Abstract
Background:
Generalized pustular psoriasis (GPP) is a rare, severe neutrophilic skin disease with high unmet clinical need. The introduction of a GPP-specific International Classification of Diseases, 10th Revision (ICD-10), code has made it possible to generate a more accurate GPP patient profile.
Objectives:
To describe the characteristics and compare the patient profile and burden of disease of patients with GPP with patients with plaque psoriasis.
Methods:
A retrospective study was conducted using a US administrative claims database, the IBM® MarketScan® Research Database. The study took place between October 1, 2015, and September 30, 2018. Patients with at least 1 inpatient or 2 outpatient L40.1 (GPP) or L40.0 (psoriasis vulgaris) diagnostic codes were included for analysis. Outcome measures included descriptions of comorbidities, medication use, and healthcare resource utilization (HCRU) among GPP, plaque psoriasis, and general population (matched to those with GPP) cohorts.
Results:
Patients with GPP had more baseline comorbidities than those with plaque psoriasis and the matched cohort, including psoriatic arthritis (20.6% vs 6.4% and <0.1%) and hyperlipidemia (20.4% vs 16.3% and 11.8%). Patients with GPP also had greater medication use and higher HCRU than those with plaque psoriasis and the matched cohort.
Conclusion:
Patients with GPP generally experience more comorbidities, with higher HCRU, than patients with plaque psoriasis. Although the large dataset permitted identification of GPP patients with longitudinal follow-up, the lack of a validation algorithm for GPP is a limitation and a potential area for future research.
Keywords
Introduction
Generalized pustular psoriasis (GPP) is a rare, severe, and potentially life-threatening disease characterized by acute flares of erythematous patches with sterile pustules. 1 –3 GPP can be relapsing, characterized by periods of remission between flares, or persistent (lasts longer than 3 months). 1 The prevalence of GPP in the USA remains unknown, and estimates vary globally. 4
Since GPP was first described early in twentieth century, the characteristics of the disease and burden of illness have not been well defined. There are currently no treatments specifically approved for GPP in the USA or Europe, highlighting an unmet need among this patient population.
An advantage of using insurance claims databases to identify key characteristics of patients with rare diseases is the ability to analyze a large and diverse cohort of patients, as well as longitudinal follow-up. 5 Two independent studies exploring the burden of disease and healthcare resource utilization (HCRU) in patients with GPP were conducted using the IBM® MarketScan® Research Database and the Optum® Clinformatics® Data Mart. 6 Here, we report the findings from the IBM® MarketScan® analysis, whereby commercial insurance and Medicare data have permitted the identification of differences in the demographics, comorbidities, concurrent medication use, and HCRU of patients with GPP compared with those with plaque psoriasis and a cohort of the general population in a real-world setting in the USA.
Methods
Both Optum® and IBM® MarketScan® studies were conducted using identical methodology; please refer to the Optum® study for full details. 6 The IBM® MarketScan® Research Database comprises the IBM® MarketScan® Commercial Database, including ∼59 million employed individuals aged <65 years and their dependents, and the IBM® MarketScan® Medicare Supplemental Database, including ∼5 million retired individuals with Medicare Supplemental insurance paid by their previous employers. Using International Classification of Diseases, 10th revision (ICD-10), codes, as well as Current Procedural Terminology (v4) and Healthcare Common Procedure Coding System codes, diagnostic codes for comorbidities and diagnostic claims during inpatient, outpatient, and emergency department (ED) visits were identified between October 1, 2015 and September 30, 2018.
Three cohorts were evaluated in this study: patients with GPP, patients with plaque psoriasis, and individuals in a general population without psoriasis (according to ICD-10 codes: L40.0, L40.2, L40.4, L40.8, or L40.9), GPP (L40.1), or palmoplantar pustulosis PPP (L40.3), but including psoriatic arthritis (L40.5) and matched 4:1 according to age category (<18, 18–64, and ≥65 years) and sex to patients with GPP. Patients with GPP and those with plaque psoriasis were defined as patients with at least 1 inpatient claim or 2 outpatient claims with an ICD-10 code of L40.1 or L40.0, respectively, in line with standard practice for identifying patients in claims databases, 7 –9 and separated by 30 days but within 365 days of one another. Eligible patients were required to have at least 6 months of continuous insurance enrollment with both medical and pharmacy coverage prior to the index date (baseline period). The date of the first qualifying diagnostic claim was used as the index date (first inpatient or second outpatient claim) to provide greater sensitivity (Figure S1). Patients with concurrent GPP and/or (PPP) and plaque psoriasis diagnosis codes were excluded from the cohort of patients with plaque psoriasis (concurrent plaque psoriasis and/or PPP was allowed in the cohort of patients with GPP). All results were analyzed descriptively using the Aetion Evidence Platform® (2020). All ICD-10 codes and pharmacy claim codes used in the analysis are shown in the Supplementary Tables S1 and S2.
Results
Patient Population and Demographics at Baseline
In total, 1,175 patients with GPP, 75,494 with plaque psoriasis, and 4,312 in the matched cohort were identified at baseline. Of these, 637 patients with GPP, 38,950 patients with plaque psoriasis, and 2,276 in the matched cohort had the required 12 months of continuous follow-up (Figure S2). The distribution of patients with Medicare Supplemental coverage was similar among patients with GPP (11.4%), plaque psoriasis (10.7%), and the matched cohort (11.8%). Most patients with GPP and plaque psoriasis were female (63.3% and 51.2%, respectively). Patients with GPP tended to be slightly older, with a mean age of 52.4 years, compared with 49.0 years among those with plaque psoriasis (Table 1).
