Abstract
Study Design:
Ameloblastoma is a benign odontogenic tumor of epithelial origin, with locally aggressive behavior; and due to its invasiveness has increased recurrence rates. It is more frequent in the mandible than the maxilla. Surgical treatment is performed with approaches ranging from conservative to radical surgery, either followed by microvascular reconstruction, or not. Adjuvant treatment has shown better local control in refractory cases. Multiple relapses are associated with BRAF gene mutation at codon 600.
Objective:
This case series aimed to describe the clinical, imaging, histopathological, therapeutic and mutational characterization of 3 patients with refractory ameloblastoma in the maxilla.
Methods:
Data of 3 patients were collected, and descriptions were provided of procedures such as clinical, imaging, surgical technique, histopathological subtype and molecular analysis for detection of BRAFV600E mutation. Results It was obtained a locoregional control after RT of two cases described. After BRAF mutation molecular analysis, no patient presented it.
Conclusion:
Surgery remains the gold standard for the treatment of ameloblastomas, even in refractory cases; however, an expanded approach to obtain free surgical margins, and reconstruction of the maxilla itself may be challenging. Adjuvant radiotherapy is still a controversial topic, but could favor reduction of the local recurrence rate in cases where the surgical margins are compromised after surgical resection. Further studies will be necessaries for analysis of the BRAFV600E mutation, for therapeutic purposes.
Keywords
Introduction
Ameloblastoma is a benign infiltrative neoplasm, of odontogenic origin, which shows a locally aggressive behavior and affects the mandible more frequently than the maxilla. 1 In the recent classification of tumors in the head and neck, by the World Health Organization (WHO) of 2017, 3 histological subtypes were established: solid/multicystic (conventional subtype), unicystic and peripheral/extraosseous, 2 the latter type is observed more frequently in younger patients.
Surgical treatment is the first-line choice for these lesions, and this will depend on the histological subtype, and surgical technique to be performed in order to obtain surgical margins free of neoplasia, which could reduce local recurrences.
Several surgical algorithms have been described in the literature 3, 4 ranging from conservative management (enucleation, marsupialization, curettage) to more radical resections (with a 1 to 1.5 cm safety margin around the surrounding tissues after hemimandibulectomy and/or hemimaxillectomy) and in refractory cases; or with extensive bone destruction, microsurgical free flap reconstructive surgery that allows better results in function and further rehabilitation,. 3
Relative to the maxilla jawbone, cases of ameloblastoma are rare. However, due to its anatomical location and the proximity to innervated structures such as the orbit, the pterigomaxilar fossa and base of the skull, surgery is limited for complete removal of the tumor. The foregoing statement also applies to refractory cases, in which reconstruction poses a challenge to the multidisciplinary team. 4 Refractory cases have been associated with molecular mutations, mainly a mutation in BRAF codon 600 (BRAFV600E), and in these cases, target therapies are considered in the initial phase, with the aim of decreasing tumor volume as well. Indication of the above-mentioned procedure will also depend on the clinical profile of the patient, tumor characteristics and tumor recurrences, as has been described in recent studies. 5
Based on the foregoing descriptions, the present study aimed to show our experience by means of clinical, imaging, surgical, histological and mutational characterization of 3 cases of refractory maxillary ameloblastoma for which, patients underwent treatment at our institution.
Case Series
Case 1
A 56-years-old man was referred to our institution with a painless swelling in the left cheek with no suspicious lymph nodes at extraoral examination. At intraoral examination, mild edema of the left soft palate was observed, in the direction toward the posterior region, close to the palatoglossal arch and palatine tonsil. He had undergone 2 previous surgical treatments in the same region, with diagnosis of maxillary ameloblastoma.
Computed tomography (CT) images revealed a solid image projected toward the left pterygoid muscles, close to the superficial cortical lamina of the ascending branch of the left mandible. The lesion measured approximately 4.3 × 4.1 × 3.2 cm. At the Magnetic Resonance Imaging (MRI) exam, multiple expansive lesions were found, composed of cystic and microcystic areas at the left masticatory space and in the left zygomatic space as shown in Figure 1A and B.
