Abstract
Chronic inflammatory diseases, including periodontitis, are the most common causes of bone tissue destruction. Periodontitis often leads to loss of connective tissue homeostasis and reduced alveolar bone levels. Human periodontal ligament stem cells (PDLSCs), a population of multipotent stem cells derived from periodontal ligament tissues, are considered as candidate cells for the regeneration of alveolar bone and periodontal tissues. Periodontitis impairs the osteogenic differentiation of human PDLSCs. Noncoding RNAs (ncRNAs), including long noncoding RNA (lncRNA), microRNA (miRNA), and circular RNA (circRNA), have been proposed as vital regulators influencing several differentiation processes including bone regeneration. Still, the molecular mechanisms of ncRNAs regulating osteogenic differentiation of human PDLSCs remain poorly understood. Exploring the influence of ncRNAs in the process of osteogenic differentiation of human PDLSCs may provide novel therapeutic strategies for tissue regeneration as the regeneration of the lost periodontium is the ultimate goal of periodontal therapy.
Introduction
Periodontal diseases, including gingivitis and periodontitis, are possibly the most common diseases in humans. 1 Periodontitis is a chronic non-communicable disease (NCD) that shares risk factors and social determinants with the major NCDs that cause approximately two-thirds of deaths including diabetes, heart diseases, chronic respiratory disease, and cancer. 2 According to the recent Global Burden of Disease Study (GBD, 1990–2010), severe periodontitis is the sixth most prevalent disease globally, with approximately 743 million people affected and an overall prevalence of 11.2%. 3,4 Individuals suffering from periodontitis are at a risk of losing multiple teeth leading to edentulism and masticatory dysfunction, hence affecting their nutrition, self-esteem, and quality of life, along with imposing huge socio-economic impacts and healthcare costs. 5,6 Periodontal diseases are accountable for 3.5 million years lived with disability. 7 As a result of severe periodontitis alone, the global cost of lost productivity has been estimated to be 54 billion USD per year. 8 The prevalence of periodontitis tends to increase with age, and the incidence escalates steeply in adults aged between 30 and 40 years. This burden will continue to rise with the increasing aging population due to increased tooth retention worldwide. 9,10
