Abstract
Autologous fat grating is a widely-accepted method to correct soft tissue deficiency. Although fat transplantation shows excellent biocompatibility and simple applicability, the relatively low retention rate caused by fat necrosis is still a challenge. The vasculature is integral after fat grafting, serving multiple crucial functions. Rapid and effective angiogenesis within grafts is essential for supplying oxygen necessary for adipocytes’ survival. It facilitates the influx of inflammatory cells to remove necrotic adipocytes and aids in the delivery of regenerative cells for adipose tissue regeneration in fat grafts. The vasculature also provides a niche for interaction between adipose progenitor cells and vascular progenitor cells, enhancing angiogenesis and adipogenesis in grafts. Various methods, such as enriching grafts with diverse pro-angiogenic cells or utilizing cell-free approaches, have been employed to enhance angiogenesis. Beige and dedifferentiated adipocytes in grafts could increase vessel density. This review aims to outline the function of vasculature in fat grafting and discuss different cell or cell-free approaches that can enhance angiogenesis following fat grafting.
Introduction
Autologous fat grafting is a procedure performed by plastic surgeons with the primary goal of restoring soft tissue defects and correcting facial deficiency 1 . In recent years, it has become a prevalent surgical method in cosmetic surgery for facial rejuvenation2,3, gluteal augmentation 3 , and breast reconstructive surgery 4 . Compared with other biologically active gels such as hyaluronic acid, it shows better biocompatibility and is relatively economical for patients3,5.
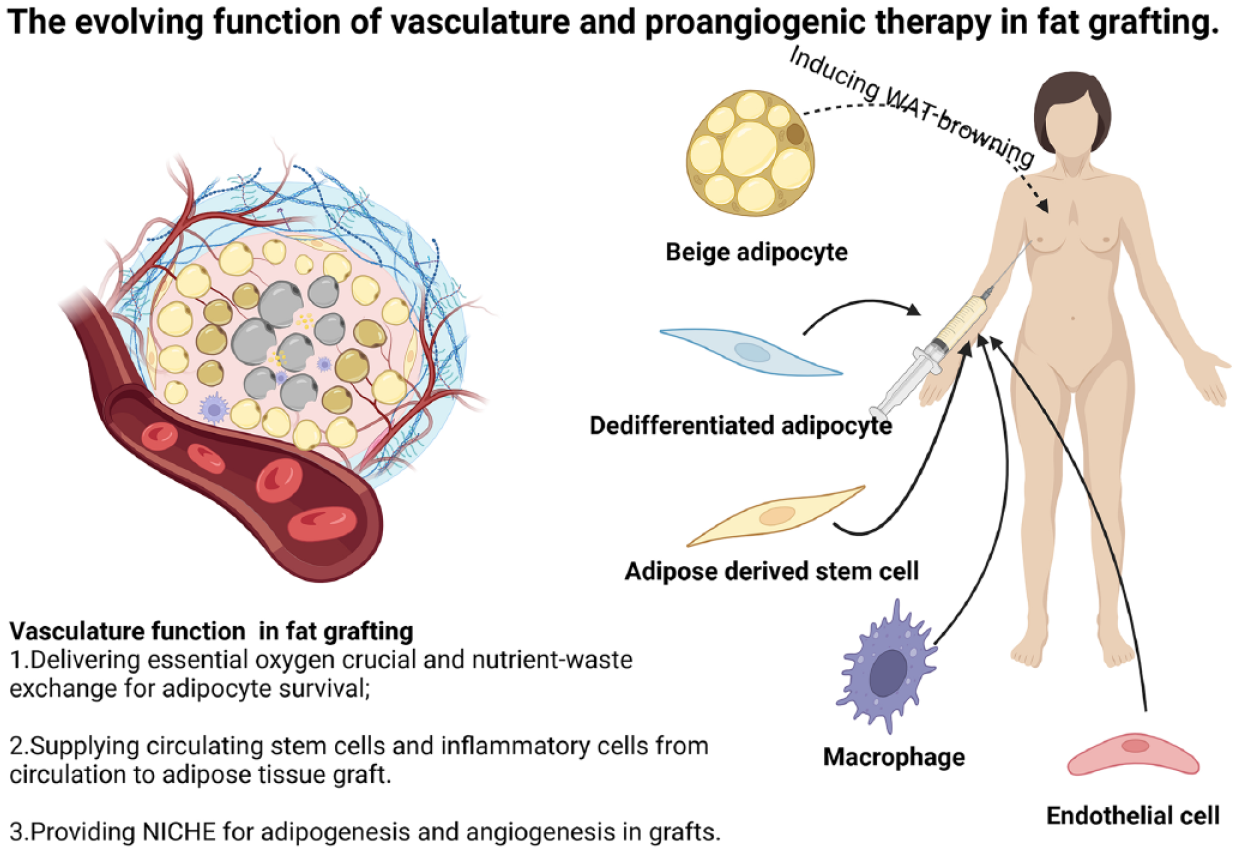
Suction-harvested adipose tissue, while effective for extraction, faces a challenge due to its lack of a natural vascular network essential for oxygenation and nutrient distribution. This deficiency can lead to undesired volume reduction, fibrosis, and the development of oil cysts due to fat necrosis6–9. Therefore, timely vascularization reconstruction is a crucial prerequisite for achieving high survival rates after fat grafting 10 . Based on the different extent of ischemic, three distinct zones emerge after fat grafting: survival (periphery), regeneration (intermediate), and necrosis (central). Initially, the boundary between the necrotic and regenerating areas is not distinctly demarcated, and the regenerative potential of the regeneration area relies on the timely ingrowth of capillaries that provide oxygen11,12. Except for providing essential oxygen, reconstructed vasculature exerts multiple functions in the fat grafting model, such as providing circulating stem cells and inflammatory cells from circulation to remodel the grafts 13 and provide a niche for the adipogenesis of perivascular cells, such as adipose-derived stem cells (ASCs), preadipocytes and pericytes, and so on (Figure 1).
Vasculature function in fat grafting. Vasculature provides a timely and enough supply of oxygen and nutrients to reduce the apoptosis of adipocytes in the early hypoxia and ischemic environment and transport unessential triglycerides to the blood to avoid lipo-toxin-induced inflammation. Vasculature delivers early-stage inflammatory cells that could clear the damaged cells and extracellular matrix to give regenerative cells a better regenerative microenvironment. Vasculature provides a perivascular niche for the mesenchymal stem cells (especially ASCs) to proliferate and differentiate into regenerated adipocytes and promote neovasculogenesis through paracrine pro-angiogenic factors or transdifferentiate into ECs. ASCs, adipose-derived stem cells; ECs, endothelial cells.
This review focuses on the vascular function that contributes to the survival and remodeling of grafts, summarizes the cellular-derived angiogenic stimulation that benefits the neovascularization after fat grafting, and provides a future perspective for the following exploration.
