Abstract
Objective
We propose a standardized approach of using the tendon of the sternocleidomastoid (SCM) muscle to locate the spinal accessory nerve (SAN) in neck dissection.
Study Design
Cross-sectional anatomic study.
Setting
Tertiary academic medical center.
Methods
Adult patients aged ≥18 years undergoing primary neck dissection for head and neck cancer were included. Anatomic measurements included the length of the SCM tendon, the distance from the mastoid tip to the entrance of the SAN into the SCM, the distance from the SAN to the distal edge of the SCM tendon, and the perpendicular distance from the anterior edge of the SCM to the SAN. Five cadaveric specimens also underwent bilateral modified radical neck dissections with the same anatomic measurements taken.
Results
Twenty-two living subjects and 5 cadavers were included. No statistical correlation was noted between patient demographics and any measurement. The mean (SD) length of the SCM tendon was 63.7 mm (11.8) in living subjects and 61.5 mm (10.4) in cadaveric specimens. The average distance from the mastoid tip to the entrance of SAN into the SCM was 51.6 mm (12.2) in living subjects and 51.6 mm (7.1) in cadaveric subjects. The distance of the SAN insertion into the SCM muscle from the anterior edge was 8.9 mm (3.4) in living subjects and 16.2 mm (7.2) in cadaver specimens. Laterality was compared in the cadaveric specimens; there was no statistically significant difference in any of the measurements between sides.
Conclusion
This study demonstrates the SCM tendon to be a reliable and safe surgical landmark to identify and preserve the SAN during neck dissection.
Since radical neck dissection was first described by Crile in 1906, patients with head and neck cancer have undergone therapeutic neck dissection for locoregional control of cervical lymphatic metastasis. 1 Since this description, advancements in lymphadenectomy have brought about modified radical and selective neck dissection techniques, which aim to preserve neurovascular and associated muscular structures. These newer techniques reduce patient morbidity associated with sacrifice of the spinal accessory nerve (SAN), internal jugular vein, and sternocleidomastoid (SCM) muscle in a radial neck dissection. In 1963, Suárez first described the modified radical neck dissection, which preserves one or more of the functionally important structures of the neck, including the SAN, while still removing of all of the contained lymph nodes from levels 1 through 5 of the neck. 2
The SAN, or CNXI, provides motor innervation to the SCM and trapezius muscles. Injury to the SAN can cause significant morbidity to the patient in what is known as “shoulder syndrome.”3-5 With diminished strength and mobility of the shoulder and upper arm, patients can experience debilitating pain in the neck, upper back, and upper extremity from the altered muscular support of the arm and shoulder. For this reason, several landmarks have been proposed to locate the SAN during neck dissections, including the great auricular nerve and SCM branch of the occipital artery.6-8
The SCM tendon, in combination with the posterior belly of the digastric muscle and SCM branch of the occipital artery, has recently been described as a reliable landmark to safely locate the SAN during neck dissection in cadaveric models. 9 In this study we present a case series in living subjects that describes the relationship of the SCM tendon to the SAN. To our knowledge, this is the only such study in living subjects that compares this relationship. We aim to provide a reliable method for trainees and surgeons to safely identify the SAN during neck dissection.
Methods
This study was approved by the University of Cincinnati Medical Center Institutional Review Board (study 2020-0253), and patients were consented. Adults (>18 years of age) who met indications for selective or modified radical neck dissection with sparing of the SAN were identified as candidates for this study by the primary surgeon in the head and neck oncology clinic. Exclusion criteria included prior lateral neck surgery or extensive disease burden resulting in mass effect on the SAN or surrounding structures.
Additionally, cadaveric specimens underwent bilateral modified radical neck dissections with preservation of the SAN in a simulated operative theater.
Surgical Technique
Each subject underwent a modified radical neck dissection (with preservation of the SAN) or selective neck dissection as indicated by the pathology. The SCM muscle and tendon were exposed by unwrapping the investing fascia off the anterior SCM over a broad front, and the SAN was skeletonized. Straight-line distances were measured from (1) the mastoid tip to the distal edge of the SCM tendon, (2) the mastoid tip to the SAN, and (3) the SAN to distal edge of SCM tendon ( Figure 1 ). The perpendicular distance in the anterior-posterior plane was measured from the anterior edge of the SCM to the crossing or entry of the SAN into the muscle ( Figure 2 ). Pictures that were taken for the figures were done with the SAN retracted. We determined all measurements of the SAN nerve distance from the SCM tendon with the nerve in a neutral position. The distance from the SCM tendon in this neutral position to the SAN, when it was found, represents the perpendicular distance that is reported.