The periodontal ligament (PDL) is a non-mineralized connective tissue that holds the tooth in place by attaching cementum to the inner wall of the alveolar bone socket. 11 Human PDL contains stem cells that are capable to differentiate into osteoblasts, cementoblasts, and adipocytes. 12,13 Because of these properties, the periodontal mesenchymal stem cells (PDLSCs) were considered to be an ideal source in the repair, regeneration, and maintenance of alveolar bone. 14 –16 However, several factors in the microenvironment, such as aging 17 and inflammation, 18 might cause dysfunction of PDLSCs. As a consequence, their osteogenic capacity might be greatly inhibited. 19 Chronic periodontitis is considered to be caused by bacterial infection and related host immunological responses, resulting in the destruction of alveolar bone and connective tissue. 19 Although researchers report that inflammation inhibits the osteogenic ability of PDLSCs, the exact mechanisms remain unclear. 18,20
Noncoding RNAs (ncRNAs) are usually considered not to possess protein-coding capabilities, rather they mainly function as key gene expression and protein regulators to govern physiological and pathological processes. 21 Different cellular processes are regulated by an array of ncRNAs, which actively contribute to lineage specification. 22 MicroRNAs (miRNAs) are the most studied type of ncRNA, and their role in stem cell maintenance and differentiation has been widely explored. 23 –25 Long noncoding RNAs (lncRNAs) are another type of ncRNA with a length of >200 nucleotides. Substantial evidence demonstrates that some lncRNAs, including MEG3, H19, and lncRNA-ANCR, could govern osteogenic differentiation of stem cells under physiological and pathological conditions. 26 –28 Circular RNA (circRNA) is a recently identified type of lncRNA and its role in several biological processes is becoming more and more evident. 22
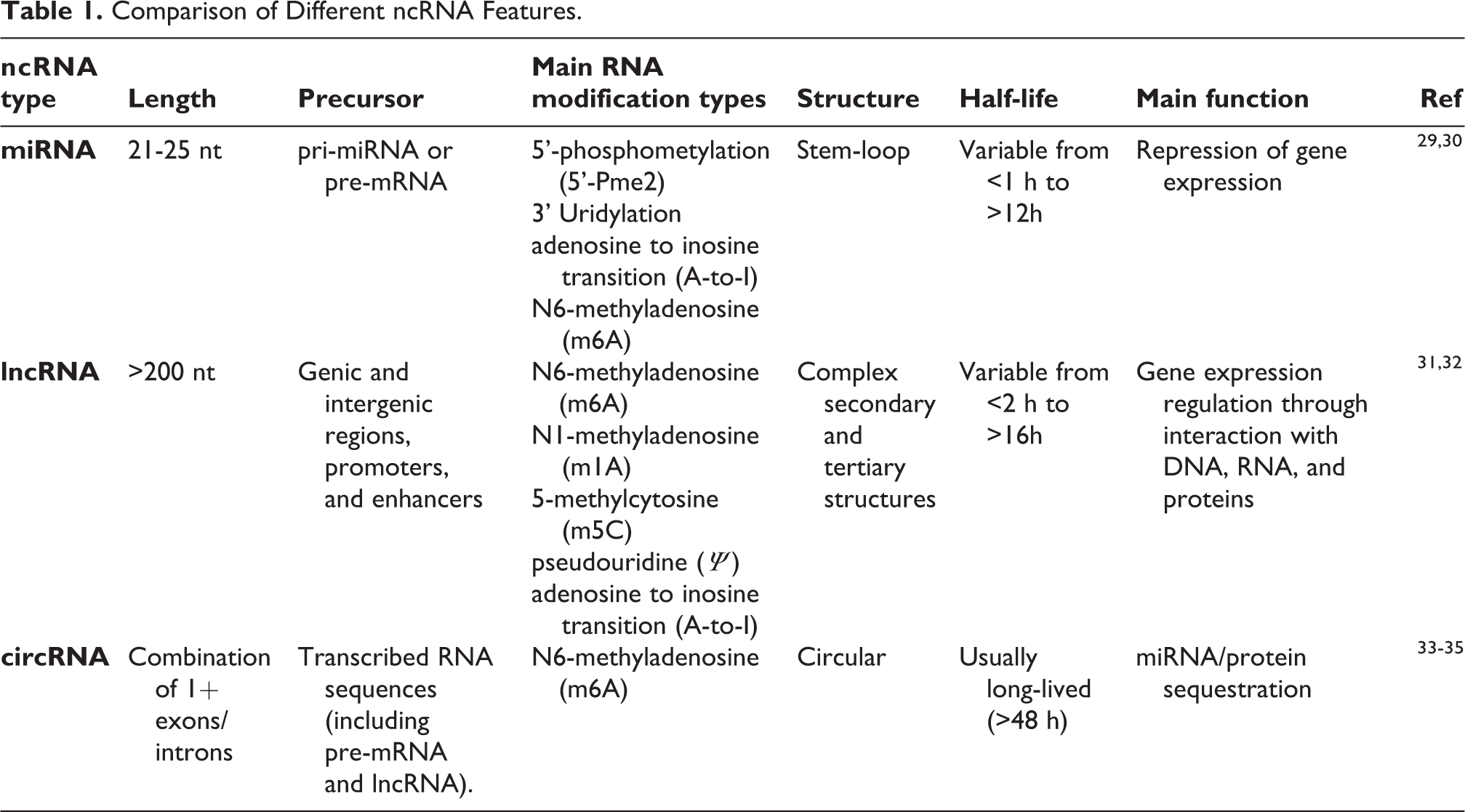
A comparison of the main features of the 3 different ncRNA types are reported in Table 1.
Comparison of Different ncRNA Features.
There is a growing body of literature on the role of ncRNAs in human PDLSCs (PDLSCs). It is imperative to comprehend the molecular mechanisms of osteogenic differentiation of human PDLSCs and how to tune it to be able to construct and replace periodontal tissues (Figure 1). The aim of this review is therefore to provide an overview on the role of ncRNA in the regulation of osteogenic differentiation of human PDLSCs.
