4w, every 4 weeks; q12w, every 12 weeks. aNot significant. bNot tested (statistical significance was not seen in previous families in the statistical testing hierarchy).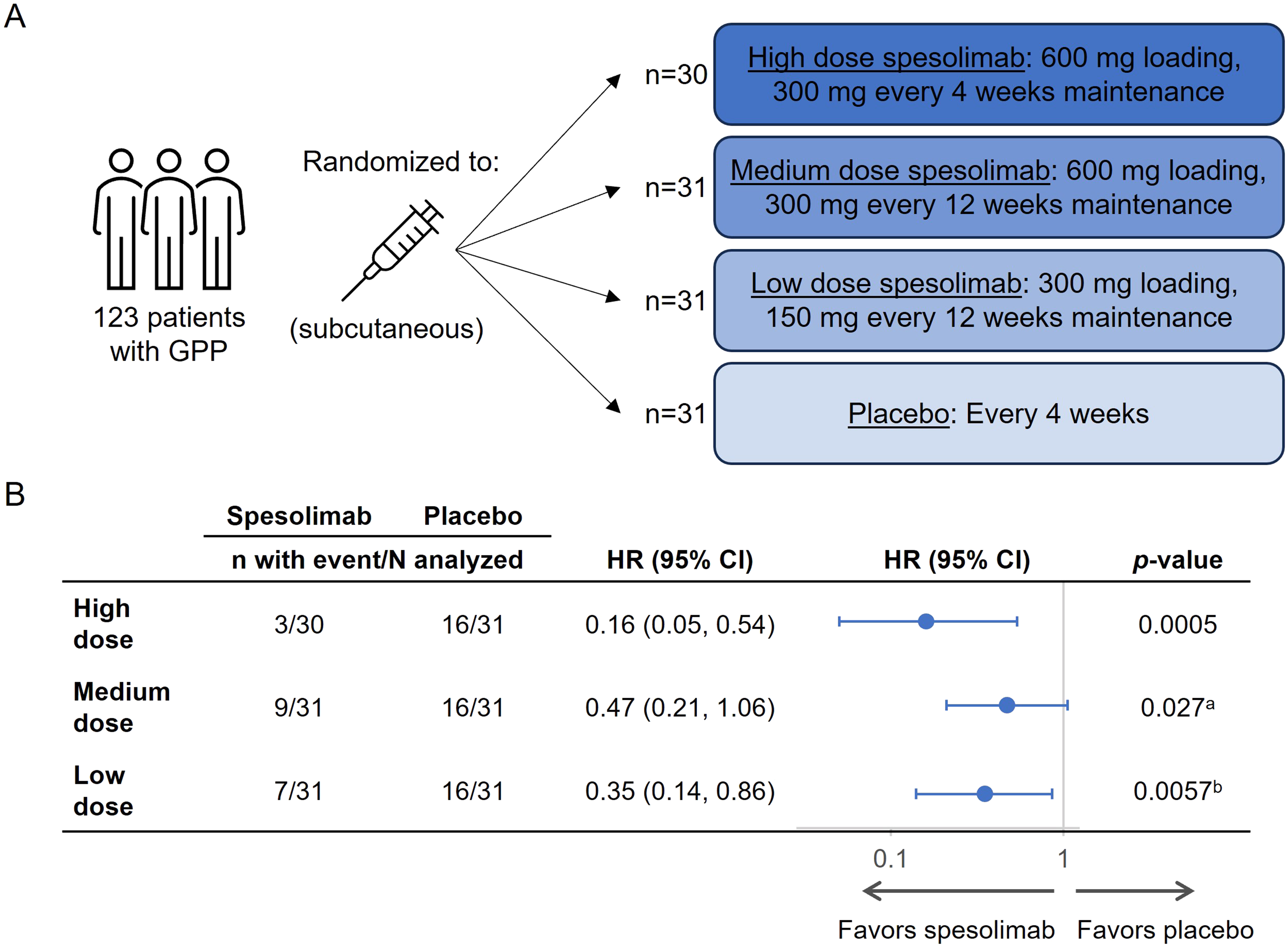
A total of 123 participants were randomized (300 mg q4w, n = 30; 300 mg q12w, n = 31; 150 mg q12w, n = 31; placebo, n = 31). Patients were either Asian (64%; 79/123) or White (36%; 44/123), 62% (76/123) of patients were female, and the mean age was 40.4 years (SD, 15.8). At randomization, patients were required to have a GPPGA total score of 0 or 1, and 86.2% had a GPPGA total score of 1 indicating some skin severity at baseline. The treatment groups were comparable regarding baseline characteristics and had a similar mean GPPGA total score. IL36RN mutation occurred in 13% (4/31) of the placebo group, vs 23% (7/31) of the 150 mg q12w group, 32% (10/31) of the 300 mg q12w group, and 23% (7/30) of the 300 mg q4w group.