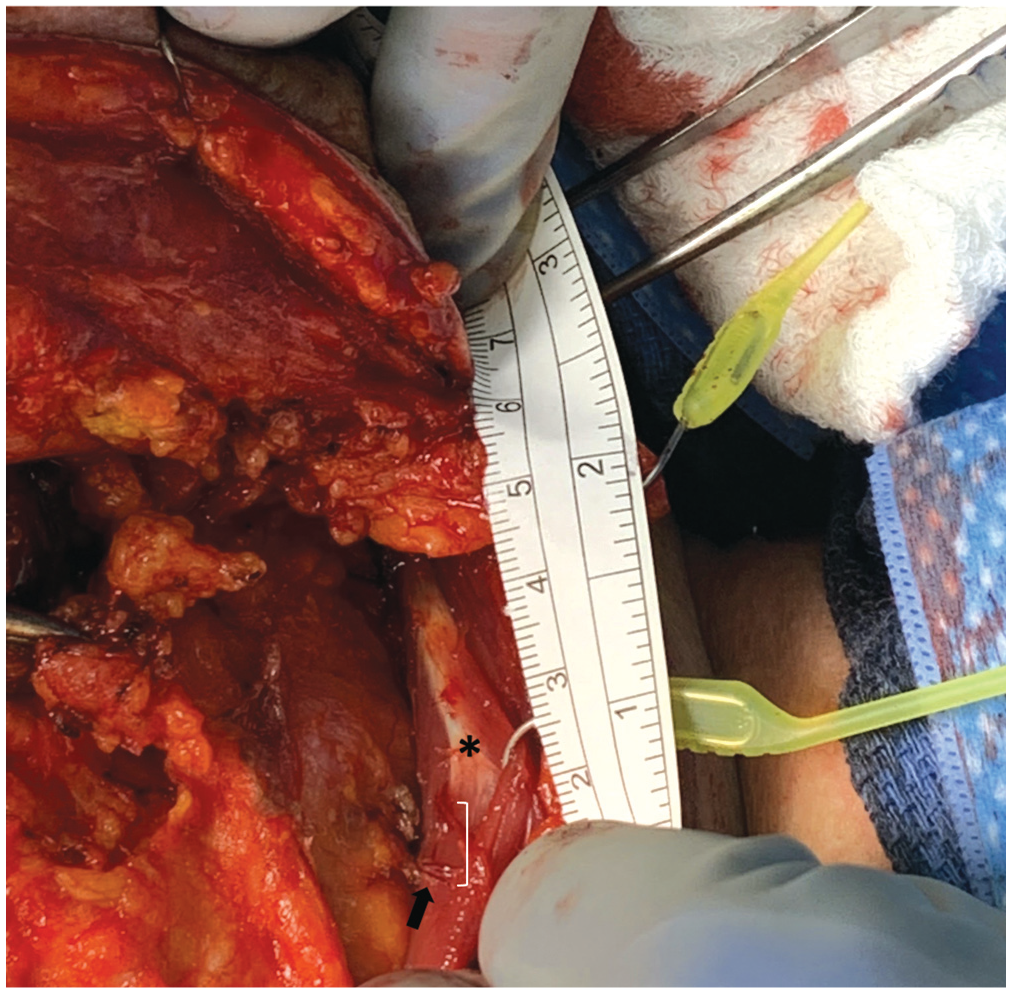
Photograph demonstrating the length of the SCM tendon (asterisk) and the distance from the SAN (arrow) to the distal edge of the SCM tendon as denoted by the bracket. SAN, spinal accessory nerve; SCM, sternocleidomastoid.
