Abstract
Background:
Factors initiating capillary conversion in the immunopathogenesis of psoriasis are not well established and quantification of vascular changes in- and outside visible psoriatic plaque has not been studied extensively.
Objectives:
To assess the pathological involvement of capillaries in the context of well-established features of psoriasis in different phases: symptomless psoriatic skin distant from a lesion (SDL), adjacent to a lesion (SAL), the margin zone of the lesion (ML) and the center of the psoriatic lesion (CL).
Methods:
In 10 patients with chronic plaque psoriasis, and 10 healthy matched controls, vascular parameters (vascular tonicity (VD), vascular surface area (VAR), microvascular density (MVD), angiogenesis (pEC)) and hypoxia in the context of well-established features (keratinocyte proliferation, neutrophils and T-cells) were analyzed within the psoriatic lesion and symptomless psoriatic skin, using immunohistochemical markers (Ki67/CD31 double-staining and HIF-1α-, Elastase- and T-Bet-staining).
Results:
VD and neutrophils were significantly increased in all sites, compared to healthy controls. The VAR, MVD, Ki67+ nuclei, HIF-1α and T-Bet start to augment in the SAL. Within the overt psoriatic plaque angiogenesis increases. Biopsies closer to the center of a plaque, exhibited increased values of the MVD, VAR, VD, Ki67+ nuclei, T-Bet+ Th1 cells and neutrophils.
Conclusion:
In SDL, vasodilatation and activation of the innate immune system (increased neutrophils) was detected. This could indicate that psoriasis is not restricted to lesional skin, but rather is a systemic disease. Closer to the psoriatic plaque, vascular and epithelial proliferation was observed, along with hypoxic cells and activation of the acquired immunity (augmented T-Bet cells).
Introduction
The notion that a vascular alteration plays a crucial role in the development and maintenance of clinical psoriasis has been debated for a long time. Modification of the microvasculature is essential in the maintenance of the psoriasis plaque in providing nutrition to the hyperplastic keratinocytes, dermal tissue and enabling migration of inflammatory cells. 1
Therefore, the microvasculature in psoriasis has been studied during many decades. Auspitz already described the pinpoint bleedings after removing the stratum corneum. 2 Pinkus and Mehregan. described the squirting papilla with elongated dilated capillaries and extravasation of neutrophils, forming micropustules of Kogoi, the pathognomonic sign in the micromorphology of psoriasis. 3 This phenomenon displays in a focal distribution pattern within the psoriatic lesion.
In addition, Kulka found convoluted and dilated capillaries in symptomless skin of psoriasis patients. 4 Detmar et al mechanistically revealed that VEGF (vascular endothelial growth factor) fulfills an important role in vascular alterations in psoriasis, while Benhadou et al found that overexpression of epidermal VEGF-A functions mediates a psoriasis-like disease. 5,6 Consequently, some authors hypothesize that the initial stimulus in psoriasis could be vascular alteration including dilatation of the cutaneous microcirculation accompanied with migration of inflammatory cells.
In contrast, Baverman and Sibley demonstrated that the percentage of labeled basal cells in the epidermis was evidently increased prior to the development of tortuous and dilated capillary loops. 7 Subsequently, epidermal turnover could be the primary defect in the psoriatic process, while the cutaneous microvasculature fulfills a modulator role. 7
Perfusion measurements of symptomless skin surrounding lesions, using laser Doppler flowmetry and speckled camera, revealed an increased perilesional perfusion. Therefore, development of new lesions could possibly be predicted by these assessments. 8 Additional studies revealed that foci of increased perfusion are associated with foci of inflammation, with increased transcription and expression of IL-17 and Th1-cells. 9
There is evidence that hypoxia plays an essential initiating role in the changes in the microvasculature of inflamed skin. Hypoxia has been suggested to be relevant to the pathogenesis of psoriasis. 10 -12 Hypoxia-inducible factor-1α (HIF-1α), an indicator of cellular hypoxia, is crucial in (micro)vascular reshaping since it regulates angiogenesis by modulation of essential factors as fibroblast growth factor-2 (FGF-2), vascular endothelial growth factor-A (VEGF-A) and VEGF-receptors. 13 Overproduction of HIF-1α and target-gene VEGF induce formation of leaky dilated vessels by, inter alia, targeting inducible nitric-oxide synthetase, an enzyme producing nitric-oxide (NO). The exact role of HIF-1α in the pathogenesis of psoriasis is still unclear.