The 300 mg q4w spesolimab group showed significant improvement vs placebo in time to GPP flare (hazard ratio [HR], 0.16; 95% CI: 0.05-0.54; P = 0.0005) (Figure 2(b)). HRs were 0.35 (95% CI: 0.14-0.86; nominal P = 0.0057) in the 150 mg q12w group, and 0.47 (0.21-1.06; P = 0.027) in the 300 mg q12w group. By week 48, 35 patients had GPP flares: 23% (7/31) of patients in the 150 mg q12w group, 30% (9/31) in the 300 mg q12w group (exposure-adjusted), 13% (3/30) in the 300 mg q4w group (exposure-adjusted), and 52% (16/31) in the placebo group. All flares in the 300 mg q4w spesolimab group occurred by week 4 following the first 300 mg SC dose at week 4. A non-flat dose-response relationship was shown for all 3 doses of spesolimab compared with placebo, with statistically significant P-values for each predefined model (linear P = 0.0022, emax1 P = 0.0024, emax2 P = 0.0023, and exponential P = 0.0034). Additionally, using a one-sided α of 0.0063 (adjusted for multiplicity), 300 mg q4w spesolimab showed significant improvement compared with placebo in occurrence of ≥1 flare by week 48 (P = 0.0013).
Adverse Events in the Safety Population of EFFISAYIL® 2. 14
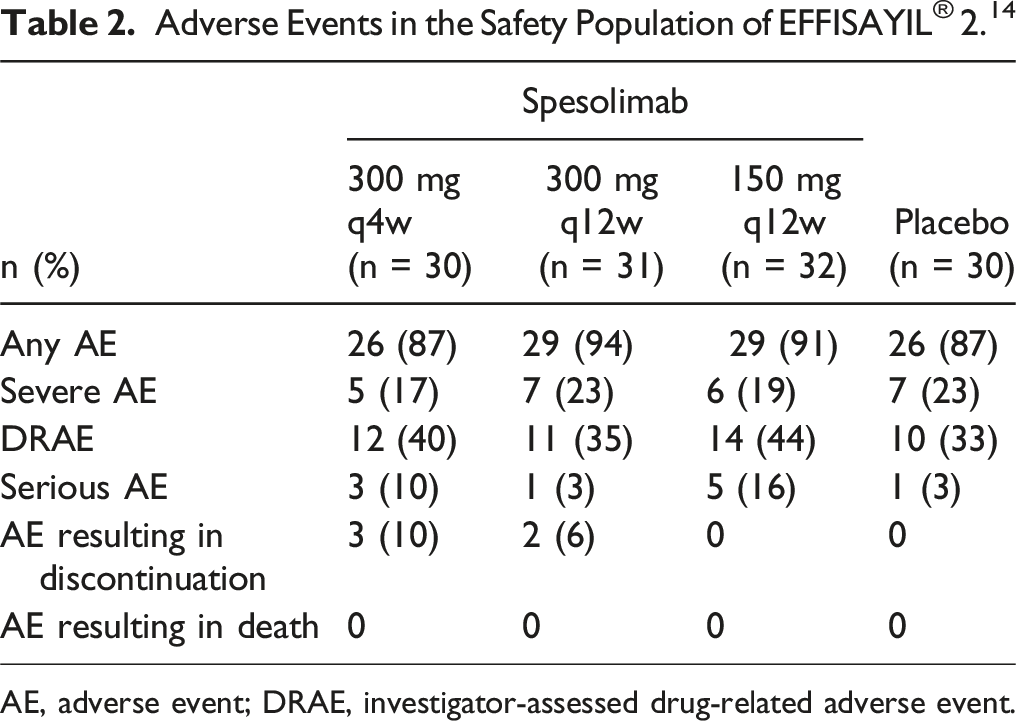
AE, adverse event; DRAE, investigator-assessed drug-related adverse event.