Baseline Patient Characteristics.
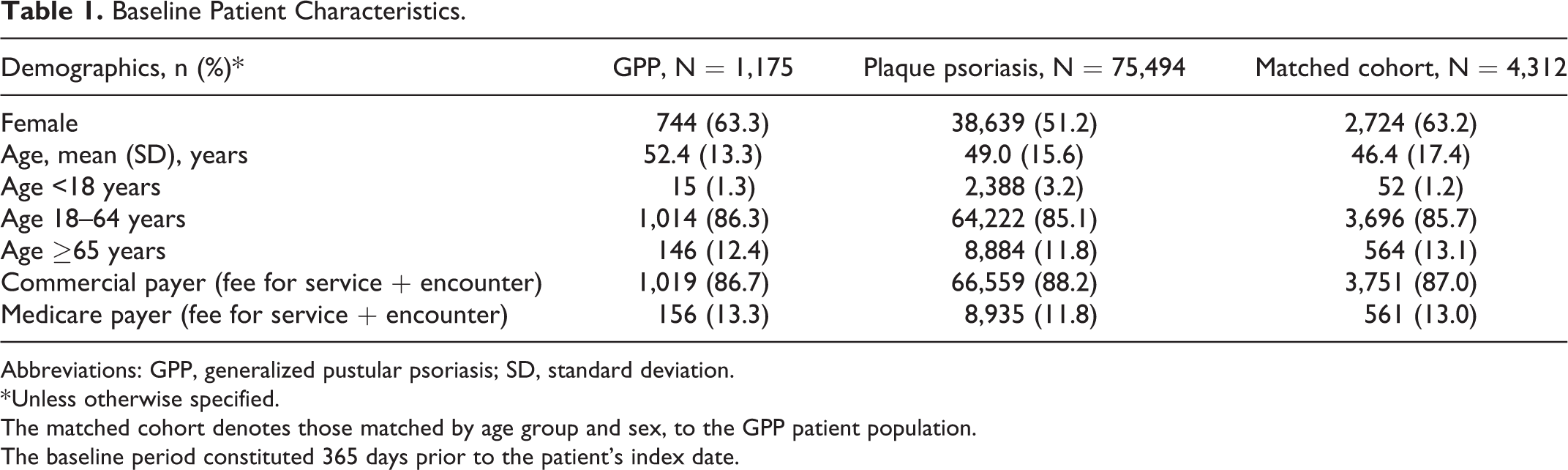
Abbreviations: GPP, generalized pustular psoriasis; SD, standard deviation.
* Unless otherwise specified.
The matched cohort denotes those matched by age group and sex, to the GPP patient population.
The baseline period constituted 365 days prior to the patient’s index date.
Comorbidities at Baseline
Several comorbid diseases occurred more frequently in patients with GPP compared with patients with plaque psoriasis and the matched cohort, notably psoriatic arthritis (20.6% vs 6.4% and <0.1%, respectively) and hyperlipidemia (20.4% vs 16.3% and 11.8%). The frequency of type 2 diabetes was similar in the GPP and plaque psoriasis cohorts (11.6% vs 10.2%), but lower in the matched cohort (6.2%) (Figure 1). A higher proportion of patients with GPP were reported to have chronic obstructive pulmonary disease than patients with plaque psoriasis and the matched cohort (6.9% vs 4.0% and 3.7%, respectively). Also, a higher proportion of patients with GPP had psychiatric disorders than patients with plaque psoriasis and those in the matched cohort, with a greater proportion of claims related to diagnoses of anxiety (9.0% vs 6.0% and 5.1%, respectively) and depression (7.0% vs 4.0% and 3.1%, respectively) (Figure 1).