So far, the order of events in the cascade resulting in the psoriatic plaque is not well understood. There is, however, some evidence that the microvasculature may be involved in the early phase of development of the lesion. 8
The aim of the present study is to assess the pathological involvement of capillaries in the context of well-established features in the pathology of the psoriatic lesion: T-cells, neutrophils and epidermal proliferation. 9,14,15 These parameters were evaluated in 4 consecutive phases of the development of the psoriatic lesion: the center of the lesion (CL) and margin zone of the lesion (ML) and symptomless skin adjacent to (SAL) and distant to (DSL) psoriatic plaques.
In this study biopsies were collected from the psoriatic plaque in the 4 phases of development.
The degree of hypoxia (HIF-1α) together with various parameters of the vasculature including the vascular network size (vascular area ratio (VAR), microvascular density (MVD)), the vessel diameter and angiogenesis (proliferating endothelial cells) were studied together with the well-established constituents of the psoriatic lesion: influx of neutrophils (elastase positive cells), Th1-cells (T-Bet positive cells) and epidermal proliferation (Ki67+ cells). The same variables were studied in skin biopsies of matched healthy controls.
Materials and Methods
Subjects
Ten patients with chronic plaque psoriasis and 10 matched healthy controls (volunteers without psoriasis) were included in this study. Written informed consent was given by all subjects before inclusion. The experiments were approved by the local ethics committee and conducted according to the Declaration of Helsinki principles.
Included psoriasis-patients had not been on a systemic anti-psoriatic drug for at least 6 weeks and all topical treatment of the target lesion was stopped 2 weeks prior to the biopsy. The healthy controls had no signs or (family) history of skin disorders; the gender, age (+/- 2 years) and localization of the biopsied skin were matched with that of the patient.
In all attendees a physical assessment was performed, including a general internal examination and inspection of the skin. The general physical examination involved assessment of the blood pressure, a Body Mass Index-score and auscultation of the heart, considering the fact these items could potentially affect the cutaneous vasculature.
Healthy volunteers were assessed to confirm that there were indeed no skin abnormalities visible, before they got included. In the psoriasis patients a PASI (Psoriasis Area Severity Index), BSA (Body Surface Area) score and a separate SUM-score (scale 0-3; mild/moderate/severe) of erythema, desquamation and induration of the target lesion were determined. A target psoriasis plaque was selected, based on the size (diameter ≥5 cm), SUM-score (SUM-score ≥5) and in consultation with the patient; with regard to the possibility of scarring or occurrence of Koebner phenomena due to skin-biopsies. The patients had stable chronic plaque psoriasis, with no evident history of progression and extension of the lesions in the last 2 weeks.
In all participants biopsies were obtained after local anesthesia (xylocaïn 2%/adrenaline 1:200,000).
In all patients four 3 mm biopsies were obtained. Two biopsies were taken from the target psoriatic plaque (ML and CL) and 2 from symptomless skin adjacent (2-3 cm) (SAL) and distant (15 cm) (SDL) to a psoriatic lesion (including the target psoriatic plaque). In healthy controls one 3 mm biopsy was performed on the same body part and visually on the exact same location as where the SDL biopsy was taken in patients to which they were matched (Table 1).
Patient Characteristics.
Abbreviations: BMI, Body Mass Index; PASI-score, Psoriasis Area Severity Index; BSA-score, Body Surface Area.
SUM-score, Erythema, desquamation and induration of the target lesion.
Immunohistochemistry
The biopsies were formalin-fixed, paraffin embedded and sectioned at 4 µm. After deparaffinizing and rehydration of the tissue samples, antibody retrieval was achieved by EDTA-buffer (pH 9.0, 10 minutes at 96°C) for the Ki67/CD31 double-staining and citrate-buffer (pH 6.0, 20 minutes at 96°C) for HIF-1α. All other staining did not require any antibody retrieval. Endogenous peroxidase activity was blocked by 3% H2O2 in phosphate-buffered saline (PBS). Sections were incubated with the primary antibodies dissolved in 1% bovine serum albumin (for all primary antibodies used and incubation times see Table 2).
