Abstract
Objective
To demonstrate the use of an anterior belly of the digastric muscle flap (ABDMF) during transoral robotic radical tonsillectomy (TORRT) with concomitant neck dissection with the intent of preventing the formation of postoperative pharyngocutaneous fistulas.
Study Design
Retrospective study.
Setting
Single academic tertiary care center.
Methods
In this study, all patients were included who underwent TORRT plus limited pharyngectomy with concomitant neck dissection and ABDMF for the treatment of oropharyngeal squamous cell carcinoma between September 2012 and September 2020. The rate of fistula formation was assessed in patients with preemptive utilization of ABDMF.
Results
A total of 43 patients underwent TORRT with neck dissection and ABDMF. No patients developed a fistula in the postoperative period or associated morbidity with the use of this flap.
Conclusion
Preemptive use of ABDMF in TORRT with concomitant neck dissection represents a reconstructive option that may help prevent the formation of pharyngocutaneous fistula by reinforcing the posteroinferior boundary of the parapharyngeal space.
Since its Food and Drug Administration approval in 2009, transoral robotic surgery (TORS) became a significantly less invasive treatment modality than open surgical approaches for addressing select oropharyngeal malignancies, while having similar oncologic outcomes. Many benefits of TORS have been researched, including improved swallowing outcomes, lower gastrostomy tube dependence rates, and increased quality of life scores, especially when compared with chemoradiation.1-7 Despite its benefits, there are still risks associated with TORS. One such concern in transoral robotic radical tonsillectomy (TORRT) is the formation of a pharyngocutaneous fistula (PCF) when it is performed with simultaneous neck dissection.
Various strategies are available to prevent or repair a PCF, such as staging the neck dissection, primary closure, or utilizing local tissue such as mucosa, fat, or muscle as a flap.8-10 However, these strategies are not without drawbacks that can place the patient at undue risk and increase costs. Nevertheless, PCF formation is a feared complication in most operations involving the pharynx due to the effects on wound healing, need for further interventions, and delays to oral feeding associated with fistula formation. 11
Because most applicable literature is limited by a small sample size and heterogeneous in its description of methods to mitigate fistula formation, the senior author began to employ an anterior belly of the digastric muscle flap (ABDMF) in patients with TORRT following fistula formation in 1 of the first 14 TORRTs performed (7% fistula rate). In this study, we present our experience with preemptive utilization of this reconstructive method to prevent PCF formation in TORRT with simultaneous neck dissection.
Materials and Methods
Patient Selection
A retrospective chart review was performed on all applicable cases between September 2012 and September 2020. These included all patients who underwent TORRT with limited pharyngectomy, including removal of adjacent superior constrictor muscle with concomitant neck dissection, and ABDMF for the treatment of squamous cell carcinoma. All procedures were performed by a single surgeon (E.V.) at the University of Arkansas for Medical Sciences. This research project was approved by the university’s institutional review board (239436). The primary endpoint of the study was to assess the rates of fistula formation. Data collection included patients’ age, sex, race, site and stage of the tumor, timing of neck dissection, PCF formation, adjuvant treatment, feeding modality, dependence on gastrostomy tube, and additional surgical treatment.
Surgical Technique
The standard sequence of surgery began with neck dissection followed by TORS, since the senior author performs prophylactic vessel ligation before the resection of the primary tumor. The extent of neck dissection encompassed levels 2A, 2B, 3, and 4, in all cases. Dissection of level 1 was avoided unless there was clinical or radiologic evidence of nodal involvement in this level. After completion of the neck dissection and prophylactic vessel ligation, the anterior belly of the digastric muscle was released from its mandibular attachment and up to its tendon. It was rotated posteriorly, with the tendon acting as the pivotal attachment, and secured to the submandibular gland superiorly, parotid gland posteriorly, and posterior belly of the digastric muscle inferiorly with 3/0 absorbable interrupted sutures to reinforce the submandibular triangle posteriorly ( Figure 1 ).