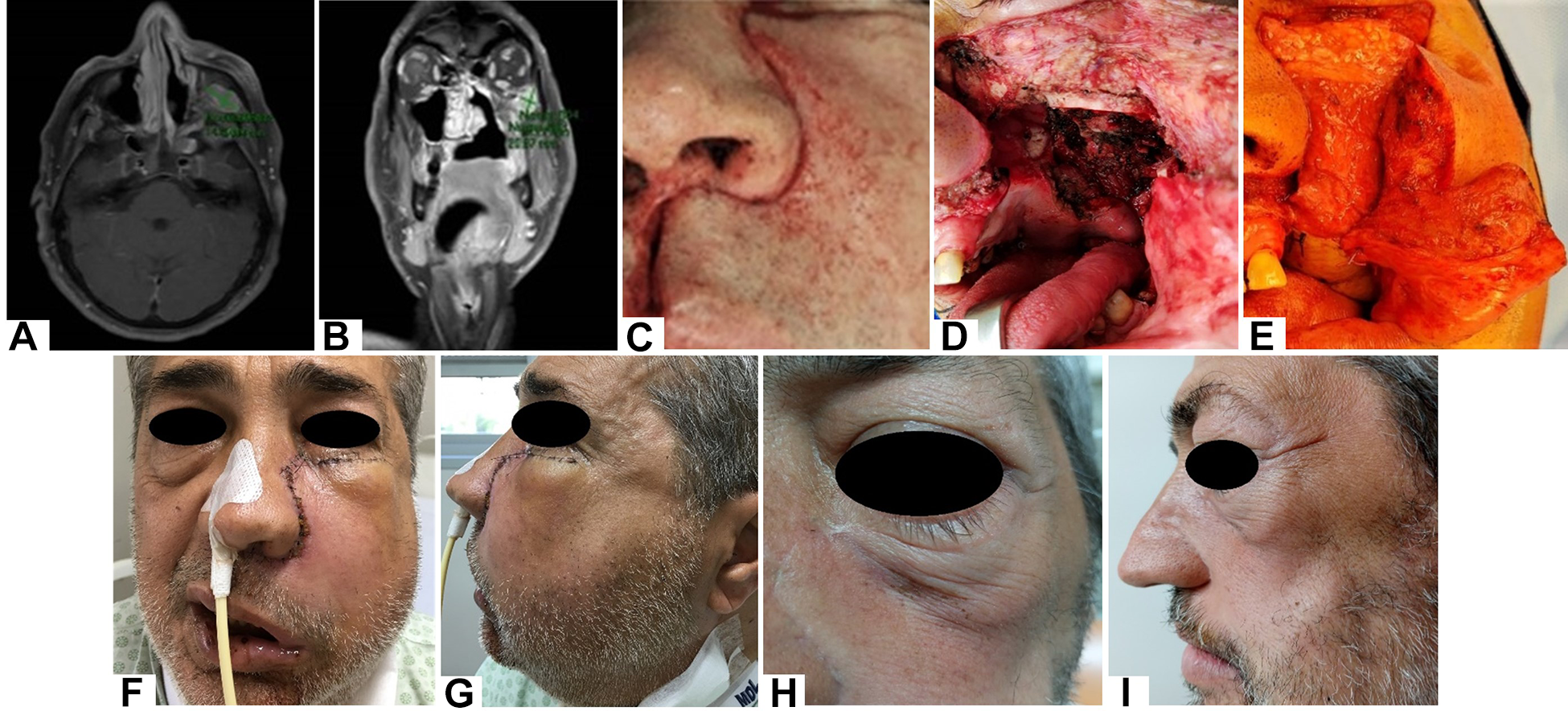
Magnetic resonance imaging (MRI) analysis: (A) axial section of 26 × 12 × 21 mm howed remnant left retrozygomatic masticator space; (B) coronal slice of 24 × 14 × 20.87 mm in left retrozygomatic masticator space; (C) surgical procedure consisted of Weber Ferguson incision made to visualize left retrozygomatic masticator space; (D) performed in subcutaneous and deep plane toward left parotid gland; (E) maxillary osteotomies were performed with sagittal micro-saw; in order to remove complete tumor mass following anatomical limits described above. Clinical aspect after ALT flap placed: (F) at the immediate postoperative phase (frontal view), (G) lateral view and after 1 year of follow-up (H) frontal and (L) lateral.