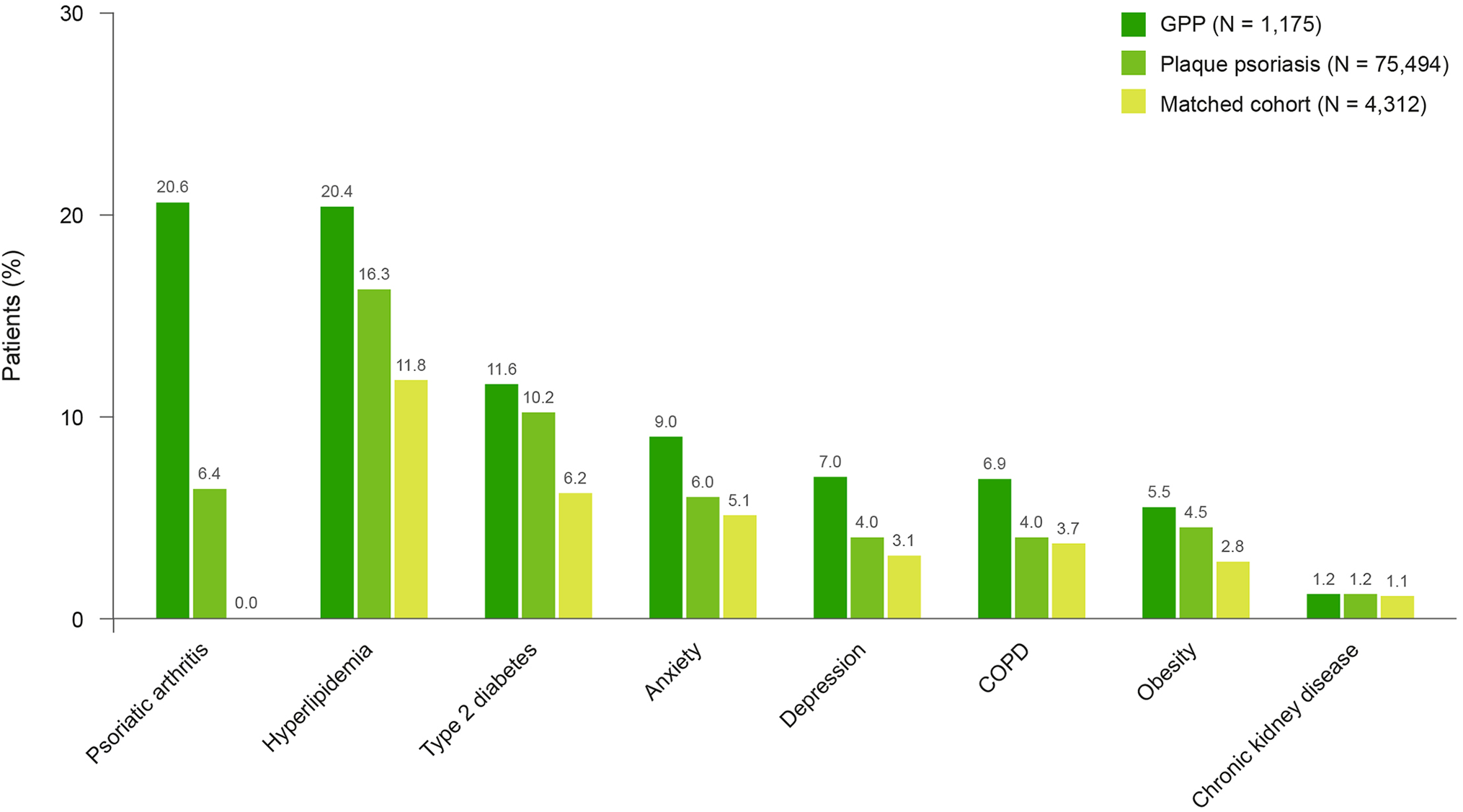
Proportion of patients with comorbidities at baseline. COPD, chronic obstructive pulmonary disease;
Concomitant Medication Use During the 12-Month Follow-Up
Concomitant medications were dispensed to a higher proportion of patients with GPP than those with plaque psoriasis and the matched cohort (Table 2). Antibiotics, psychiatric medications, and antihypertensives were the most frequently dispensed medications in all cohorts (Table 2). In addition, 36.1% of patients with GPP had a dispensing for opioid pain medication (pharmacy claim for ATC N02AJ or N02AX, Table S2) compared with 24.3% of patients with plaque psoriasis and 19.8% of individuals in the matched cohort (Table 2).
Most Common Concomitant Treatments During the 12-Month Follow-Up.
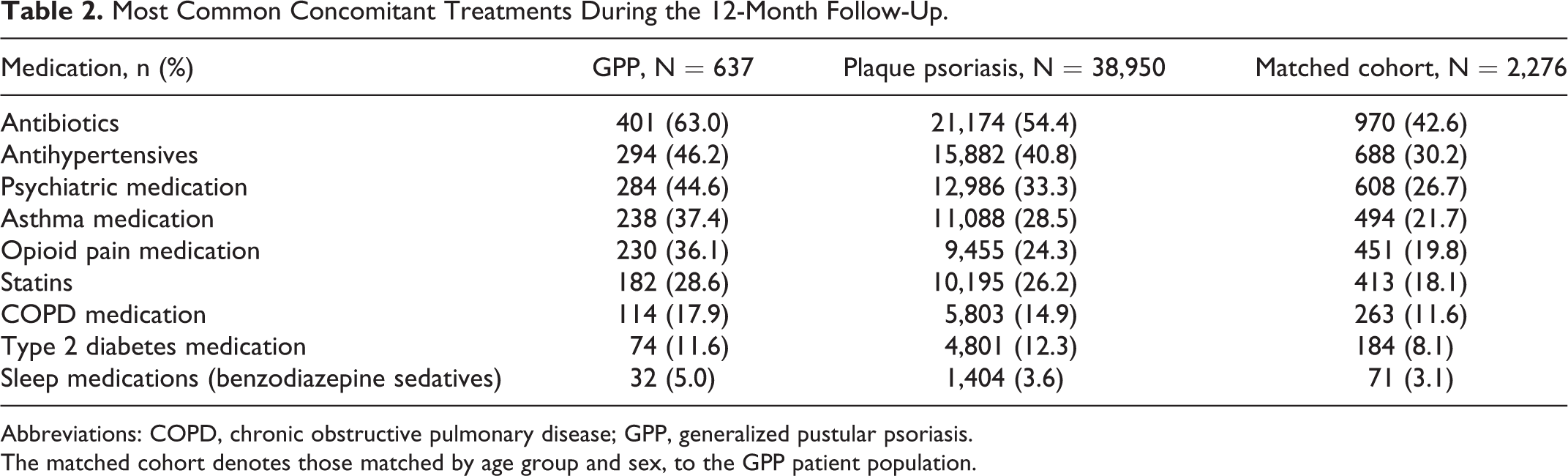
Abbreviations: COPD, chronic obstructive pulmonary disease; GPP, generalized pustular psoriasis.