Subgroup analyses of EFFISAYIL® 2 demonstrated consistent efficacy of spesolimab in preventing flares in patients with and without IL36RN mutation, with the presence or absence of plaque PsO at baseline, and in all body mass index categories. 68 There were rapid improvements in DLQI among patients who received 300 mg q4w spesolimab compared with placebo, and this effect was sustained through week 48. 69
Evidence suggested that study participants were experiencing chronic GPP burden prior to treatment with spesolimab. Patients exhibited underlying GPP disease activity at baseline despite receiving biologic therapies, systemic therapies, or both. 13 In a subgroup analysis of 15/31 placebo-group patients who did not experience a flare over the study duration, a DLQI score of moderate (6-10) or very large (11-20) impact on QoL was reported in 67% (10/15) and 40% (6/15) of patients, respectively, during ≥1 study visit. 21 Moreover, 40% (6/15) of placebo-group patients who did not experience a flare had a GPPGA total score of 2 (ranking of severity from 0-4) during ≥1 study visit, with 4 patients reporting a score of 2 at ≥4 visits, and several patients reporting a moderate (7/15) or severe (3/15) score on the pain Visual Analog Scale during ≥1 study visit. 21 These data provide further evidence that GPP negatively affects patients who are not currently experiencing a flare or receiving adequate long-term management. The main limitation of EFFISAYIL® 2 was its limited participant diversity, as study participants were either Asian or White and predominantly female. Evidence from more patients and for a longer duration is needed to determine the long-term effects of continuous management with spesolimab in GPP. Overall, the results from EFFISAYIL® 2 confirmed that spesolimab was effective and safe to treat patients with GPP.
Conclusion
GPP is a chronic disease characterized by persistent disease burden, recurrent flares, high incidence of chronic comorbidities, and enduring impact on patient QoL. Thus, GPP requires a long-term continuous management approach. Improved recognition among healthcare providers of GPP as a chronic disease and a multidisciplinary treatment strategy are needed. Now that spesolimab is an approved IV and SC treatment for patients with GPP, more data are needed on the outcomes of multi-year spesolimab treatment, including the results of the EFFISAYIL® 2 open-label extension study. 70 Additionally, more information is needed regarding the effects of long-term management of GPP on patient QoL. The disease burden of GPP affects most patients beyond the duration of flares, and there remains a need for guidelines on how to manage chronic GPP, including flare prevention and treatment of sustained symptoms. Data from EFFISAYIL® 2 demonstrated that 300 mg q4w SC spesolimab after a 600 mg loading dose significantly reduced the risk of flare occurrence over 48 weeks vs placebo and had a reassuring safety profile, leading to FDA approval to treat GPP when not experiencing a flare in adult patients and pediatric patients aged 12 and over and weighing at least 40 kg.
Expert Opinion
A major unmet need in GPP is the establishment of guidelines for a multidisciplinary approach to the long-term management of GPP, including standardized measurements to assess disease severity, QoL and pain, flare treatment, flare prevention, and management of comorbid conditions. This review demonstrates that chronic management strategies should mitigate both the persistent disease burden patients experience between flares as well as the potentially life-threatening nature of GPP flares. It is important for GPP to be treated using FDA-approved medications because of its impact on QoL, high morbidity, and high mortality rate. Long-term management with SC spesolimab is a new FDA-approved strategy to treat GPP when not experiencing a flare, which could improve QoL and reduce or improve comorbidities in patients who experience recurring flares and chronic disease burden. Continuous management of GPP may also prevent hospitalization and other serious, potentially life-threatening consequences of flares. Unfortunately, access to treatment may be limited by barriers associated with insurance and the healthcare system. The establishment of official guidelines that recommend therapy for long-term management of GPP could help ease the logistical burden that causes delays in accessing treatment.
