Abstract
Objective
To investigate whether lymph node ratio (LNR) and log odds ratio (LODDS) have prognostic significance for overall survival (OS) and disease-free survival (DFS) in patients with laryngeal squamous cell cancer (LSCC) treated with curative intent.
Study Design
Case-control study.
Setting
University hospital.
Subjects and Methods
Records of 229 patients with LSCC who underwent surgery with a curative intent with or without adjuvant treatment from 2000 to 2014 were reviewed. The clinicopathological parameters LNR and LODDS were analyzed; univariate and multivariate analysis was performed to evaluate the prognosis of each for OS and DFS.
Results
The 5-year OS was 81.7% for LNR ≤0.233 and 47.1% for LNR >0.233, and the 5-year OS was 79.6% for LODDS ≤–0.1 and 51.8% for LODDS >–0.1, respectively. In the univariate analysis, the independent variables were subsites, pT stage (pT1 and pT2 vs pT3 and pT4), pN, pTNM, alcohol consumption, and LNR and LODDS (P < .05). By multivariate analysis, we determined that subsites, pT stage, alcohol consumption, LNR, and LODDS were independent prognostic predictors of survival (P < .05). Univariate and multivariate models identified that both LNR and LODDS were significant prognostic factors for survival. However, the hazard ratio (HR) for LNR >0.233 vs ≤0.233 was 8.95 (95% confidence interval [CI], 3.18-25.16; P < .001) in OS, and the HR was 11.37 (95% CI, 4.02-32.15; P < .001) in DFS. The risk of LNR was noticeably greater than other factors.
Conclusions
LNR and LODDS were both prognostic factors for OS and DFS. However, LNR was confirmed as a more reliable indicator for evaluating the prognosis, and it can be used to increase the prognostic value of the traditional TNM classification of LSCC.
The estimated incidence and mortality of laryngeal cancer was 26,400 and 14,500 cases, respectively, in China in 2015. 1 More than half of patients presented with advanced disease, with an overall 5-year survival rate of approximately 50%. 2 Despite improvements in the evaluations and management of laryngeal squamous cell cancer (LSCC), the 5-year relative survival rate has not significantly improved in the past 30 years. 3 Tumor staging systems have been developed to assist in estimating prognosis after surgery and to compare results across institutions. 4 Many questions have been raised regarding which clinical feature is the most important for the TNM (tumor-node-metastasis) staging systems’ ability to estimate prognostic outcomes for LSCC. It would be useful to identify other parameters that could provide more useful information regarding prognosis after treatment.
The prognosis of patients with LSCC has been determined primarily by the stage at presentation, with the single most important factor being the presence of neck node metastases, which reduces long-term survival by 50%. 5 The status of regional lymph node metastasis has been widely considered an important prognostic factor to plan subsequent postoperative treatment. However, the current approach does not take into consideration the number of positive lymph nodes as accurately as the lymph node ratio (LNR), which may be a limiting factor in the precision of the prognostic estimates. Recently, several studies have shown that LNR and log odds ratio (LODDS) are useful predictors of prognosis in various cancers, such as head and neck squamous cell cancer, 6 hypopharyngeal cancer,7,8 oral cancer, 9 lung adenocarcinoma, 10 breast cancer, 11 and gastric cancer. 12 In addition, compared with numerous other prognostic factors, the advantages of LNR and LODDS are that they are simple, widely available, and inexpensive parameters derived from the postoperative evaluation of the pathological specimen. However, to our knowledge, only a few studies have examined the prognostic value of LNR in patients with LSCC,13,14 and the prognostic value of LODDS has yet to be determined in these patients. The aim of this study was to evaluate the relationship of LNR and LODDS to the prognosis of patients with LSCC who underwent surgery with a curative intent.
Patients and Methods
This was a retrospective study including 229 patients with LSCC who underwent curative surgery for biopsy-proven laryngeal squamous cell cancer between January 2000 and December 2014. Patients with synchronous or metachronous malignancies, recurrent tumors, unresectable disease/macroscopic incomplete resection, follow-up period <3 years, pN3 disease, the number of dissected lymph nodes <6, and distant metastases were excluded from the study. In patients with a pN3 neck stage, the lymph node metastases usually present as a conglomerate of lymph nodes so that calculating an LNR is not reliable. Moreover, due to the need to ensure a comprehensive neck dissection and complete pathological examination of the specimen, patients with a limited selective neck dissection with <6 lymph nodes were retrospectively excluded. 15 The patients were classified by pathological stage (pTNM) according to the seventh edition of the Union for International Cancer Control (UICC) classification, published in 2010. 16 Primary surgery was carried out via either partial or total laryngectomy, and this was most often supplemented with a standardized unilateral or bilateral-selective neck dissection of levels II to V. The indication for a unilateral or bilateral neck dissection was dependent on the tumor location and the TNM staging. Each specimen was evaluated by 2 independent pathologists without knowledge of the clinical features of the cases. In locally advanced tumors, postoperative irradiation was administered and combined with chemotherapy, if indicated, in the presence of 2 or more lymph node metastases, capsular perforation, or positive resection margins. 17 All cases were discussed in a multidisciplinary head and neck–specific tumor board to guide surgical and adjuvant therapy recommendations. The study was approved by the Review Board of Sun Yat-sen University Cancer Center, China.