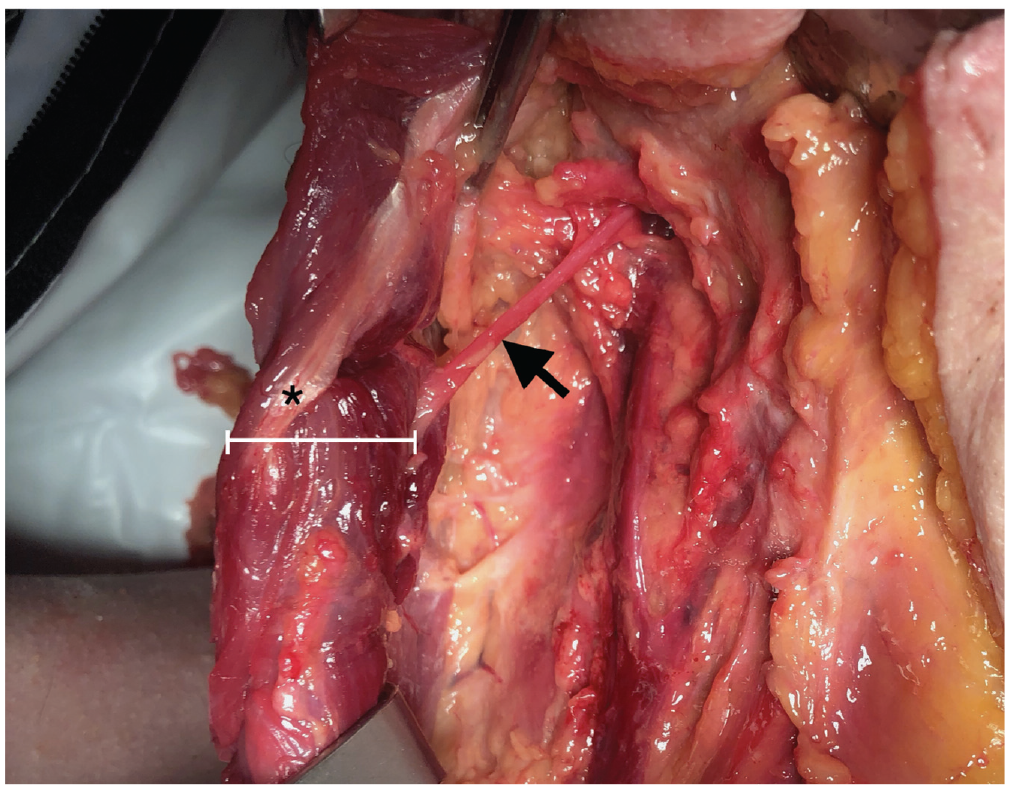
Photograph demonstrating the proximity of the SAN (arrow) to the distal edge of the SCM tendon (asterisk). The error bar denotes the perpendicular distance from the anterior edge of the SCM to the SAN. SAN, spinal accessory nerve; SCM, sternocleidomastoid.
All living patients in this case series had neck dissection preserving the SCM and SAN; measurements were taken at the very beginning of the neck dissection as SCM fascia was being dissected and the SCM tendon was identified.
All values were obtained with a standard ruler and recorded to the nearest millimeter. Laterality was not included in data collection for living subjects, as most did not have bilateral neck dissections; however, laterality was tabulated for cadaveric specimens.
Statistical Analysis
Descriptive statistics were calculated on variables and reported via mean and standard deviation for continuous variables as well as frequency and percentage for categorical variables. The Kolmogorov-Smirnov test was used to test for normal distribution of variables in living subjects. A Mann-Whitney U test was performed to compare the anatomic measurements based on age, sex, and prior radiation therapy (α = 0.05). To compare laterality in the cadaveric specimens, a paired t test was performed to compare right vs left sides (α = 0.05). P values were calculated and reported.
Results
A total of 22 living subjects were included in this study, of which 16 (73%) were male with an average age of 63 years (range, 40-85 years). Five cadaver subjects underwent bilateral neck dissections: 3 female (60%) and 2 male (40%; Table 1 ).
Subject Characteristics.
When living subjects were evaluated by sex, we found significant differences for the length of the SCM tendon (P = .027) and the distance from the mastoid tip to the entrance of the SAN into the SCM (P = .023). We did not find significant differences by age or previous radiation ( Table 2 ). However, the relative position of the nerve to the tendon, from the distal edge of the tendon and measured in perpendicular, was similar between the sexes.