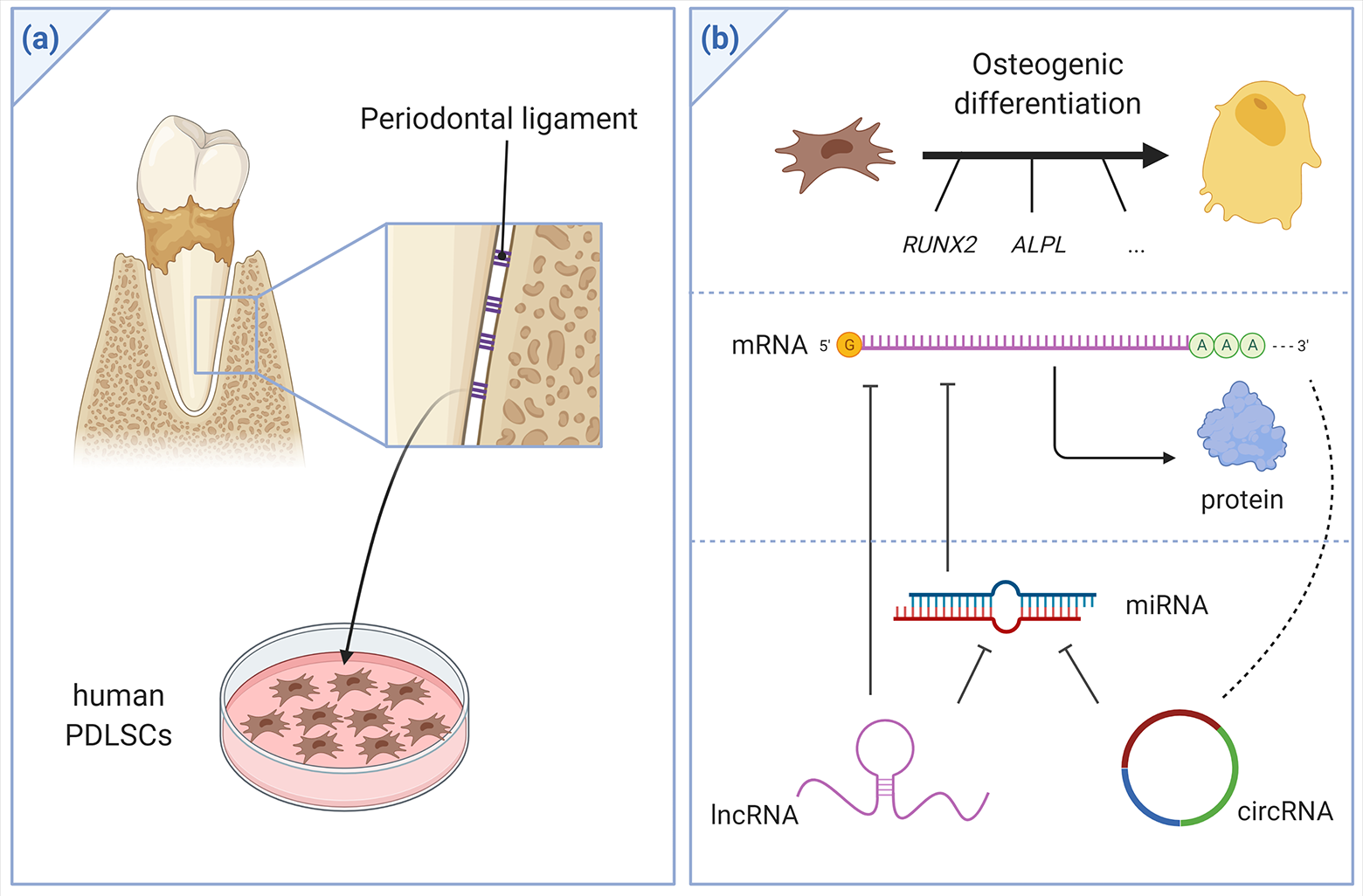
(a) Periodontal Ligament stem cells (PDLSCs) can be isolated from the periodontal ligaments of extracted teeth from healthy subjects (e.g. extracted premolar or third molars) or periodontitis patients. The isolated cells can be cultured and expanded
Expression and Function of miRNA in Osteogenic Differentiation of Human PDLSCs
MiRNAs represent a subset of small ncRNAs (∼22 nucleotides in length on average) that negatively regulate the expression of target genes and play a vital role in cell differentiation. 12,36,37 As a general mechanism, they are able to inhibit mRNA translation by binding to the 3’ untranslated regions (3’-UTR) of target genes and promote mRNA degradation (Figure 2). 38 Therefore, they play a crucial part in diverse biological processes, including but not limited to development, cell differentiation, proliferation, and apoptosis. 39 Numerous miRNAs, such as components of the miR-30 family, 40 have been reported to regulate the osteogenesis of human mesenchymal stem cells. Recently, miRNAs have also been shown to be involved in periodontal diseases, 41,42 and some were investigated as potential salivary biomarkers for periodontal disease and other oral pathologies. 43
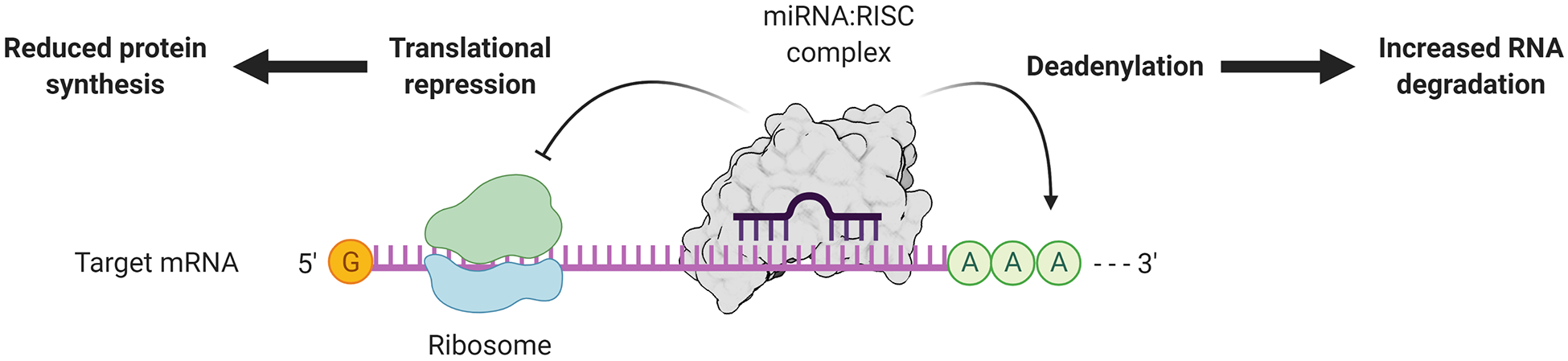
Simplified schematic representation of miRNA-dependent post-transcriptional regulation of gene expression. After biogenesis, exportation from the nucleus and maturation, miRNAs in the cytoplasm are incorporated into the RNA-induced silencing complex (RISC) and direct target recognition. In animal cells, the mechanism of target recognition is mainly through imperfect base pairing. The presence of mismatches directs repression of gene expression through 2 main mechanisms: repression of protein translation via interaction with the ribosomes, and promotion of deadenylation that leads to destabilization of the RNA molecule. Figure created with BioRender.com
Different miRNAs have been identified as playing a role in the regulation of osteogenesis of human PDLSCs. Table 2 summarizes the main findings and focuses on studies that identified both the miRNAs and their target genes/pathways, thus providing a comprehensive explanation of their mechanisms of action.