Vasculature Function After Fat Grafting
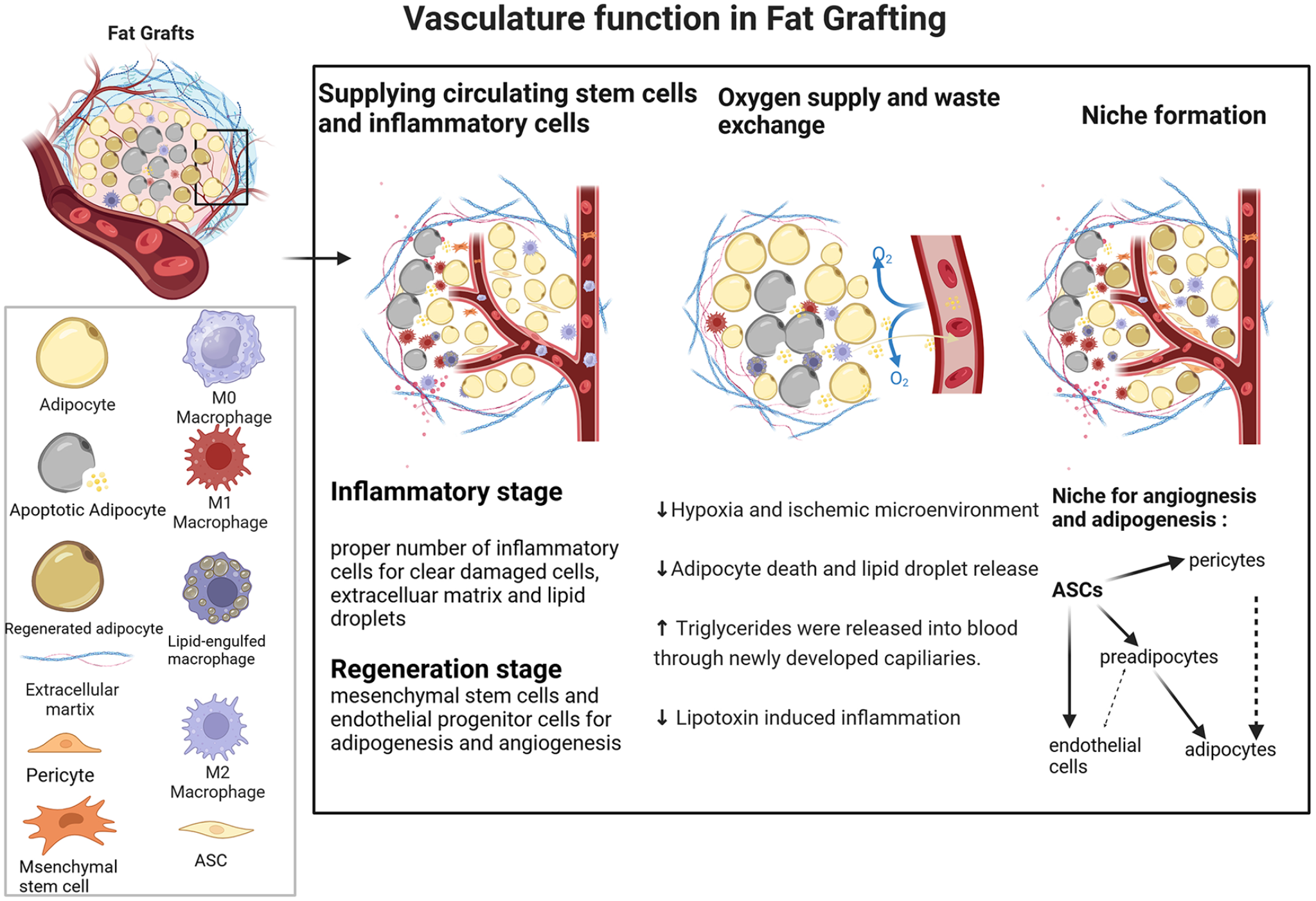
Delivering Essential Oxygen Crucial and Nutrient-Waste Exchange for Adipocyte Survival
Following grafting, adipose tissue enters a state of severe ischemic and hypoxia, characterized by low oxygen levels 13 . Most adipocytes in the graft begin to die within 24 h ASCs remain viable for up to 72 h but still demonstrate limited resistance to hypoxia. Hence, the angiogenesis process within the first 72 h after grafting assumes paramount importance. This period is crucial for ensuring the timely delivery of oxygen and nutrients to rescue the transplanted cells, ultimately influencing their fate13,14.
Before angiogenesis occurs, grafted adipose tissue can only receive oxygen through plasmatic diffusion from surrounding vessels 12 . It takes 3–7 days for the peripheral vessels to ingrow into the outer layer of the grafts 15 . Moreover, newly formed blood vessels in grafts provide nutrients and exchange metabolic products after 2 days of implantation16,17. To guarantee sufficient blood and oxygen delivery to the graft, each fat graft droplet must interface with a capillary recipient site in a 1:1 ratio to establish a successful fat–recipient complex. Excessive fat droplets relative to capillary recipient sites can result in inadequate neovasculogenesis, potentially leading to fat resorption and necrosis 18 . Therefore, smaller grafts tend to exhibit better survival rates compared with larger grafts due to the higher surface-to-volume ratio, which allows for a larger area of the graft to be in contact with the vascular bed and facilitates more efficient vascularization16,19. Adequate oxygen supply is also essential for macrophages to effectively clear dead adipocytes and lipid droplets by maintaining proper fatty acid oxidation 20 . Moreover, timely vascularization provides a channel for transporting excess triglycerides into the bloodstream, thereby reducing lipotoxin-induced massive inflammation21–24.
Supplying Circulating Stem Cells and Inflammatory Cells From Circulation to Adipose Tissue Graft
After grafting, macrophages infiltrate into the grafts in the early stage to promote remodeling by clearing free fatty acids, phagocytosing dead cells, and digesting damaged extracellular matrix (ECM) components. These activities create space for regenerative cells such as ASCs, endothelial progenitor cells (EPCs), and other stem cells, which can enhance angiogenesis and adipogenesis25–27. Traditional perspectives considered that macrophages infiltrated into the inflammatory site are mainly from circulation 28 . In recent investigations, it has been established that during the initial week post-surgery, the proportion of infiltrated macrophages derived from the fascia surpasses that of macrophages originating from circulation. Notably, experiments involving the removal of fascia from the back of recipient mice in the fat grafting model have revealed a subsequent reduction in the number of early-stage macrophage infiltrations 29 .