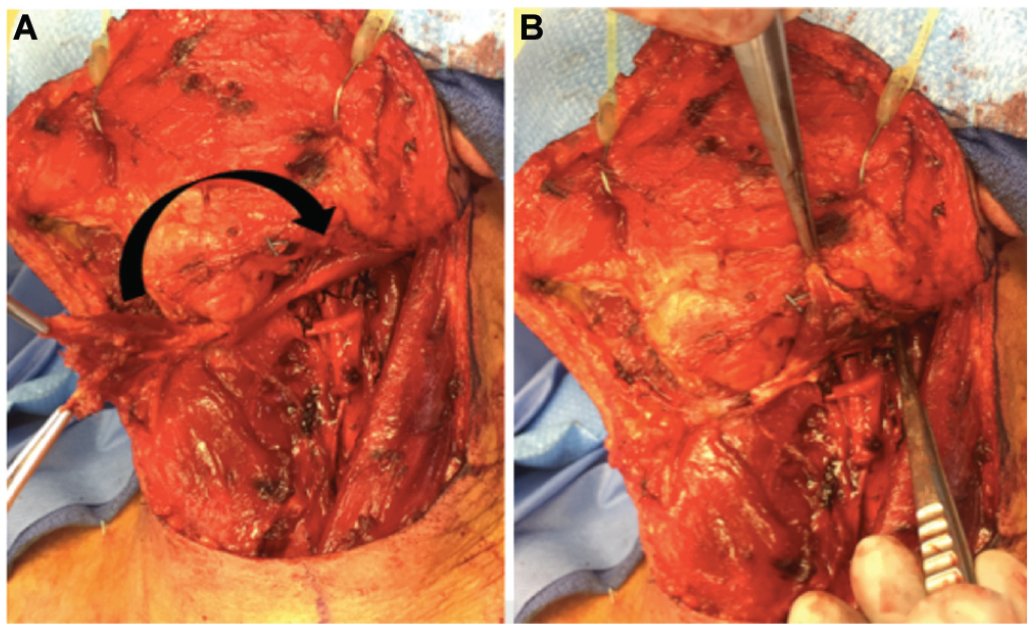
The anterior belly of the digastric muscle is released from the mandible, rotated posteriorly upon its tendon (A), and secured to the submandibular gland superiorly, parotid gland posteriorly, and posterior belly of the digastric muscle inferiorly (B).
Results
Forty-three patients underwent ABDMF during TORRT with limited pharyngectomy with exposure of parapharyngeal fat and a simultaneous neck dissection. Patient demographics, staging information, final margin status, adjuvant treatment information, and timing for oral intake are summarized in Table 1 . None of the 43 patients who underwent ABDMF developed a fistula in the immediate or late postoperative period. There was no associated morbidity with the use of ABDMF noted within the study period. Of these patients, 33 underwent postoperative external beam radiation. Three of these patients had primary chemotherapy and radiation prior to surgical intervention; the rest were postoperatively treated at an appropriate interval after surgery.
Demographic and Surgical Outcome Data for TORRT With Anterior Belly of Digastric Muscle Flap Reconstruction.
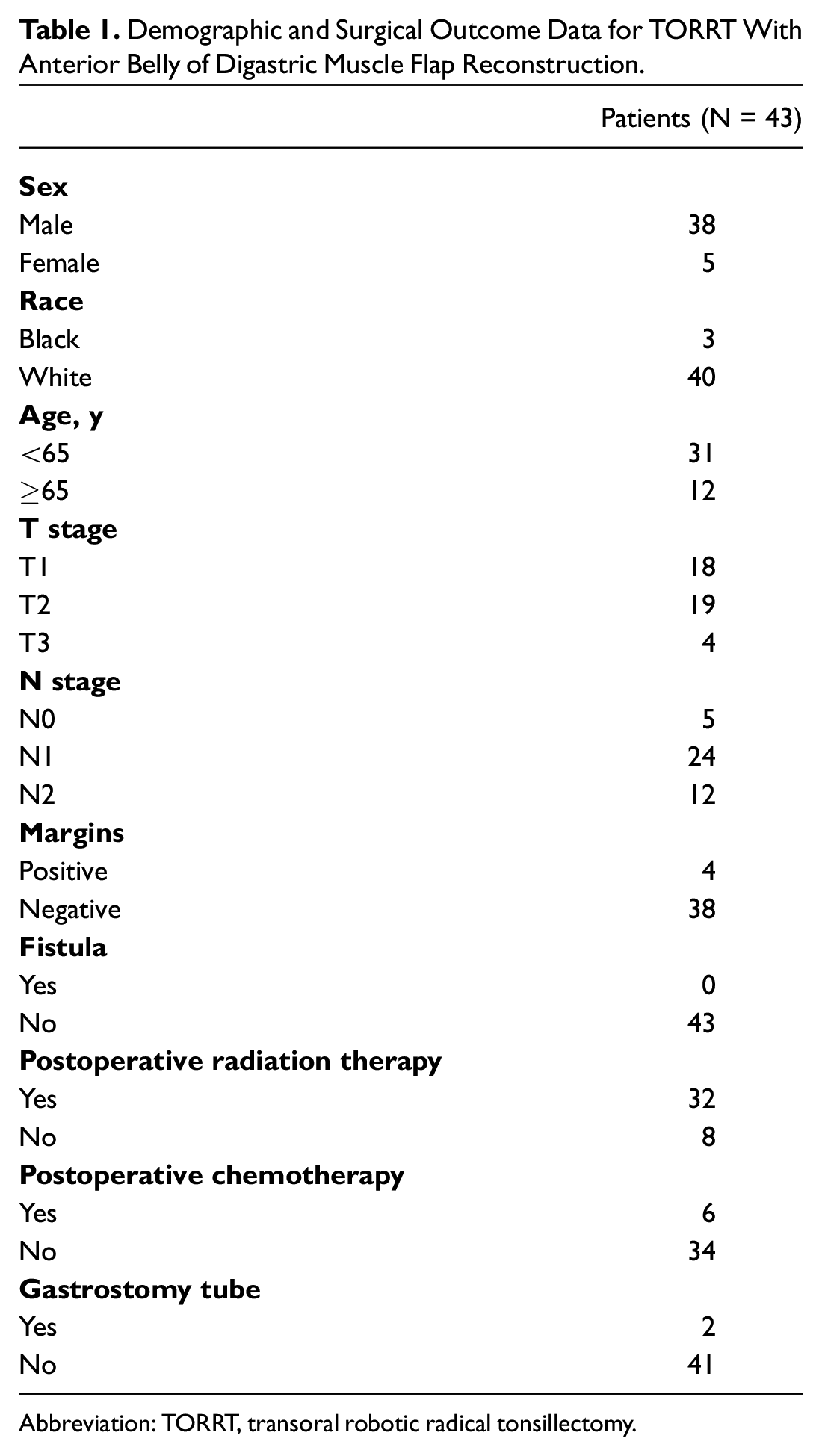
Abbreviation: TORRT, transoral robotic radical tonsillectomy.
Only 2 patients necessitated the use of a gastrostomy tube, while most were able to resume oral intake on the first postoperative day after having the Dobhoff tube removed. However, 1 of these patients who required enteral tube feedings had a gastrostomy tube placed during initial chemotherapy/radiation and underwent salvage surgery with dysphagia postoperatively. This patient eventually returned to oral feeding without any fistula formation. The other patient who required a gastrostomy tube needed prolonged tube feedings and underwent percutaneous gastric tube placement before his postoperative radiation treatment. Two patients were discharged with Dobhoff tube feedings, which were switched to oral intake at their 1-week postoperative visit ( Table 1 ).
Discussion
Although the true incidence of PCF is not known, fistula rates as high as 9.5% have been reported when TORS is performed concomitantly with neck dissection. 12 Moore et al reported a 29% rate of communication between the pharynx and cervical compartment in TORS with simultaneous neck dissections, with only 14% of these connections becoming a true fistula. 13