In addition to access to FDA-approved therapies, early detection and diagnosis of GPP are vital for providing timely treatment. The rarity and unpredictability of GPP flares and lack of awareness of standardized diagnostic criteria for GPP are obstacles to prompt diagnosis and treatment in settings where patients are most likely to present with flares (i.e., non-dermatologist settings such as the emergency department and the hospital). 71 Patients are frequently misdiagnosed with acute infection until negative cultures are confirmed, delaying flare treatment and underscoring the need for widespread physician education on the clinical appearance of GPP. Recent consensus statements from the International Psoriasis Council (IPC) and the National Psoriasis Foundation have established standardized diagnostic criteria for GPP, and both indicate that the potentially life-threatening nature of GPP necessitates immediate treatment.17,72 The IPC identified the primary clinical diagnostic criteria as “macroscopically visible sterile pustules on erythematous base and not restricted to the acral region or within psoriatic plaques.” Because ensuring timely access to FDA-approved therapies is critical to reducing morbidity and mortality during GPP flares, diagnosis should be made without relying on biopsy,17,72 nor should treatment be delayed by tuberculosis testing. 73 Currently, spesolimab is the only medication approved in the US to specifically treat GPP, based on its efficacy and safety profile. GPP flares may be treated with a single 900 mg IV infusion with an optional second infusion 1 week afterwards for persistent flare symptoms, and GPP may be treated while not experiencing a flare with a 600 mg SC loading dose followed by 300 mg SC injections q4w.11-13
For patients to receive optimal treatment, GPP must be recognized by dermatologists and physicians of all specialties as a chronic disease. Further long-term research and follow-up with patients with GPP will allow for better understanding of the chronic pathophysiology of GPP and may help identify additional flare triggers and how best to avoid them. More research on the long-term effects of GPP treatment is also needed, including a multi-year follow-up of patients taking spesolimab for long-term management, as well as other therapies.
The approval of SC spesolimab will enable patients to self-administer maintenance doses of the medication at home and alleviate the burden of traveling to clinics for IV infusions. Patients with GPP should have easier access to long-term therapies that relieve persistent symptoms and ease the anxiety and QoL burden associated with GPP. Ongoing and future studies should emphasize that GPP is a chronic disease associated with multiple comorbidities, and treatment guidelines should target the treatment of GPP both chronically and during flare episodes, as well as outline stepwise therapy approaches for the management of patients with and without chronic disease burden.
Footnotes
Acknowledgements
The authors met criteria for authorship as recommended by the International Committee of Medical Journal Editors (ICMJE). The authors did not receive payment related to the development of this manuscript. Boehringer Ingelheim Pharmaceuticals, Inc. (BIPI) was given the opportunity to review the manuscript for medical and scientific accuracy as well as intellectual property considerations. Jia Gao, PharmD, of Elevate Scientific Solutions LLC, provided medical writing, editorial support, and formatting support, which were contracted and funded by BIPI.
Author Contributions
BE and MGL contributed to conception of this review article, interpretation of data, and writing the manuscript. All authors reviewed and approved the final version of the manuscript.
Declaration of Conflicting Interest
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: BE is an investigator for AbbVie, Amgen (previously Celgene), AnaptysBio, Bausch Health (formerly Valeant Pharmaceuticals), Boehringer Ingelheim, Bristol Myers Squibb, Eli Lilly, Incyte, LEO Pharma, Menlo, Merck, Novartis, Pfizer, Regeneron, Sun Pharmaceutical Industries, UCB, and Vanda; and is a consultant for Amgen (previously Celgene), Arcutis, Bausch Health (formerly Valeant Pharmaceuticals), Boehringer Ingelheim, Bristol Myers Squibb, Eli Lilly, LEO Pharma, Novartis, and UCB. MGL is an employee of Mount Sinai and receives research funds from: Abbvie, Amgen, Arcutis, Avotres, Boehringer Ingelheim, Cara Therapeutics, Dermavant Sciences, Eli Lilly, Incyte, Inozyme, Janssen Research & Development, LLC, Ortho Dermatologics, Pfizer, Sanofi-Regeneron, and UCB, Inc., and is a consultant for Almirall, AltruBio Inc., AnaptysBio, Apogee, Arcutis, Inc., AstraZeneca, Atomwise, Avotres Therapeutics, Brickell Biotech, Boehringer-Ingelheim, Bristol-Myers Squibb, Castle Biosciences, Celltrion, Corevitas, Dermavant Sciences, EPI, Evommune, Inc., Facilitation of International Dermatology Education, Forte Biosciences, Foundation for Research and Education in Dermatology, Galderma, Genentech, Incyte, LEO Pharma, Meiji Seika Pharma, Mindera, Pfizer, Sanofi-Regeneron, Seanergy, Strata, Takeda, Trevi, and Verrica.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: this work was supported by Boehringer Ingelheim Pharmaceuticals, Inc., Ridgefield, CT, USA. The authors received no payment related to the development of the manuscript.