New vasculature also provides channels for the circulating stem cells, new vessels within the graft predominantly arise from recipient vessels growing into the graft, rather than from the reassembly of donor endothelial cells (ECs) or the reconnection of recipient and donor vessels 30 . In the nascent phase of angiogenesis within the grafts, conduits are established to facilitate the ingress of blood-derived hematopoietic cells and ASCs, thereby enabling their pivotal contribution to the initial stages of regeneration 31 . From a long-term perspective, most of the CD34+ cells are derived from the recipient during adipogenesis rather than in the initial grafts 30 . Besides, bone marrow-derived endothelial progenitors are also capable of migrating to the ischemic location, then differentiate into ECs and finally become capillaries in the superficial graft zones32,14.
Providing NICHE for Adipogenesis and Angiogenesis in Grafts
Mounting evidence indicates that ASCs along with other types of mesenchymal stem cells (MSCs) are closely linked to vascular and perivascular niches in different tissues 33 . During human embryonic development, adipocytes first appear from emergent vascular networks 34 . Lineage-tracing studies have demonstrated that adipocyte progenitors reside within the walls of adult mouse adipose tissue capillaries35,36. Co-implantation of adipose tissue with ASCs differentiated toward an endothelial phenotype significantly enhances both the survival and angiogenesis of the transplanted adipose tissue 37 . Coculturing EPCs with ASCs enhances EPCs’ tube formation capacities and angiogenesis markers such as tie2 and vascular endothelial growth factor (VEGF)-A. ASCs’ combination enhances the function of EPCs in the fat transplant rather than directly differentiating into them 38 . ASCs, often found alongside microvessels, stabilize the perivascular niche by differentiating into pericytes and expressing key pericyte markers such as α-smooth muscle actin (α-SMA), neural/glial antigen 2 (NG2), or platelet-derived growth factor receptor β (PDGFRβ) 39 . Indeed, perivascular cells such as vascular smooth muscle cells and pericytes have been proven to give rise not only to white adipocytes but also to differentiate into beige adipocytes40,41, which are located around the vasculature, sharing the same location as pericytes 42 . Inducing more pericytes in transplanted adipose tissue could also enhance the adipocytes’ viability and increase the survival of adipocytes 43 , which could also shed light on the function of pericytes as the orchestrators of vasculature and adipogenesis 40 . Besides, the reciprocal interaction between preadipocytes and endothelial cells (ECs) was promoted by the infiltration of blood vessels 44 . Preadipocytes induce and guide EC migration through fibroblast growth factor (FGF)- and vascular endothelial growth factor (VEGF)-dependent pathways 45 . Conversely, the VEGF-triggered signaling pathway in ECs promotes preadipocyte differentiation via a paracrine mechanism 46 . Differentiation from preadipocytes to mature adipocytes is associated with increased production of angiogenic factors that induce robust angiogenic responses 47 .
Cell-Enrichment Therapy in Fat Grafting
Enhancing fat grafts with diverse cell types that demonstrate significant angiogenic activity is presently recognized as a highly effective approach for enhancing the neovasculogenesis of transplanted adipose tissue. To achieve this objective, macrophages, ASCs, and other cells from the stromal vascular fraction (SVF) have been proposed 10 .
Endothelial Progenitor Cell
EPCs obtained from humans can integrate into sites of active angiogenesis, thereby enhancing the growth of collateral vessels in ischemic tissues 32 . The sprouting of the peripheral vessels toward the graft typically spans a period of 3–7 days with the recruitment of EPCs15,48–50. Bone marrow-derived EPCs migrate to a target ischemic site, then differentiate into ECs, and finally become capillaries for the superficial graft zones32,14. However, the EPCs cannot migrate through the outer graft layers to access the ischemic center, meaning that the inner part depends on its progenitor cells to induce capillary formation51,52. Combining EPCs with fat grafts has been shown to enhance early-stage angiogenesis while minimizing the development of oil cysts and fibrosis. In their pursuit of EPCs with heightened regenerative potential, Asahara et al. utilized a serum-free medium comprising five different vasculogenic components (VEGF, stem cell factor, Flt-3 ligand, thrombopoietin and interleukin 6)—designed as a Quality and Quantity culture (QQ culture) for cell expansion and vasculogenic of EPCs 53 . Maxim et al. discovered that when transplanted with QQ cultured c-Kit+ Sca-1+ Lin− (KSL) EPCs, grafts get better angiogenesis and less fibrosis and inflammation levels 52 . Their further study proves that the QQ culture method also enhanced the vasculogenic potential of peripheral blood mononuclear cells (PBMNCs) by enriching them with EPCs and anti-inflammatory M2-monocyte/macrophages. Grafts enriched with MNC-QQ exhibit the highest vessel density and secrete increased levels of VEGF paracrine 51 .
Macrophage
In the normal response to injury of multiple tissue types, monocytes infiltrate the wound site and differentiate into macrophages that exhibit a highly pro-inflammatory phenotype, at later stages, the macrophage population undergoes a shift in phenotype to pro-regenerative status54,55 (Figure 2). The conventional classification of macrophage phenotype can be delineated into two primary types: the pro-inflammatory M1-polarized phenotype and the regenerative, pro-angiogenic M2-polarized phenotype 56 . Another article has challenged this viewpoint, both M1 and M2 macrophages can facilitate the vascular reconstruction and vascular remodeling process. Primary human M1 macrophages secrete high levels of VEGF and increase the expression of VEGFR2 on EC 57 . In the M2 macrophage subset, M2a macrophages secrete PDGF-BB, stabilizing pericytes and enhancing EC sprouting. Meanwhile, M2c macrophages release MMP9, contributing to basement membrane degradation and ECM remodeling. Besides, M2c-derived osteopontin promotes EC proliferation and migration 58 .