Overview of the Primary Antibodies.

In the CD31/Ki67 double-staining, the Ki67-antibody was first visualized using a peroxidase-based Envision kit (DAKO, Glostrup, Denmark) 3-3′-diaminobenzidine metal-enhanced (Metal-Enhanced DAB substrate-kit, Thermo Scientific, US). Subsequently, the visualization of CD31 was achieved with BrightVision (BrightVision Poly-AP anti-Ms IgG from ImmunoLogic, the Netherlands). HIF-1α-, T-bet- and the Elastase-antibody were stained using the same peroxidase-based EnVision kit and 3-3′-diaminobenzidine metal-enhanced kit as described in the Ki67-staining. All sections were counterstained with Mayer’s haematoxylin (Sigma-Aldrich). Substitution with 1% PBS served as negative controls.
Quantification of Surface Area and Cells
Immunologically stained tissue samples were captured using the Mirax Midi Scanner (3DHISTECH, Budapest, Hungary). Slides were mechanically scanned with fully automated focusing and tissue finding, using a x20 objective lens Plan-Apochromat (Carl Zeiss, Jena, Germany), NA = 0.8] and images were stored in jpg-compression based format (3DHistech) and set to an 80% compression ratio (pixel size 0.23 µm). 16
Processing package Fiji 1.47 was used to evaluate all tissue samples. Regarding the epidermis an area across the whole section was measured, excluding the stratum corneum. Within the dermal compartment, the region of interest was defined as the surface from the basement membrane down to 350 µm.
HIF-1α+ keratinocytes were determined and expressed as percentage of the total count of keratinocytes. Keratinocyte-nuclei positive for Ki67 were described as Ki67+ cells per millimeter length of the basement membrane. T-bet+ (Th1) and Elastase+ (neutrophil) cells were counted and expressed as number of positive cells per mm2 dermis.
The vasculature was evaluated using several characteristics of the blood vessels and vascular bed (from the basement membrane down to 350 µm). The vascular network size was examined using the microvascular density (MVD) and the vascular area ratio (VAR). The MVD represents the number of vessels in the selected area. The VAR is defined as the area occupied by (micro)vessel; the area stained for CD31 within the selected area. The vessel diameter (VD) is determined as the smallest part of a vessel-lumen (in µm) surrounded by endothelial cells (ECs).
Since the vessel diameter could potentially be influenced by the location of dissection of the vessel in the tissue sample, multiple tissue slices with multiple visual fields were examined in every biopsy in every individual. 17 -19
Endothelial cells that double-stain CD31 (endothelial marker) and Ki67 (proliferation marker) are considered as proliferating ECs (pECs). pECs were counted and expressed as Ki67+ ECs relative to the total endothelial surface (mm2).
Statistics
Results were expressed as mean ± standard error of the mean (SEM). For the statistical analysis SPSS version 25 was used (SPSS, Benelux BV, the Netherlands). Differences in the immunohistochemical variables were assed using a Wilcoxon matched pairs signed rank test. P-values of < 0.05 were considered statistically significant.
Results
Subject Characteristics
In this study, 10 psoriasis patients and 10 matched healthy controls were included. The composition of the 2 groups of volunteers, psoriasis patients and healthy matched volunteers is equal. In both groups there are 6 males and 4 females; aged 20-64 years in the psoriasis group and 21-63 years in the matched healthy volunteers. The characteristics of all subjects, patients and controls, are displayed in Table 1.
Size of the Vascular Network
The size of the vascular network was determined on 2 parameters: the vascular area ratio (VAR) and the micro-vascular density (MVD).
The MVD, the number of blood vessels in the selected area of the skin in healthy volunteers and in SDL of psoriasis patients did not differ from each other (30.7 ± 1.5 vs 33.8 ± 1.6 vessels/mm2; p = 0.24) (Figure 1). The MVD expanded considerable and significantly in SAL (43.3 ± 3 vessels/mm2; p = 0.02) and extended even further in the psoriatic plaque (ML and CL (77.0 ± 3.8 and 81.4 ± 5 vessels/mm2; both P < 0.01) (Figures 1 and 2).

