Abstract
This review discusses the field of coprocytobiology, defined as a combined method of cell preservation, isolation, and cytology, which has applications to the investigation of noninvasive fecal screening for colorectal cancer. In the decade since the field was last reviewed, cell isolation has progressed rapidly via the development of technologies such as microfluidic and magnetic cell sorting. The landscape of cytology has also advanced in this time with the emergence of novel cytological methods and cell preservation strategies. Previous reviews present an outdated and incomplete view of coprocytobiology, summarizing a limited number of early publications, ignoring the principle of cell preservation and focusing on a single method of isolation rather than the field as a whole. In contrast to these publications, this review presents an updated, comprehensive, and unbiased representation of the technical aspects of coprocytobiology and provides unique insight into the common methodological pitfalls, best practice, and future directions of cytological screening for colorectal cancer.
Graphical Abstract
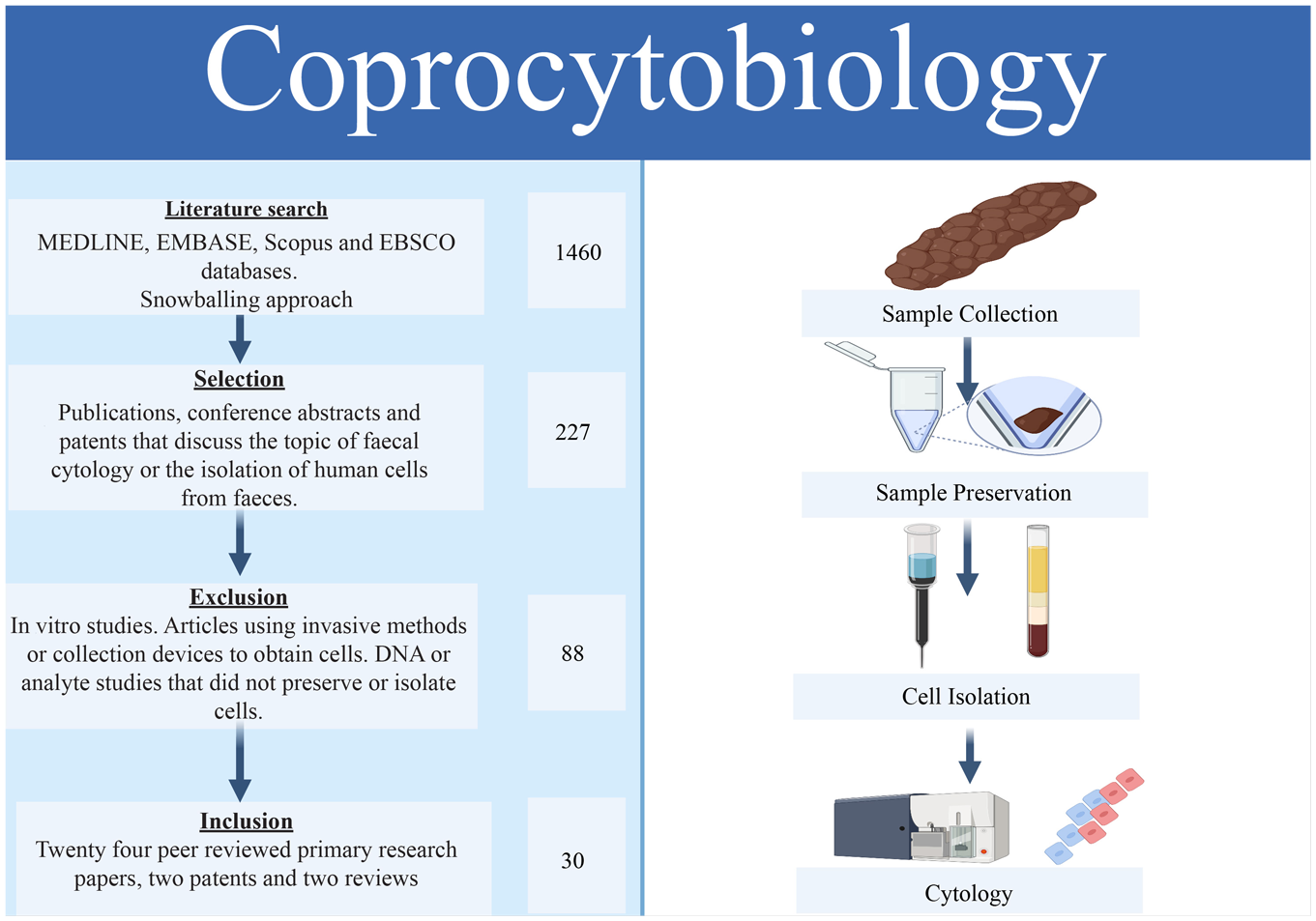
Introduction
Diagnostic sampling is a vital component of medical practice. Timely and appropriate collection of relevant samples allows clinicians to gain an understanding of a patient’s health and design tailored treatment programs. The most common forms of sampling are, however, often invasive. For example, in the assessment of possible underlying colorectal cancer, colonoscopic biopsies provide reliable data and are considered the gold standard of investigation. However, these investigations can be painful and cumbersome and require specialized medical knowledge to minimize the risk of complications such as infection, bleeding, hematoma, or bowel perforation. 1 These factors impact the healthcare budget, patient choice, and outcome.1,2 In response to these limitations, new forms of screening using liquid biosampling have been implemented.
Liquid biosamples are biological fluids that reflect the pathophysiological state of an individual. 3 The ideal liquid biosample can be collected in small volumes by the patient, stored with ease, and transported to the clinic with minimal effort. Feces is an example of a liquid biosample specimen. Perhaps the most advantageous aspect of fecal biosampling is the predictable nature of production and potential for home collection. Therefore, the collection of feces does not require the specialized and expensive clinical knowledge inherent with other forms of sampling, such as colonoscopic biopsy. Fecal samples contain many medically informative constituents that reflect the physiological state of the colon. 4 With the appropriate collection, transport, and storage methods, home fecal collection could have utility in the investigation of colorectal cancer and further minimize the invasive experience of sampling in clinical settings.