The matched cohort denotes those matched by age group and sex, to the GPP patient population.
Dermatologic Medication Use During the 12-Month Follow-Up
Overall, 70.0% of patients with GPP were dispensed a systemic therapy (biologic, non-biologic, or both), compared with 62.1% of patients with plaque psoriasis. A similar proportion of patients with GPP and those with plaque psoriasis had claims for a biologic monotherapy (11.0% and 12.3%, respectively), while a higher proportion of patients with GPP received a non-biologic systemic monotherapy than those with plaque psoriasis (13.2% and 7.2%) (Table S3). A lower proportion of patients with GPP received a topical medication alone compared with those with plaque psoriasis (Table S3).
Of the biologic therapies, a similar proportion of patients in the GPP and plaque psoriasis cohorts were prescribed tumor necrosis factor (TNF) inhibitors (Figure 2). A slightly lower proportion of patients with GPP were prescribed interleukin inhibitors compared with those with plaque psoriasis (Figure 2).
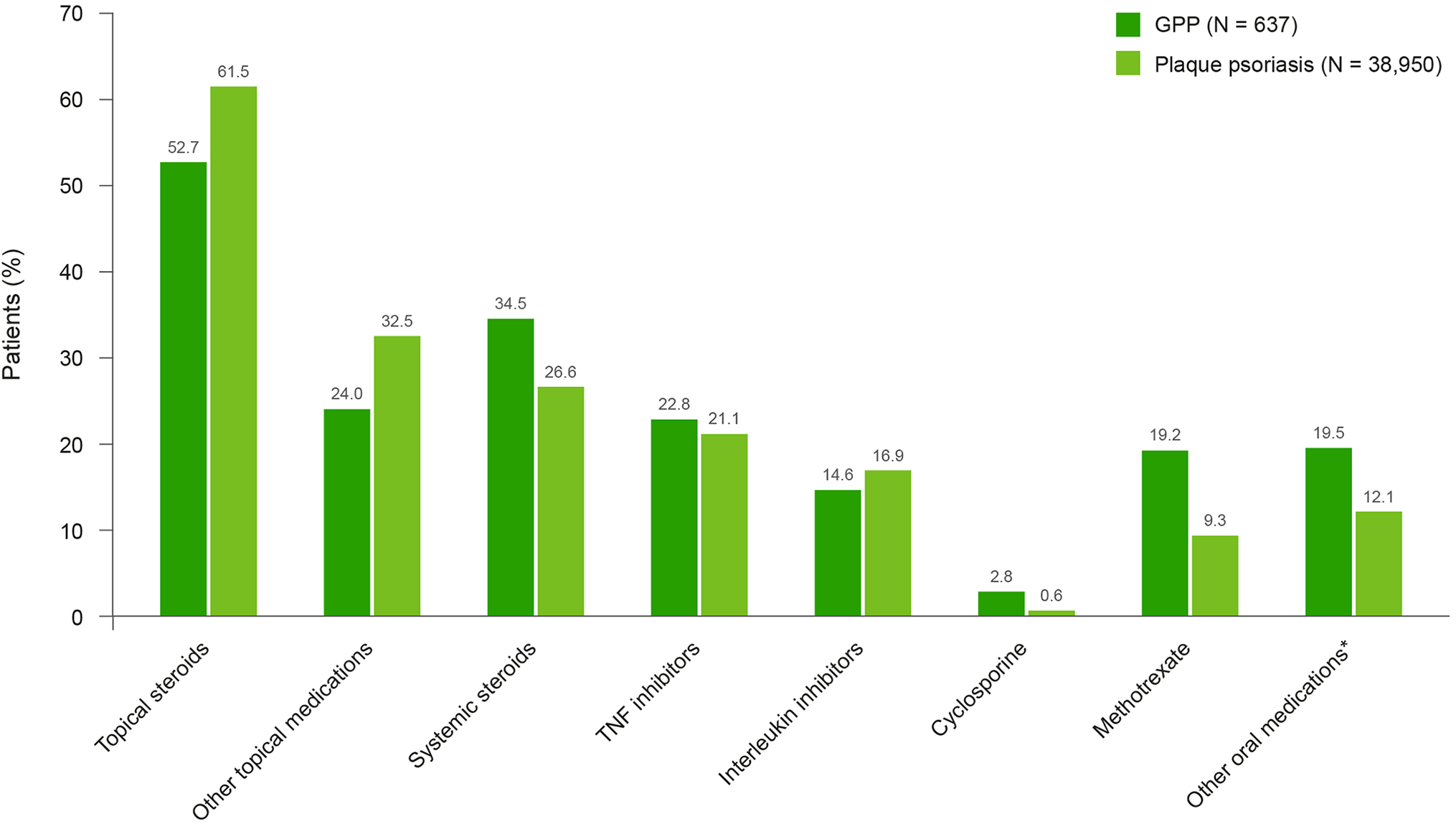
Medication use by class during the 12-month follow-up. *Other oral medications include 6-thioguanine, acitretin, apremilast, hydroxyurea, mycophenolate mofetil, and sulfasalazine. Topical steroids, other topical medications, and systemic steroids are representative of different classes of drug that include various types of medication. GPP, generalized pustular psoriasis; TNF, tumor necrosis factor.