A total maxillectomy was done for extensive tumor resection that covered the left maxillary sinus region, performed by means of the Weber Ferguson (WF) incision and rhinopharyngectomy associated with masticator space removal before tracheostomy. After resection, the facial nerve and the internal maxillary artery were repaired. Maxillary osteotomies were performed in order to demarcate the following limits: laterally, the temporal zygomatic process, medially through the orbital floor and in the nasal bone, after maxillary synthesis in the midline region, and mid palate suture (raphe) in the hard palate. As regards the soft palate, partial resection of the palatopharyngeal and palatoglossal arch was performed toward the rhinopharynx, through to the lower-posterior region, extended to the coronoid process, and removal of the ipsilateral third molar (Figure 1C to F). Finally, reconstruction of the defect was done using anterolateral thigh (ALT) flap.
Adjuvant Radiotherapy (RT) was indicated in this patient for locoregional control, with the dose of 60 Gy delivered both in the region of the tumor bed and flap area. After 30 sessions completed, Grade 3 oral mucositis was observed and treated by photobiomodulation (PBM) sessions (laser therapy) for the relief of symptoms.
Case 2
A 22-year-old woman previously submitted to conservative surgical procedures such as curettage and enucleation since 2014 and February 2018 and October 2018, respectively, was referred to our institution due to another local recurrence.
A total maxillectomy was performed with resection of the floor of right orbit enlarged to the skin of the face, infratemporal fossa, partial parotidectomy, ethmoid and sphenoid sinusectomy and microsurgical ALT flap was done.
In March 2020, there was suspicion of another recurrence. At MRI, there was evidence of an expansive lesion centered on the posterior and upper margin of the flap (in the maxillary tumor bed), as shown in Figure 2A to D, with multilocular cystic characteristics, measuring 62 × 55 × 42 mm. In fact, the lesion involved the wing of the right sphenoid at the base of the skull, dura mater infiltrated, lateral wall of the right orbit and extended into the extra-cone space of the orbit, where the rectus muscle (from the eyeball) was compressed and displaced.
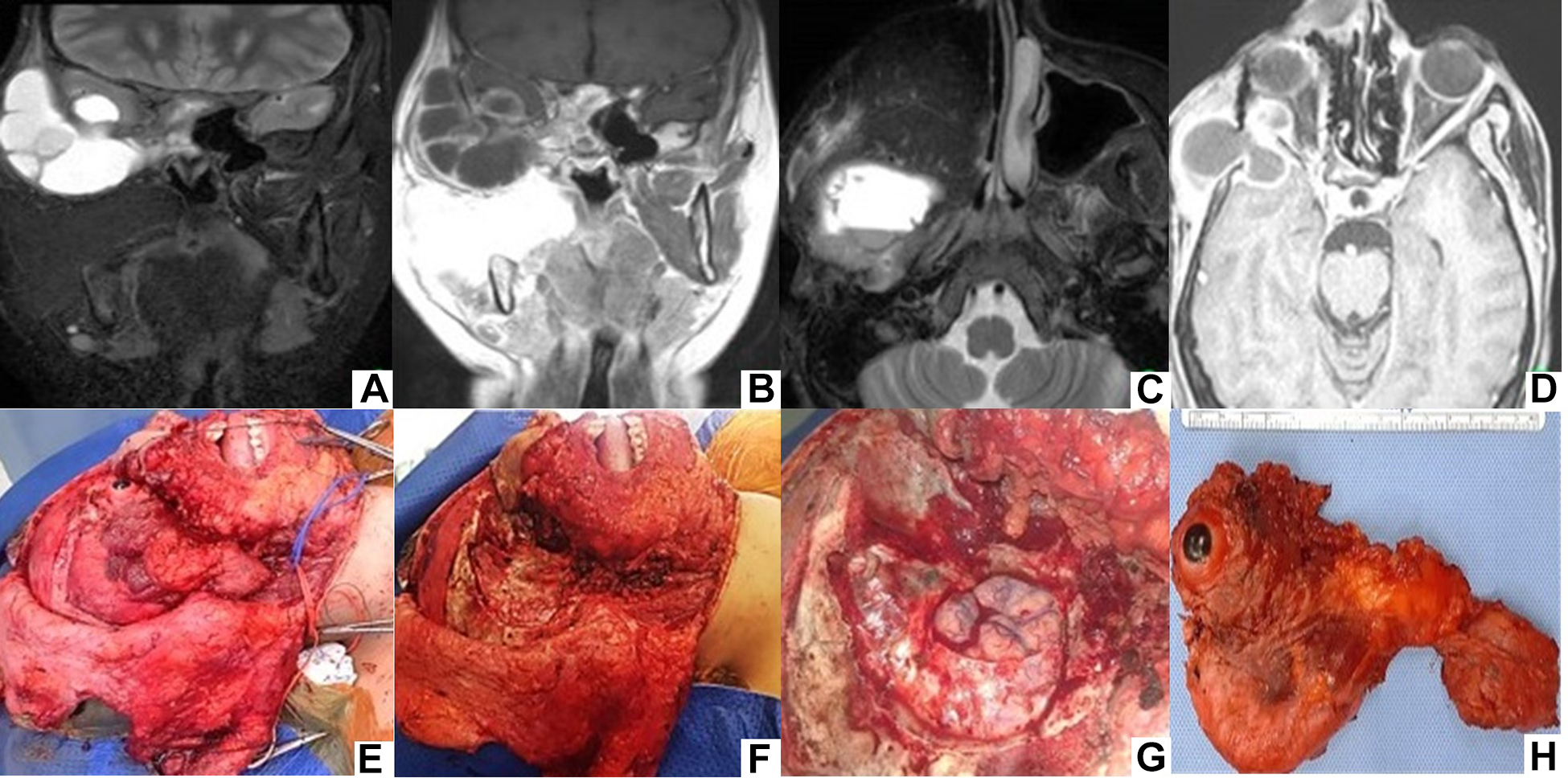