On the Origin of Feces
Feces is a diverse mass of several components, many of which can be measured and investigated to gain insight into the internal workings of the gastrointestinal tract. The majority of a fecal sample is composed of water 5 ( Fig. 1 ). The dry component of feces is mostly split evenly between bacterial cells and proteins from food or produced by bacteria. However, a very small proportion of the dry weight is also made up of epithelial cells—approximately 1010 epithelial cells are shed from the host’s gastrointestinal tract every day.6–9
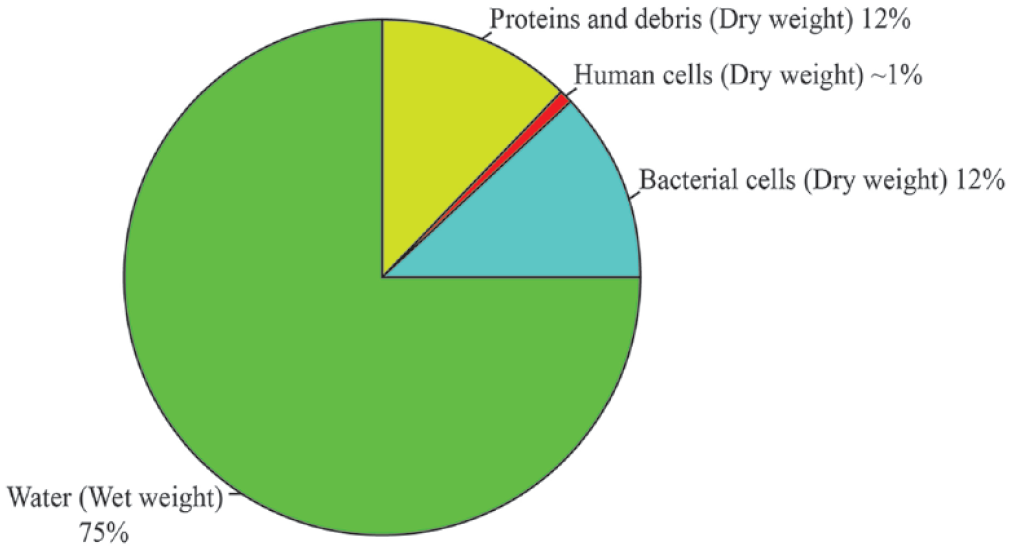
The composition of a fecal sample. Fecal samples are made up of wet and dry components; the wet component (green) accounts for ~75% of the weight of a fecal sample and is composed of water and soluble molecules such as lipids and some polysaccharides (e.g., mucins). 10 Approximately 9 × 1010 bacterial cells are present per gram of stool (blue). 11 These cells comprise ~50% of the dry mass of a fecal sample. The other 50% of the dry mass is made up of undigested proteins and insoluble polysaccharides such as cellulose (red). A relatively small number of host cells shed from the epithelial lining of the colon are also present (purple) (5 × 104 to 4 × 107 cells per sample).12,13
Feces in the lower gastrointestinal tract is surrounded by viscous polysaccharide chains that form gastrointestinal mucins, 6 which play essential roles in fecal transit, immune defense, and nutrient reabsorption.14,15 Mucin captures shed epithelial cells and bacterial cells; these cells are driven into the fecal mass via a process known as peristalsis, wherein contraction of the colon mixes feces and mucin.6,16 In the lower gastrointestinal tract, much of the body’s defense from bacterial infection is achieved by the physical barrier of gastrointestinal mucus that prevents penetration of bacteria from stools into the epithelial layer. 17 Immune cells are mainly located in the subepithelial layer of the colon lining; they are occasionally identified in feces; however, they do little to modulate the presence of bacteria in the fecal mass itself.17,18 It has also been proposed that the transit of fecal matter through the large intestine causes friction against the lining of the gut and collects host epithelial cells in its viscous mass as it passes through.19,20 Variations in the rate of cell shedding occur between individuals and can be indicative of the presence of a gastrointestinal disorder.6,19
Fecal Biosampling and Cancer
Colorectal cancer is responsible for almost 1 million deaths per year, making it the second-highest cause of cancer-related mortality worldwide. 21 As a result, detecting colorectal cancers is one of the most frequently cited motivations for examining fecal samples. 22 The physiological conditions and proliferative processes of the colon contribute to the efficacy of fecal samples for colorectal cancer screening.
In the colon, cell proliferation is common and maintains the integrity of the epithelial layer by encouraging turnover of old cells, which are released into feces via extrusion and shedding. 8 Hyperproliferation leads to the development of dysplasia, an abnormal morphology that composes colonic growths known as adenomas. 6 Adenomas themselves typically do not cause illness, although they may bleed; however, epithelial adenomas can often develop into adenocarcinoma, 23 and in doing so, adenomas modulate the surrounding environment, leading to an increased crowding of villi that is associated with increased extrusion of colonocytes.6,8,20 Ninety percent of colorectal cancers originate from the epithelial layer; 23 the cellular changes produced by these epithelial cell tumors are reflected in feces by several biomarkers, such as DNA, protein, and hemoglobin content.6,23–25
Noninvasive screening of fecal biomarkers for colorectal cancer has had a far-reaching impact on early detection and treatment, reducing mortality by 18%.26,27 Current screening regimes that rely upon home sample collection mostly ignore cellular components of biosamples, instead focusing on molecular methods that attempt to detect and analyze DNA, analytes, or chemical factors. 25
The current screening program for colorectal cancer relies on the immunochemical fecal occult blood test (iFOBT). 25 In Australia, the iFOBT’s specificity is high, correctly identifying 95% of individuals without precancerous adenoma or colorectal cancer.25,28 However, the ability to detect affected individuals (sensitivity) is low, at 59%. 25 Affected individuals who are misidentified by a screening test are known as false negatives. 25 Such false negatives arise because iFOBT relies on detecting small amounts of heme, a component of blood released by highly vascularized neoplastic formations that are indicative of adenocarcinoma. 25 This is problematic as not all malignant colorectal neoplastic formations bleed, resulting in incorrect negative iFOBT results. 29 False negatives can have an immense impact on patient outcomes, as crucial early treatments will be delayed until symptoms become apparent or an accurate test is completed. 29
Noninvasive tests based on genomic markers, such as Cologuard (Exact Sciences Corporation, Madison, WI), identify multiple genomic and epigenomic aberrations in key genes related to colorectal cancer, including KRAS and BMP3, APC, and TP53.22,30,31 Cologuard also incorporates a detection method for heme in the same manner as the iFOBT. 32 Genomic tests have been shown to have a high sensitivity for colorectal adenocarcinoma; however, the expense of these tests greatly reduces patient compliance and has therefore limited their impact on a population level.33,34 Cheaper tests such as iFOBT are often imprecise and prone to misclassification. In a clinical sense, these false classifications can have a lasting effect on patient well-being or lead to unnecessary and costly invasive procedures. 35
Cell-Based Screening
Cell-based screening approaches provide an alternative to analyte or DNA-based methods and are accepted practice in many current clinical screening programs, such as those used for cervical cancer screening in some countries.36,37 The Papanicolaou smear program has been largely responsible for the 70% decline in cervical cancer mortality over the last 70 years and is considerably cheaper per test than DNA-based screening.4,25,38–40 In many developed nations, such as Australia, the cost of DNA-based methods has meant that the iFOBT is still currently the primary form of bowel cancer screening. 25 Here cervical cytology was still being applied as recently as 2018, at a per-test cost of $19.60 (AU). 39 Contrasted against this is the cost of a fecal DNA test at $877.50 (AU) and an iFOBT home test kit at $32 (AU). 25 We propose that cytological methods could potentially be adapted to the analysis of feces and provide an economic, accurate, and noninvasive alternative to current screening regimes in these nations.
Cytology uses methods to investigate the physical properties of exfoliated cells in a biofluid and draw inferences about the state of these cells and their tissue of origin. 41 When collected and stored under appropriate conditions, cell surface properties are relatively stable, and they maintain their morphology. 42 Cytology readily identifies important physical characteristics, such as cell structure, cell number, nuclear state and morphology, which are also mostly ignored by molecular methods.25,40 This is mainly practiced on suspensions of individual cells, although it can also be applied to clumps of cells.16,43 Cytological methods have seen limited application to feces, mostly due to the difficulty of preserving cells in the presence of bacteria and mucins.43,44 Cytology is also hindered by some of the intrinsic properties of fecal samples, listed below, which can obstruct inspection of cells in these samples. 16
Limitations of Cytological Screening in Stool
The application of cytology to fecal samples has been limited by poor visualization of gut-derived cells due to the high proportion of debris and non-host cell material;16,19 a lack of established baseline measures of host cells (see the “Cytology” section) and unfavorable sample characteristics, which damage host cells and alter their morphology.4,45 These limitations may be addressed through the discipline of coprocytobiology. 4 With new advances in laboratory methods and preservative approaches, self-collection of biosamples from home is beginning to become a more realistic possibility for clinical cytological screening approaches. The field of coprocytobiology has made headway into making this a reality; this field has developed several innovative methods that have the potential to change the landscape of colorectal cancer screening.
Coprocytobiology: The Key to Cytological Screening in Feces
Early study of the human cellular component of stool was pioneered initially by the groups of Iyengar and Nair, the latter coining the term “coprocytobiology” for their specialized methods of studying purified cellular components of fecal samples.4,46 Throughout the nineties, cellular isolation from fecal samples followed by cytological assessment was applied experimentally to pathologies such as irritable bowel syndrome and colorectal cancer.12,47 The field of coprocytobiology has advanced since it was first reviewed in 2003, producing novel methods and a series of contemporary publications.4,7,13,16,18,43,47–56 The various cellular characteristics of colonic adenocarcinoma provide a rich landscape for the implementation of coprocytobiological techniques. Innovative isolation methods using existing and recent technologies could be further applied to improve the efficacy of fecal cytology and develop new screening regimes for colorectal cancer.