miRNA Involved in the Regulation of Osteogenic Differentiation of Human Periodontal Ligament Stem Cells (PDLSC).
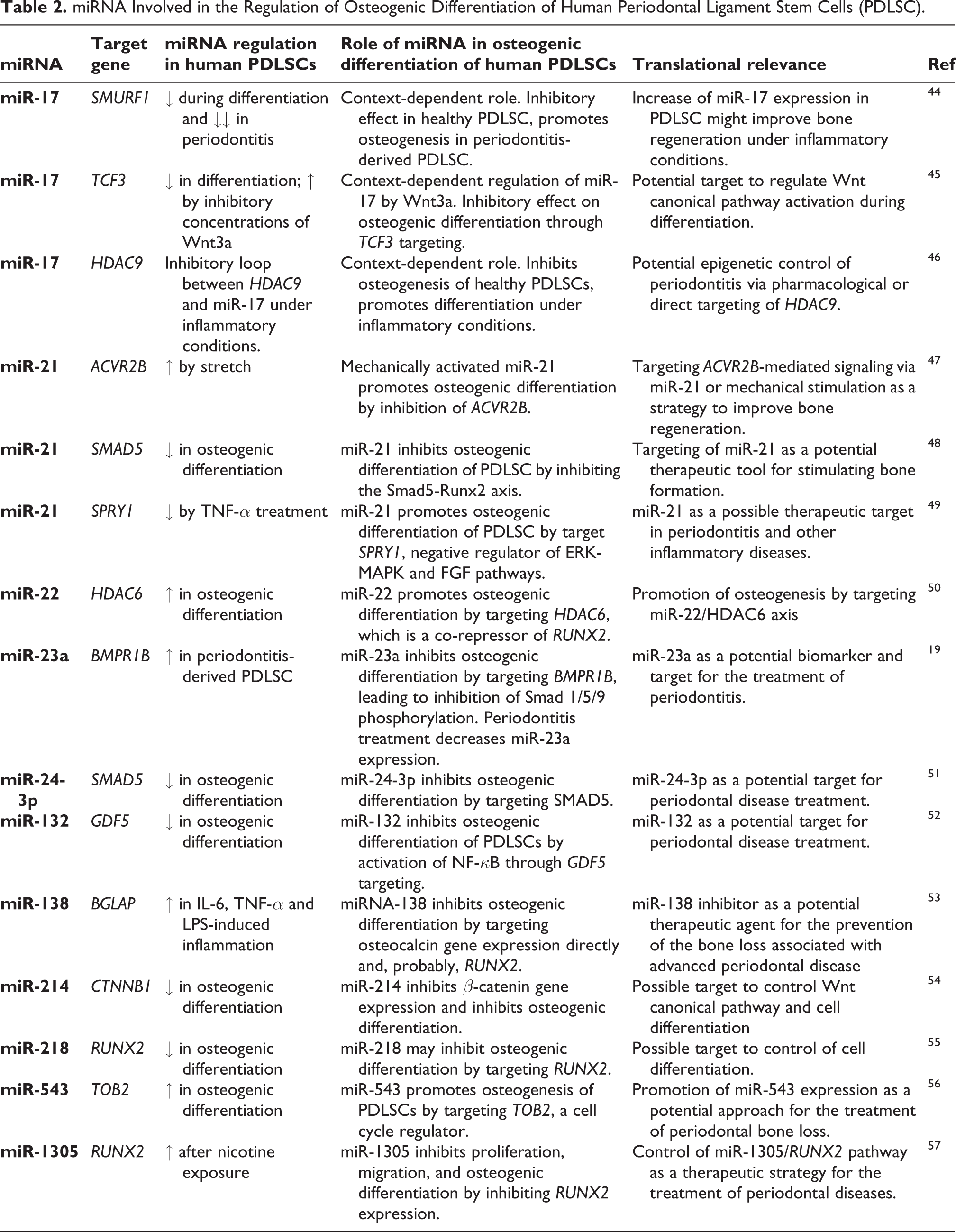
While some miRNAs appear to have a clearly defined role in the promotion or inhibition of osteogenesis, at least 2 of them show a context-dependent type of activation or effect on differentiation. In particular, miR-17 seems to inhibit osteogenic differentiation of healthy human PDLSCs, but it has a promoting effect when the cells are isolated from periodontitis-affected patients or cultured under inflammatory conditions. 44 –46 Similarly, miR-21 shows different effects depending on whether the differentiation potential of PDLSCs is investigated under mechanical stimulation or during inflammation. 47 –49 These results highlight the complexity of gene expression regulation in response to different stimuli, and the importance of addressing an appropriate research question when investigating mechanisms of differentiation at the molecular level.