Definition of LNR and LODDS
LNR represented the probability of positive lymph nodes in the retrieved lymph nodes and was calculated as the positive lymph nodes divided by the total number of retrieved lymph nodes. 18 LODDS was calculated as the natural logarithm of the quotient between number of positive lymph nodes and the number of negative lymph nodes. 19 To avoid rare numbers, 0.5 was added when necessary to both the positive lymph node and negative lymph node numbers. The formula was log [(PNOD + 0.5)/ (TNOD – PNOD + 0.5)], where PNOD was the number of positive lymph nodes and TNOD was the total number of lymph nodes retrieved.
Statistical Analysis
Recurrence was defined as “local” for occurrences at primary sites, “regional” for neck lymph node involvement, and “metastasis” for occurrences in other organs. Overall survival (OS) was measured from the date of initial surgery to the death of the patient or the end of follow-up, whichever occurred first; disease-free survival (DFS) was measured from the date of initial surgery to the time of recurrence or the end of follow-up, whichever occurred first. The Kaplan-Meier estimates were evaluated using the log-rank test, and a P value of <.05 was considered significant. The cut-point for optimal separation of LNR and LODDS was obtained using the method described by Yildiz et al 20 with the log-rank test applied to measure separation between the groups produced at the cut-point. The cut-point that minimized the P value obtained from the log-rank test was used as the best separation of LNR and LODDS into high- and low-risk categories. The analyses were performed using the software package SPSS Statistics 20 (SPSS, Inc, an IBM Company, Chicago, Illinois).
Results
Patients and Tumor Characteristics
A total of 229 patients were treated with primary surgery. The clinicopathological characteristics are outlined in Table 1 . The median age was 60 years (range, 35-85 years), and the median follow-up period was 62.4 months (range, 4.0-190.5 months). The site of the primary tumor was divided into glottic cancer (n = 119, 51.97%), supraglottic cancer (n = 95, 41.48%), and subglottic cancer (n = 15, 6.55%). The number of pN-positive patients was 145 (63.31%), and 84 patients (36.69%) were pN-negative. Twenty-one (9.17%) patients were at pathological stage II, 67(29.26%) patients were at pathological stage III, and 141 (61.57%) patients were at pathological stage IV. The number of patients with smoking history was 164 (71.62%), and 84 (38.71%) patients consumed alcohol (a patient had 1 alcoholic beverage in his or her lifetime). In addition, 2 patients with positive margins were treated with postoperative chemoradiotherapy.
Clinicopathological Characteristics of Patients with LSCC (n = 229). a
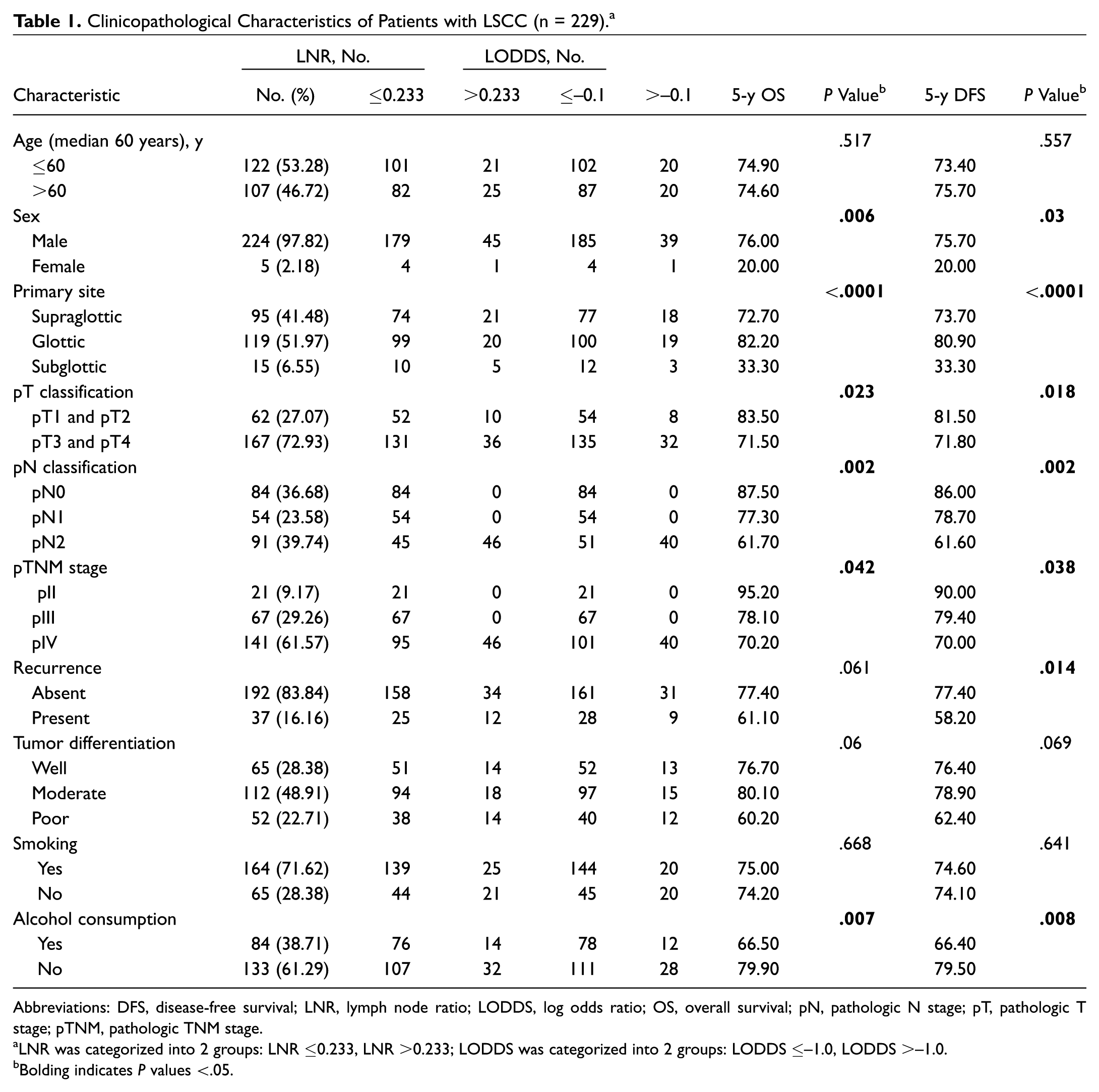
Abbreviations: DFS, disease-free survival; LNR, lymph node ratio; LODDS, log odds ratio; OS, overall survival; pN, pathologic N stage; pT, pathologic T stage; pTNM, pathologic TNM stage.