MRI assessment: (A, C) MRI with contrast solution, enabled observation in coronal and axial sections in T2/STIR sequence, of expansive, cystic and multilocular lesion centered on posterior and upper margin of flap and formation at liquid level, with peripheral and septal enhancement. (B) In T1-weighted sequence, lesion is observed extending to middle fossa. (D) In the T1/SPAIR sequence, lesion involving right wing of sphenoid is visualized, lateral wall of orbit with infiltration of dura. (E) Surgical sequence for placing musculocutaneous flap from anterolateral thigh region. (F) Tumor bed after surgical removal of lesion. (G) Craniotomy/craniectomy of sphenoid bone with resection of infiltrated dura mater. (H) Surgical piece removed, observed macroscopically.
In April 2020, she underwent surgical removal of the relapse at the right maxilla and infratemporal fossa associated with the exenteration of the right orbit, greater sphenoid wing, resection of the dura mater, anterior and posterior ethmoidectomy of remaining cells, sphenoid sinusectomy, total parotidectomy including facial nerve resection.
Reconstructive surgery consisted of substitution of the dura mater, and microvascularized reconstruction by placement of ALT flap as shown in Figure 2E to H.
Adjuvant RT was performed at tumor bed with a total dose of 60 Gy delivered in 30 daily sessions of 200 cGy, which caused a moderate degree of toxicity observed in the oral mucosa that covered the irradiated field with PBM that improved the local condition.
Case 3
A 74-year-old man, diagnosed with ameloblastoma since 1994, who underwent multiple surgeries (12 registered in total) all with compromised surgical margins, reported that the last recurrence was detected in 2017. At our institution, the main complaint referred was the presence of cutaneous fistulae and ulcer measuring 10 × 10 mm at the medial corner of the right eye that started on November 2019; and at MRI, lesion progression was observed.
At MRI analysis, the lesion was centered in the left nasal fossa with areas of cystic/necrotic degeneration, with invasion from the anterior cranial fossa toward dura mater. In addition, edema of adjacent parenchyma was observed extending across the anterior ethmoidal region on right side. This infiltrated the medial wall of the right (extraconal) orbit and subcutaneous area from the medial epicanthus to the roof of the left orbit with extension to the left maxillary sinus, measuring almost 61 × 54 × 48 mm in the main axes (Figure 3).