Of those who received systemic therapies, slightly more patients with GPP (45.9%) were treated with combination therapies than those with plaque psoriasis (42.6%) (Table S3). Adalimumab was the most commonly prescribed TNF inhibitor for both patients with GPP (13.7%) and plaque psoriasis (15.6%), and ustekinumab was the most prescribed interleukin inhibitor in both cohorts (7.7% and 11.5%, respectively). T-cell inhibitors were not often prescribed in either the GPP or plaque psoriasis cohorts. Of the non-biologic systemic therapies, methotrexate was the most commonly prescribed individual therapy in both patients with GPP (19.2%) and plaque psoriasis (9.3%), where as systemic steroids were the most commonly prescribed class of medications (34.5% vs 26.6%) (Figure 2).
For combination therapies, 9.1% of patients with GPP received a topical medication with a biologic systemic treatment compared with 11.1% patients with plaque psoriasis, while the same proportion of patients in each cohort (18.1%) received a topical medication with a non-biologic systemic treatment (Table S3). More patients with GPP (8.8%) received a non-biologic therapy with a biologic therapy compared with patients with plaque psoriasis (5.0%). Overall, 9.9% of patients with GPP received a combination of topical, non-biologic, and biologic treatments compared with 8.4% of patients with plaque psoriasis (Table S3). See Table S4 for included therapies.
A similar proportion of patients with GPP and plaque psoriasis did not receive any treatment (14.3% and 14.0%, respectively) over the course of the study (Table S3).
Medication Use by Age
More patients with GPP aged ≥65 years received systemic non-biologic treatment monotherapy (14.8%) than patients with plaque psoriasis (7.7%). Substantially fewer patients with GPP aged ≥65 years received a biological monotherapy compared with those aged <65 years across the combined <18 and 18–64 years age groups (6.8% vs 11.9%); a similar trend was seen in patients with plaque psoriasis (5.1% vs 13.3%, respectively) (Table S3). Of the biologic treatments, adalimumab was the most commonly prescribed treatment in both patients with GPP and those with plaque psoriasis in those aged ≥65 years and those <65 years; however, patients aged <65 years received adalimumab more frequently than patients aged ≥65 years: 14.8% versus 6.8% with GPP, respectively, and 16.9% vs 6.8% with plaque psoriasis, respectively.
Of the 88 patients with GPP who were aged ≥65 years, 17.0% did not receive any treatment during follow-up (Table S3). Similarly, 15.6% of patients with plaque psoriasis aged ≥65 years did not receive treatment during follow-up. Of the patients <65 years, 13.8% of patients with GPP and 13.7% of patients with plaque psoriasis did not receive treatment.
HCRU During the 12-Month Follow-Up
Overall, patients with GPP had more outpatient visits (median [interquartile range (IQR)]:17.0 [9.0–32.0]) than those with plaque psoriasis (14.0 [8.0–25.0]) and the matched cohort (7.0 [3.0–15.0 ]) (Table 3). Although the median number of inpatient visits was the same across all cohorts (Table 3), more patients with GPP required at least 1 inpatient visit (12.2%) than those in the plaque psoriasis (6.5%) and the matched cohorts (6.2%).The duration of inpatient stays was also longer for patients with GPP than those with plaque psoriasis and the matched cohort (Table 3). The most common reasons for inpatient hospitalization for patients with GPP were sepsis (other), cellulitis and acute lymphangitis (10.3% each). For those with plaque psoriasis, the most common reason for inpatient hospitalization was osteoarthritis of the knee (7.5%) (Table S5).
Overall HCRU During the 12-Month Follow-Up.