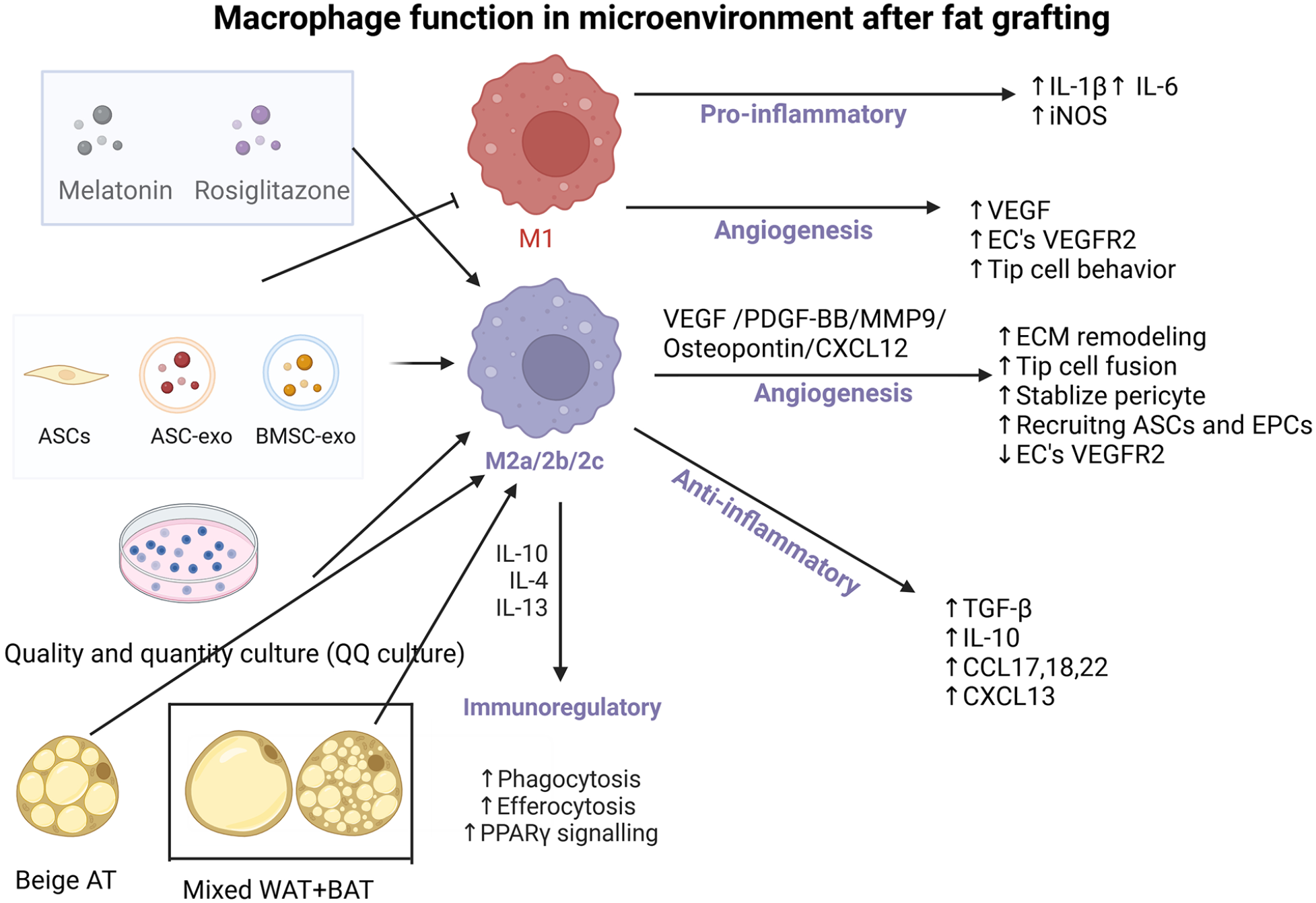
Macrophage function in microenvironment after fat grafting. IL, interleukin; iNOS, inducible nitric oxide synthase; VEGF, vascular endothelial growth factor; VEGFR2, vascular endothelial growth factor receptor 2; ECM, extracellular matrix; TGF-β, transforming growth factor-β; CCL, C-C motif chemokine ligand; CXCL, C-X-C motif chemokine; PPARγ, peroxisome proliferatoractivated receptor γ; PDGF-BB, platelet-derived growth factor subunit B.
Monocyte infiltration into the grafts shows the same pattern as the common injury. During the first week after surgery, grafts enter an “inflammatory phase,” M1 macrophages emerge within the first week to facilitate the clearance of necrotic tissue and release pro-inflammatory factors26,59,60. Then in the regeneration stage (weeks 2–4 and beyond), there is a gradual transition as M1 macrophages are gradually replaced by M2 macrophages, which shift toward regulating angiogenesis and suppressing inflammation59,61,62. M2 macrophages facilitate the sprouting of pre-existing blood vessels and their subsequent fusion to form new functional vasculature. This process is mediated by the direct localization of M2 macrophages at the fusion sites, where they engage with integrins on the EC surfaces, thereby promoting the connection of adjacent tip cells 63 . Moreover, as previously mentioned, osteopontin derived from M2 macrophages also contributes to promoting endothelial tip cell linkage by interacting with integrins on the surface of ECs 64 . Numerous factors in fat grafting contribute to the enhancement of M2 macrophage polarization (as depicted in the figure), indirectly fostering angiogenesis and shifting the pro-inflammatory microenvironment toward an anti-inflammatory state post-fat grafting65–67.
Manipulating macrophage infiltration by using liposome-clodronate/Macrophage-Colony Stimulating Factor (M-CSF) resulted in either incompetent or improved angiogenesis after grafting 68 . Early macrophage infiltration in grafts demonstrates a robust correlation with the onset of early neovasculogenesis. The early presence of macrophages plays a pivotal role in attracting more Sca-1+/CD45+ stem cells capable of generating ECs and neovessels possibly through the CXCL12/CXCR4 axis 68 , and CXCL12 are also capable of promoting EPCs mediated angiogenesis and reduce the fat apoptosis 69 . Chen et al. highlight the reciprocal mechanism that early angiogenesis is also essential for CXCL12-dependent ASC recruitment. Besides, inhibiting CXCL12 expression at the donor site in the autologous fat grafting model promotes increased ASC infiltration into the recipient site, thereby raising graft retention rates 31 .
Macrophage cell-enrichment therapy increases the vascularization of grafts. Both M0, M1, and M2a could enhance the revascularized ability in transplanted vascular grafts or glutaraldehyde-crosslinked scaffolds57,58. Mixing M2 macrophage with white adipose transplant also increases the angiogenesis after 3 months of grafting 70 . QQ-cultured PBMNCs including EPCs and M2 macrophage also increase the neovascularization in the grafts both in 1 week or 7 weeks after grafting. Moreover, QQ-cultured PBMNCs promote angiogenesis through upregulating the expression of VEGF-A in the graft and QQ-cultured MNCs are capable of becoming a part of the newly formed vessel 51 .
ASCs’ Function in Cell-Assisted Lipotransfer (CAL)
Advanced regenerative therapies focus on harnessing the potential of stem cells, for direct application to damaged sites, known as cell therapy, or for tissue engineering by utilizing suitable scaffolds as carriers for these cells which holds distinct advantages over differentiated cells 71 . Adipose tissue has been historically considered a passive energy storage. However, currently, it is well known that it is also a rich source of multipotent ASCs72,73. ASCs express the surface MSC markers CD73, CD90, CD105, and STRO-1 and display a broad range of regenerative properties including a high replication capacity and potential to differentiate into various cell lineages, as well as the ability to secrete numerous pro-angiogenic growth factors, which are essential for promoting the formation of vascular networks and supporting the vasculature hemostasis74,75.
Yoshimura’s group pioneered the utilization of exogenous ASCs in fat grafting and named this procedure “cell-assisted lipotransfer (CAL).” By elevating the local concentration of ASCs during the initial phase of grafting, CAL enhances angiogenesis and consequently improves graft retention rates 76 .