Methods of Coprocytobiology
Coprocytobiology enables the cytological inspection of cells in fecal samples by using the processes of cell preservation and isolation to counter the effects of unfavorable fecal characteristics. Variations in the technical aspects of an isolation or preservation method can drastically alter the number and/or morphology of exfoliated cells in a fecal sample ( Table 1 ).4,12,16,42,43,57 The number of cells exfoliated into stool varies greatly depending on these methods and the population sampled. For example, Kaeffer et al. used a magnet–antibody-based method to isolate 20–40 cells/g of wet stool from neonates, 18 whereas Nair et al. captured 20–40 million cells/g of wet stool from adult participants using an alternate technique. 4 Chandel et al. used the same method as Nair et al. to isolate 50,000–2,000,000 cells/g of wet stool from neonates. 47 Bandaletova et al. further investigated colonocyte numbers in feces, finding that numbers of exfoliated cells were highly variable between adult individuals with up to sevenfold differences. 19 Although the number of human cells recovered from fecal matter is widely variable, these publications conclusively show that a human cell component makes up a significant portion of dry stool. The intricacies of coprocytobiological techniques therefore have a considerable influence on the success of a fecal cytological investigation and must be understood in order to build optimized screening tools for colorectal cancer.
Technical Variations in Cytobiological Technique.
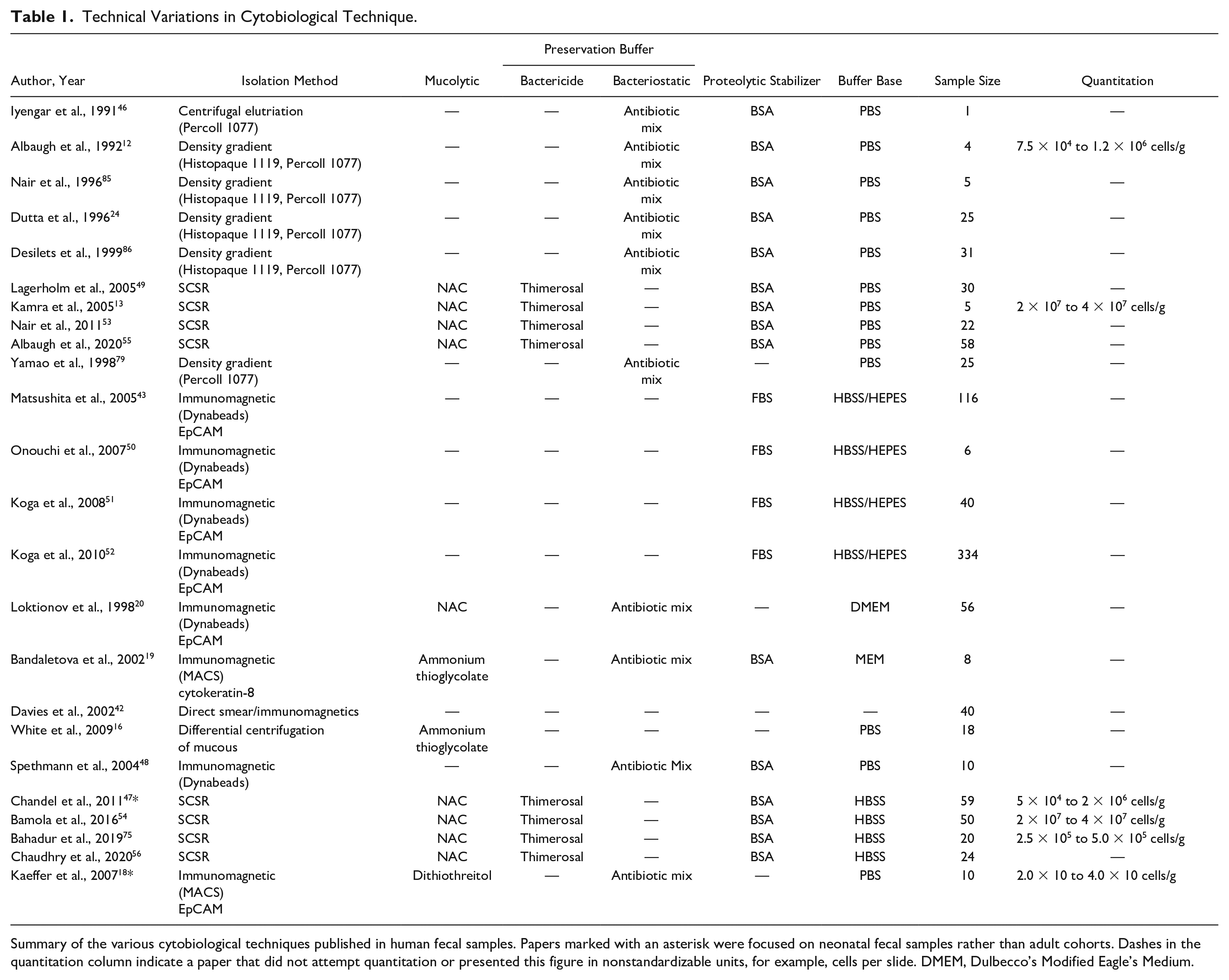
Summary of the various cytobiological techniques published in human fecal samples. Papers marked with an asterisk were focused on neonatal fecal samples rather than adult cohorts. Dashes in the quantitation column indicate a paper that did not attempt quantitation or presented this figure in nonstandardizable units, for example, cells per slide. DMEM, Dulbecco’s Modified Eagle’s Medium.
Preservation and Collection of Fecal Samples
Preservation and collection of fecal specimens can have a strong influence over the efficacy of isolation methods for downstream analyses, as cells can be degraded and lost if not adequately preserved.
Cold-chain management is a method of preserving cells and DNA in a sample. The use of low temperatures (<4 °C) reduces damage to the integrity of cells present for up to 24 h. 58 Cold-chain management is the preferred method of collection in microbiome and DNA-based studies of feces. 58 Davies et al. compared optimal storage temperatures and times of fecal samples with HT-29 colonic epithelial carcinoma cell spike-ins. It was established that cell loss was minimal in this cell line when samples were kept at 0 °C for less than 24 h. 42 Cold-chain preservation, however, is a costly and cumbersome process and unsuitable for any clinical practice that requires patients to self-collect fecal samples. 59 In response, a series of ambient temperature buffers have been developed to negate the effect of the fecal environment on exfoliated cells at room temperature.
Traditional Cytological Preservative Buffers
Ambient preservation buffers are an essential factor in the successful implementation of coprocytobiological techniques for colorectal cancer screening. Indeed, preservation buffers are employed in current screening approaches, such as the iFOBT and Cologuard, to prevent DNA or hemoglobin from being degraded by enzymes and bacteria in fecal samples. 60 Tris-ethylenediaminetetraacetic acid (TE) is incorporated in these designs to preserve and suspend DNA or analytes.61–64 However, TE is unsuited to cell preservation approaches as it solubilizes membrane proteins and lyses cells. 65
Several routinely used cytological cell preservation approaches have been trialed in fecal samples. Formalin and methanol are often used to preserve tissue samples when the desired outcome is a tissue-based or cellular analysis. 66 These approaches can cause crosslinking of DNA and proteins, preventing the application of downstream techniques that rely on cell surface receptors or the analysis of molecular markers. 66
Methanol-based cytological buffers, such as Thinprep and Preservcyt, were instrumental in the development of liquid-based cytology for cervical cancer screening. 67 Methanol preservation dehydrates cells while preserving surface epitopes (proteins required for immunological techniques). Several groups have attempted to use these formulations as preservative buffers for fecal samples;16,57 however, fecal mucins prevented cell isolates from adhering to slides and therefore hindered sample processing and accurate visualization. 16 Methanol and formalin are also highly toxic, rendering them unsuitable for applications that allow patients to collect samples from the comfort of their home. Traditional cytological cell preservatives appear to be unsuited to the complex nature of fecal samples, resulting in the design of several innovative buffers.