Overview of immunohistological results. A, Micro vascular density (MVD): the number of (micro)vessels in the selected area of the dermis (per mm2). B, Vascular Area Ratio (VAR): fraction of the area occupied by (micro)vessel within the selected dermal area (in %). C, Vessel diameter: the smallest part of vessel-lumen (in µm). D, Endothelial Cell Proliferation (Ki67+ EC nuclei per CD31+ mm2). E, Ki67+ keratinocytes per mm length of basement membrane. F, T-BET: the number of Th1-cells expressed as the number of positive cells per mm2 dermis. G, Elastase: the number of neutrophils expressed as the number of positive cells per mm2 dermis. H, HIF-1α: HIF-1α + keratinocytes (in %). The Bar charts represent the results of the analyzed skin biopsies including the psoriatic lesion, the symptomless skin and the skin of matched healthy controls. The psoriatic lesion includes the center of the lesion (CL), margin of the lesion (ML), the symptomless skin adjacent to the lesion (SAL), the symptomless skin distant to the lesion (SDL). Matched healthy controls (HC). Means ± SEM. *p < 0.05: statistically significant compared to skin of the matched healthy controls.  : Statistically significant compared to 2 subsequent biopsies.
: Statistically significant compared to 2 subsequent biopsies.
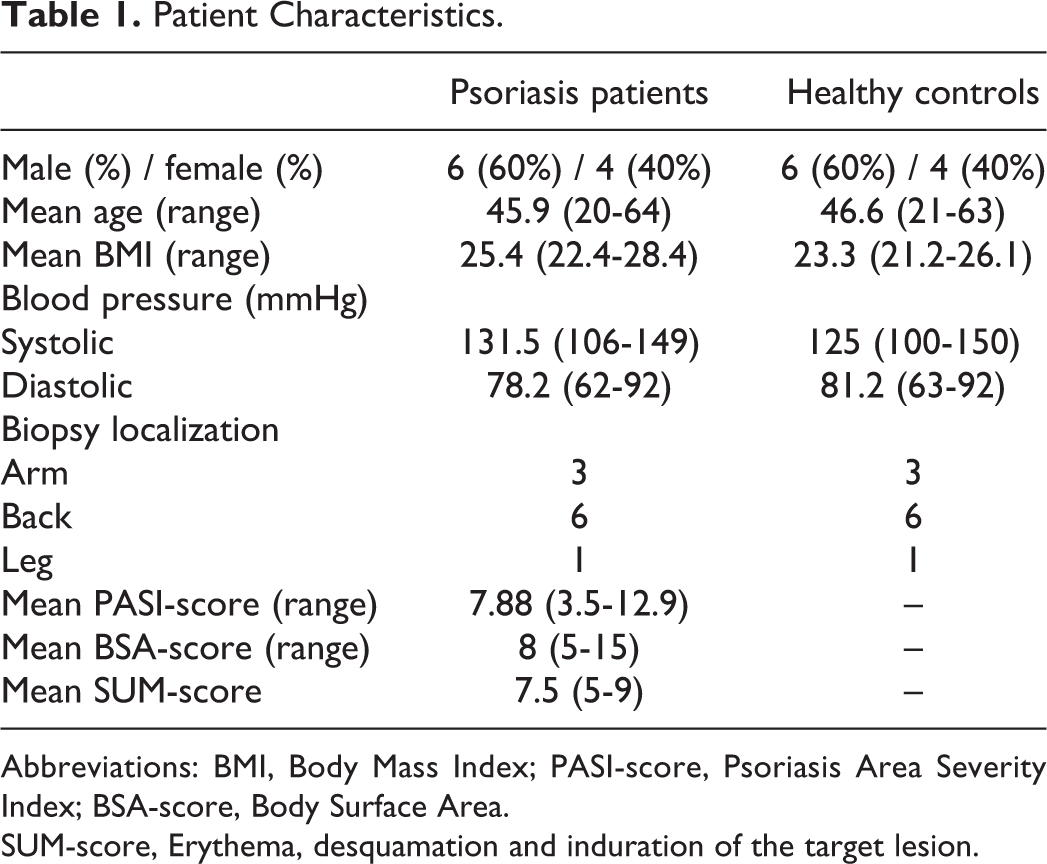
Representative photographs of the CD31/Ki67 double-staining in the psoriatic skin. The psoriatic lesion includes the center of the lesion (CL), margin of the lesion (ML), the symptomless skin adjacent to the lesion (SAL), the symptomless skin distant to the lesion (SDL). Matched healthy controls (HC).