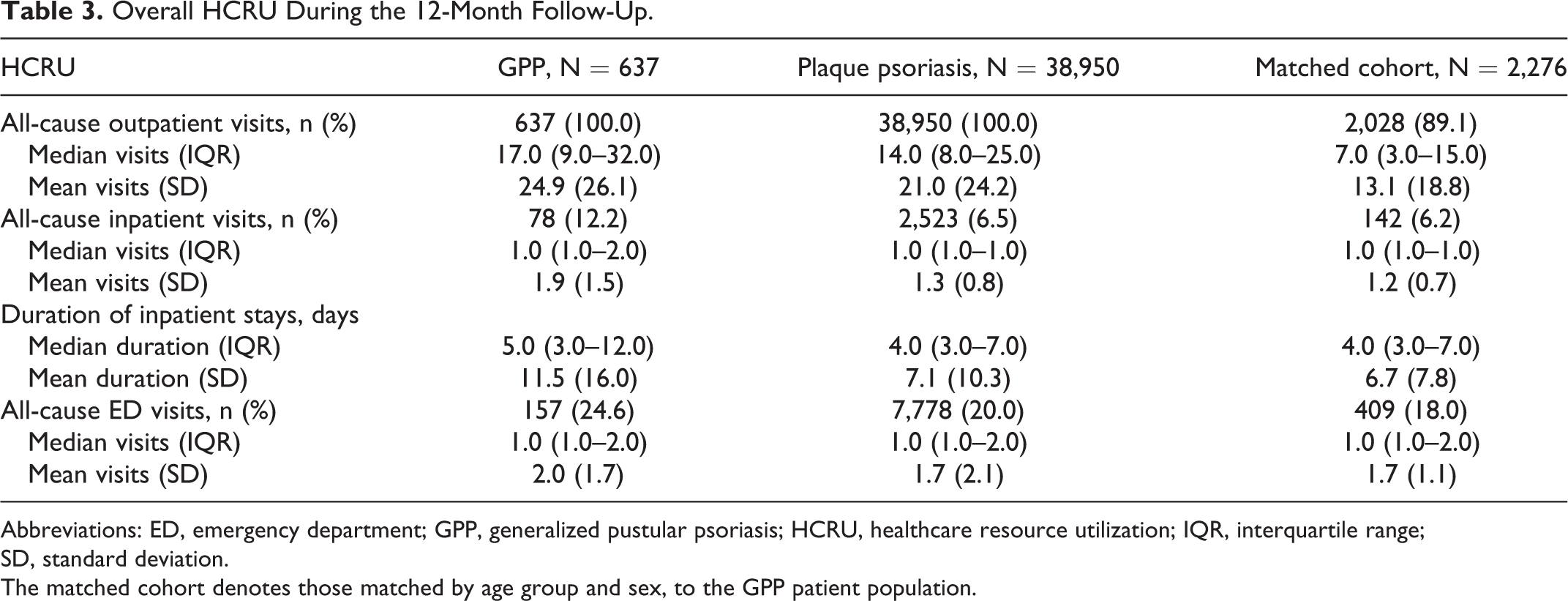
Abbreviations: ED, emergency department; GPP, generalized pustular psoriasis; HCRU, healthcare resource utilization; IQR, interquartile range;
SD, standard deviation.
The matched cohort denotes those matched by age group and sex, to the GPP patient population.
The median number of ED visits was the same across all groups, although more patients with GPP (24.6%) needed at least 1 ED visit than those in the plaque psoriasis (20.0%) and matched cohorts (18.0%) (Table 3).
Specialty of Treating Physicians
During the 12-month follow-up, fewer patients with GPP visited a dermatologist for any reason (inpatient, outpatient, or ED) compared with those with plaque psoriasis (62.3% vs 84.1%), whereas more patients with GPP than patients with plaque psoriasis visited family practice (58.7% vs 52.1%) and internal medicine physicians (43.6% vs 38.5%), as well as rheumatologists (36.7% vs 11.7%) (Table S6).
Discussion
This study summarizes the clinical characteristics of 1,175 patients with GPP, using real-world data in the USA; these included patient characteristics, medication use, and HCRU using the IBM® MarketScan® Research Database.
Patients with GPP had a higher prevalence of comorbidities than patients with plaque psoriasis, including hyperlipidemia and psoriatic arthritis. Prevalences of the specific comorbidities identified in this study for patients with plaque psoriasis are comparable to those identified in previous studies. 10,11 Notably, a greater prevalence of hyperlipidemia in patients with GPP than in those with plaque psoriasis was also observed in the Optum® study. 6 It is possible that these comorbidities are associated with the defective inflammatory pathways underlying the pathogenesis of GPP. The systemic inflammation in GPP could be perpetuated further by the increased deposition of low-density lipids in hyperlipidemia, which triggers the interleukin-1β inflammatory pathway. 12 Psoriatic arthritis is also a chronic inflammatory disease caused by alterations in inflammatory mediators. 13 Furthermore, comorbidities may be impacted by disease severity; unfortunately, disease severity of GPP and plaque psoriasis is not captured in this database. Despite this limitation, disease severity can be inferred by the proportion of patients receiving systemic therapies. Approximately 70% of patients with GPP and 60% of patients with plaque psoriasis received systemic therapies throughout the duration of the study, suggesting comparable disease severity in the 2 cohorts. It is likely that the 2 cohorts included a heterogeneous population of disease severity such that the presence of milder disease within each cohort may have diluted the prevalence of certain comorbidities.
In contrast to the Optum® study, 6 the IBM® MarketScan® Research Database includes only a small proportion of data from patients receiving Medicare Advantage (though includes a higher proportion of Medicare Supplemental data) and comprises person-specific data from mostly employed, commercially insured individuals. Collectively, patients had a lower mean age in this study than in the Optum® study. 6 The Optum® database comprises single-payer source claims data, with a higher proportion of patients receiving Medicare Advantage than in the IBM® MarketScan® Research Database. 14 This could account for the higher mean age and thus the differences in treatment selection and access; this may also affect the prevalence and types of comorbidities experienced, as well as treatments received, in both databases. Medication use in patients between 18 and 64 years was similar in both databases, highlighting that age, Medicare insurance, and the use of multiple insurers in the IBM® MarketScan® Database are likely driving the differences in treatment.