In addition to those reported in Table 2, further studies identified miRNAs that are likely involved in the regulation of differentiation. Hao et al 58 identified by microarray analysis a miRNA profile for human PDLSCs induced to osteogenic differentiation. A total of 116 miRNAs were found to be differentially regulated, and the expression of 6 of them was validated by qPCR: miR-654-3p, miR-4288, and miR-34c-5p were confirmed as upregulated, while miR-218-5p, miR-663a, and miR-874-3p were downregulated during osteogenesis. Another paper described a miRNA signature for PDLSCs subjected to tension (10% equibiaxial strain at 1.0 Hz for 12 h). 59 The authors identified 53 differentially regulated miRNA between normal and stretched PDLSCs, and validated the expression of miR-1246, miR-5096, miR-638, miR-663, miR-21, miR-4492, miR-4734 as upregulated and of miR-3195, miR-4281, miR-3178 as downregulated.
In accordance with the study by Chen reported in Table 2,
57
miR-1305 together with miR-18b were found to be upregulated both after nicotine exposure
miR-210 was found to be upregulated in human PDLSCs by the presence of hydroxyapatite granules (Endobon® Xenograft). 62 The authors suggested a possible association between miR-210 and VEGF expression/secretion, though the target has not been clearly identified.
Finally, Zhou et al 14 showed that ibandronate, a nitrogen containing bisphosphonate, might promote proliferation and osteogenic differentiation of human PDLSCs and regulate the expression of osteogenesis-related miRNAs, such as miR-130a, miR-18a, mir-125b, and members of the miR-133 and miR-200 family.
Expression and Role of lncRNA in Osteogenic Differentiation of Human PDLSCs
LncRNAs represent a heterogeneous class of non-protein-coding transcripts, which are more than 200 nucleotides in length and can be several kilobases long. LncRNAs have limited or no encoding capability due to the lack of an extended open reading frame, 63 but they are capable of regulating transcriptional complexes and chromatin structure. 64,65 Therefore, lncRNAs regulate the expression of genes and miRNA through a number of different mechanisms 66 (Figure 3) and are now recognized to play an important role in the regulation of key cellular processes, such as cell growth, differentiation, and apoptosis. 67 -69 LncRNAs have an important role in determining cell fate, and some key members of this family have been recognized to regulate osteogenesis. 70
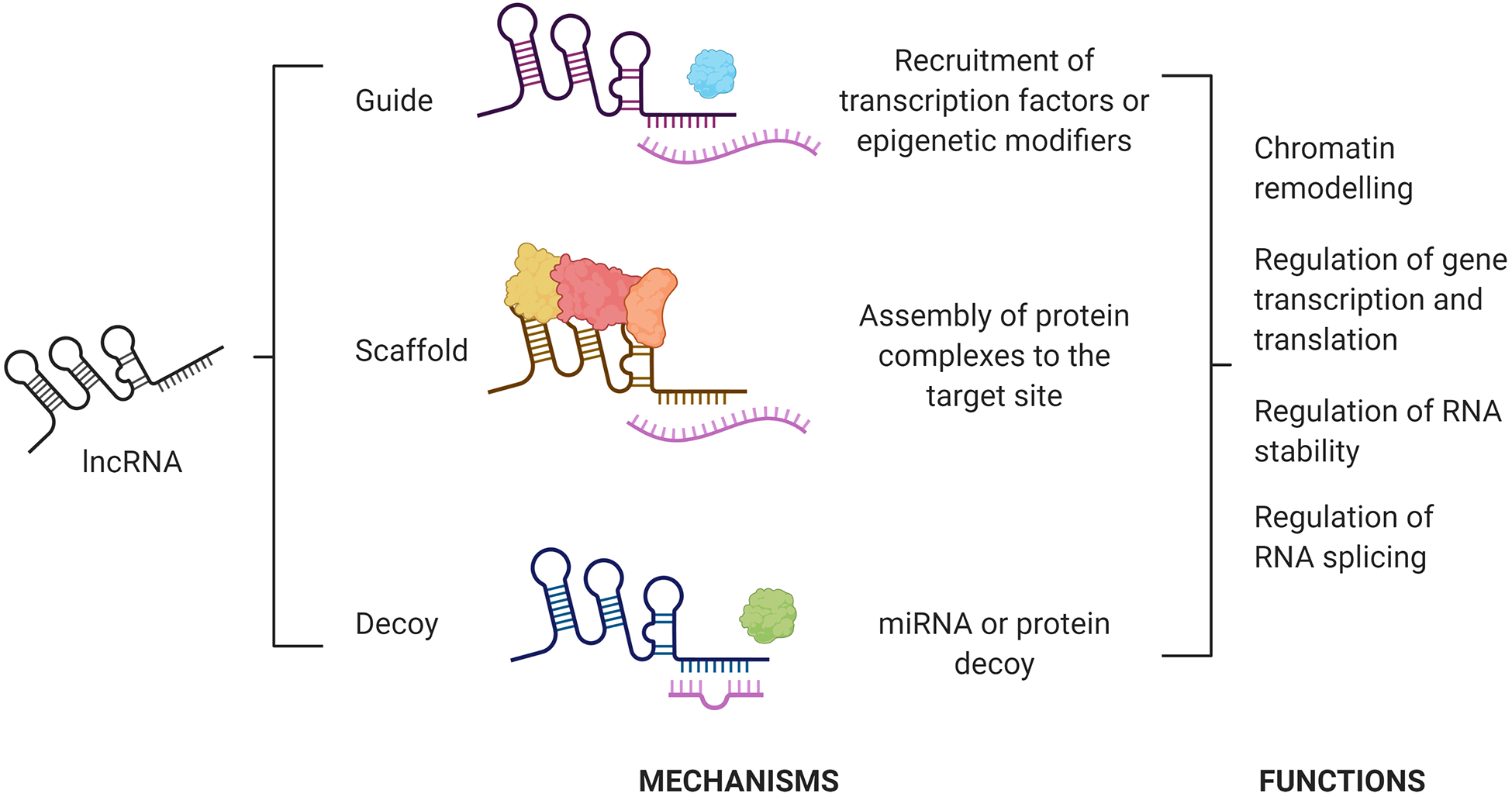