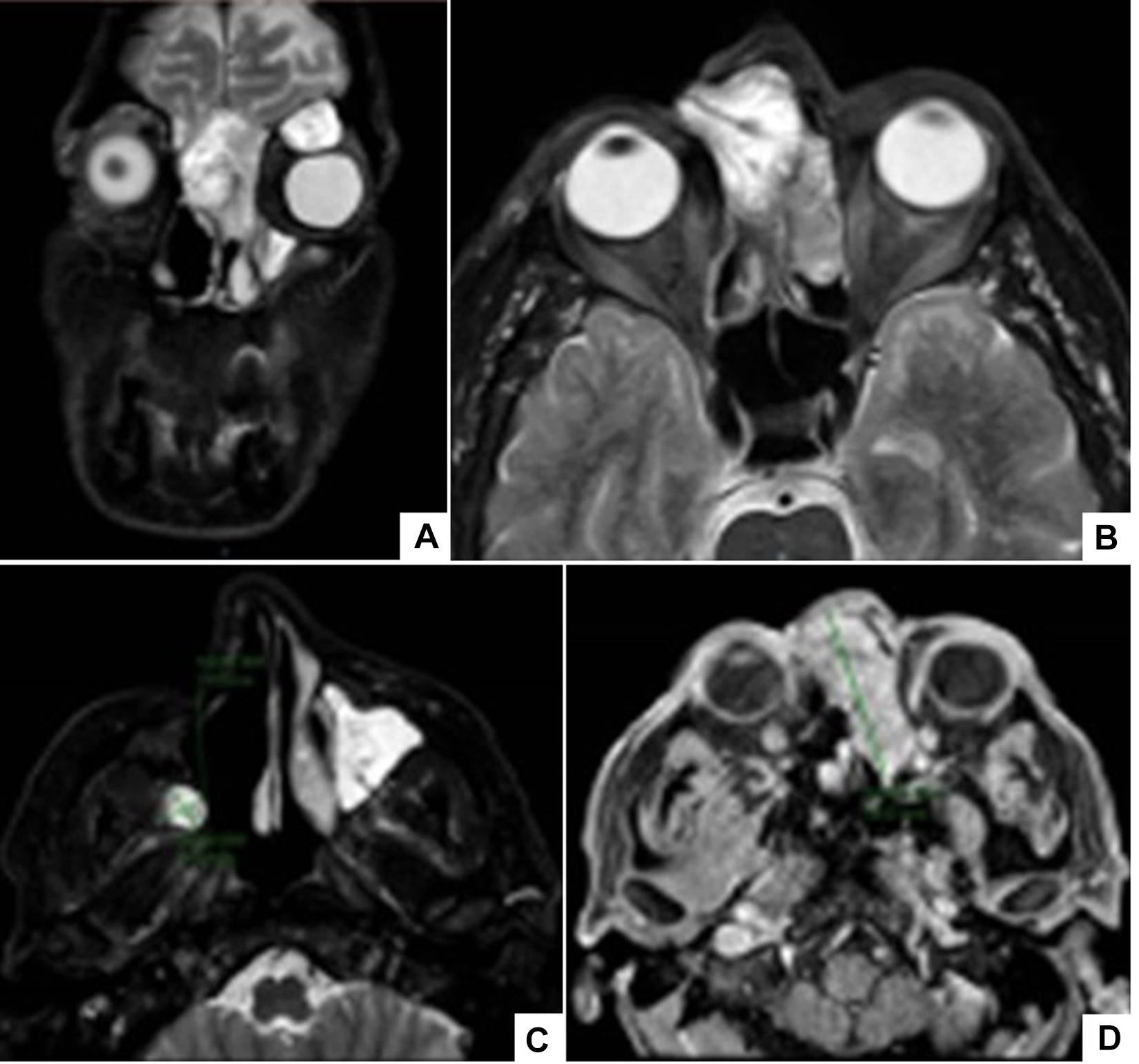
MRI showed: (A) coronal slice showed lesions centered at left nasal cavity surrounded by areas of cystic and necrotic degeneration, with (B) invasion of anterior cranial fossa and (C) extending toward anterior right ethmoidal region, invading right (extraconal) orbital ball and subcutaneous region from medial corner and (D) orbital roof with lowering of eyeball with extension to left maxillary sinus.
After histopathological analysis, multicystic ameloblastoma was confirmed. Thus, facial osteotomy, total maxillectomy on the right, meso-suprastructural maxillectomy on the left, tracheostomy, passage of NET (nasal-enteral tube) and cutaneous tumor resection were performed (Figure 4A to D).