As shown in Supplementary Table S6, more patients with GPP were hospitalized for sepsis (streptococcal or other) than those in the plaque psoriasis cohort (11.6% vs 4.3%), suggesting that patients with GPP may be more susceptible to severe infections. The mean duration of inpatient hospitalizations in the GPP cohort was longer than in both the plaque psoriasis and matched cohorts. Additionally, inpatient hospitalization was more frequent in patients with GPP than those with plaque psoriasis. Collectively, these results suggest that, on average, a patient with GPP has higher HCRU than a patient with plaque psoriasis, highlighting the high disease burden in patients with GPP.
Fewer patients with GPP visited dermatology specialists in this study compared with those with plaque psoriasis; this could be because GPP may be more frequently managed by inpatient hospitalization, rather than in the clinic.
In this study, the increased use of antihypertensives in patients with GPP compared with those with plaque psoriasis suggests an increased prevalence of hypertension, despite hypertension not being reported as a frequent comorbidity in patients with GPP. A likely explanation for this is that antihypertensive medication is often used to treat conditions other than hypertension (e.g. arrhythmias). The use of antihypertensives suggests that there may also be an increased risk of cardiovascular complications in patients with GPP. Increased levels of cardiovascular disease (CVD) were not seen, however; this could be because patients with comorbid conditions linked to CVD (e.g. type 2 diabetes and obesity) may not have developed CVD over the duration of the study. Obesity is a known comorbidity of GPP, 15 supporting the observation that patients with GPP were more likely than those with plaque psoriasis to have a diagnostic claim indicating obesity in this study. However, we cannot be certain of the association because body mass index (BMI) is not captured in this database.
In addition to antihypertensives, psychiatric medications are one of the most highly prescribed medications. Although anxiety and depression were reported as prevalent comorbidities, it should be noted that some psychiatric medications have dual use. Some medications are also potent antihistamines so it is possible, although unlikely, that these treatments may be being prescribed for intractable pruritis rather than for psychiatric reasons. As with most claims-based data sources, diagnosis codes were not linked to prescriptions so it is not possible to ascertain why certain medications were prescribed to individual patients, particularly for medications approved for use in more than 1 indication and those with dual uses.
In this study patients with GPP were treated with concomitant medication more often than those with plaque psoriasis, suggesting that these patients have a higher prevalence of comorbidities, further highlighting that these 2 patient populations have different clinical profiles. 15 Patients with GPP were as likely to receive biologic systemic therapies as those in the plaque psoriasis cohort, but less likely to receive topical medications, suggesting that topical medications may not be considered to be as practical or effective in patients with GPP as they are in those with plaque psoriasis. The low frequency of biologic use in the IBM® MarketScan® Research Database is likely due to formulary restrictions, given that there are no biologics specifically approved for GPP in the USA. The Optum® study showed markedly less biologic use in the GPP patient cohort compared with this study, which may reflect the inclusion of patients enrolled in Medicare Advantage in the Optum® study. 6 This suggests that biologics may not be as widely prescribed in older patients because of safety concerns (such as contraindication) or issues with medication access.
Notably, some patients were reported as not receiving any treatment during the follow-up period. It is probable that some patients with a history of GPP did not experience a significant flare of disease during the time of the study. It is also likely that some therapies may not have been recorded, for example, the use of phototherapy was not captured in this database.
Limitations of this study include the retrospective nature of data collection. Data are from an administrative patient insurance claims database that relies on patients and clinicians submitting separate claims, and data were collected for reimbursement purposes rather than research. As such, there is an inability to capture data on the severity of GPP disease or flares (including frequency, remission, or relapse). It is likely that some of the hospitalizations and certain medication use are in response to flares, though this is not possible to ascertain. Despite these limitations, the data on treatments and HCRU are generally considered to be of high quality as these data are collected based on what the patient received at the pharmacy, and HCRU in the outpatient, inpatient, and ED settings, respectively. Patients enrolled in this database also have the propensity to change health insurance plans if they change jobs, which may have adversely affected the number of patients with 12 months of follow-up. In addition, some topical medications are available over the counter, which could have led to under-representation of topical medication use in the database.
Owing to the nature of the IBM® MarketScan® Research Database, the lower mean age of those enrolled compared with those in the Optum® study 6 also means that those of retirement age (≥65 years) are under-represented, which may impact the reported comorbidities and medications received. The database also does not include measures for ethnicity and BMI. Therefore, it is not possible to describe these measures or consider describing other key characteristics stratified by these measures. In addition, the lack of a validation algorithm, as well as a lack of consensus to identify cases of GPP specifically, presents a limitation in studies of this kind; however, stratification was based on standard procedures for retrospective claims database studies.
As GPP has no specifically approved treatment in the USA, 16 the type of commercial insurance a patient has may impact treatment decisions, as well as the physician seen. First-line treatments used by physicians for GPP have previously often been restricted to formularies without biologics, but the gradual increase in the use of biologics, as well as their increased acceptance into payer formularies, over the duration of this study and reflected in the slightly increased use of adalimumab by patients with GPP compared with other treatments, suggests that this may be changing.
In combination with the Optum® study, these analyses demonstrate the differences between the patient profiles of those with GPP and those with plaque psoriasis. 6 The ability to extrapolate data from multiple insurance plans in the IBM® MarketScan® Database allows for a highly detailed characterization of patients with GPP. Here, results indicate that patients with GPP generally have a higher disease burden, with an increased incidence of comorbidities, and HCRU than patients with plaque psoriasis and the matched general population cohort.