The VAR, the area fraction of ECs in a selected tissue area, was 2.3 ± 0.3% in the skin of healthy volunteers. The VAR in SDL revealed similar results (2.6 ± 0.3%; p = 0.21). In SAL, a small but significant increase (3.0 ± 0.2%; p = 0.01) was observed. The VAR in the psoriatic plaques itself (ML and CL) is evidently increased.
Vessel Diameter (VD)
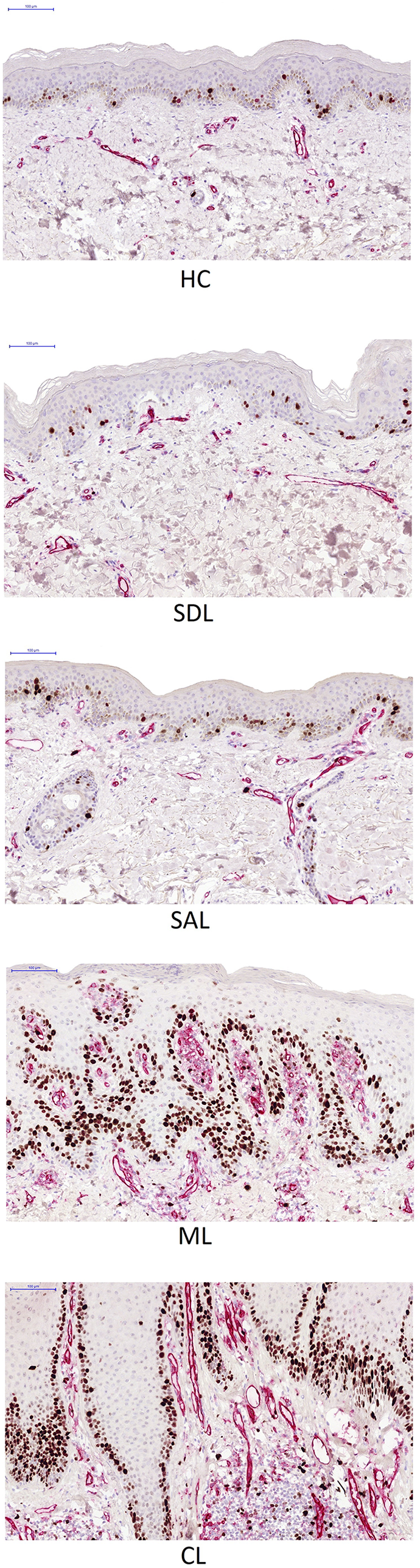
The mean diameter of the vessel lumen (VD), the smallest part of the vessel-lumen (in µm) surrounded by endothelial cells (ECs), in healthy skin is 7.0 ± 0.3 µm. Results demonstrated an increased VD in all psoriatic skin (lesional and symptomless skin) compared to healthy skin (SDL: 8.1 ± 0.4 µm; p = < 0.05; SAL 11.6: ± 0.6 µm, ML: 12.3 ± 0.5 µm, CL: 14.7 ± 0.4 µm; in SAL, ML and CL: p < 0.05) (Figure 1, Table 3).
Vessel Diameter (VD) in Biopsies in Psoriasis Patients Versus Biopsy of Matched Healthy Control per Body Location.
The mean vessel diameter (VD) of all biopsies in biopsies performed in psoriatic patients and their healthy matched controls. Biopsies in the psoriatic patients include the SDL (symptomless skin 15 cm distant form the target psoriatic plaque), SAL (symptomless skin 2-3 cm from the target psoriatic lesion), ML (margin of the psoriatic plaque) and CL (center of the psoriatic lesion). In the healthy matched control (HC) the biopsy was taken at visually the exact same location on the same body part as the SDL in the matched psoriatic patient.
Results are presented as mean ± standard error of mean. Wilcoxon matched pairs signed rank test. P-values of < 0.05 were considered statistically significant.