VEGF (also known as VEGF-A) 77 serves as an excellent target for enhancing the paracrine function of ASCs in promoting the formation and stabilization of vascular networks within grafts. VEGF induces proliferation, sprouting, migration, and tube formation of ECs and also keeps the ECs survive77–79 (Figure 3). Oxygen deprivation activates hypoxia-inducible factor 1α (HIF1α) in macrophages and ASCs, leading to the expression of VEGF-A, which stimulates the specification of ECs into tip cells and stalk cells. These tip cells, characterized by their high motility, actively sense environmental guidance cues and guide the nascent sprouts by forming filopodia. Stalk cells provide structural support to the nascent capillary as part of the body of the nascent sprout. Mechanistically, VEGF binds to the VEGFR2 receptor expressed on tip cells, promoting their proliferation, migration, and adhesion. Concurrently, the phosphorylation of the VEGFR2 receptor activates the DLL4-NOTCH1 signaling pathway in adjacent ECs, which inhibits VEGFR2 expression in these neighboring cells. This inhibition promotes the stalk cell phenotype, which matures and stabilizes the nascent sprout by recruiting more pericytes and ECM 77 .
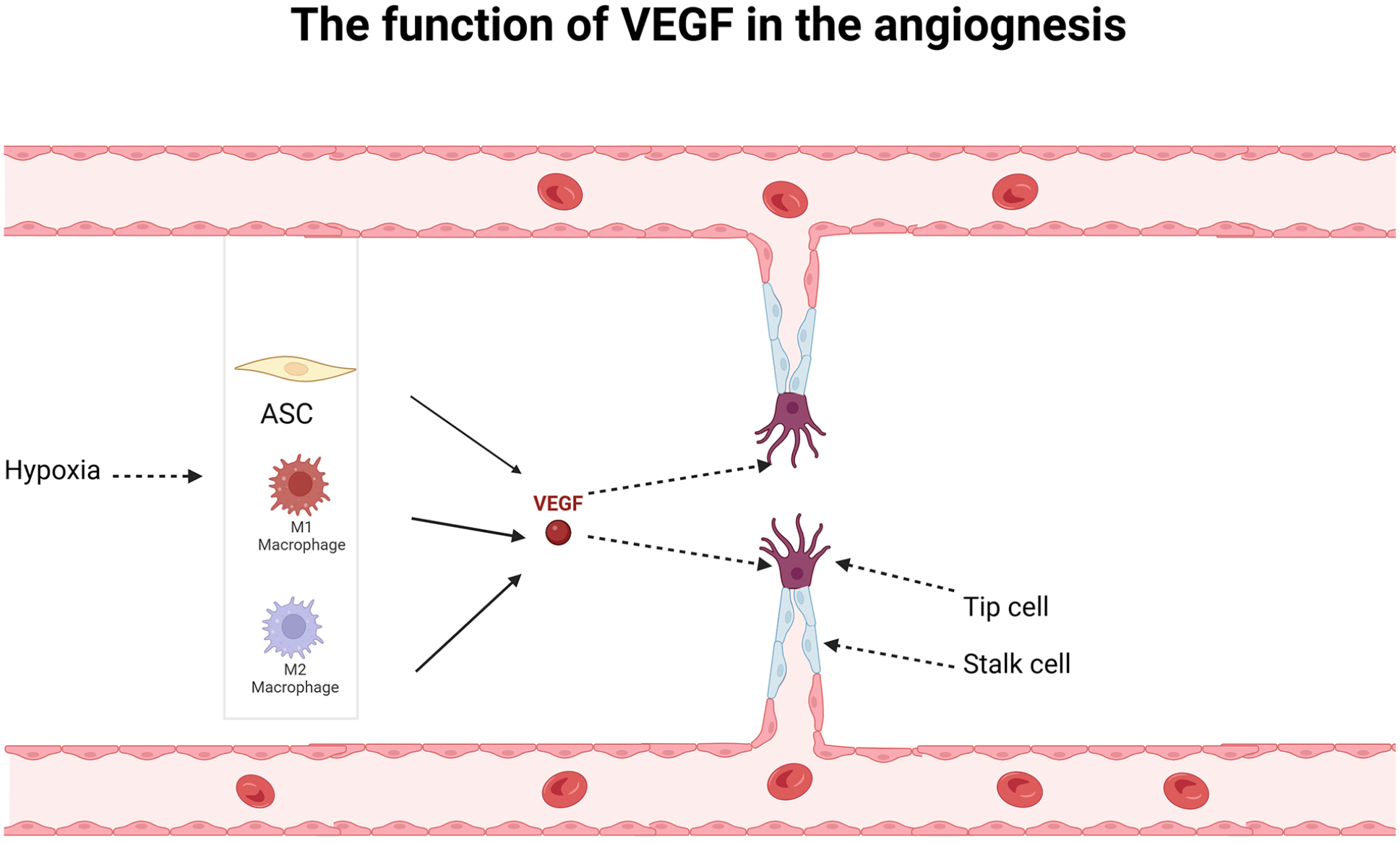
The function of VEGF in the angiogenesis. Under hypoxia conditions, macrophages and ASCs-derived VEGF specify the expression of endothelial cells into tip cells and stalk cells. Tip cells function as a guider to build connections with the adjacent vessels and stalk cells function as a supporter to give structural support.
HIF1α-VEGF axis in ASCs mediates the angiogenesis in grafts. C-reactive protein (CRP) affected the proliferation and pro-angiogenic paracrine activity of ASCs through upregulating VEGF-A expression by activating HIF-1α 80 . Moreover, inhibiting the negative regulator of VEGF-A with a DII4-neutralizing antibody enhances the angiogenesis of grafts and exhibits synergistic effects when combined with concurrent ASC supplementation 81 .
Besides, the overexpression of miR-126 in ASCs results in increased expression of VEGF. Transplanting adipose tissue with exogenous ASCs that overexpress miR-126 enhances the angiogenic capacity of grafts. This effect is mediated through the Erk/Akt pathway rather than via only VEGF signaling 82 . Besides, transplanting white adipose tissue (WAT) with exogenous ASCs transduced with VEGF also increases the angiogenesis and capillary density in the grafts 75 .
The trans-differentiation of ASCs into ECs and pericytes represents another mechanism contributing to angiogenesis in grafts following CAL. EC-derived basic FGF (bFGF) triggers endothelial differentiation in ASCs by inhibiting miR-145 through the PI3K/AKT/FOXO1 signaling pathway. This inhibition relieves suppression of the angiogenic transcription factor ETS1, thereby promoting the differentiation of ASCs into ECs 83 . Downregulation of miR-145 in ASCs induced vascular network formation and neovessels incorporation in ischemic muscle in vivo 83 . Combined treatment of ASCs with VEGF and bone morphogenetic protein-4 (BMP4) under hypoxia conditions induces the differentiation of ASCs into vascular EC lineage and acquires the ability of mature EC such as tube formation, low-density lipoprotein (LDL) uptake, and nitric oxide secretion84,85. Besides, treating ASCs with TGF-β induces their differentiation into pericytes. Transdifferentiating more ASC into pericytes could enhance neovascularization in CAL 43 . Moreover, culturing ASCs with endothelial growth medium (EGM-2) in vitro and transplanting them with human umbilical vascular ECs (HUVEC) subcutaneously into immunodeficient mice increases the HUVEC survival and enhances the vasculature function 86 .