Composite Ambient Temperature Buffers
Complex biosamples, such as feces, contain unique factors that have the potential to render traditional preservation approaches inadequate for downstream applications. Gastrointestinal mucins, degradative enzymes, and a constant state of bacterial metabolism in feces can interfere with cytological analysis and cell isolation protocols.16,43 For greater clinical efficacy, preservative buffers can be composed of several elements to combat the specific limitations of the biosample and target a desired outcome. Furthermore, the constituents of these buffers must be safe for patients to collect samples from the comfort of their home.
Proteolytic Stabilization
Fecal samples contain a mass of free-floating bacterial proteolytic enzymes as well as native host enzymes that degrade and lyse exfoliated cells over time.4,42 Animal serum proteins are often used in preservation buffers as proteolytic stabilization agents. 44 Organic compounds, such as bovine serum albumin (BSA), or the closely related fetal bovine serum (FBS), saturate preservative solutions with protein, buffering vital cellular proteins from degradation by free-floating enzymes in the fecal mass. 44 Animal proteins such as BSA occupy the active site of proteolytic enzymes, preventing them from binding and degrading vital cellular proteins found in the membranes of exfoliated cells.4,44 These agents are preferable protease stabilization agents as they are used in the running buffers for downstream processes, such as fluorescence-activated cell sorting (FACS) or magnetic-activated cell sorting (MACS), and therefore do not interfere with these processes. 68
FBS and BSA have been employed as proteolytic stabilization agents in a series of composite buffers used for the preservation of fecal host cells and are widely regarded as a necessary component of any fecal transport or preservation medium.12,19,43,44,46 Matsushita et al. used spike-ins of a colon cancer cell line (HT-29) to measure the retrieval rates of preservation and isolation methods in stool under ambient storage conditions. 43 The group was able to measure a 20% improvement in cells retrieved when using 10% FBS in Hank’s Buffered Saline Solution (HBSS) mix instead of HBSS alone. 43 Interestingly, these results were replicated by Onouchi et al., who found that the buffer was capable of preserving fecal DNA for up to 48 h at room temperature. 50
In fecal samples, the somatic cell sampling and recovery (SCSR) kit is the only commercially available buffer that claims to preserve cellular morphology.4,44,69 The SCSR preservation buffer is a composite of several reagents used to achieve cell preservation at ambient temperatures in fecal samples.4,44 The U.S. patent application for this preservation buffer contains several possible compositions as well as a “preferred transport medium,” which employs BSA as a proteolytic stabilization agent. 44
Bactericidal and Bacteriostatic Agents
Bacterial cells in feces metabolize the cells around them and produce degradative by-products. 44 Bacteriostatic agents can be employed in cell preservation buffers to mitigate degradation. 44 These agents are used to disrupt the function and replication of bacterial cells while leaving host cells or analytes intact. 70 Antibiotic mixes suspended in saline solution have been employed to this effect in fecal samples.12,19,20,46 Three groups have used an antibiotic mix containing amphotericin, gentamicin, streptomycin, and penicillin suspended in a composite buffer composed mainly of saline solution or Eagle’s Minimum Essential Medium (MEM).12,19,20,46 These antibiotic formulations were capable of preserving high yields of viable fecal colonocytes (>80%) for more than 24 h at ambient temperatures. 12 However, the solutions still had to be chilled on ice until analysis, likely because the bacteriostatic agents had a limited penetrance into the mass of the stool and could not disperse in high enough concentrations to halt the replication of bacteria deep in the mass of stool and gastrointestinal mucus.12,19,44,46
Bactericidal agents are used to lyse bacterial cells and release their cellular contents into the surrounding samples, leaving host human cells intact. 71 The release of bacterial enzymes and cellular contents into the solution could produce further cellular degradation of host cells; however, when coupled with a proteolytic stabilization agent, such as BSA, the effect of this release can be reduced, “mopping up” free-floating enzyme. 72 This design is used in SCSR’s ambient preservation buffer, which uses low concentrations of the mercury-alkyl compound Thimerosal as a bactericidal agent, together with BSA as a proteolytic stabilization agent. 44 Nair et al.’s SCSR design has had successful results in preserving host cells at ambient temperatures for more than 24 h.13,47,53–56
Mucin Dispersal
The solid mass of a stool is held together by gastrointestinal mucins, a viscous matrix that creates a barrier preventing the flow of fluids, such as antimicrobials and proteolytic stabilization agents, throughout the solid mass while also harboring bacterial cells.16,44,73 Consequently, the efficacy of host cell preservation buffers is significantly impeded.16,44 Incomplete penetrance of an antimicrobial solution can result in bacterial cells degrading and metabolizing the epithelial cells around them. 44 Furthermore, the viscosity of mucins can inhibit downstream processes that rely on the channeling of cells through tight membranes or filters.16,47 To overcome these limitations, mucin-degrading compounds can be included in preservation buffers. Examples are n-acetyl-cysteine (NAC) and potassium iodide.16,44 NAC was used in Nair et al.’s SCSR buffer, breaking disulfide bonds of mucins, effectively disbanding the viscous matrix. 74
White et al. trialed the use of NAC to disperse gastrointestinal mucins and capture colonic epithelial cells from feces. 12 Surprisingly, the approach was found to be inefficient for separating the mucin matrix that prevented immunohistochemical analysis of fecal-derived cells. 16 White et al. instead opted to use a fibrin clotting method to suspend the cells. 12 Their isolation approach failed to adequately remove fecal debris, a limitation common in coprocytobiological publications.16,19,43,47,48 It is possible that the presence of debris in White et al.’s method may have diluted or prevented the action of the mucolytic compound present.
In contrast to White et al.’s work, Bandaletova et al. were able to examine cells using immunocytochemistry after employing a similar mucolytic agent (5% ammonium thioglycolate) suspended in a composite buffer. 19 They also employed an alternative method of cell isolation, which may have contributed to the success of the method (see “Immunomagnetic Separation” section). NAC has also been used to considerable effect in a series of publications that relied on alternate cytological methods of examination rather than immunocytochemical approaches.13,47,53–56 Further investigation is required to determine the reasons for the disparity in reporting on the efficacy of mucolytic agents in fecal samples.