Proliferating Endothelial Cells (pECs)
In healthy skin proliferating Endothelial Cells (pEC) are almost non-existent (0 ± 0.0 pECs/CD31 mm2). Although an increase in the number of proliferating ECs is found in the SDL, these results do not significantly differ from the skin of healthy volunteers (3.6 ± 3.7 pECs/CD31 mm2; p = 0.32). The level of proliferating ECs expanded to nearly significant in SAL (35 ± 18 pECs/CD31 mm2; p = 0.10). In the psoriatic plaques the rise becomes substantial (ML: 138.7 ± 25 and CL: 116 ± 22 pECs/CD31 mm2; p < 0.05).
HIF-1α Levels
HIF-1α expression is limited and quite similar in SDL compared to skin of healthy volunteers (in both groups: 0.7% ± 0.2 of the total number of keratinocytes; p = 0.8).
HIF-1α expression is evidently increased in SAL (1.2% ± 0.2; p = 0.04) and within the psoriatic plaque (ML: 12.4% ± 0.8; CL: 10.9% ± 0.7; both p < 0.01%). A substantial expansion in HIF-1α is noticed comparing the results of SAL to SDL.
Inflammatory Cells: T-Bet / Elastase
Regarding the T-Bet+ Th1-cell subset, the number is similar in SDL compared to healthy skin (22.7 ± 1.7 vs 20.4 ± 1.4; p = 0.2). In the psoriatic plaque a firm higher average is observed, with a maximum number of cells detected in the center of the psoriatic plaque (CL: 170.7 ± 9.0). The number of Th1-cells increases significantly in each of the subsequent biopsies; the closer to the center of the psoriasis plaque the biopsy was taken, the higher the amount of T-Bet+ cells.
The influx of elastase+ neutrophils is augmented in all psoriatic skin sites compared to healthy skin. In
SDL the increase of the number of neutrophils is small, but statistically significantly different from healthy skin (p = 0.02). SAL reveals a tendency of a mild, significant, increase in neutrophils (SAL: 32.8 ± 2.5 versus SDL: 9.9 ± 1.6; p = 0.01) (Figure 3). However, a very significant surge of elastase+ neutrophils is observed in the clinically affected skin ML: 113.2 ± 4 and CL: 159 ± 7; p < 0.01).
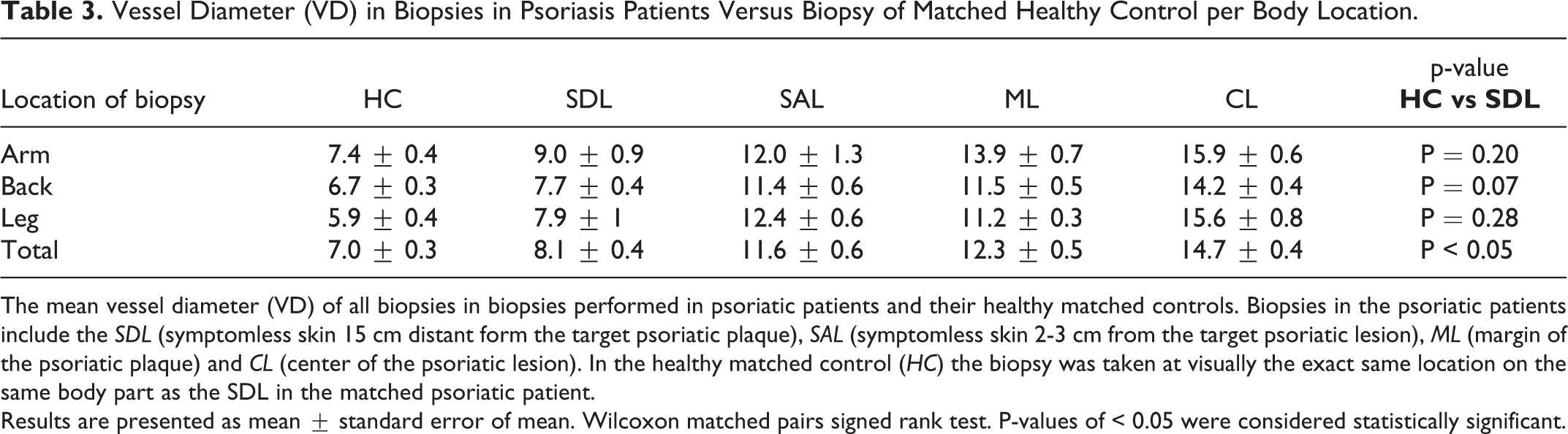
Representative photographs Elastase-staining SDL versus HC. Representative photographs of the Elastase-staining in symptomless psoriatic skin 15 cm distant from a psoriatic lesion (SDL) in 2 patients and in the 2 matched healthy controls (HC). These biopsies were performed on visually the exact same location and same body part in the psoriatic patients and their matched healthy controls.