Measurement Comparison by Groups: Sex, Age, and Radiation. a
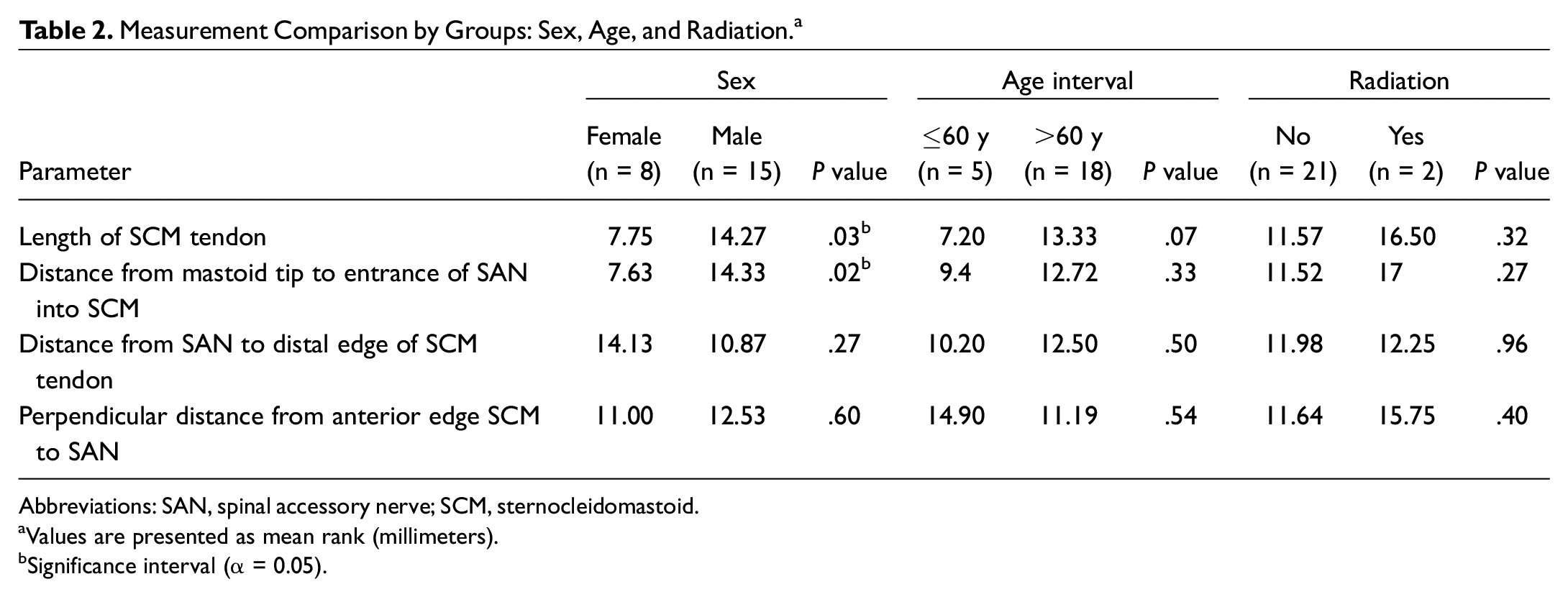
Abbreviations: SAN, spinal accessory nerve; SCM, sternocleidomastoid.
Values are presented as mean rank (millimeters).
Significance interval (α = 0.05).
Of the 22 subjects, 2 had prior radiation to the neck. No statistical difference in measurements was appreciated between radiated and radiation-naïve cases ( Table 2 ). One subject had prior neck surgery; however, this was a thyroidectomy and central neck dissection, and the level 2 neck was not involved. None of the 5 cadaveric specimens had prior radiation to the neck or previous neck surgery.
From the 22 living subjects, 23 measurements were taken as 1 patient underwent bilateral neck dissection. The average length of the SCM tendon was 63.7 mm (SD, 11.8; Table 3 ). The mean distance from the mastoid tip to the entrance of the SAN into the SCM muscle was 51.6 mm (SD, 12.2). The average distance from the SAN to the distal edge of the SCM tendon was 12.1 mm (SD, 4.9). In the anterior-posterior dimension, the mean distance from the anterior border of the SCM to the SAN was 8.9 mm (SD, 3.4). Laterality was not investigated in these subjects since all but 1 patient underwent unilateral neck dissection.