There remains uncertainty regarding the mechanism by which exogenous ASCs become involved in the angiogenesis process during CAL, particularly in the crucial early stages following surgery. Yuan et al. gave a hypothesis that exogenous ASCs in free fat transplantation may not directly engage in angiogenesis through differentiating into mature adipocytes or ECs. However, they are believed to enhance the survival rate of graft-resident interstitial cells through paracrine effects 87 . Gan et al. 38 also demonstrated that ASCs combined with EPCs enhance the function of EPCs in the fat transplant rather than directly differentiate into them. Conversely, using a tracing method to reveal the differentiation of donor ASCs in the CAL at 4 weeks after transplantation, Hong et al. 88 demonstrated that donor-derived ASC can directly differentiate into ECs and adipocytes to take part in angiogenesis. Evidence suggests that ASC-derived ECs can be assembled into newly formed vessels 89 . CD34+CD31– cells sorted from SVF cells can differentiate into ECs in vitro and incorporate into the ischemic hindlimb vasculature in vivo 90 . While intravenously injecting ASCs immediately after surgery does increase vascular density in the graft, their primary mechanism is through paracrine signaling 91 . Yan Huang et al. further demonstrated that intravenous injection of ASCs immediately after surgery and again at 2 weeks post-surgery led to increased retention of graft volume and vascular density. Simultaneously, grafts that are intravenously injected with ASC exhibit higher expression levels of CXCL12 and CXCR4 92 .
ASC-exosome is another strategy to enhance early angiogenesis. Recent research findings indicate that exosomes derived from stem cells exhibit comparable or even superior performance to that of MSCs93–95. In comparison with ASCs, ASC-exos offer several advantages. These exosomes are small extracellular vesicles (50–200 nm) that can be sterile filtered and stored as safe, ready-to-use biological products with high biological activity. Furthermore, their use circumvents issues associated with cell therapy, such as limited cell survival, senescence-induced genetic instability, and the risk of unfavorable differentiation 10 .
ASC-exos also facilitate angiogenesis within grafted adipose tissue through both direct and indirect means96–98. Directly, ASC-exos promote angiogenesis and upregulate early-stage inflammation, which is beneficial for clearing dead cells and creating a pro-regenerative environment 96 . ASC-exos enhance the proliferation, migration, and tube formation capacity of HUVEC through the HIF-1α/VEGF signaling pathway 99 . Moreover, hypoxia-treated ASC-exo contains more pro-angiogenic growth factors than normal ASC-exos 98 , which fits the microenvironment after grafting 13 . Injectable thermosensitive hydrogels show significant promise as carriers for exosomes 100 . Exosomes encapsulated within hydrogels can be utilized to regulate their release over extended periods, enhancing their therapeutic effects and efficacy 99 . Human ASCs-derived exosomes (hASC-Exos) encapsulated in PF-127 hydrogel have been shown to enhance the neovascularization of autologous fat grafts 99 .
Indirectly, ASC-exosomes may increase the vessel density and browning of WAT in grafts by inducing more M2 macrophage differentiation and suppressing M1 macrophage differentiation101–103.
Adipocytes’ Pro-angiogenic Function in Fat Grafting
Brown and Beige Adipocytes
Adipose tissue consists of three primary types of adipocytes. White adipocytes, predominant in WAT, are unilocular cells specializing in lipid storage. In contrast, beige and brown adipocytes are multilocular, mitochondria-rich cells specialized in energy dissipation through non-shivering thermogenesis 104 .
Beige adipocytes arise from subcutaneous WAT (sWAT) depots in response to β-adrenergic stimulation, dietary factors, or exposure to cold, a reversible process known as browning or beiging 105 . Although originating from myogenic transcription factor 5 (Myf5)-negative mesodermal stem cells like white adipocytes, beige adipocytes functionally resemble brown adipocytes. They possess the capacity to convert chemical energy into heat in response to specific stimuli 106 .
Transplantation of brown adipose tissue has been widely utilized in the treatment of diabetes, obesity, and their associated complications107–110, owing to the high secretion of autocrine and paracrine factors 111 . Compared with the transplantation of white adipocytes, the transplantation of rosiglitazone-induced beige adipocytes also offers metabolic benefits by alleviating obesity-induced inflammation and increasing the proportion of high-density lipoprotein (HDL) 112 . Moreover, the beigeing of perivascular adipose tissue caused by vascular injury alleviates inflammation by expressing neuregulin 4, which activates M2 macrophage polarization 113 .
Brown and beige adipocytes with a higher surface-to-volume ratio can facilitate the process of diffusion and increase the relative exposure area to oxygen114–116 (Figure 4). In comparison with traditional white adipocytes, PPAR-γ-induced beige adipocytes exhibit higher tolerance to hypoxic and inflammatory environments 115 . Beige adipose grafts are demonstrated with increased angiogenesis, recruiting fewer macrophages in the early stages, and consequently, exhibiting enhanced survival after the transplantation 115 . Overexpressing VEGF-A in adipocytes enhances its angiogenesis and induces beige fat formation in the doxycycline(dox)-inducible, VEGF-A overexpressing transgenic (VEGF-Tg) mouse model. Additionally, transplantation of subcutaneous adipose tissue from dox-inducible VEGF-Tg mice into the interscapular region of wild-type C57/BL6J mice, followed by a 3-week high-fat diet regimen, demonstrates enhanced adipocyte survival and reduced macrophage infiltration in VEGF-A-overexpressing adipose grafts 117 . Moreover, transplantation of cold-stimulated induced beige adipose tissue reveals heightened angiogenesis levels and a relatively diminished inflammatory state 116 .