LNR was categorized into 2 groups: LNR ≤0.233, LNR >0.233; LODDS was categorized into 2 groups: LODDS ≤–1.0, LODDS >–1.0.
Bolding indicates P values <.05.
Interventions
Primary surgery as a single treatment modality was performed in 143 patients (62.4%), surgery followed by adjuvant radiotherapy was performed in 39 (17.0%) patients, and surgery with adjuvant chemoradiotherapy was performed in 47 (20.5%) patients. Bilateral neck dissection was performed in 75 patients (32.72%), ipsilateral neck dissection in 152 patients (66.38%), and contralateral neck dissection 2 patients (0.9%).
LNR and LODDS
Evaluation of the histopathological reports showed that a median of 16 lymph nodes (mean, 18.51; range, 6-69) was dissected, with a median of 1 metastasis (mean, 2.13; range, 0-20) being detected positive. LNRs (range, 0-1.0) were divided into 2 groups by the cutoff value: LNR ≤0.233 and LNR >0.233; LODDSs (range, –4.62 to 2.71) were divided into 2 groups by the cutoff value: LODDS ≤–1.00 and LODDS >–1.00. The treatment regimens between LNR and LODDS groups for patients are shown in Table 2 .
Treatment Regimens for Different LNR and LODDS Groups. a

Abbreviations: LNR, lymph node ratio; LODDS, log odds ratio.
LNR was categorized into 2 groups: LNR ≤0.233, LNR >0.233; LODDS was categorized into 2 groups: LODDS ≤–1.0, LODDS >–1.0.
Lymph Node Metastasis Classification and Prediction of OS and DFS
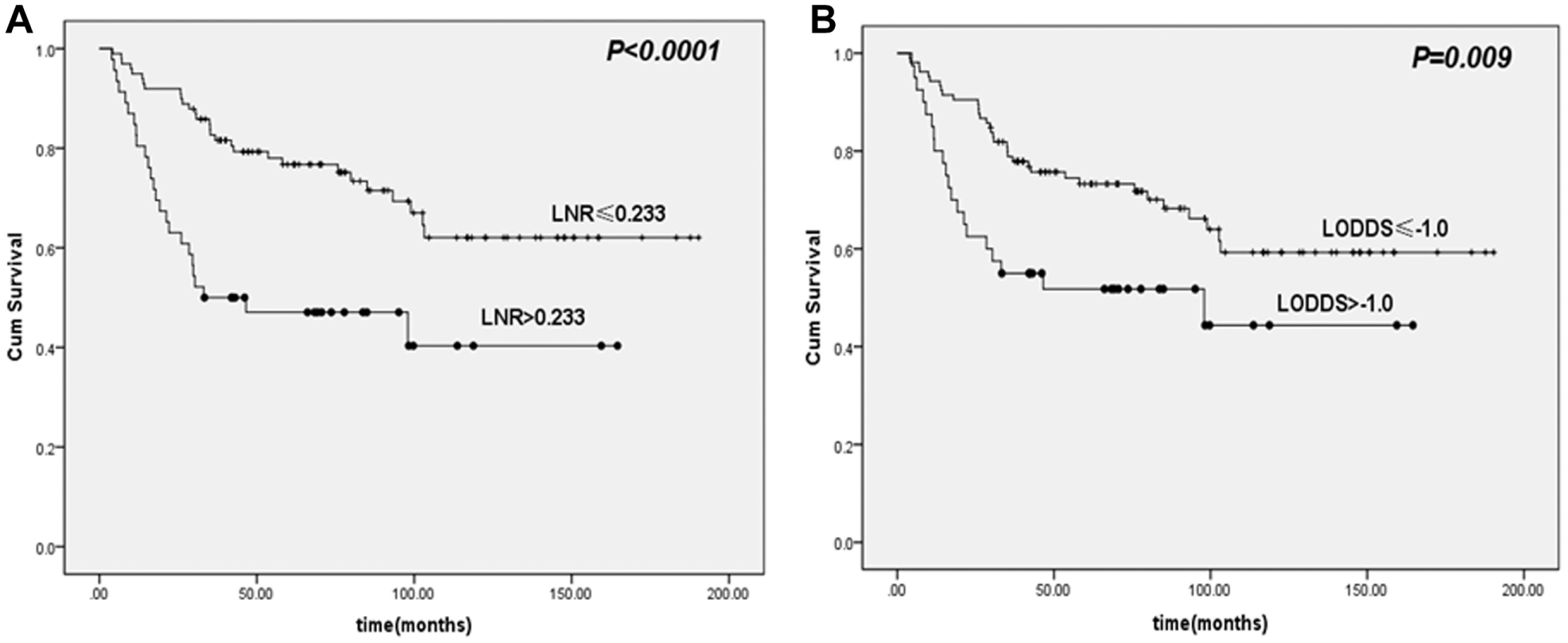
The 5-year OS for the entire study sample was 74.8% (range, 3.91-190.4 months; median ± SD, 69.6 ± 2.79 months). The 5-year OS was 81.7% for LNR ≤0.233 and 47.1% for LNR >0.233 of lymph node ratio, respectively (P < .0001); the 5-year DFS was 81.4% for LNR ≤0.233 and 46.8% for LNR >0.233 of lymph node ratio, respectively (P < .0001). The 