Cell Isolation
Cytological assessment of feces has been previously limited by low numbers of cells in samples. 19 Isolation methods can be used to reduce the debris present in a sample and enrich a target cell type to improve visual cellular analysis. 68 Several methods have been used traditionally to target the properties of specific cell types to isolate them from mixed populations. 68
Size Filtration
Size filtration has been applied for decades to limit the flow of larger objects using a network of tightly arranged fibers or a porous membrane. 68 This method separates the resulting liquid and smaller structures (the filtrate) from the mass of larger material impeded from moving through the tight fibers or pores of the filter (the residue). 68 Filtration is a quick, cheap approach to separating host cells from debris and bacterial constituents, although often ineffective in highly viscous samples. Fecal samples contain a number of large particles, such as food-based proteins and cells, which clog filters and reduce their efficacy as a lone isolation method.57,68
Size filtering approaches have been used as pretreatments for more complex isolation methods. Notably, White et al. and Matsushita et al. used filters with distinctively large pore sizes (512 and 125 µm, respectively).16,43 Matsushita et al.’s and White et al.’s experimental methods were unique in their ability to isolate clumps of malignant cells that maintained structural features and tissue architecture. This was not reported in publications that used pore sizes <60 µm ( Fig. 2 ).13,19,47,53–56,75 Matsushita et al. also noted a significant reduction in the retrieval of cultured cells spiked into fecal matter when comparing filters of 96 or 48 µm with their 512 µm filter.43,57 However, no mucolytic agent was present in their design; therefore, the clogging observed in their filter may have been due to the presence of excess mucin and not the pore size of the filter. 43
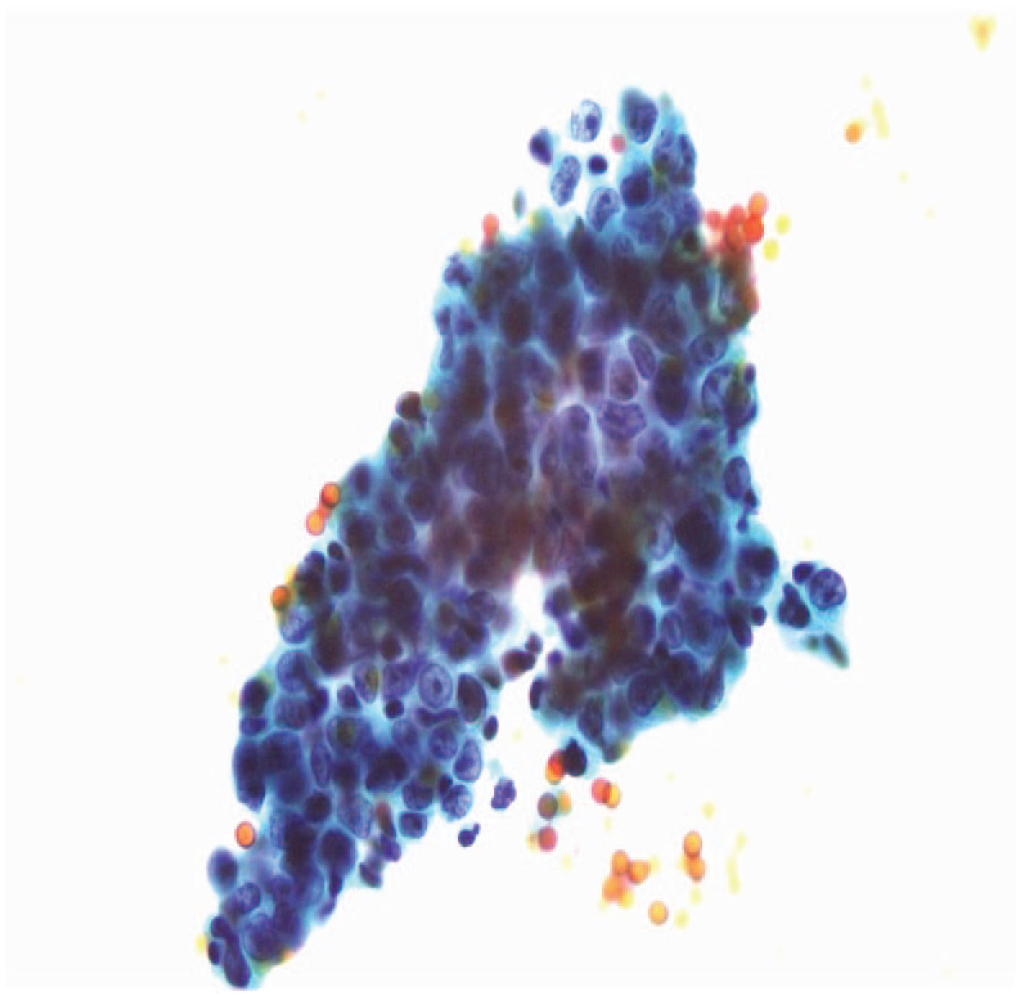
Clump of cells observed in host cell-enriched isolates of fecal samples. Papanicolaou staining of a fecal cell isolate from a patient with rectal cancer (Dukes’ stage C) visualized under 400× light microscopy. [No scale provided.] Reproduced from Matsushita et al. 43 with permission from Elsevier.
Density Gradients
Density gradients are the most widely used cell isolation method and rely on a slightly more complex principle than simple filtration. 76 Gradients separate cells based on density; cells are layered atop a specific solution before centrifugation is applied to implement gravitational pressure to the sample and gradient ( Fig. 3 ). Cells will migrate to different regions of the gradient under centrifugation, as the density of cells will be equal to the density of that region of the gradient. 68 Removing fractions of the gradient allows cells of differing density to be separated ( Fig. 3 ). 68
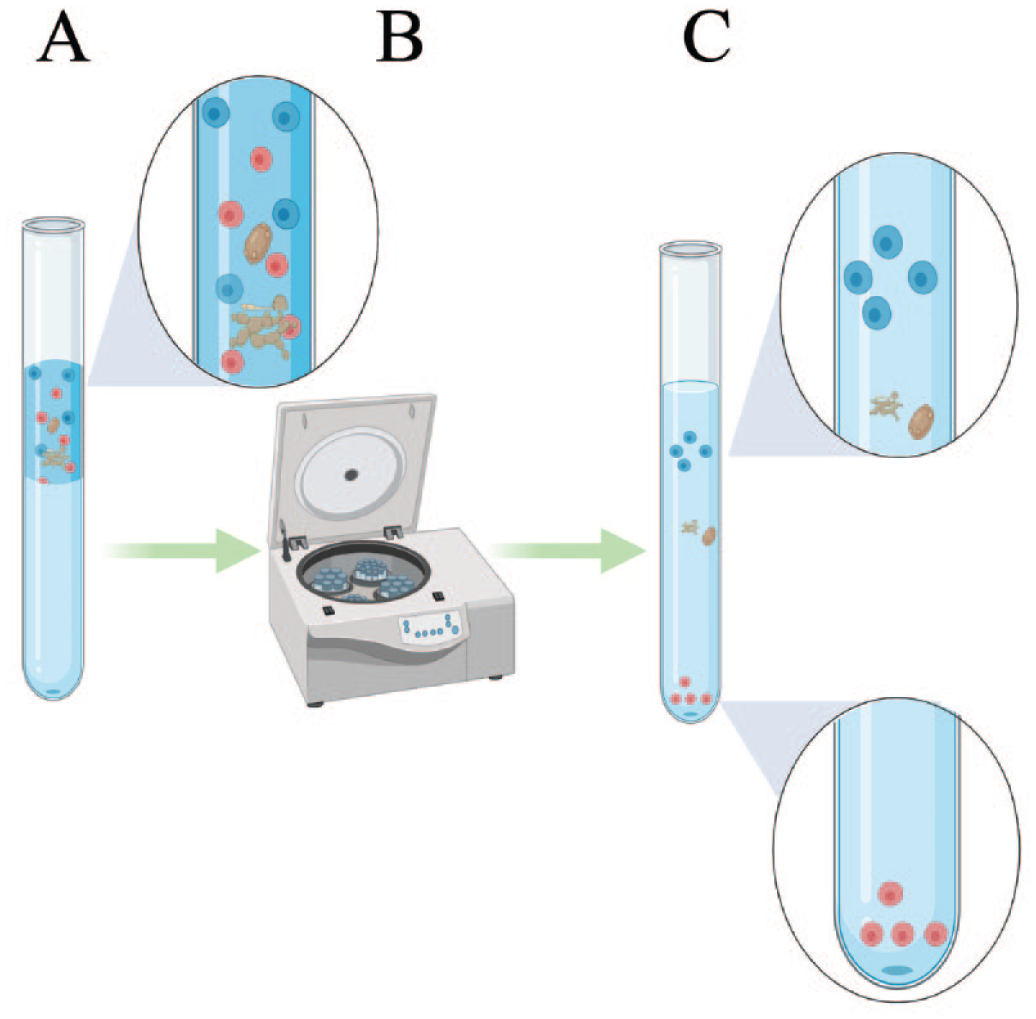
The density gradient separation process. (
Somatic Cell Sampling and Recovery
Iyengar et al. were able to isolate colonocytes in fecal specimens after Percoll gradient centrifugation. 46 When coupled with a preservation buffer and simple filtering pre-preparation, this method became the basis for the SCSR procedure. 4 SCSR has been applied to fecal samples in a range of settings for the past 30 years and laid the groundwork for noninvasive testing of exfoliated cells in stool samples. SCSR cell isolates have been used in several proof-of-concept applications, including the investigation of colorectal cancer, irritable bowel disorder, and mucosal immunity.13,53,54 Despite the number of notable publications that used the method, SCSR has never been applied as a clinical screening method. Publications that use SCSR have received some criticism for their reports on the number of cells isolated using the method.19,77 Discrepancies in the reporting of cell morphology and size are another possible explanation for the method’s lack of clinical adoption (discussed in the “Cytology” section). Furthermore, SCSR is commercially available at the cost of $33 AU, doubling the cost of current cytological screening measures. 44 Alternatively, commercially available density gradient media can be combined with generic preservation buffer formulations to produce colonic epithelial cells for less than $5 AU. 19
Immunomagnetic Separation