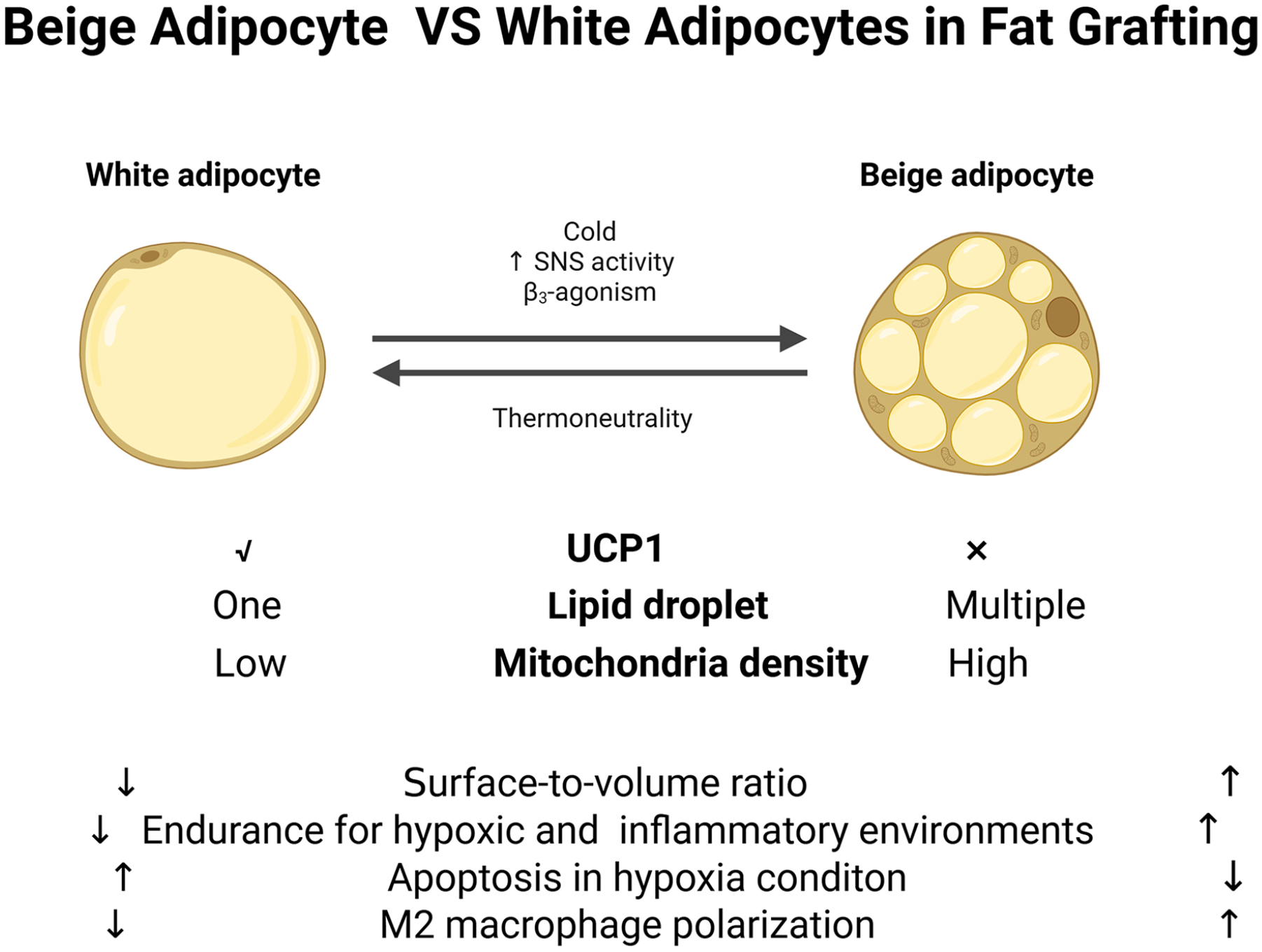
The difference between beige adipocyte and white adipocyte. White adipocytes are unilocular cells specializing in lipid storage with one big lipid droplet and low mitochondria density. The beige adipocytes are multiple lipid droplets with the expression of UCP1 and a high level of mitochondria density. Under hypoxia conditions, beige adipocytes show better endurance and less apoptosis compared with white adipocytes. higher surface-to-volume ratio makes it more easily to get enough blood supply and therefore more easily to survive after fat grafting. Beige adipocytes could also induce more M2 macrophage polarization. UCP1, uncoupling protein 1.
However, brown adipocytes within grafts may struggle to withstand hypoxic and ischemic conditions, leading to sustained high levels of inflammation due to the “whitening” of brown adipose tissue which results in adipocyte death and apoptosis95,118. Transplanting brown adipose tissue (BAT) typically exhibits lower retention rates compared with beige adipose tissue and even WAT, partially due to the heightened inflammatory state115,119. However, Zheng et al. 120 found that transplanting grafts that contain a mixture of BAT and WAT increased the recruitment of M2 macrophage and the expression of VEGF-A. Moreover, inducing the browning of white adipose transplant after grafting could also increase the expression of VEGF in the grafts and enhance angiogenesis 119 .
Dedifferentiated Adipocytes (DAs)
Traditional views depict adipogenic differentiation as an irreversible process where MSCs transition into mature adipocytes. Mature adipocytes were believed to possess limited plasticity, primarily responding to stimuli with changes in size rather than undergoing further dedifferentiation121,122. Through the “ceiling culture” method, mature adipocytes exhibit dedifferentiation, ultimately transforming into progenitor cells expressing markers characteristic of adipose stem cells. Similar to stem cells, these progenitor cells exhibit proliferative and redifferentiation capabilities. Additionally, they demonstrate multilineage differentiation potential, differentiating into various cell types both in vitro and in vivo123–126 (Figure 5).