5-year OS was 79.6% for LODDS ≤–0.1 and 51.8% for LODDS >–0.1 of log odds ratio, respectively (P < .0001); the 5-year DFS was 79.3% for LODDS ≤–0.1 and 51.6% for LODDS >–0.1 of log odds ratio, respectively (P < .0001). The results of the univariate Cox regression model for clinicopathologic parameters are shown in Table 1 . In the univariate analysis, the independent variables were sex, primary subsites, pT stage (pT1 and pT2 vs pT3 and pT4), pN stage, pTNM stage, alcohol consumption, LNR, and LODDS. Those variables with P < .05 in the univariate regression model were included in the multivariate regression model in Table 3 . In the multivariate analysis, we observed that sex, primary sites, alcohol consumption, LNR, and LODDS were independent predictors of 5-year OS (all P < .05; Figure 1 ); however, pT classification (pT1 and pT2 vs pT3 and pT4) as an independent predictor did not reach statistical significance (P = .05). The 5-year DFS was 74.5% (range, 3.91-190.4 months; median ± SD, 67.62 ± 2.82 months) in our study. In univariate analysis of DFS, the independent variables were sex, primary subsites, pT stage (pT1 and pT2 vs pT3 and pT4), pN stage, pTNM stage, recurrence, alcohol consumption, and LNR and LODDS. In multivariate analysis, we determined that sex, primary subset site, pT classification (pT1 and pT2 vs pT3 and pT4), alcohol consumption, LNR, and LODDS were independent predictors of 5-year DFS (all P < .05; Figure 2 and Table 4 ).
Univariable and Multivariable Cox Model of Prognostic Factors for Overall Survival. a
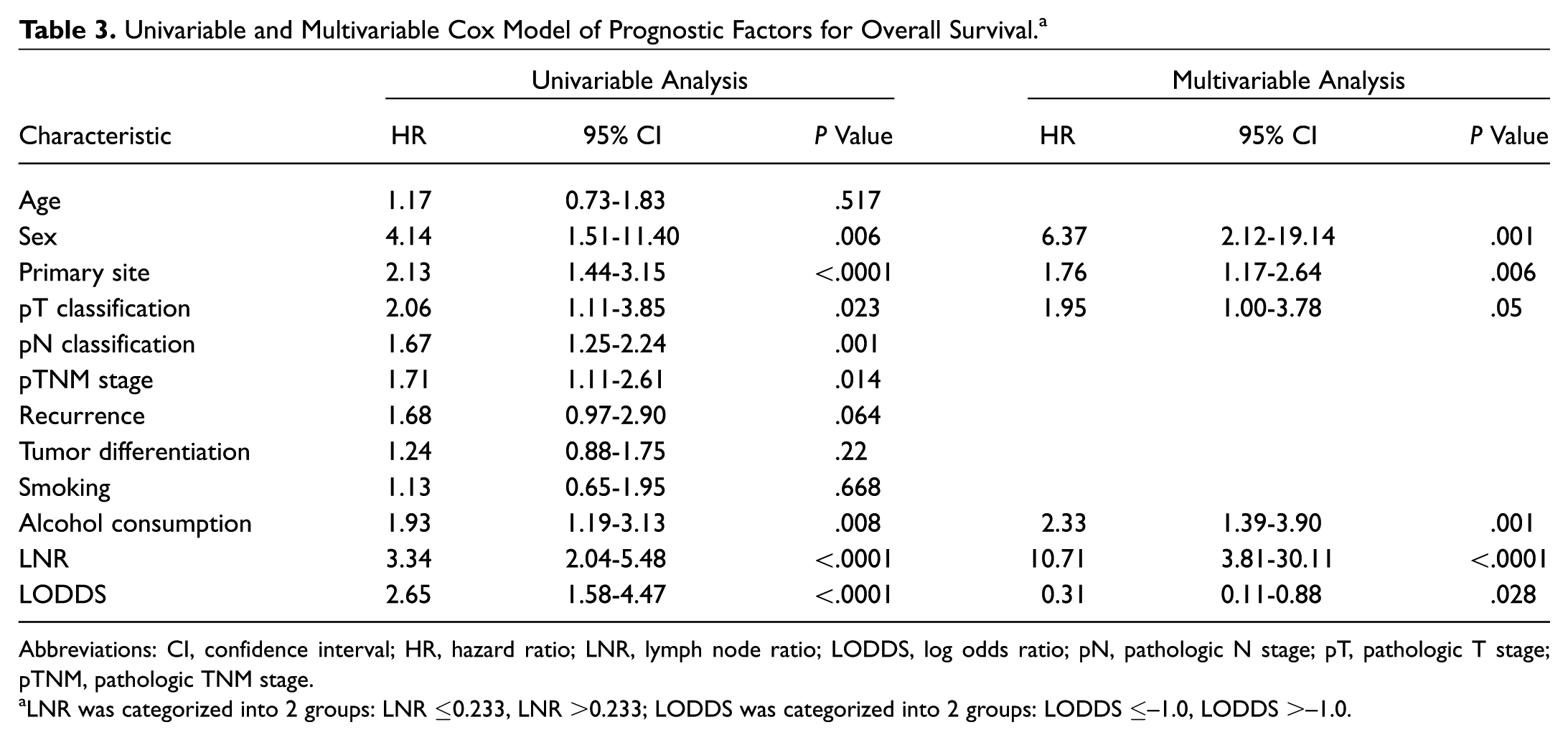
Abbreviations: CI, confidence interval; HR, hazard ratio; LNR, lymph node ratio; LODDS, log odds ratio; pN, pathologic N stage; pT, pathologic T stage; pTNM, pathologic TNM stage.