The most traditional way of PCF prevention in TORS has been staging the neck dissection. 4 However, this approach causes exposure of the patient to additional general anesthesia, increased cost, and delay in adjuvant treatment. 9 Furthermore, it was shown that there was no significant difference in PCF rates when neck dissection was performed simultaneously or staged.4,14
Another preventative measure for PCF is avoiding the dissection of level 1b, since the contents of this level, including the submandibular gland, may provide a natural barrier between the oropharynx and the neck compartment.15,16 Unless there is clinical involvement of this nodal basin, avoiding the dissection of level 1 where TORS is performed in conjunction with simultaneous neck dissection would be feasible, especially in patients who are HPV+.16,17
Intraoperative pharyngocervical communications can be managed in various ways, including primary closure, local muscle flap, fibrin glue, submandibular gland transposition, digastric muscle flap, and sternocleidomastoid (SCM) flap.13,15,18 Using such simple measures is generally sufficient since most communications between pharyngeal and cervical compartments are small. 13 Utilizing local muscle flaps in such instances would provide adequate sealing with minimal morbidity, and ABDMF seems to be a reasonable choice since it provides a readily accessible local tissue source for either closing an existing defect or reinforcing the area at risk for fistula formation, especially when level 1 content is preserved. Additionally, as noted, no morbidity was appreciated within our cohort, making this option deserving of consideration in these patients. A superiorly based SCM flap may be another reasonable option due to its larger surface coverage proportional to its size, especially when level 1 dissection was performed. Obviously, the necessity of more complicated reconstructive measures would contradict the minimally invasive aspect of TORS and may be the result of poor initial patient selection.