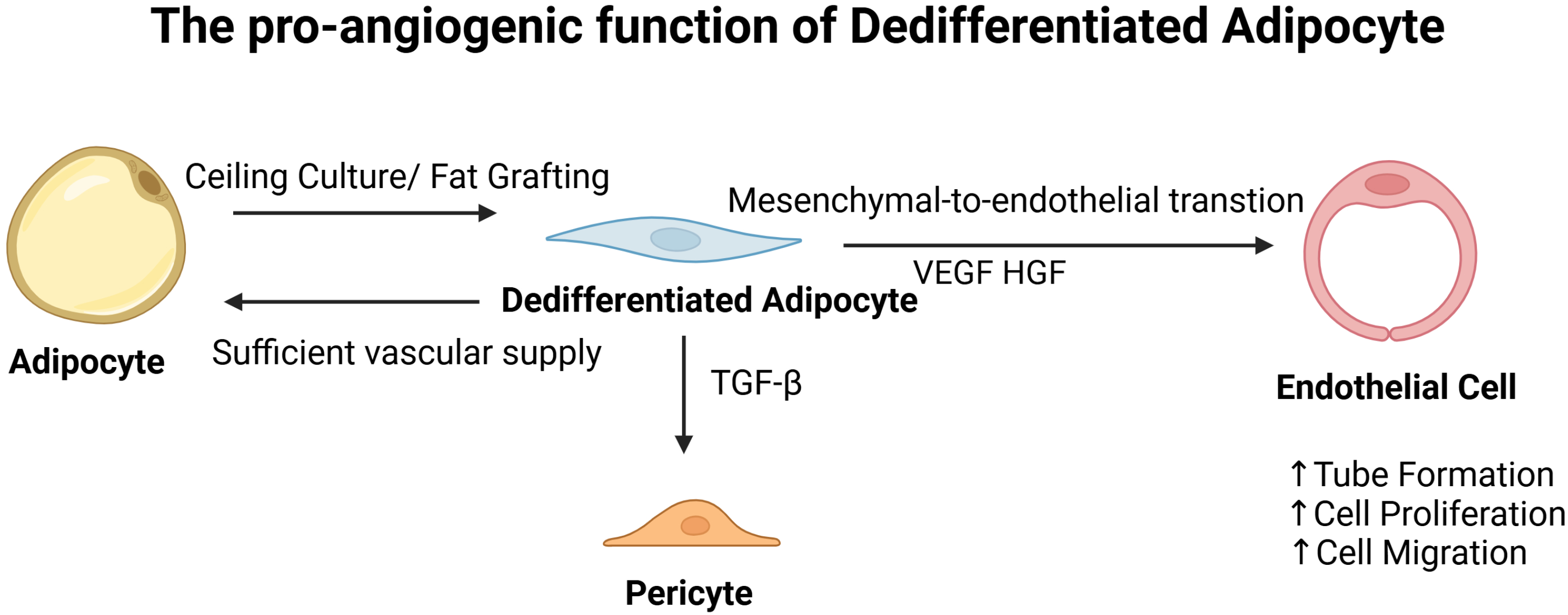
The pro-angiogenic function of dedifferentiated adipocyte. DAs in was founded in the SVFs in the grafts after 1 week of fat transplantation. In vitro ceiling culture also induces adipocyte dedifferentiation. With enough vascular supply, DAs could redifferentiate into mature adipocytes. DAs acquire the ability to differentiate into ECs or pericytes and also enhance the proliferation, cell migration, and tube formation of ECs through secretory proteins such as VEGF and HGF. HGF, hepatocyte growth factor; VEGF, vascular endothelial growth factor.
DAs are demonstrated to have the same adipogenesis and angiogenesis potential compared with ASCs in fat grafts 127 . DAs can promote EC tube formation in vitro and facilitate angiogenesis in ischemic hindlimbs, potentially through the secretion of factors such as VEGF-A and hepatocyte growth factor (HGF). Moreover, when co-cultured with ECs, DAs express pericyte markers. TGF-β can also induce DAs to differentiate into mural cells in vitro 128 . In 1 week after fat grafting, the mature adipocytes in grafts were found to differentiate into DAs 129 , which can also spontaneously undergo mesenchymal-to-endothelial transition and differentiate into ECs. This transition enables them to participate in neovascularization, particularly in response to BMP4 and BMP9 signaling 130 . Interestingly, host-derived adipocytes contribute to fat graft regeneration by migrating from the recipient inguinal fat pad into the grafts and undergoing dedifferentiation, which can be augmented by internal expansion to enhance the retention rate of fat grafts 131 . Moreover, given a sufficient vascular supply, DAs have the potential to undergo redifferentiation into mature adipocytes132,133, which might be a way for them to survive in the regeneration area.
Future Perspective of Vascularization in Fat Grafting
As mentioned above, timely and complete neovascularization in the grafts determines the fate of cells in the grafts. Therefore, numerous efforts have been made to enhance early-stage revascularization by adding different kinds of angiogenic cells such as EPCs, ASCs, or other in vitro QQ cultured ECs and peripheral mononuclear cells31,51,52. A more detailed understanding of the precise mechanism through which these cells integrate into the microenvironment’s vasculature after grafting, and their interactions with other progenitor cells to support adipogenesis, ECM remodeling, and adipocyte regeneration, remains a crucial focus for future research. Finding a stable source of vessel-forming progenitor cells and adding them to the grafts provides a novel strategy to rescue the ischemic microenvironment after grafting 134 . Enriching the ASCs filtered with more angiogenic potential might be another choice to get better vascularization after grafting. A subpopulation of human ASC marked by CD34+CD146+ enhances the induction of tube formation of HUVECs in vitro. Enriching fat with CD34+CD146+ ASCs shows better vascularization compared with CD34+ unsorted SVF cells 135 . Another two populations of distinct vascular stem/progenitor cells (VSPCs) are identified from the ASCs subpopulation, marked respectively by CD45–Ter119–Tie2+PDGFRa–CD31+CD105highSca1low and CD45–Ter119–Tie2+PDGFRa+CD31CD105lowSca1low. Cotransplantation of these two types of cells forms functional vessels that improve perfusion of the mouse hindlimb ischemia model. However, there is still little article reporting that applying the different angiogenic potential of ASCs in the CAL 134 . Identifying novel progenitor cells that can develop into fully functional blood vessels, continues to be a significant challenge136–138. The significant variability in angiogenic potential among identical types of progenitor cells, such as distinct subtypes of ASCs, underscores the need for further investigation136,139.
In recent years, single-cell RNA sequence (scRNA-seq) has become a popular method to detect the deconvolve tissue heterogeneity140–142. Quite a lot of studies have been applied in analyzing the cells of SVFs in the WAT and brown adipose tissue 143 . Although susceptible to certain technical noise, single-cell RNA sequencing (scRNA-seq) offers vital insights into cellular populations, gene expression patterns, and variations in cellular states that may occur under diverse physiological conditions141,144. It is quite useful to filter the subpopulations according to their functions in the tissue and identify more angiogenic subpopulations in the SVFs 145 . The latest spatial transcriptomic to build a high-resolution cellular map for further exploration 146 . However, there is still no research applying the scRNA-seq in fat grafting to reveal the dynamic cellular map and the function of different stem and progenitor cells.
Another emerging technology in recent years is 3D-culture in vitro, compared with cells cultured as monolayers (2D), 3D spheroids can more accurately mimic the in vivo cell growth environment while retaining their biological characteristics. 3D-cultured ASCs demonstrate increased proliferation potential and reduced senescence compared with conventional culture methods. Co-culturing 3D-cultured ASCs with HUVECs enhances their neovascularization capabilities. Employing 3D-cultured ASCs in CAL improves the survival and neovascularization of ASCs 147 .
Conclusion
This review summarizes the current knowledge about vasculature function in fat grafting as well as the different cellular compositions that improve revascularization during the process of fat regeneration. The results provide a greater understanding of the application of different cell-enrichment therapies. However, there are still a lot of unknown mechanisms for vasculature function in fat grafting 44 .
Footnotes
Acknowledgements
Authors’ Note
Zhang Xining as an Independent First Author in this review.
Author Contributions
Z.X. performed the literature search and wrote the manuscript and figures. L.S. conceived the project and revised the manuscript.
Availability of data and material
The authors confirm that the data supporting the findings of this study are available within the article.
Ethical Approval
This study was approved by our institutional review board.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The author(s) was supported by the National Natural Science Foundation of China (82102356), Outstanding Young Medical Talents Training Funding Project of The First Affiliated Hospital of Harbin Medical University (2021J05).