LNR was categorized into 2 groups: LNR ≤0.233, LNR >0.233; LODDS was categorized into 2 groups: LODDS ≤–1.0, LODDS >–1.0.
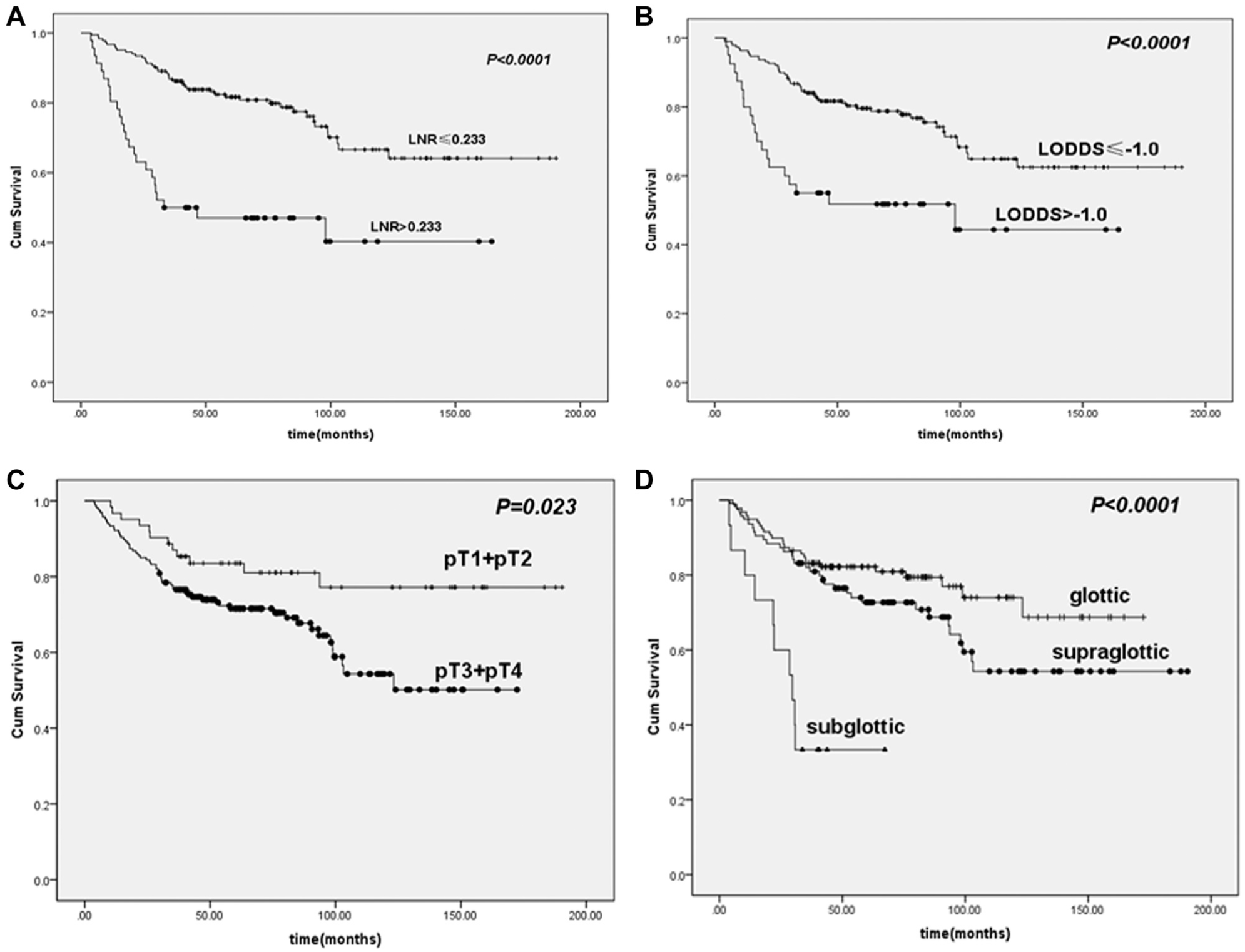
Overall survival (OS) curves according to lymph node ratio (LNR), log odds of positive lymph nodes (LODDS), pT classification (pT1 and pT2 vs pT3 and pT4), and primary subsites. (A) OS curves stratified based on different LNR groups. (B) OS curves stratified based on different LODDS groups. (C) OS curves stratified based on different pT classification (pT1 and pT2 vs pT3 and pT4). (D) OS curves stratified based on different subsites.