Immunomagnetic separation employs a bead–antibody conjugate ( Fig. 4 ) to isolate a specific cell type based on a cell surface receptor. External magnetism then draws the conjugate-bound cells away from the rest of the sample to be purified. This method became widely used in the 21st century with the commercialization of MACS and is used in a range of sample types, such as blood and tissue. 68 Although usually very specific, some nonspecific binding to the conjugates can occur in samples containing large numbers of degraded cells and proteins. 78
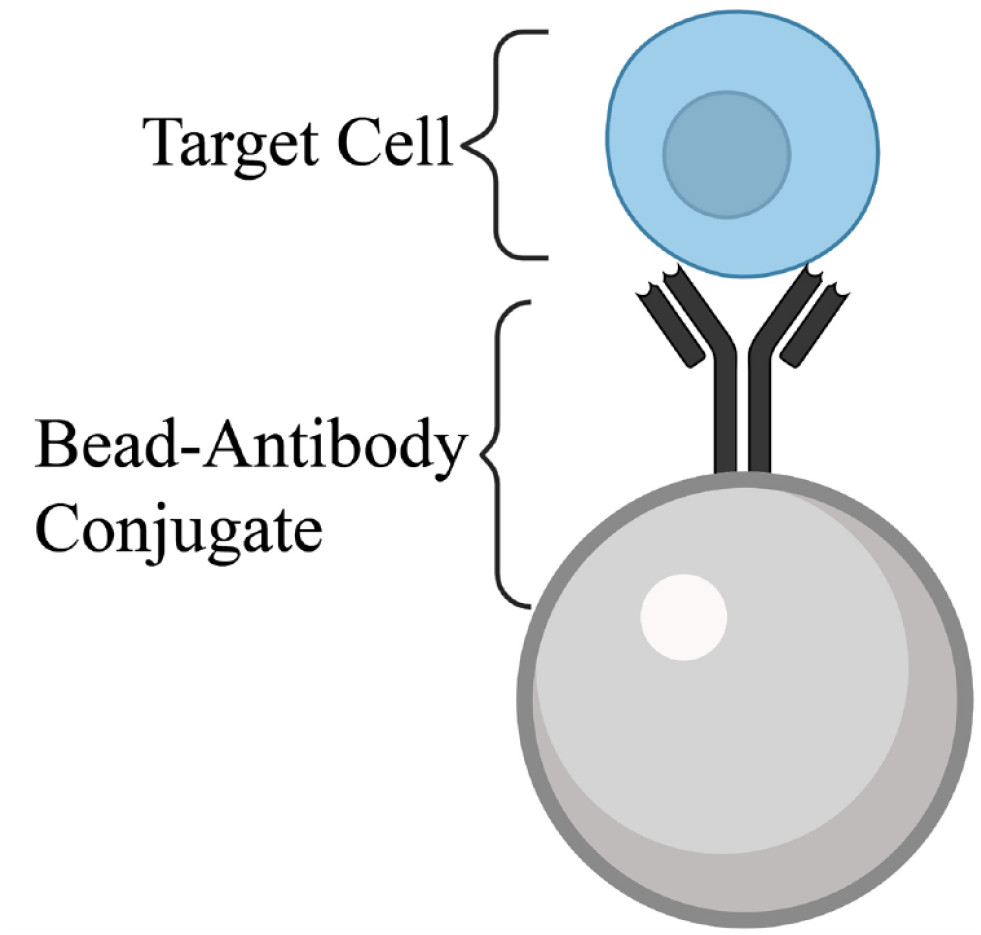
Bead–antibody conjugate. An antibody conjugated to a ferrous bead complexes with complementary surface markers to bind a target cell. This complex can then be pulled out of heterogenous samples via an external magnetic field.
Immunomagnetic Separation in Fecal Samples
Magnetic capture started to be applied to a range of samples types throughout the early 2000s. With the commercialization of MACS and Dynabead conjugates, immunomagnetic techniques became common practice lab methods.16,19,43,50,51 Immunomagnetic separation in fecal samples has become almost as common as density gradients, and this isolation approach has been utilized in a range of studies.16,19,43,50,51 Bandetelova et al.’s application of anti-cytokeratin 8 monoclonal antibody (mAb) MACS beads to stool surface washes provided ones of the first reliable characterizations of colonocytes in stool samples. 19 Davies et al. also reported similar morphological characteristics in cells isolated using epithelial cell adhesion molecule (EpCAM) conjugated to magnetic beads, establishing a cytological method for discriminating subtypes of colonic growths using minichromosome maintenance protein 2 (MCM2) immunocytochemistry. 42 Spethmann et al. compared similar EpCAM-conjugated beads to density gradients. 48 Spethmann et al. used reverse transcriptase (RT)-PCR analysis of isolated cells to verify earlier claims that immunomagnetic approaches produced results with greater reproducibility.48,79 The improved morphological characterization between density gradients and immunomagnetic methods was later cited by Matsushita et al. as well as Koga et al. in a series of papers based on optimization and implementation of immunomagnetic techniques in stool samples.43,51 Matsushita et al. provided the most promising evidence that cytology and human cell pulldown from stool samples could be clinically employed to investigate pathology while at the same time concluding that DNA-based methods were more easily tailored to the detection of colorectal cancer. 43 At present, fecal sample diagnostics are dominated by molecular methods, and yet a lack of performance still exists in the accurate detection of colorectal cancer. Further investigation of immunomagnetic techniques alongside improved cell sorting approaches and newly developed technologies is a possible avenue for optimization in coprocytobiology.
Microfluidic Devices
Microfluidics refers to the study of particle flows at sizes of less than a micrometer. 80 This field has been instrumental in the development of “lab-on-a-chip” technologies, a term given to a range of different physical devices that can separate cells based on various physical and chemical factors. 80 The devices are most commonly constructed of polymethylmethacrylate (PMMA) and use a range of innovative methods to separate cells, including physical methods, such as filtration, and affinity-based methods, such as aptamer binding. 80 A range of designs and approaches to filtering and construction are reviewed in Dalili et al. 80 Lab-on-a-chip designs are a particularly advantageous method of cell enrichment because of their specific design and ability to incorporate multiple cell isolation and engineering principles into a single device. Sample-specific factors such as viscosity and presence of debris can also be compensated for using specialized channels, filters, or other design features. 80 Furthermore, these devices can be mass manufactured for less than $0.1 (AU) per device, making them an extremely economic alternative to current cell isolation methods. 81 Tailored microfluidic designs have been applied to a range of mixed samples to great effect and appear to be a fledgling field in the world of coprocytobiology. Many microfluidic designs have been tailored to the detection and isolation of cancer cells and therefore have relevance to colorectal cancer screening.