Supplemental Material
Supplemental Material, sj-pdf-1-jps-10.1177_24755303211021779 - Healthcare Resource Utilization and Baseline Characteristics of Patients With Generalized Pustular Psoriasis: Real-World Results From a Large US Database of Multiple Commercial Medical Insurers
Supplemental Material, sj-pdf-1-jps-10.1177_24755303211021779 for Healthcare Resource Utilization and Baseline Characteristics of Patients With Generalized Pustular Psoriasis: Real-World Results From a Large US Database of Multiple Commercial Medical Insurers by Jeffrey M. Sobell, Ran Gao, Amanda K. Golembesky, Nirali Kotowsky, Elizabeth M. Garry, Erin O. Comerford, Rhonda L. Bohn, Wendell C. Valdecantos, Steven R. Feldman and Craig Leonardi in Journal of Psoriasis and Psoriatic Arthritis
Footnotes
Authors’ Note
To ensure independent interpretation of clinical study results, Boehringer Ingelheim grants all external authors access to summarized results and other relevant material as needed by them to fulfill their role and obligations as authors under the International Committee of Medical Journal Editors (ICMJE) criteria. Furthermore, clinical study documents (e.g., the study report, study protocol, and statistical analysis plan) and participant clinical study data are available for sharing after publication of the primary manuscript in a peer-reviewed journal and if regulatory activities are complete and other criteria met, as per the Boehringer Ingelheim policy on transparency and publication of clinical study data (see https://trials.boehringer-ingelheim.com/transparency_policy.html). Prior to providing access, documents will be examined, and, if necessary, redacted and the data will be de-identified, to protect the personal data of study patients and personnel, and to respect the boundaries of informed consent of the study patients. Clinical study reports and related clinical documents can be requested via the link https://trials.boehringer-ingelheim.com/trial_results/clinical_submission_documents.html. All such requests will be governed by a document sharing agreement. Bona fide, qualified scientific and medical researchers may request access to de-identified, analyzable participant clinical study data with corresponding documentation describing the structure and content of the datasets. Upon approval, and governed by a data sharing agreement, data are shared in a secured data-access system for a limited period of 1 year, which may be extended upon request. Researchers should use ![]() to request access to study data.
to request access to study data.
Acknowledgments
This study was funded by Boehringer Ingelheim. Editorial and writing assistance in the preparation of this manuscript was provided by Amy Pashler, PhD and Leigh Church, PhD of OPEN Health Communications (London, UK) and funded by Boehringer Ingelheim. The listed authors have authorized the submission of their manuscript via third party and approved any statements or declarations. We wish to acknowledge Irisdaly Estevez, Julia A. Pisc, and Jocelyn R. Wang, contracted by Boehringer Ingelheim, for their contributions to the analytic implementation via the Aetion Evidence Platform. Agreements between Boehringer Ingelheim and the authors included the confidentiality of the study data.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: JM Sobell received research, speaking and/or consulting support from Amgen, AbbVie, Janssen, Lilly, Novartis, Sun Pharma, BMS, Celgene, Sanofi, and Regeneron. AK Golembesky was an employee of Boehringer Ingelheim during the implementation of this study and is now employed by GlaxoSmithKline. EM Garry is an employee of Aetion, Inc with stock options. EO Comerford was employed by Aetion, Inc during the implementation of this study, has equity in the company, and is now employed by Sanofi. RL Bohn is the owner and principal of Bohn Epidemiology, LLC. SR Feldman received research, speaking, and/or consulting support from Galderma, Almirall, Alvotech, Leo Pharma, BMS, Boehringer Ingelheim, Mylan, Celgene, Pfizer, Ortho Dermatologics, AbbVie, Samsung, Janssen, Lilly, Menlo Therapeutics, Merck, Amgen, Helsinn, Arena, Novartis, Regeneron, Sanofi, Novan, Qurient, National Biological Corporation, Caremark, Advance Medical, Sun Pharma, Suncare Research, Informa, UpToDate, and National Psoriasis Foundation. SR Feldman is founder and majority owner of ![]() and founder and part owner of Causa Research. C Leonardi has received honoraria or fees for serving on advisory boards, as a speaker, and as a consultant, as well as grants as an investigator from AbbVie, Actavis, Amgen, Boehringer Ingelheim, Celgene, Coherus, Dermira, Eli Lilly, Galderma, Janssen, Leo, Merck, Novartis, Pfizer, LEO Pharma, Sandoz, Stiefel, UCB, Vitae, and Wyeth. R Gao, N Kotowsky, and WC Valdecantos are employees of Boehringer Ingelheim.
and founder and part owner of Causa Research. C Leonardi has received honoraria or fees for serving on advisory boards, as a speaker, and as a consultant, as well as grants as an investigator from AbbVie, Actavis, Amgen, Boehringer Ingelheim, Celgene, Coherus, Dermira, Eli Lilly, Galderma, Janssen, Leo, Merck, Novartis, Pfizer, LEO Pharma, Sandoz, Stiefel, UCB, Vitae, and Wyeth. R Gao, N Kotowsky, and WC Valdecantos are employees of Boehringer Ingelheim.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Boehringer Ingelheim sponsored this study and provided funding for the conduct, data analysis, and medical writing assistance for the study’s publication.
Supplemental Material
Supplemental material for this article is available online.
Abbreviations
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