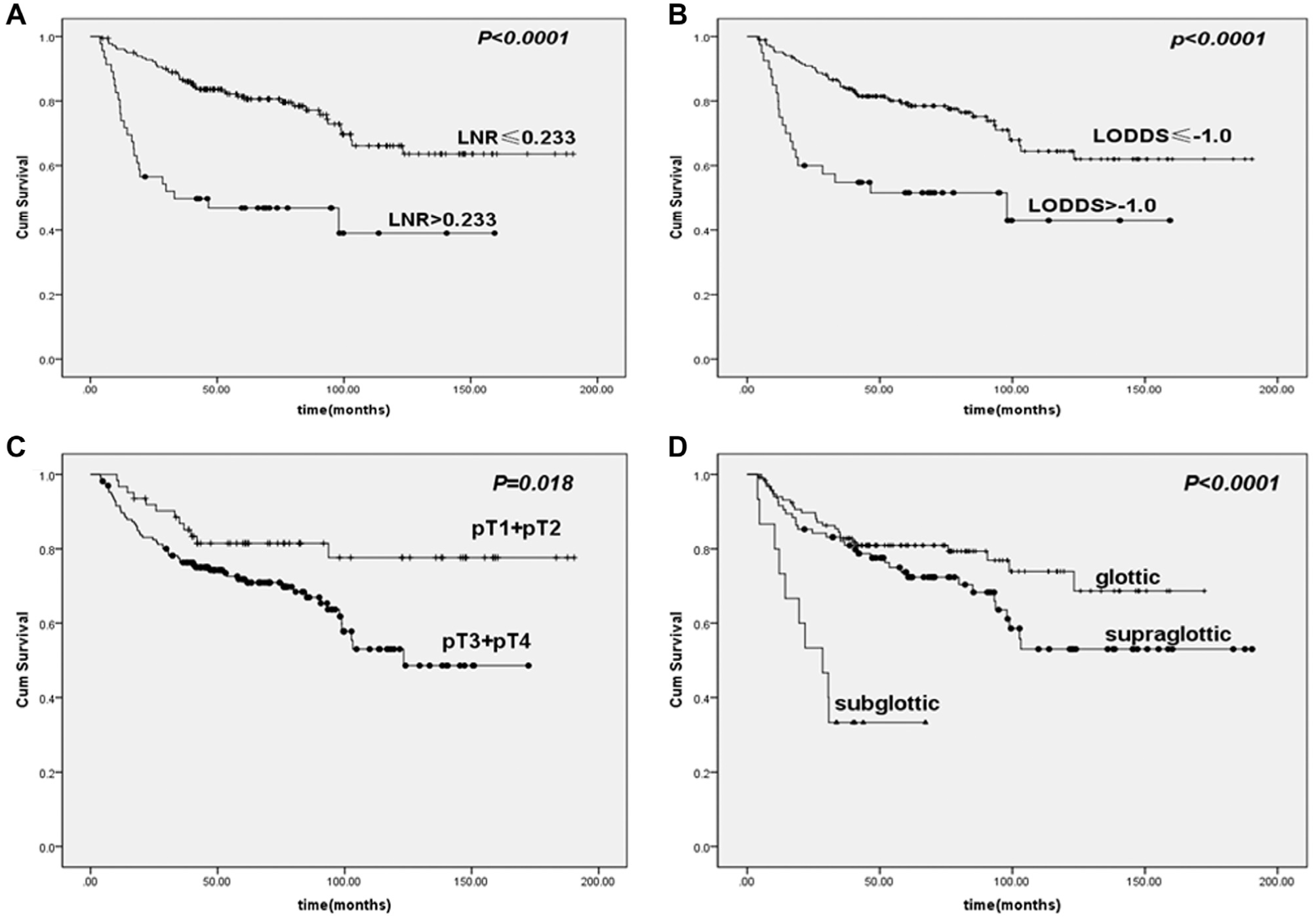
Disease-free survival (DFS) curves according to lymph node ratio (LNR), log odds of positive lymph nodes (LODDS), pT classification (pT1 and pT2 vs pT3 and pT4), and primary subsites. (A) DFS curves stratified based on different LNR groups. (B) DFS curves stratified based on different LODDS groups. (C) DFS curves stratified based on different pT classification (pT1 and pT2 vs pT3 and pT4). (D) DFS curves stratified based on different subsites.