Microfluidic Devices in Feces
Lab-on-a-chip devices are fast becoming a widespread method for cell isolation, enumeration, and sorting. These devices have recently been applied to fecal samples and could potentially be applied as an isolation approach to improve coprocytobiological techniques. A recent device described by Zhao et al. holds promise for the field of coprocytobiology. 82 The authors applied innovative passaging and filtering designs that used channeling and acoustic stimulation to dilute viscous fecal samples ( Fig. 5 ). The device was able to effectively liquefy and homogenize human stool samples, subsequently filtering large debris and obtaining a purified sample that contained a high proportion of live bacterial species. 82 Samples were not examined for the presence of human cells; however, large cellular structures of morphology similar to that of human epithelial cells can be observed among scanning electron microscope images of the resulting homogenate. 82 Devices such as those described by Zhao et al. could potentially be applied to liquefy, isolate, and enrich colonocytes for further study of exfoliated epithelial cells and possibly detect circulating tumor cells via cytological means.
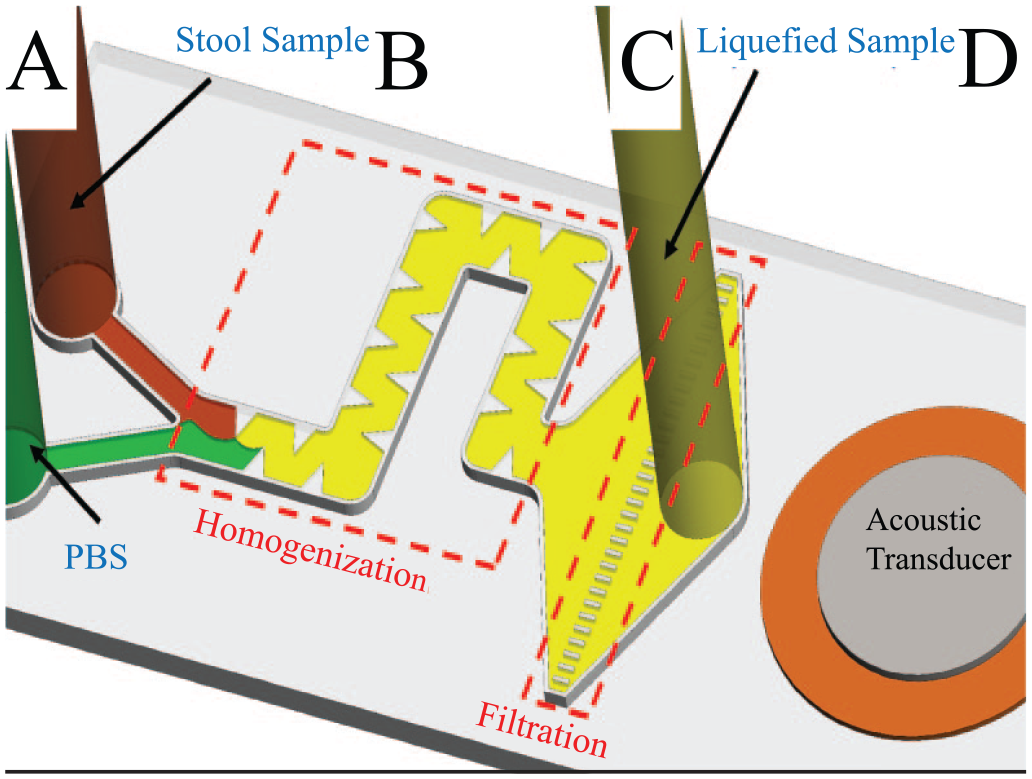
Diagram of the acoustofluidic stool liquefication device. (
Fecal Host Cell Isolation from Surface Washings
The most recent innovation in coprocytobiological cell isolation was made by White et al. 16 The method combined several existing methods into one isolation and cytology protocol. With a modified filtering and differential centrifugation approach, colonocytes were isolated from stool surface washes and observed using MCM2 immunocytochemistry. In their cell isolates, White et al. noted similar morphology to that previously described.16,19,42 The surface washing isolation method used by Bandetelova et al. and White et al. used fluid movement on the outer surface of a stool to remove the fecal mucus and cells from this layer.16,19 This method resulted in roughly one-tenth of the number of cells per slide required by Matsushita et al.’s screening method,16,43 possibly because surface washing ignores the effect of peristalsis, which mixes the contents of a stool, resulting in the internalization of cells that have been shed onto the surface of a sample. However, White et al. showed that relatively low numbers of viable colonocytes could be used to identify colorectal adenocarcinoma with a 50% sensitivity and 100% specificity, providing an important basis for further investigation of the many previously applied coprocytobiological techniques ( Table 1 ). In the last decade, novel preservation and isolation techniques such as those described by White et al. and Zhao et al. have changed the landscape of coprocytobiology. Applying well-established cytological techniques to these novel methods could be used to develop cost-effective screening regimes for colorectal cancer.
Cytology
Morphological cellular changes are a hallmark of colorectal cancer development; measurement of these cellular changes has been, for the most part, indirect. Current screening regimes focus on subtle molecular differences in fecal composition rather than the obvious cellular changes that accompany them. Cytology provides an economic and simple alternative to molecular screening methods. Previous publications have trialed several differing coprocytobiological methods to make this a possibility ( Table 1 ). However, inconsistencies in coprocytobiological methodologies have resulted in variable levels of success.
Cytological Examination of Feces
The SCSR isolation method has been an influential isolation method in the world of coprocytobiology. Cytological data obtained from SCSR isolations have influenced many papers in the field and have been the sole focus of the two early reviews of coprocytobiology.4,77 Although the efficacy of this method has been divisive, Loktionov’s 2007 mini-review identified a lack of cytological evidence to explain the apparent success of the SCSR method.