TORRT often necessitates a limited pharyngectomy in the form of removal of the adjacent constrictor muscle as the deep margin, which poses a risk of PCF formation due to exposure of parapharyngeal fibrofatty tissues to contaminated oropharyngeal environment and secretions. However, exposure of parapharyngeal fibrofatty tissues may not translate into an actual fistula formation in every case. The rate of secondary healing plays an important role in the prevention of fistula in these cases, where fibrinous exudate and granulation tissue formation act as protective natural barriers to keep pharyngeal and cervical compartments separate. However, any delay in secondary healing in patients undergoing TORRT may pose the risk of fistula formation. Preemptive utilization of local tissues seems to be a reasonable approach to support the pharyngeal wall defect until secondary healing provides a sufficient seal. Prophylactic use of SCM muscle flap to prevent fistula formation in TORS has been reported with success and minimal morbidity. 18 This would be a useful approach especially in cases where level 1 dissection is performed and more voluminous local tissue support is needed. If level 1 dissection is not performed, ABDMF would be an alternative to SCM muscle, possibly with even less morbidity, if any at all.
The digastric muscle has a reliable vascular supply via perforators from the submental branch of the facial artery. 19 When the anterior belly is released from its surroundings as a flap, the perforators supplying the muscle are severed, but the fascial capillaries may still provide adequate blood supply, which may form the basis of its utilization in other purposes, such as rehabilitation of marginal mandibular nerve paralysis.20,21 Furthermore, it has been shown that small-volume muscle fragments used as avascularized graft material are viable after replantation in older studies. 22 However, even if the ABDMF does not survive via its native blood supply or imbibition, it may still serve as a biological dressing to the area to be reinforced and may help to prevent fistula formation by promoting scar formation.
Formation of a PCF in TORS may be multifactorial: factors that include performing simultaneous neck dissection, the extent of oropharyngeal resection, the inclusion of level 1 in neck dissection, the amount of native tissue left between oropharynx and neck compartments, and the healing capacity of the patient via secondary intention. The technique described in this article aims to reinforce the posteroinferior boundary of the parapharyngeal space at the conclusion of the neck dissection to prevent a potential communication after resection of the superior constrictor in cases where the anteroinferior boundary was left intact by avoiding level 1 dissection. Utilization of ABDMF without any fistula formation in this series, despite the initiation of oral feeding on the first postoperative day in the majority of patients, indicates the benefit of this technique. Naturally, this study is limited by its small sample size and the inherent inability to draw statistically or clinically significant conclusions in comparison with patients who did not have the described technique utilized in their surgery (1 of 14). However, the lack of fistula formation in this cohort after implementation of this technique, as compared with an approximate 7% rate of fistula in patients without an ABDMF, has encouraged the senior author to continue to use this technique for all patients with TORRT. Further limitations exist in the possible selection bias of the patient population studied, which is formed by patients with mostly T1 and T2 primaries. However, it should also be noted that more advanced primary tumors may not be the best candidates for TORRT. Additional limitations exist in the retrospective nature of this study to fully categorize all patient variables due to what is available in the electronic medical record. Although descriptive, our study is the first case series of the preemptive use of ABDMF as an effort to prevent or decrease the rate of PCF.
Conclusion
Preemptive use of ABDMF may help to prevent the formation of PCF by reinforcing the posteroinferior boundary of the parapharyngeal space in patients undergoing TORRT with simultaneous neck dissection preserving level 1, without any significant morbidity. SCMMF seems to provide a good alternative in cases where level 1 is included in neck dissection due to its larger volume, which allows it to cover the entirety of level 1.
Author Contributions
Disclosures
Footnotes
This abstract was presented at the AAO-HNSF 2020 Virtual Annual Meeting & OTO Experience; September 13-16, 2020.