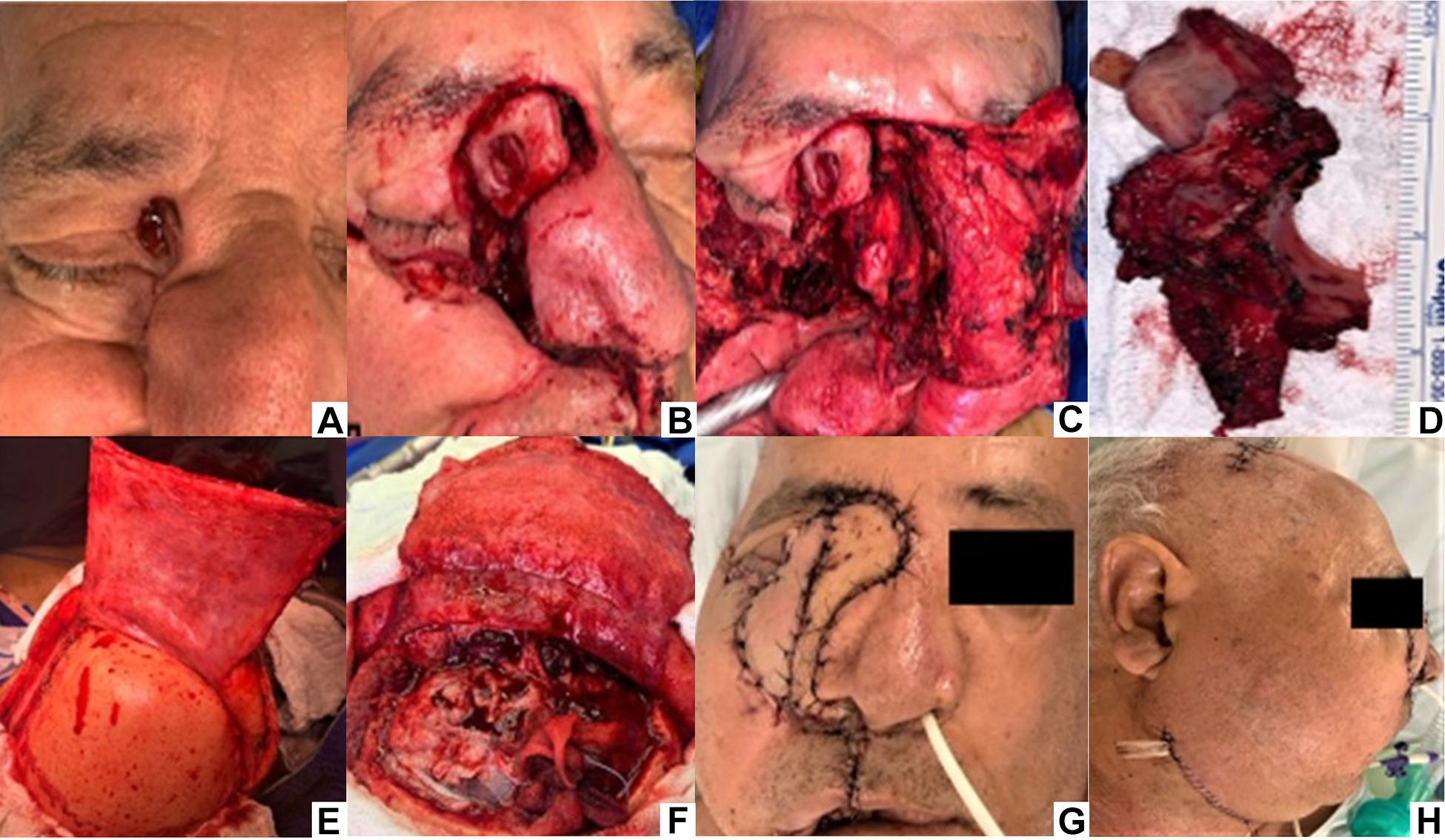
(A) At extraoral examination, an ulcerated lesion was observed in upper medial corner of right eye; (B) modified Weber Ferguson approach was performed; (C) meso-suprastructure maxillectomy to expose infiltrated region; (D) surgical specimen resulting from resection. (E) Scalp lifted flap for dural and craniofacial reconstruction with pericranial detachment; (F) dura mater exposure at anterior frontal dural region through which this entire portion was resected; (G) microsurgical reconstruction (frontal view) and (H) lateral view after flap positioned.
After this, a bicoronal approach to access the frontal sinus (Figure 4E) was performed for complete removal of this infiltrated tissue (Figure 4F). Finally, through the transfacial access, microsurgical reconstruction of the orbital-maxillary complex was performed by placing an ALT flap, obtained from the left thigh, with a bypass between the microsurgical anastomoses. In addition, a full thickness skin graft was placed on the right hemi-face and a fascia-lata graft on the medial face for reconstruction of the orbits and bone graft on the nasal base for reconstruction of the columella as shown in Figure 4G and H.
For these 3 refractory cases of maxillary ameloblastoma, the histological subtype was solid / multicystic according to the recent classification (Figure 5). Finally, as mentioned above, mutation codon 600 (BRAFV600E) was not detected in this case series, (Figure 6A to D).