Simplified schematic summarizing main lncRNA mechanisms of action and function. LncRNAs exert their actions in virtue of their sequence and their complex secondary and tertiary structure, which lead to binding DNA, RNA, and proteins. This results in lncRNA acting as guides, scaffolding backbones, or decoys to govern molecular processes involved at different levels in the expression of protein-coding genes, such as chromatin remodeling and epigenetic modifications, regulation of RNA transcription, stability, splicing, and translation. Figure created with BioRender.com.
During the osteogenic differentiation of human PDLSCs, several lncRNAs are differentially expressed.
71
-73
For instance, downregulation of anti-differentiation noncoding RNA (ANCR) inhibits osteogenic differentiation of human PDLSCs.
74
Moreover, lnc Taurine upregulated gene 1 (TUG1) promotes the expression of runt-related transcription factor 2 (
In the paper by Qu et al, 76 994 upregulated and 1177 downregulated lncRNAs were identified by microarray analysis, and those fell into different types, including antisense, enhancer-like lncRNA, and long intergenic noncoding RNA (lincRNA). Interestingly, 5 different transcript isoforms of lncRNA MEG3 were found to be significantly upregulated, supporting a positive role of this lncRNA in osteogenic differentiation in accordance with other studies in PDLSCs 77 and in bone marrow MSCs derived from multiple myeloma patients. 26
Another study used RNA sequencing to identify 960 differentially expressed lncRNA in PDLSCs osteogenesis. Together with differentially expressed circular RNAs (which will be discussed more in details within the next section), many of these transcripts are predicted to bind several miRNAs, leading to the regulation of hundreds of messenger RNAs involved in key signaling pathways of osteoblast differentiation and stem cell pluripotency. 72 LncRNA differential expression was also observed during osteogenic differentiation in a TNF-α induced inflammatory environment. 73
The identification of lncRNA which are differentially expressed during differentiation is of pivotal importance to identify targets that should be studied more in details. However, the validation of expression, and the identification of precise molecular mechanisms is required in order to better understand and exploit the function of ncRNAs. Table 3 summarizes the role of specific lncRNA, with identified targets and specific functions, in osteogenic differentiation of human PDLSCs.
Long Noncoding RNA (lncRNA) Involved in Regulation of Osteogenic Differentiation of Human Periodontal Ligament Stem Cells (PDLSC).