Measured Distances of Living Subjects. a
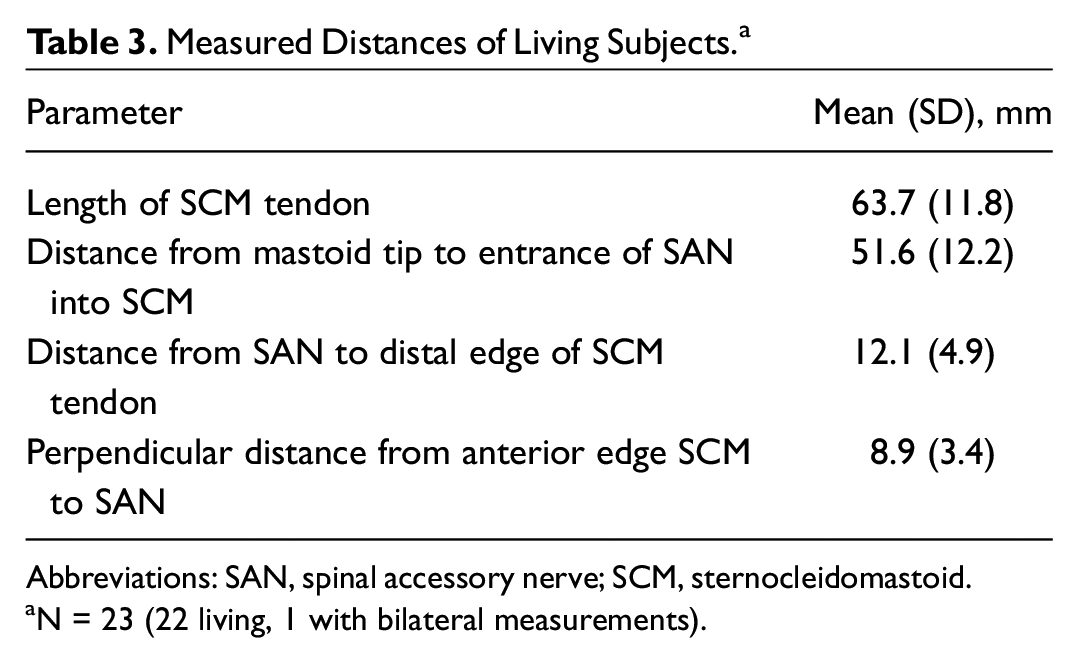
Abbreviations: SAN, spinal accessory nerve; SCM, sternocleidomastoid.
N = 23 (22 living, 1 with bilateral measurements).
From the 5 cadaveric specimens, 10 measurements were taken as each underwent bilateral procedures. The average length of the SCM tendon was 61.5 mm (SD, 10.4; Table 4 ). The mean distance from the mastoid tip to the entrance of the SAN into the SCM muscle was 51.6 mm (SD, 7.1). The average distance from the SAN to the distal edge of the SCM tendon was 9.9 mm (SD, 10.7). In the anterior-posterior dimension, the mean distance from the anterior border of the SCM to the SAN was 16.2 mm (SD, 7.2). There was no statistically significant difference in any of the measurements when compared between sides ( Table 5 ).
Measured Distances of Cadaver Subjects. a
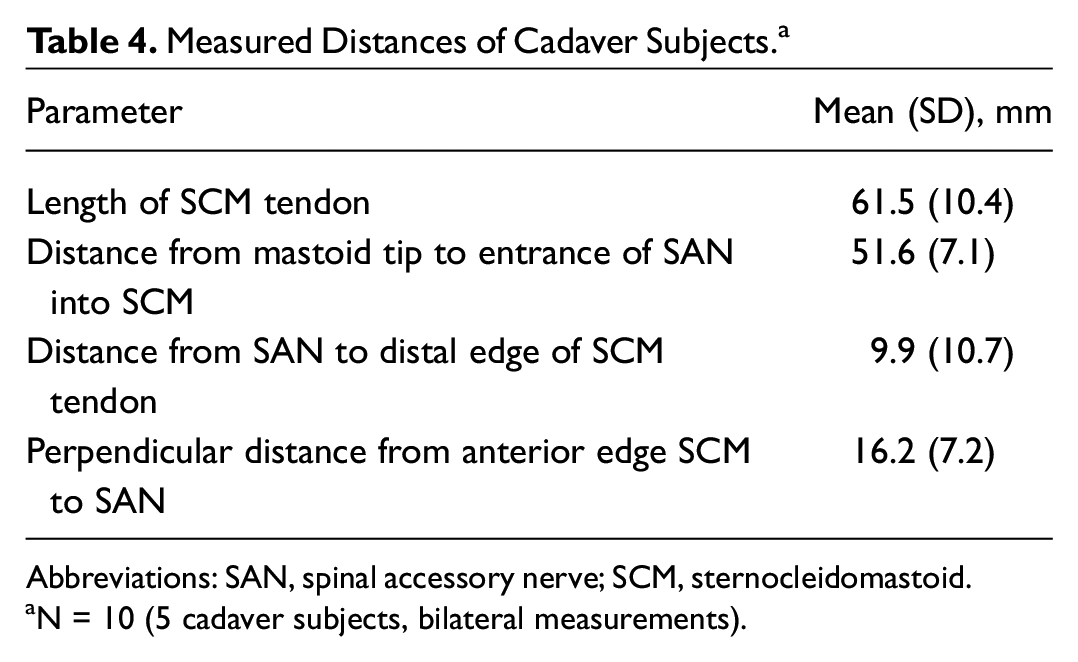
Abbreviations: SAN, spinal accessory nerve; SCM, sternocleidomastoid.