77
Furthermore, a discrepancy in the reporting of cytological images obtained from the method exists. Albaugh et al.’s paper from 2020 reprinted the only published image of an SCSR “colonocyte” from Nair et al.’s 2003 paper and renamed this cell a “gastrointestinal progenitor cell (GIPC)” (
Fig. 6C
),4,55 describing it as a specialized epithelial cell that “in addition to acquiring progenitor stem cell characteristics, exhibits cytotoxicity against tumour cells.”4,55 To date, no cytological evidence has been provided to indicate that SCSR isolates contain cells other than the small, circular GIPC and the goblet cell depicted in Nair et al.’s 2003 review (
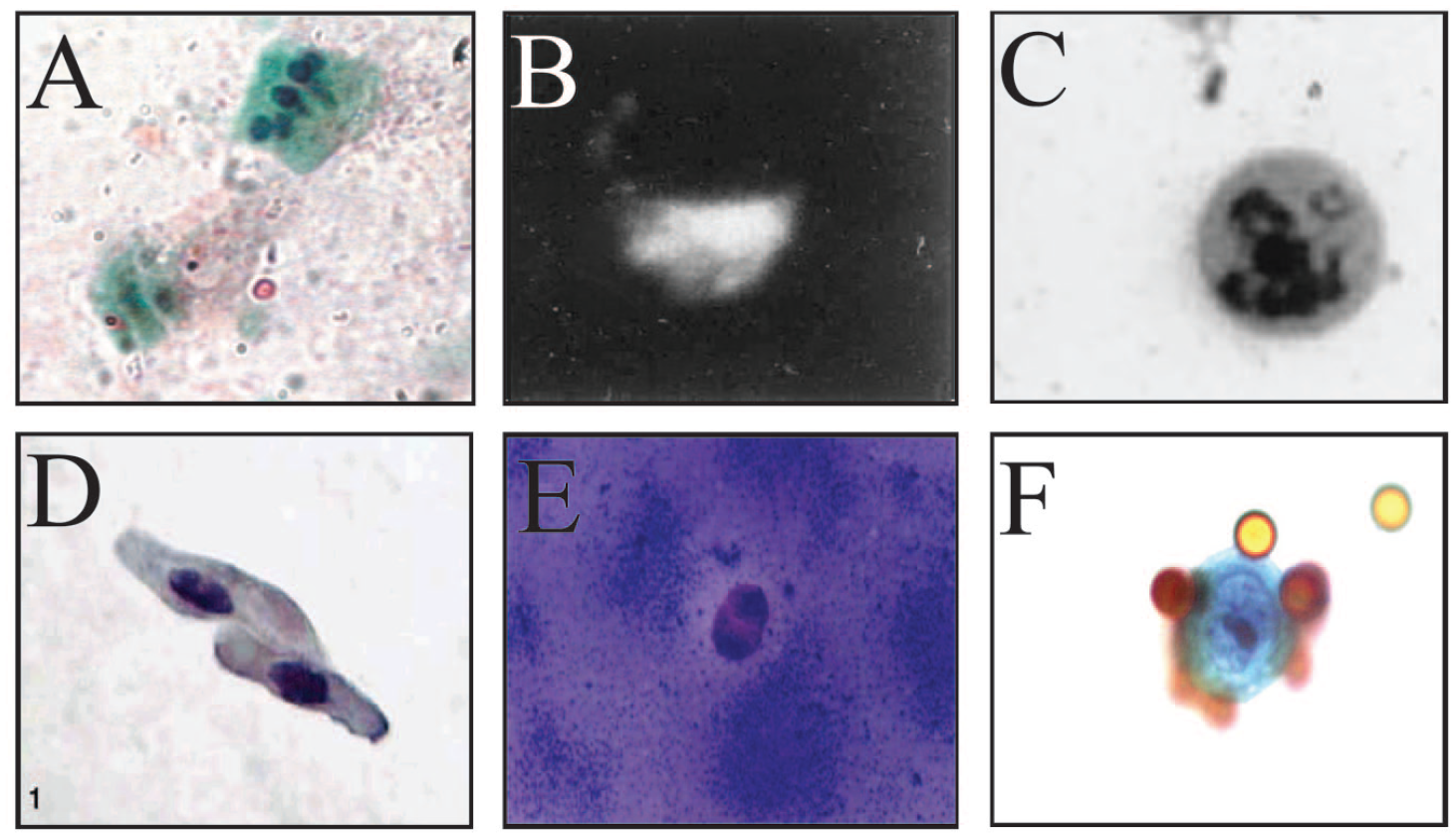
Images of exfoliated host cells after isolation from fecal samples. (
Albaugh’s, Nair’s, and Iyengar’s groups have also consistently asserted that human cells isolated from stools via SCSR exist in two size populations of 2–5 µm and 5–8 µm, making the largest cells measured in their FACS analyses about 50% of the size of colonocytes identified by other groups, such as White et al., Bandaletova et al., and Davies et al. (
Multichromatic staining using a series of counterstains is utilized in the Papanicolaou method. 83 Matsushita et al. used this method in cell isolates to demonstrate that malignant cells could indeed be identified in fecal samples. 43 Coupled with cell isolates obtained by surface binding of specific features, this approach could be adapted to stratify colorectal abnormalities based on their selective binding and morphology. Figure 2 shows an example of a cell clump found in the stool of a subject with colorectal adenocarcinoma. Dysregulation of apoptosis and variation in EpCAM binding are characteristic of colorectal cancer progression and the likely cause of these features. 84 Matsushita et al. also used cytological measures, such as the nuclear-cytoplasmic ratio, to identify malignant cells in the fecal samples of subjects ( Fig. 6F ).
Matsushita et al.’s cytological method had limited sensitivity (28%) and high specificity (100%) for colorectal cancer. 43 Matsushita et al. compared the host cell retrieval of Percoll density gradients to magnetic bead isolation, reporting much higher retrieval rates in the immunomagnetic method (33.5% vs 0.8%). This claim is in contradiction to many of the colonocyte number estimates made with density gradient techniques and findings on the recovery rates of magnetic beads given by other publications.12,46,47,54–56,85 Interestingly, no bacteriostatic or bactericidal agent was applied in their preservation buffer, as has been standard in other publications ( Table 1 ).4,12,13,16,18,19,24,43,46–49,51,53–56,79,85,86
Notably, the Dynabeads Epithelial Enrich (Dynal, Oslo, Norway) used by Matsushita et al. contains 0.1% of sodium azide, a bacteriostatic agent.44,87 The presence of sodium azide in the immunomagnetic method could explain the unprecedented disparity between the two isolation approaches. Incidental lysis of bacterial cells in the immunomagnetic approach could have reduced cell degradation due to the presence of low concentrations of sodium azide. 88 However, it is unlikely that such a low level of bactericide completely halted the bacterial metabolism of epithelial cells in the successful method. These incidental variations in bactericide concentrations may have been partly responsible for the low sensitivity of Matsushita et al.’s overall cytological method.
The low retrieval rates of Matsushita et al.’s cell isolation approaches were provided as an explanation for the low sensitivity of their cytological method. This method, however, hinged upon a rudimentary preservation buffer design that may have influenced the results of their study. 43 Further review and optimization of coprocytobiological techniques could be used in the future to increase cell yield and achieve better sensitivity using the Papanicolaou method. Overall, the work serves as an important proof of concept and outlines areas for future improvement in the field.
Immunocytochemistry of Isolated Cells
Cell immunostaining has also been applied to investigate cell surface markers for pathological investigations of fecal samples.16,42 The method uses interactions between cellular features or surface markers and cytological dyes to identify clinically relevant features of cells. 42 Minichromosome maintenance proteins have been applied in this manner for the investigation of colorectal pathologies ( Fig. 6A ). 42 These proteins are vital for DNA replication and highly upregulated in the case of symptomatic colorectal cancer. 42 Davies et al. were able to differentiate neoplastic from healthy patients in 37 of 40 fecal cell isolates using this method coupled with immunoperoxidase staining. 42 Furthermore, Davies’ approach identified 25 of 25 healthy patients from their fecal isolates. This approach was then replicated by White et al., who again correctly identified all healthy patients, also finding that 50% of affected individuals could be identified via MCM2 immunocytochemistry of fecal cell isolates. 16 Both Davies et al. and White et al. used cell isolation and preservation processes with a relatively low yield. By implementing improved high-yield coprocytobiological processes, immunocytochemistry could be implemented as a supplementary approach to the Papanicolaou method, and data from both sources could be used to develop a highly sensitive and specific screening method.
While instances of these hallmark features have been pinpointed, little has been published to characterize the heterogenous cell populations found in the stools of individuals affected by colorectal disorders. Further clinical examination of these features using novel isolation methods should be conducted in order to establish cytological guidelines to develop clinically relevant processes for the identification of colonic abnormalities.
Conclusion
The landscape of coprocytobiology has changed since the term was first coined in the early 2000s. The utility of these processes has increased greatly, as the costs of molecular techniques remain high and new technologies have become available. The future of the field depends on the development of novel and economic methods. This can be achieved through the integration of several existing and emerging technologies and techniques. Ambient temperature preservation should be the focus of cytological screening programs, which may be aided by further investigation into the use of composite buffers tailored to the characteristics of feces. The availability of advanced microfluidic designs paves the way for new cell separation and sorting techniques. Cell separation and cytology have the potential to be developed into integrated designs that detect and analyze multiple cytological features of host cells. At the same time, cost should be the determining factor of future screening programs, and coprocytobiology should be reinvestigated with a focus on minimizing per-test patient expenses.
Footnotes
Acknowledgements
The authors would like to acknowledge the support afforded by the Australian Rotary Health and Rotary Club of Mornington through the Reg Kilborn Funding Partner PhD Scholarship, which allowed investigator Ryan to participate in this work.
The authors would also like to acknowledge that
We would like to thank Richard Alexander for his assistance and expertise in adapting
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