LncRNA-
Other lncRNA have been reported to have a promoting role in osteogenic differentiation of human PDLSCs, such as
Expression and Role of circRNA in Osteogenic Differentiation of Human PDLSCs
CircRNAs are RNA molecules with a wide size range, with ends covalently closed to form the peculiar circular structures. 33 CircRNAs have several regulatory functions, including that of miRNA and RNA binding-protein sponges, 34 which hinder their accessibility to target molecules (Figure 4). The role of circRNAs in regulating key cellular processes during stem cell differentiation has recently started to be acknowledged. Differential expression profiles of circRNAs in osteogenic differentiation of bone marrow 22,80 or adipose 81 derived MSCs was identified, and the function of some of them is being studied in more details. 82,83
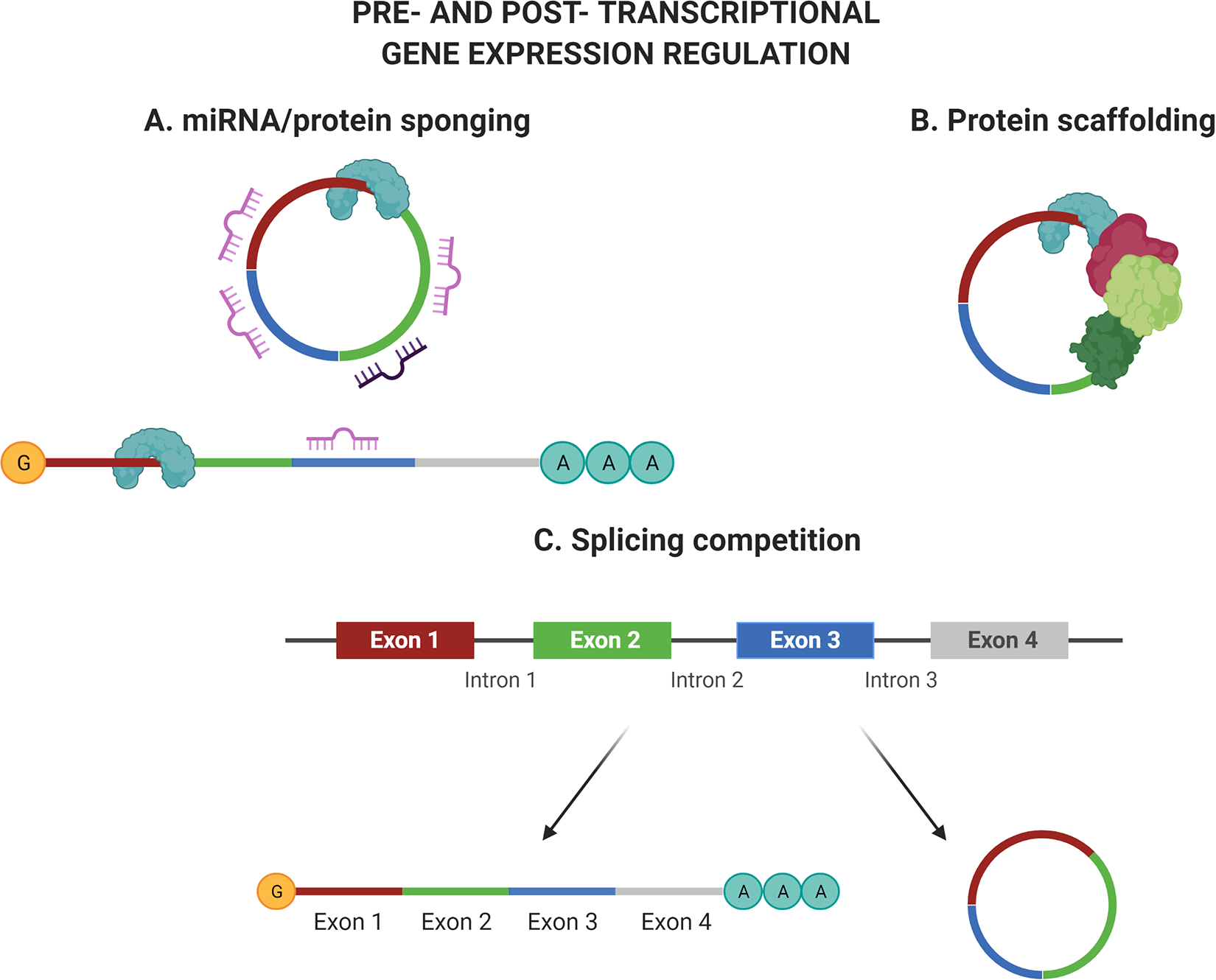
The schematic summarizes some of the main mechanisms of action of circRNA to regulate gene expression, both pre- and post-transcriptionally. (A) Mainly studied is the miRNA/protein sponging function. The sequence derived from the pre-mRNA retains multiple miRNA and protein binding sites. Therefore, the presence of the circRNA acts as a decoy and hinders the ability of miRNA/proteins to reach their target sites on the mRNA. (B) Similar to lncRNA, circRNA can help recruiting and assembling protein together, that could be used for example to facilitate transcription factors complex interaction with the target gene promoter. (C) It is also believed that circRNA formation can compete with the linear mRNA for splicing, therefore regulating the cognate mRNA expression. Figure created with BioRender.com.
Only a few studies identified circRNA expression profiles or functions in osteogenic differentiation of human PDLSCs. Zheng et al
84
analyzed the expression profile of circRNA (together with mRNA and miRNA) during osteogenic differentiation of human PDLSCs, resulting in the construction of a circRNA-miRNA-mRNA network. In the above-mentioned paper by Gu et al,
72
the authors analyzed both lncRNA and circRNA as possible competing endogenous RNAs. In particular, circRNAs derived from
Circular CDR1as, also known as cIRS-7, is one of the most studied circRNA, and it possesses several binding sites for different miRNAs, including at least 70 repeated miR-7 seed sequences.