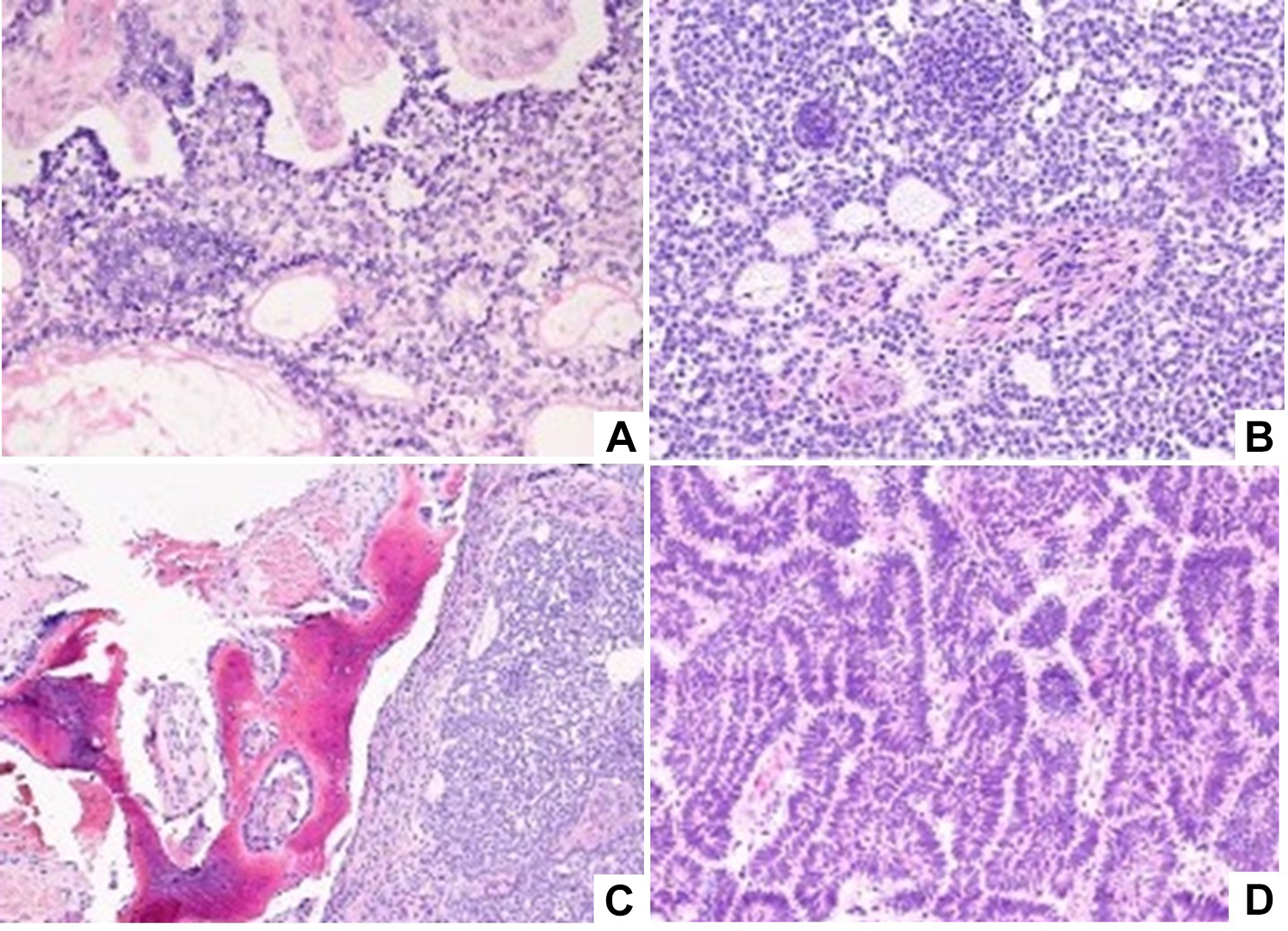
Histological analysis of surgical specimens by Hematoxylin & Eosin (H&E). (A) Hyperchromatic palisading epithelial cells forming islands and cords within a fibrous stroma, with multiple cystic areas that characterized Ameloblastoma, conventional type. (Case 1); (B) another case showing similar cytological and architectural characteristics (Case 2); (C) lower magnification microscopy (100×)demonstrating extensive bone infiltration with larger extent (Case 2); and finally, (D) higher power magnification (400×) microscopy evidencing nuclear palisading and hyperchromia in the tumor cells, arranged prominently in cords (Case 3).