N = 10 (5 cadaver subjects, bilateral measurements).
Subgroup Analysis by Side: Cadaver Subjects.

Abbreviations: SAN, spinal accessory nerve; SCM, sternocleidomastoid.
Discussion
Identification and preservation of the SAN form a key step in modern neck dissection. Preservation of the SAN diminishes the morbidity associated with shoulder syndrome. Various landmarks have been described to help identify the SAN. While the SCM tendon has been described as a reliable landmark in cadaver studies, we present the first in vivo patient anatomic case series demonstrating the utility of the SCM tendon in safely identifying the SAN.
In a recent publication, Eastwood and George suggested the use of a triangle space bordered by the SCM branch of the occipital artery, superior SCM tendon, and digastric muscle to locate the SAN in cadaveric models. 9 The average distance of the SAN from the distal edge of the SCM tendon that they described was 2.31 ± 1.72 mm. The authors cited the use of cadavers as a potential weakness, as tissue preservation techniques could change the size and appearance of the SAN, possibly explaining the discrepancies between their findings and our data. We would argue that since the largest discrepancy in measurements between the studies was less than a centimeter, the tendon is a reliable gross landmark during neck dissection. They reported locating the SAN in this triangle space in 95.8% of cases. However, a previous neck dissection or radiation may render the occipital artery difficult or even impossible to find, which is why we did not include this landmark in our study. In our opinion, the distal tendinous portion of the SCM is reliably identified independent of prior neck oncologic treatment and is sufficient to identify the SAN.
Other existing anatomic landmarks used to identify the SAN are inconsistent and unreliable. Chaukar et al described a small vein in the anterior triangle, near the junction of the upper and middle thirds of the SCM, which drains the SCM and passes lateral to the SAN. The authors stated that the SAN lies approximately 2 mm deep to this vein. 10 However, there has been no documented standardization of this approach in identifying the SAN. 3 Furthermore, the consistency of these landmarks may be severely diminished in the setting of prior oncologic treatment of the head and neck region.
An alternate approach proposed by Deschler and Singer suggests identifying the nerve at the jugular foramen by dividing the attachment of the SCM to the mastoid and then identifying the posterior belly of the digastric muscle superiorly, prior to opening the carotid sheath. They then suggested dissecting the internal jugular vein up to the skull base to reveal the SAN, often on the anterolateral aspect of the vein. 11 The study does not address methods of identifying the SAN if it crosses medial to the internal jugular vein, which may occur in up to 15% to 40% of cases. 3
Stearns and Shaheen described yet another technique to identify the SAN by dividing the SCM into thirds and dissecting the posterior border of the SCM at the junction of the middle and upper thirds. 12 Their technique provides a general indication of the location of the nerve without describing any specific landmark to use for identification of the SAN.
Our results must be taken in the context of the study limitations, namely the small sample size and single-surgeon experience in the included neck dissections. Additionally, the lack of laterality in living patients in this study may not identify differences in SAN positioning between sides within individual patients. Finally, the results of this study may not be applicable to patients with bulky neck disease that might cause displacement of the SAN from mass effect.
Conclusion
Knowledge of the relationship of the SAN to its surrounding structures and reliable landmarks is crucial in preserving shoulder function in patients undergoing neck dissection. The findings of this anatomic study case series corroborate data from prior cadaveric studies and demonstrate that the SCM tendon can be a reliable and safe method of identifying the SAN during neck dissection. This is paramount in the surgical education of trainees in otolaryngology.