85
Recently, CDR1as was found to be significantly upregulated during osteogenic differentiation of human PDLSCs and this was associated to miR-7 downregulation,
86
highlighting the miRNA-sponging mechanism in this system as also reported in other contexts. One of the targets of miR-7 appears to be
Overall, circRNA holds promise for the development of new therapeutic strategies to improve osteogenic differentiation of PDLSCs and to control response to inflammation. Definitely, more studies are required in this new field of molecular biology and gene expression regulation.
Clinical Utility and Current Limitations in ncRNA Research and Use
As of January 2020, around 300 actively recruiting clinical studies that include the study or use of miRNA and/or other ncRNAs are registered on ClinicalTrial.gov. This number indicates that there is a large interest in the introduction of ncRNA into the clinical practice; however, further considerations are needed. First of all, a large proportion of the clinical studies fall within the categories of cancer, cardiovascular, respiratory, and gastrointestinal diseases, and no study is directly related to any craniomaxillofacial condition. Also, most of the studies seem to focus on the use of miRNA (and in a much smaller proportion of lncRNA and circRNA) for diagnosis, prognostic evaluation, or drug follow up. While this represents an essentially important tool for precision medicine and for a better understanding of underlying pathophysiological mechanisms, the potential of ncRNA as therapeutic agents seem not to be extensively exploited to date outside basic research and preclinical studies. The development of miRNA-based therapeutics is nonetheless advancing, and recent reviews sum up the state of art about artificial miRNAs challenges and opportunities, plus drugs and companies currently working with miRNA for clinical practice. 88 -90
The reason for this might rely in the relatively recent discovery of the different ncRNA types. The mechanism of RNA interference by small interfering (si)RNAs was first identified in 1993 in the nematode
LncRNAs have important roles in the pathogenesis of periodontal disease via regulation of many signaling pathways, some of them being revealed through high-throughput next generation sequencing methods. 92 Further evaluation of functional links between lncRNAs and their role in mediating osteogenic differentiation of PDLSCs may facilitate the determination of complex molecular mechanisms to better treat periodontal diseases. These comprehensive protocols would also aid in designing personalized procedures in this aspect. 93 Besides, lncRNAs may be considered as therapeutic targets in periodontal disease. Forced siRNA-mediated downregulation of some lncRNAs or their overexpression could influence the pathologic process of periodontal disease, yet these protocols have limited clinical applications so far because of the delivery methods and safety concerns. 93
circRNA were discovered recently and more in-depth basic research still needs to be conducted in order to better understand how to exploit and tune their properties. circRNAs represent a new area for investigation in the field of gene expression regulation, and more rigorous research is needed to clarify their role in osteogenic differentiation of PDLSCs and in periodontitis. Also, once circRNA functions and role will be clearer, they can be engineered, or their expression manipulated, 94 -100 making circRNA as promising new tools for biotechnological approaches in CMF bone regeneration and tissue engineering. Using engineered circRNAs as therapeutic agents also poses a further challenge, as circular RNA structures can be detected by the immune system as they can be recognized as potential threats, such as of viral origin. Thus, the design of therapeutic agents based on circRNAs should be also done carefully to address this problem. Some studies focused specifically on this aspect, 101 -104 realizing that the presence of the m6A modification 102 and of human introns 104 might be important to suppress innate immunity.
Altogether, it is foreseeable that the advances in basic research within the field of ncRNA, unravelling new and still not completely clear molecular mechanisms, in the future will bring to better patient care. A closer connection between the clinicians and researchers with different backgrounds (e.g., cell and molecular biologists, chemists and chemical engineers, bioinformaticians, computational scientists), resulting in an interdisciplinary approach, will benefit and accelerate the translation of basic research results to clinical practice.
Conclusions
This review has provided insights into the role of different ncRNA types and their modulatory network in regulating osteogenic differentiation of human PDLSCs. Owing to their osteogenic differentiation ability, PDLSCs demonstrate efficient potential in the clinical application of periodontium repair and regeneration. A better understanding of the molecular mechanisms involved in bone regeneration or in the pathogenesis of periodontal bone loss will benefit future clinical applications. Indeed, this might help in the identification of novel molecular actors which can be potentially used as biomarkers for disease detection and follow up procedures, or as therapeutic targets/agents to treat certain bone-related oral diseases.
Moreover, since PDLSCs are often considered as potential sources of cells for bone tissue engineering strategies, it is necessary to understand how the microenvironment might affect them at a molecular level in order to design the best strategies of intervention.
Footnotes
Authors’ note
E.D.B. is Deputy Editor for the Basic Science & Molecular Biology section of CMTRO.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This Investigation was supported by AO Foundation and AO CMF.