Sequencing readings by PCR through IGV (Integrative Genomics Viewer) software for each sample evaluated, which showed absence of mutation at codon 600 of BRAF (BRAF V600E).
Discussion
Ameloblastoma exhibits a locally aggressive behavior, and when refractory, after multiple local recurrences, there has been increased involvement of the surrounding structures, with more frequent occurrence in the mandible than maxilla jawbone. 3,4,6
Refractory ameloblastomas in the maxilla were observed in 3 patients, whose medical records, age and type of presentation were different. According to previous retrospective cohort studies, the histological subtype with the highest recurrence rate is the solid/multicystic type, and in a lower percentage, other subtypes (unicystic, peripheral). Solid/multicystic ameloblastoma is generally found in the population between the third and fourth decade of life, while the peripheral/extraosseous pattern has been described mainly in younger patients. 1,3,5,7 However, in our case series, the 3 patients were diagnosed with the solid/ multicystic pattern associated with multiple relapses, as described previously.
Management of ameloblastoma is primarily surgical and can be divided into conservative and radical surgeries, which will depend on the histological subtype and its clinical presentation. In the case of unicystic and peripheral subtypes, they are usually treated conservatively, while the solid or multicystic pattern is often treated with radical resection surgery with free margins. 3,6
All surgeries described for the patients at the beginning of diagnosis were performed by means of a conservative approach, such as curettage and enucleation that showed compromised surgical margins, which demonstrated that, an inadequate surgical approach increased the risk of local failure. 3,4 Whereas radical resective surgery, such as segmental resection or with margins up to 1.5 cm including healthy bone, are associated with lower rates of recurrence; however, a locoregional reconstruction becomes necessary, 7,8 as shown in these cases submitted to hemimaxillectomy with microsurgical free flap reconstruction. All patients showed extensive soft tissue defects and our first option for reconstruction of those defects was the anterolateral thigh flap. Its flexibility allows for optimized coverage with the possibility of simultaneous reconstruction of skin and mucous defects while the pedicle vessels allow for cervical vessels anastomosis without graft interposition. 9
The use of radiotherapy (RT) for ameloblastoma is controversial for locoregional control; however, it could be indicated in patients who are not fit for resection of recurrent lesions and or with post-surgical microscopic disease or macroscopic residual disease. 10 Therefore, this therapeutic modality is preferably offered via IMRT (Intensity modulated radiation therapy). The total dose should cover the original tumor bed (bone damage and soft tissue involved) plus a safety margin of 0.5 to 1.0 cm. The final dose ranges from 55 to 66 Gy delivered in 20 to 35 daily fractions in the postoperative period, and 65 to 70 Gy for inoperable cases (primary tumor and recurrences).
In this case series, 2 patients (Cases 1 and 2) underwent RT after surgery using the IMRT technique with total dose of 60 Gy at the tumor bed, and covering part of the flap positioned, in order to decrease the risk of local recurrence. Side effects of radiation were observed in both patients, and this was controlled by local measures low-level laser therapy and oral care. At present, both patients have completed RT with satisfactory resolution of local side effects caused by radiation. 10
Finally, several studies have associated the presence of BRAFV600E mutation in patients with refractory ameloblastoma, of different ethnicities, including the Brazilian population. 5 A recent study conducted in Korean patients with diagnosis of refractory ameloblastoma, 90% of patients showed this mutation, and multicystic subtype was identified in the majority of patients. 11 In fact, initial diagnosis of BRAFV600E could be considered a prognosis of neoadjuvant or adjuvant treatment established, such as Vemurafenib drug for decreasing tumor volume in cases that were non-surgically removed, due to their location or increased surgical morbidity due to the multiple recurrences. 12 In the present case series, no patient presented BRAF mutation after molecular analysis.
Conclusion
Refractory ameloblastoma located in maxilla bone is rare, and surgical removal of it could be a challenging task, due to its aggressive infiltrated local behavior, associated with higher rates of recurrence and increased surgical morbidity. RT is still controversial for local control, therefore, further studies would be necessary to clarify its role in locoregional control. Although BRAFV600E mutation has been detected in several populations worldwide, and directly associated with refractory cases, it was not detected in this study, therefore a larger number of patients involved in cohort studies is required for detailed analysis.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Wilber Edison Bernaola-Paredes is a student fellow and supported with a scholarship by the National Council for Scientific and Technological Development of Brazil (CNPq) (140071/2019-9).