Univariable and Multivariable Cox Model of Prognostic Factors for Disease-Free Survival. a
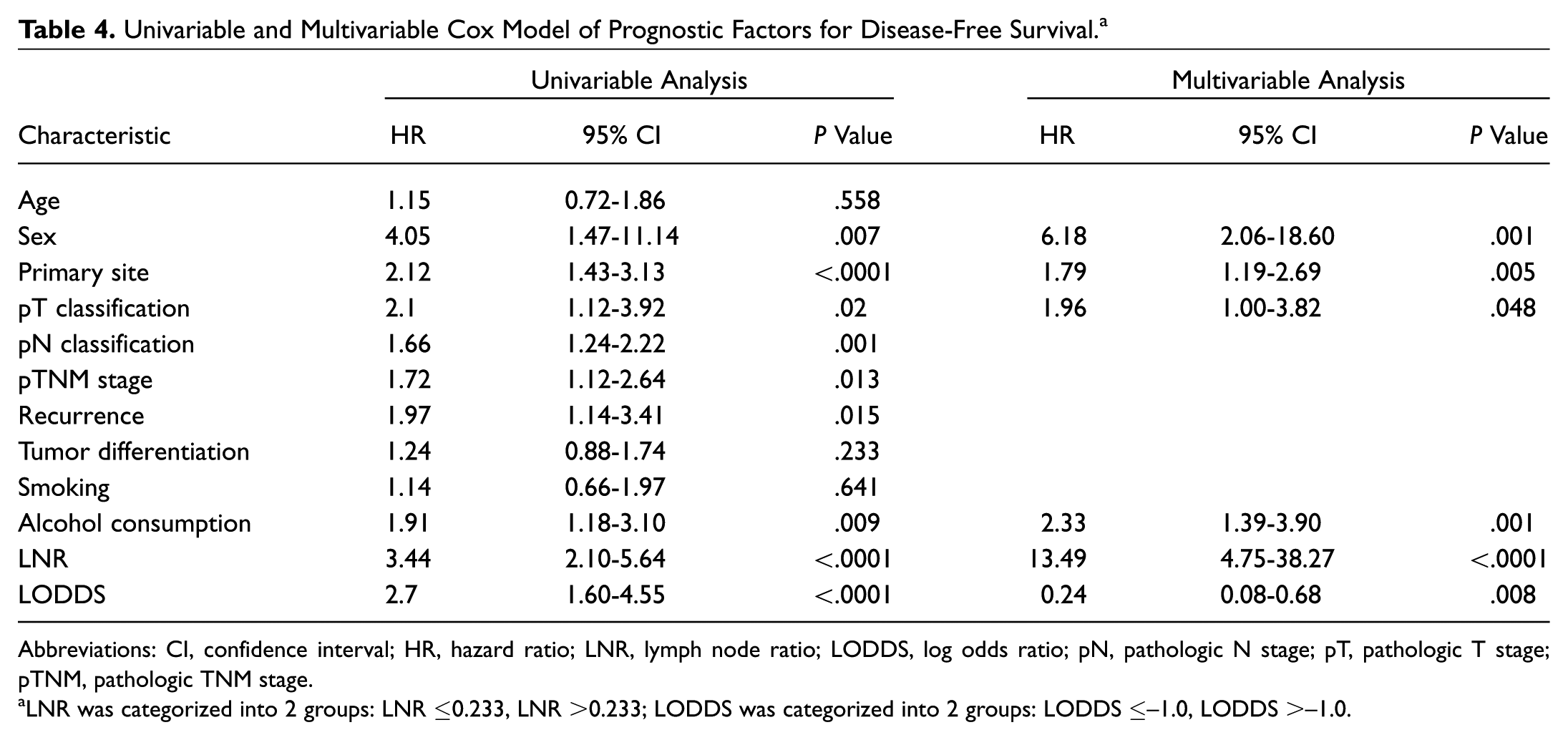
Abbreviations: CI, confidence interval; HR, hazard ratio; LNR, lymph node ratio; LODDS, log odds ratio; pN, pathologic N stage; pT, pathologic T stage; pTNM, pathologic TNM stage.
LNR was categorized into 2 groups: LNR ≤0.233, LNR >0.233; LODDS was categorized into 2 groups: LODDS ≤–1.0, LODDS >–1.0.
The hazard ratio (HR) for patients with LNR >0.233 vs ≤0.233 was 10.71 (95% confidence interval [CI], 3.81-30.11; P < .001) for OS and 13.49 (95% CI, 4.75-38.27; P < .001) for DFS. The HR of LNR was noticeably greater than other factors (see Table 3 and Table 4 for details of the multivariable analysis). LNR and LODDS remained significant factors for OS (P < .001) and DFS (P = .007) in the positive lymph node (pN+) patients ( Figure 3 ).
Overall survival (OS) curves according to lymph node ratio (LNR) and log odds of positive lymph nodes (LODDS) for positive node metastasis (pN+) patients. (A) OS curves stratified based on different LNR groups. (B) OS curves stratified based on different LODDS groups.
Patterns of Failure
During the follow-up period, 37 of 229 patients (16.12%) experienced disease relapse, including 10 patients (4.37%) with local LSCC recurrence, 11 patients (4.8%) with regional recurrence, 4 patients (5.24%) with locoregional recurrence, and 12 patients (5.24%) with distant metastasis.