Ki67+ keratinocyte Nuclei
SDL reveals an approximately equivalent number of proliferating keratinocytes compared to healthy skin; in both groups ± 20 Ki67+ keratinocytes per mm basement membrane were found (p = 0.8).
In the psoriatic plaque the Ki67+ keratinocytes were evidently increased (ML: 113 ± 4.0; CL: 159 ± 7.1 (in both p < 0.01)). However, an increase in proliferating keratinocytes is already present in the SAL (33.0 ± 2.6; p < 0.01) (Figure 1).
Discussion
In the present study, a statistically significant increase of the vascular diameter in all psoriatic skin sites including the SDL was observed. This result is in line with the findings of Kulka, who described the finding of dilated capillaries in symptomless psoriasis skin. 4
Furthermore, already in SDL a statistically significant influx of neutrophils is observed; the other parameters including the hypoxia did not show statistically significant differences between the SDL and skin of normal volunteers.
In symptomless skin more adjacent to a lesion (SAL) other parameters significantly increased as a concerted appearance, including vascular network size (microvascular density (MVD) and microvascular area ratio (VAR)), keratinocyte proliferation and influx of T-Bet+ Th1-cells. This is consistent with Hendriks et al who also observed an increase in vasculature in the SAL. 8,9 These changes appear also to be associated with increased oxygen consumption, given the increase of the HIF-1α expression (a marker for hypoxia). Within the psoriatic plaque itself (ML and CL) the proliferating endothelial cells significantly increase.
In the selection of immune markers we decided to have T-bet as a marker for acquired immunity and neutrophils as a marker for innate immunity. We have studied these markers in several inflammatory conditions. 17,18 However, Th17 is generally considered a more important T-cell in the pathogenesis of psoriasis. Therefore, in future studies on psoriasis we will evaluate Th17 cells (ROR-γt) in combination with IL-17 and -23 as these cytokines drive the immune pathogenesis of psoriasis.
In summary, in symptomless psoriatic skin distant from a psoriasis plaque, activation of the innate immune system, witnessed by neutrophil increases and subclinical vasodilatation can be recorded.
Closer to the psoriatic plaque, which is more downstream in the psoriatic pathogenesis, vascular and epithelial proliferation are recorded, along with HIF-1α (hypoxic) cells and activation of the acquired immunity witnessed by T-Bet cells.
Footnotes
Authors’ Note
The experiments were approved by the local ethics committee and conducted according to the Declaration of Helsinki principles. Written informed consent was given by all subjects before inclusion.
Acknowledgments
The authors would like to thank all volunteers who participated in this study and I. Otte-Holler and U. Flucke for their valuable contribution to the immunohistochemical procedures during this project.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Van der Vleuten CJM is involved as a medical consultant at Pierre Fabre. Seyger MMB received grants from/was involved in clinical trials from Abbvie, Almirall, Astellas, Janssen, Leo Pharma, Lilly and Pfizer. She served as a consultant for Abbvie, Admirall, Boehringer Ingelheim, Janssen, Lilly and Pfizer. She gave lectures for Abbvie, Janssen, Lilly and Pfizer and traveled with Abbvie, Lily, Pfizer and Leo Pharma to meetings; fees were paid directly to the institution. Van de Kerkhof PCM has carried out consultancy services for Celgene, Centocor, Almirall, Amgen, Pfizer, Philips, Abbott, Eli Lilly, Galderma, Novartis, Janssen Cilag, Leo Pharma, Sandoz, Mitsubishi, Sandoz. He carried out clinical trials for Basilea, Pfizer, Eli Lily, Amgen, Abbvie, Philips Lighting, Janssen Cilag and Leo Pharma.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was sponsored by Dermatology BV Radboud UMC Nijmegen.