Discussion
Despite the improvements in comprehensive treatment strategies, the survival outcomes for LSCC are still unsatisfactory. The prognosis for patients with laryngeal cancer is currently estimated based on the TNM system, which assigns nodal metastasis based on the involved lymph node station. These staging systems have been created to assist with prognosis, treatment selection, and research efforts, including comparisons of outcomes and efforts to drive clinical trial methodology 21 ; however, they are limited by their inability to predict success on an individual basis. Besides, the current TNM staging system is based on the number, size, and laterality of positive cervical lymph nodes in LSCC to identify patients at high-risk recurrence, and poor prognosis might not be enough. 22 For this reason, LNR has been postulated to be a predictor of patient outcome: the numerator (the number of metastatic nodes) of LNR reflects tumor burden, and the denominator (the total number of lymph nodes resected) of LNR represents a marker for adequate dissection. 23
An initial report on the prognostic value of LNR after primary surgical treatment for advanced laryngeal cancer was published in the literature. 24 Increasing evidence is establishing the prognostic role of LNR in LSCC 13 and hypopharyngeal carcinoma.7,25,26 Our results support these previous findings, and those patients with an LNR ratio of 0.233 or higher had significantly worse OS and DFS. Even after adjustment by multivariate analysis, the risk for mortality was noticeably greater for patients with a higher LNR than those with a lower LNR among these patients with LSCC. The consistency of the results obtained from different populations reinforced the prognostic value of LNR. Subsequently, we categorized the patients with pN+ stage according to their LNRs, assigning them to low- and high-risk LNR groups, respectively. As expected, there were significant differences among the Kaplan-Meier survival estimates for the 2 LNR groups ( Figure 3 ). Other studies have also found that within subgroups of patients with pathological N1 or N2 disease, LNR was able to identify low-risk and high-risk patients with oral cavity cancer.27,28 Pathological nodal stage based on the TNM staging system, although a predictor for OS and DFS on univariate analysis in our study, did not remain significant in multivariate analysis. Similar findings14,25 have indicated that LNR could predict the outcomes better than counting the positive lymph nodes, suggesting that LNR may have greater prognostic significance than the traditional TNM nodal staging.
Künzel et al 14 reported that LNR is a mathematical figure that is influenced by the extent of neck dissection and the number of lymph nodes in the pathological specimen. As such, LNR may be of limited value in the decision-making process in the treatment of patients with LSCC when a small number of lymph nodes are removed and examined. As a matter of fact, the reliable and standardized neck dissection procedure (levels II-V) is usually used in patients with advanced laryngeal cancer, 24 and selective neck dissection has been employed successfully in selected cases, particularly for N1 or occasionally N2 nodal involvement. 14 In contrast, patients who have negative lymph node (N0) status may benefit from elective neck dissection for staging and treatment purposes, and the risk of occult metastasis without selective neck dissection is greater than 20%. 29
The LNR reflects the degree of lymph node metastasis and the extent of the neck dissection, and it was found to improve prognostic information in head and neck squamous cell cancer, hypopharyngeal cancer, oral cancer, and others. According to the UICC/American Joint Committee on Cancer recommendations, 15 only when the number of retrieved and examined lymph nodes is ≥6 (nose/paranasal sinus ≥10) should it be regarded as sufficient for classification. For these reasons, we included only patients who had ≥6 retrieved and examined lymph nodes and found that an LNR >0.233 predicted a poorer OS and DFS.
Based on our data, the results indicated that the high LNR (P = .041) was close to recurrence; in particular, the results showed that the patients with a high LNR had a 2-fold higher risk (26.1%, 12/46) of recurrence than patients with a low LNR (13.7%, 25/183). Prabhu et al 30 suggested that patients with an LNR >20% should be defined as high risk for locoregional recurrence and death and should be considered for adjuvant chemoradiotherapy as a method to improve locoregional control. Currently, patients with pathologic factors that include advanced tumor (T) classification, advanced nodal classification, perineural invasion, lymphovascular space invasion, close/involved margins, and nodal extracapsular extension 31 are considered candidates for postoperative adjuvant (chemo)radiotherapy to increase disease control.
Most recently, another new classification system has been proposed—the log odds of positive lymph nodes (LODDS)—which is calculated by the log of the ratio between the positive and negative lymph nodes.18,32 LODDS was analyzed in different solid carcinoma types, such as colorectal cancer 18 and perihilar cholangiocarcinoma. 32 To our knowledge, until now, there has been no such analysis on laryngeal cancer. After adjustment by multivariate analysis, LODDS remained an independent risk factor for OS and DFS in our study, but the HR of LNR (OS, 10.71; DFS, 13.49) was obviously greater than LODDS (OS, 0.287; DFS, 0.235), and LODDS (P = .230) had no relationship to local recurrence. Our results were similar to those of other authors 26 in the evaluation of the relationship between LNR and patients’ recurrence. Therefore, patients at high risk of locoregional recurrence from either LNRs or the classic high-risk factors might benefit from intensified adjuvant therapy.
In addition, multivariate analysis demonstrated that primary sites, pT classification (pT1 and pT2 vs pT3 and pT4), and alcohol consumption were independent prognostic factors for survival outcomes. These results were similar to those of other authors 33 regarding the main prognosticators in LSCC.
There were several limitations to this study. This was a single-center retrospective study based on 229 eligible patients with LSCC. Most patients were male. Further research validating the results in large databases of patients with LSCC and prospective trials are needed to see how reliable LNR is as a potential prognostic predictor in LSCC.
In conclusion, LNR and LODDS were both predictors of OS and DFS in this cohort. LNR was a more reliable indicator of patient prognosis and appears to be a parameter that could expand the prognostic value of the traditional TNM classification of LSCC.
Author Contributions
Disclosures
Footnotes
Sponsorships or competing interests that may be relevant to content are disclosed at the end of this article.
